Chapter 4 The Bohr Model of the Atom

Chapter 4 The Bohr Model of the Atom Part 1
Visible Light


Niels Bohr • In 1913 Bohr published a theory about the structure of the atom based on an earlier theory of Rutherford's. • Bohr expanded upon this theory by proposing that electrons travel only in certain successively larger orbits.
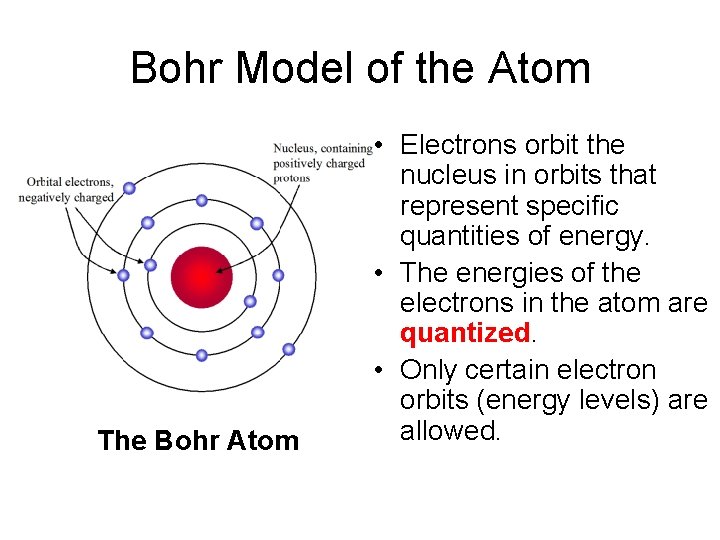
Bohr Model of the Atom The Bohr Atom • Electrons orbit the nucleus in orbits that represent specific quantities of energy. • The energies of the electrons in the atom are quantized. • Only certain electron orbits (energy levels) are allowed.

Niels Bohr • Bohr also described the way atoms emit radiation by suggesting that when an electron jumps from an outer orbit to an inner one, that it emits light.
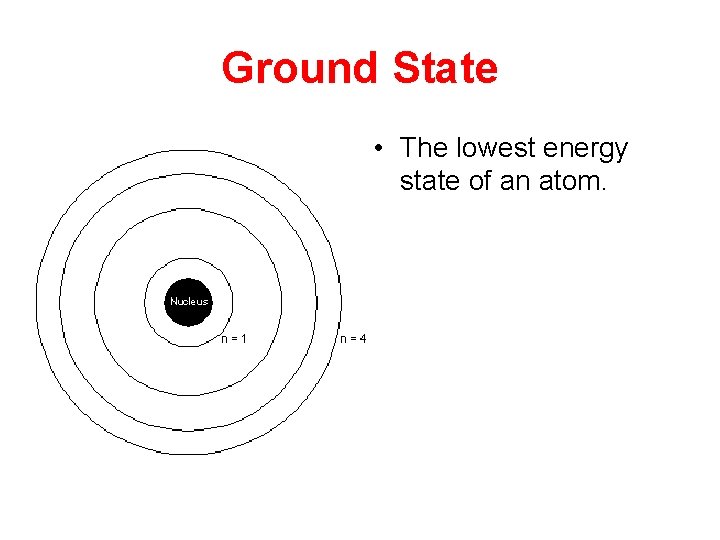
Ground State • The lowest energy state of an atom.
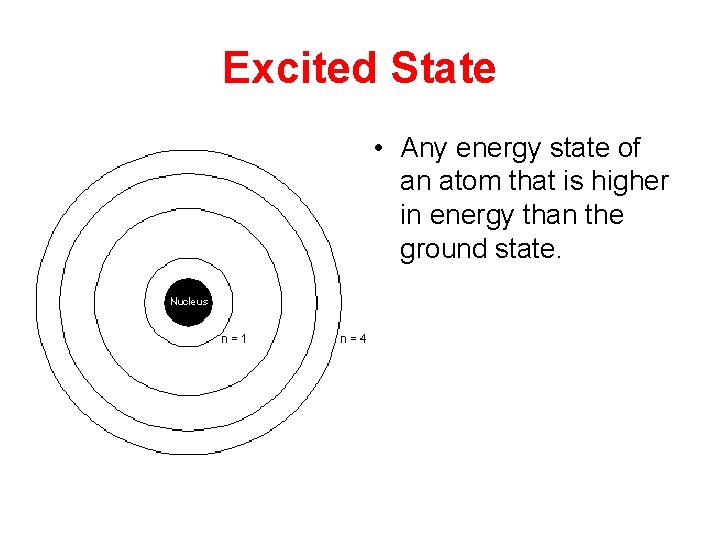
Excited State • Any energy state of an atom that is higher in energy than the ground state.
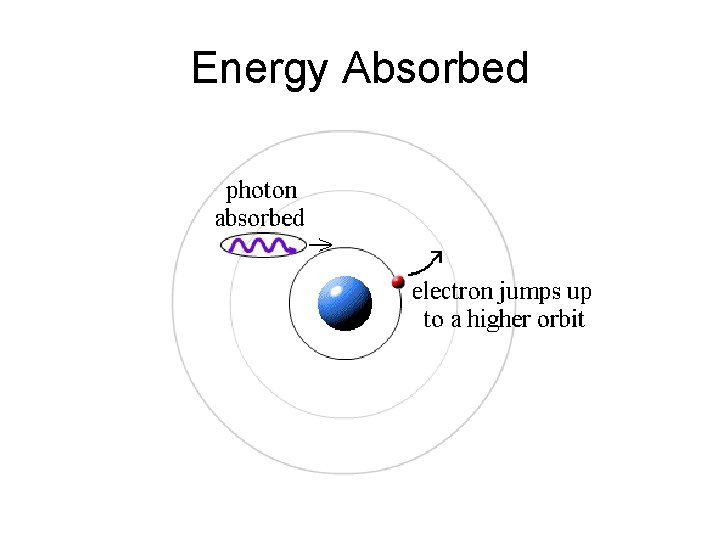
Energy Absorbed

Absorption (Dark – Line) Spectra
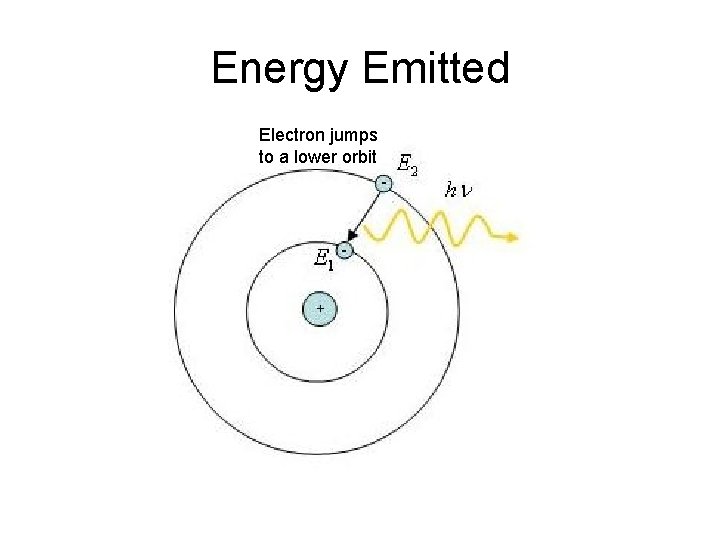
Energy Emitted Electron jumps to a lower orbit

Emission (Bright – Line) Spectra
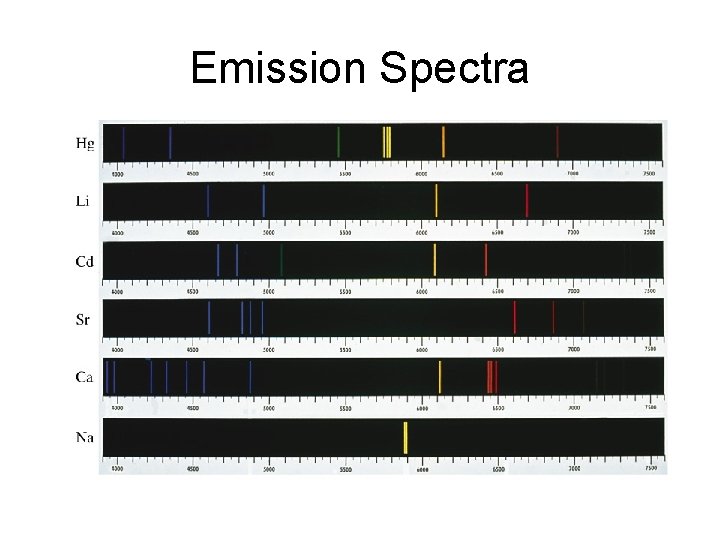
Emission Spectra

The lines present in an emission spectrum are the lines missing in an absorption spectrum.
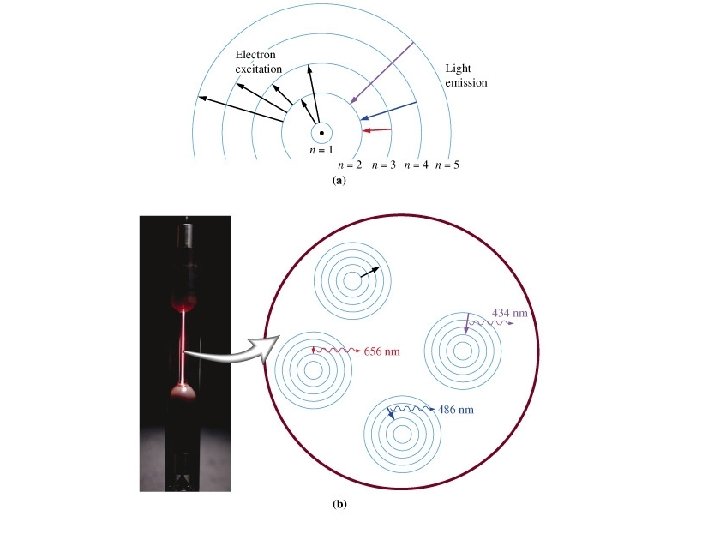
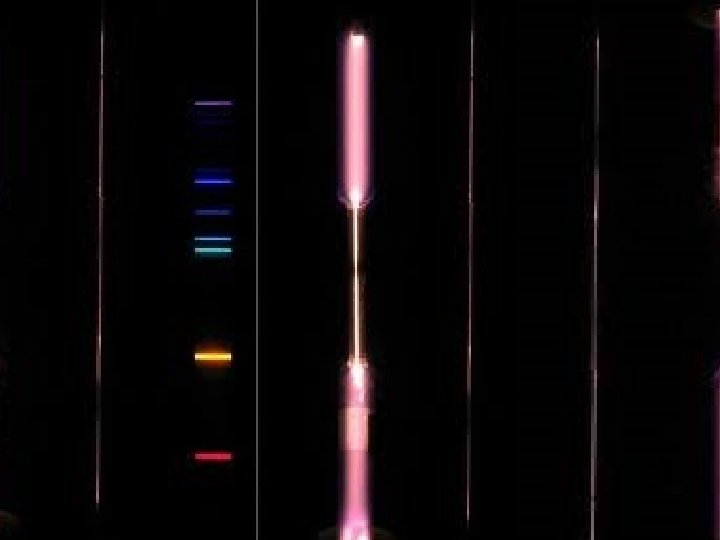

Describe the process that occurs inside atoms that produces the spectra? • Energy is absorbed by the atom. • The electrons jump into higher orbits and the atom becomes “excited”. • The electrons return to their original orbits and the atom returns to the ground state. • The electrons release energy in the form of visible light. • The specific energies (wavelengths, frequencies) of visible light absorbed and released produce the spectra.

Northern lights (Aurora’s) are one of nature's most beautiful manifestations of the ability of electrons to jump between quantum states
Northern Lights Video

Star Finder Video - Fingerprints of Light

Homework • Chapter 4: Worksheet 3 • Continue to work on your study guide for chapter 4 (2 pages front and back).
- Slides: 21