Chapter 4 Terpenes Biosynthesis of Monoterpenes Terpenes generally
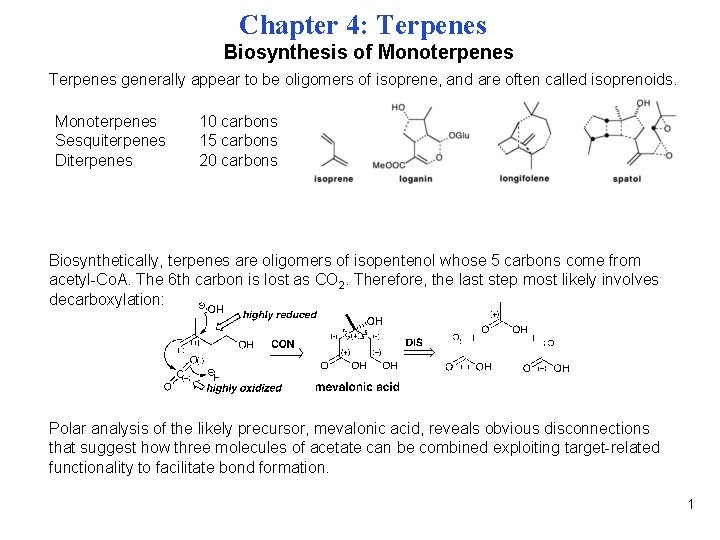
Chapter 4: Terpenes Biosynthesis of Monoterpenes Terpenes generally appear to be oligomers of isoprene, and are often called isoprenoids. Monoterpenes Sesquiterpenes Diterpenes 10 carbons 15 carbons 20 carbons Biosynthetically, terpenes are oligomers of isopentenol whose 5 carbons come from acetyl-Co. A. The 6 th carbon is lost as CO 2. Therefore, the last step most likely involves decarboxylation: Polar analysis of the likely precursor, mevalonic acid, reveals obvious disconnections that suggest how three molecules of acetate can be combined exploiting target-related functionality to facilitate bond formation. 1
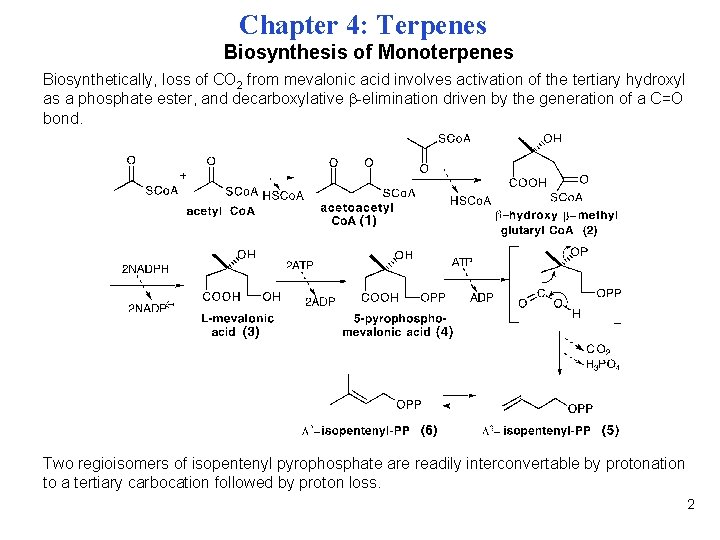
Chapter 4: Terpenes Biosynthesis of Monoterpenes Biosynthetically, loss of CO 2 from mevalonic acid involves activation of the tertiary hydroxyl as a phosphate ester, and decarboxylative -elimination driven by the generation of a C=O bond. Two regioisomers of isopentenyl pyrophosphate are readily interconvertable by protonation to a tertiary carbocation followed by proton loss. 2
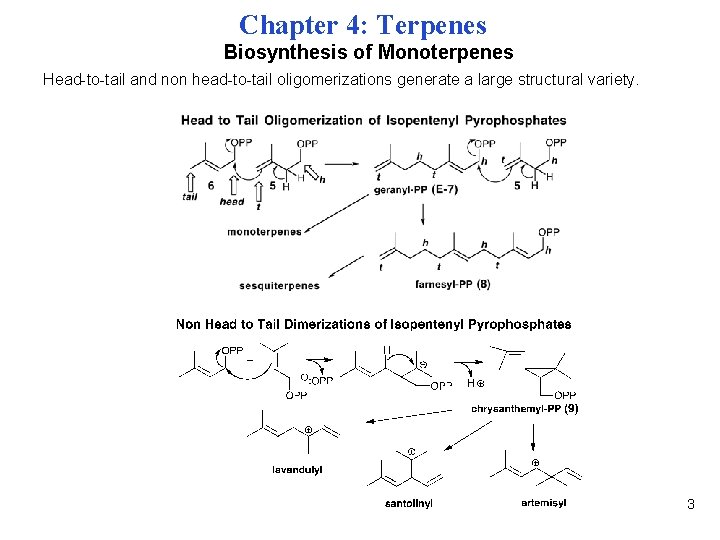
Chapter 4: Terpenes Biosynthesis of Monoterpenes Head-to-tail and non head-to-tail oligomerizations generate a large structural variety. 3
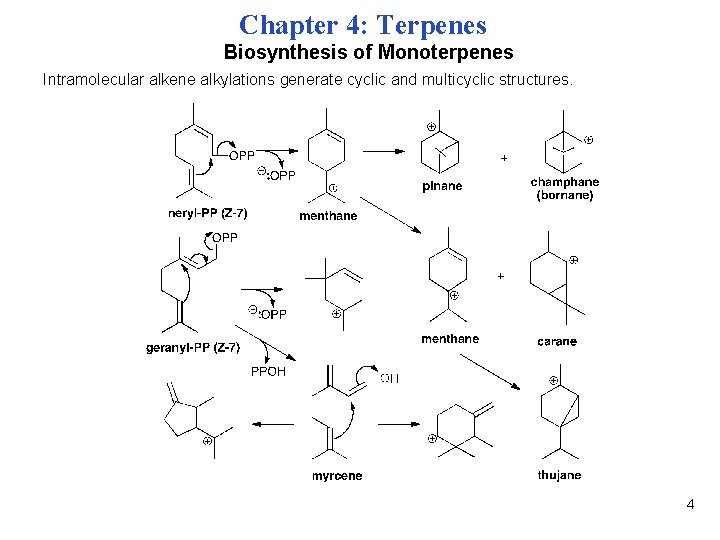
Chapter 4: Terpenes Biosynthesis of Monoterpenes Intramolecular alkene alkylations generate cyclic and multicyclic structures. 4
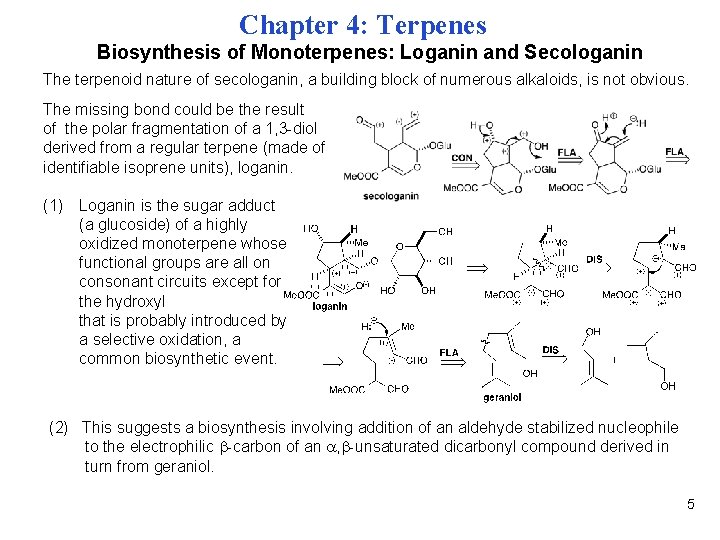
Chapter 4: Terpenes Biosynthesis of Monoterpenes: Loganin and Secologanin The terpenoid nature of secologanin, a building block of numerous alkaloids, is not obvious. The missing bond could be the result of the polar fragmentation of a 1, 3 -diol derived from a regular terpene (made of identifiable isoprene units), loganin. (1) Loganin is the sugar adduct (a glucoside) of a highly oxidized monoterpene whose functional groups are all on consonant circuits except for the hydroxyl that is probably introduced by a selective oxidation, a common biosynthetic event. (2) This suggests a biosynthesis involving addition of an aldehyde stabilized nucleophile to the electrophilic -carbon of an , -unsaturated dicarbonyl compound derived in turn from geraniol. 5
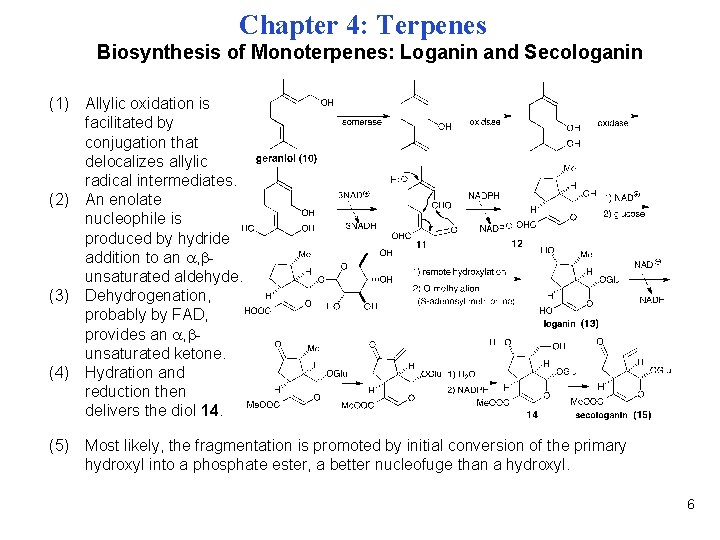
Chapter 4: Terpenes Biosynthesis of Monoterpenes: Loganin and Secologanin (1) Allylic oxidation is facilitated by conjugation that delocalizes allylic radical intermediates. (2) An enolate nucleophile is produced by hydride addition to an , unsaturated aldehyde. (3) Dehydrogenation, probably by FAD, provides an , unsaturated ketone. (4) Hydration and reduction then delivers the diol 14. (5) Most likely, the fragmentation is promoted by initial conversion of the primary hydroxyl into a phosphate ester, a better nucleofuge than a hydroxyl. 6
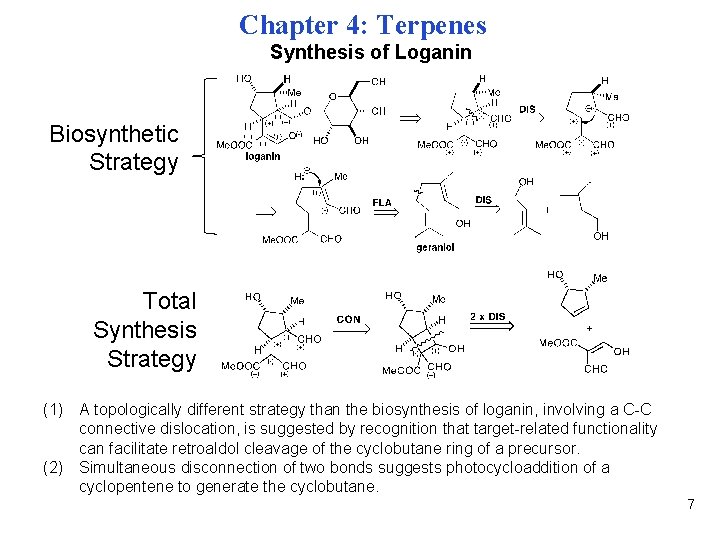
Chapter 4: Terpenes Synthesis of Loganin Biosynthetic Strategy Total Synthesis Strategy (1) A topologically different strategy than the biosynthesis of loganin, involving a C-C connective dislocation, is suggested by recognition that target-related functionality can facilitate retroaldol cleavage of the cyclobutane ring of a precursor. (2) Simultaneous disconnection of two bonds suggests photocycloaddition of a cyclopentene to generate the cyclobutane. 7
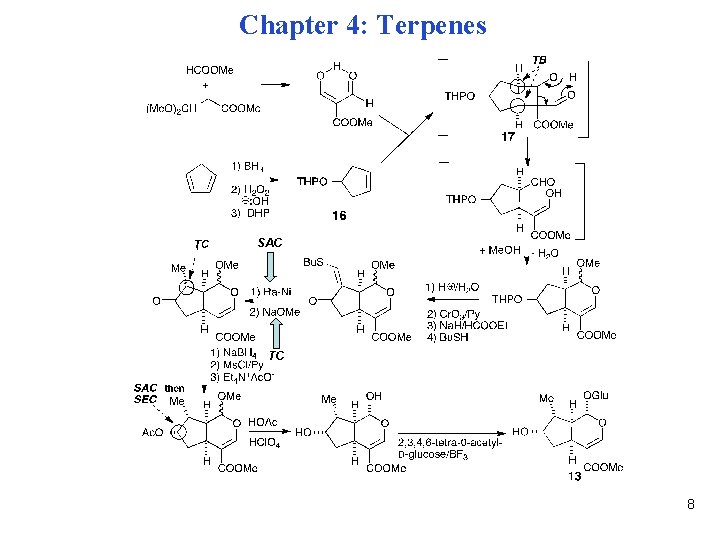
Chapter 4: Terpenes SAC TC 8
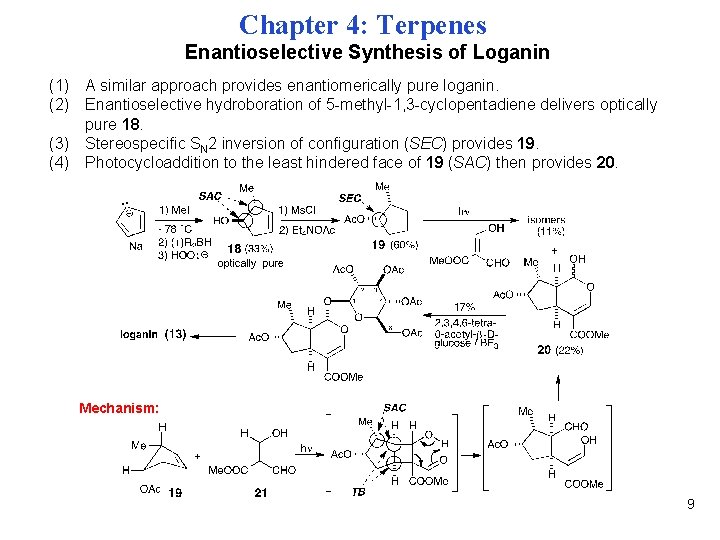
Chapter 4: Terpenes Enantioselective Synthesis of Loganin (1) A similar approach provides enantiomerically pure loganin. (2) Enantioselective hydroboration of 5 -methyl-1, 3 -cyclopentadiene delivers optically pure 18. (3) Stereospecific SN 2 inversion of configuration (SEC) provides 19. (4) Photocycloaddition to the least hindered face of 19 (SAC) then provides 20. Mechanism: 9
Chapter 4: Terpenes Sesquiterpenes 10
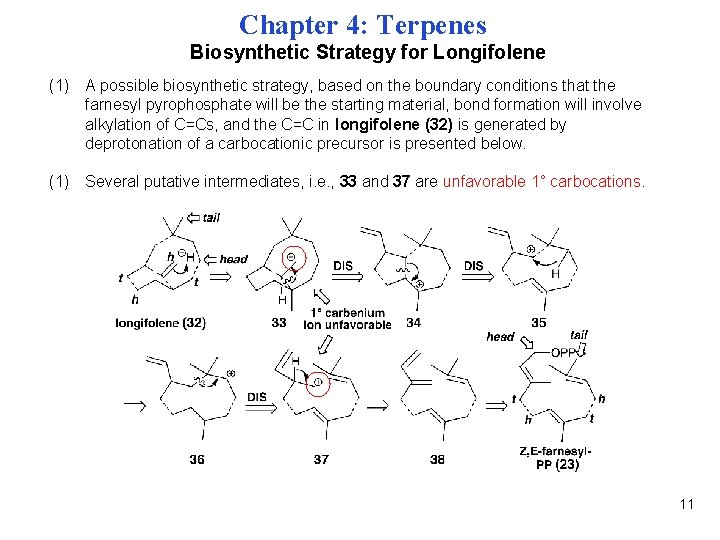
Chapter 4: Terpenes Biosynthetic Strategy for Longifolene (1) A possible biosynthetic strategy, based on the boundary conditions that the farnesyl pyrophosphate will be the starting material, bond formation will involve alkylation of C=Cs, and the C=C in longifolene (32) is generated by deprotonation of a carbocationic precursor is presented below. (1) Several putative intermediates, i. e. , 33 and 37 are unfavorable 1° carbocations. 11
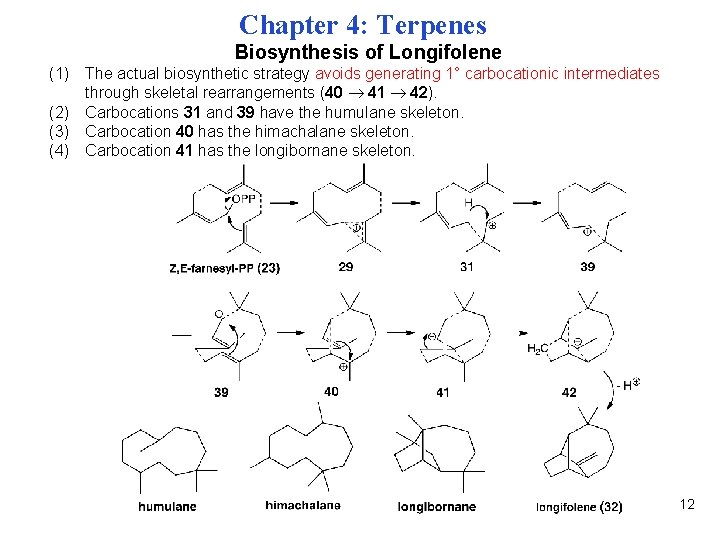
Chapter 4: Terpenes Biosynthesis of Longifolene (1) The actual biosynthetic strategy avoids generating 1° carbocationic intermediates through skeletal rearrangements (40 41 42). (2) Carbocations 31 and 39 have the humulane skeleton. (3) Carbocation 40 has the himachalane skeleton. (4) Carbocation 41 has the longibornane skeleton. 12
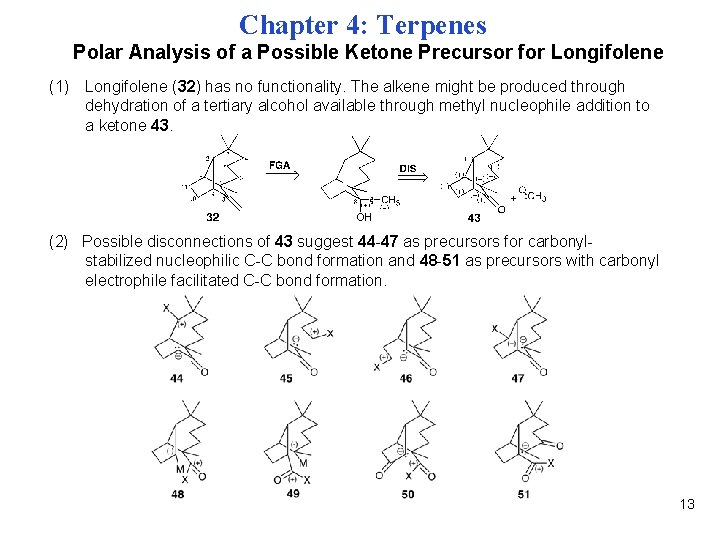
Chapter 4: Terpenes Polar Analysis of a Possible Ketone Precursor for Longifolene (1) Longifolene (32) has no functionality. The alkene might be produced through dehydration of a tertiary alcohol available through methyl nucleophile addition to a ketone 43. (2) Possible disconnections of 43 suggest 44 -47 as precursors for carbonylstabilized nucleophilic C-C bond formation and 48 -51 as precursors with carbonyl electrophile facilitated C-C bond formation. 13
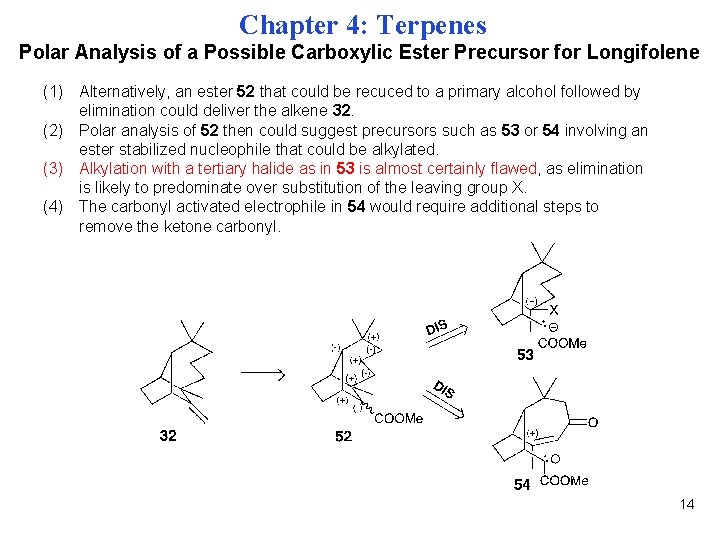
Chapter 4: Terpenes Polar Analysis of a Possible Carboxylic Ester Precursor for Longifolene (1) Alternatively, an ester 52 that could be recuced to a primary alcohol followed by elimination could deliver the alkene 32. (2) Polar analysis of 52 then could suggest precursors such as 53 or 54 involving an ester stabilized nucleophile that could be alkylated. (3) Alkylation with a tertiary halide as in 53 is almost certainly flawed, as elimination is likely to predominate over substitution of the leaving group X. (4) The carbonyl activated electrophile in 54 would require additional steps to remove the ketone carbonyl. 14
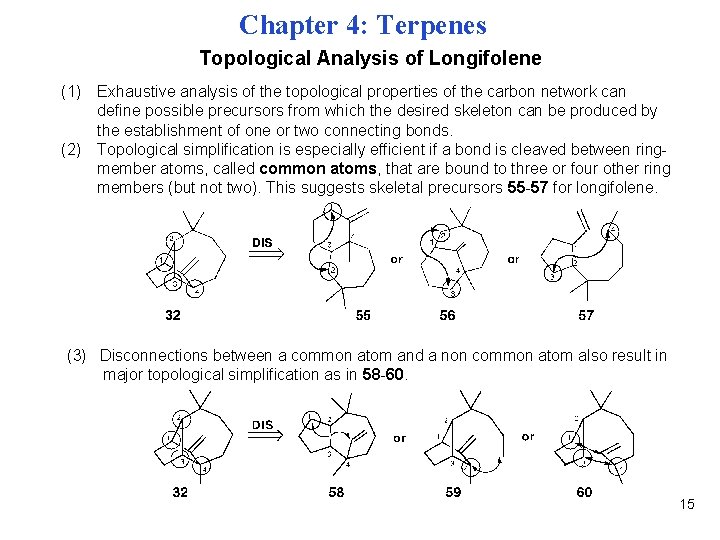
Chapter 4: Terpenes Topological Analysis of Longifolene (1) Exhaustive analysis of the topological properties of the carbon network can define possible precursors from which the desired skeleton can be produced by the establishment of one or two connecting bonds. (2) Topological simplification is especially efficient if a bond is cleaved between ringmember atoms, called common atoms, that are bound to three or four other ring members (but not two). This suggests skeletal precursors 55 -57 for longifolene. (3) Disconnections between a common atom and a non common atom also result in major topological simplification as in 58 -60. 15
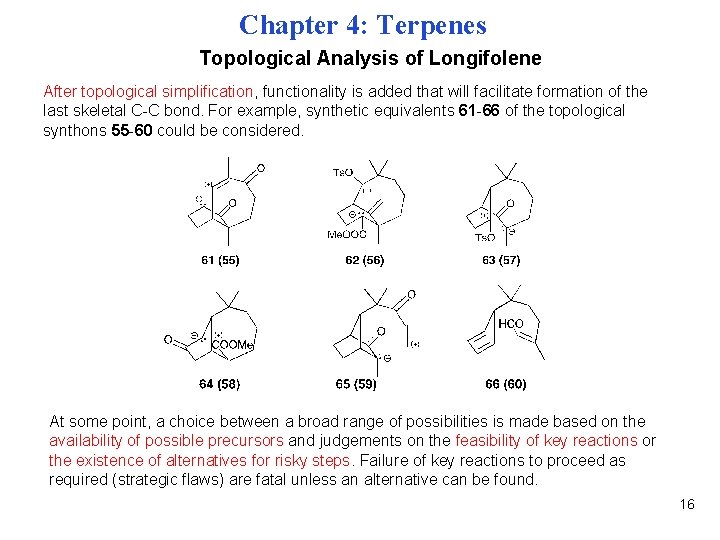
Chapter 4: Terpenes Topological Analysis of Longifolene After topological simplification, functionality is added that will facilitate formation of the last skeletal C-C bond. For example, synthetic equivalents 61 -66 of the topological synthons 55 -60 could be considered. At some point, a choice between a broad range of possibilities is made based on the availability of possible precursors and judgements on the feasibility of key reactions or the existence of alternatives for risky steps. Failure of key reactions to proceed as required (strategic flaws) are fatal unless an alternative can be found. 16
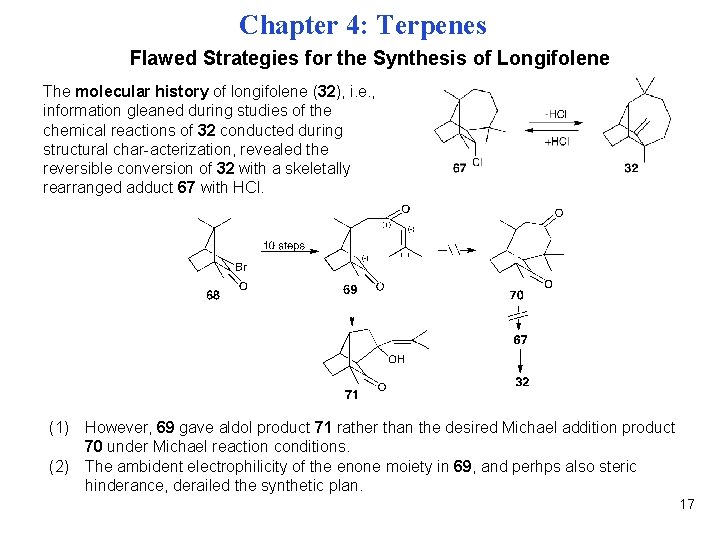
Chapter 4: Terpenes Flawed Strategies for the Synthesis of Longifolene The molecular history of longifolene (32), i. e. , information gleaned during studies of the chemical reactions of 32 conducted during structural char-acterization, revealed the reversible conversion of 32 with a skeletally rearranged adduct 67 with HCl. (1) However, 69 gave aldol product 71 rather than the desired Michael addition product 70 under Michael reaction conditions. (2) The ambident electrophilicity of the enone moiety in 69, and perhps also steric hinderance, derailed the synthetic plan. 17
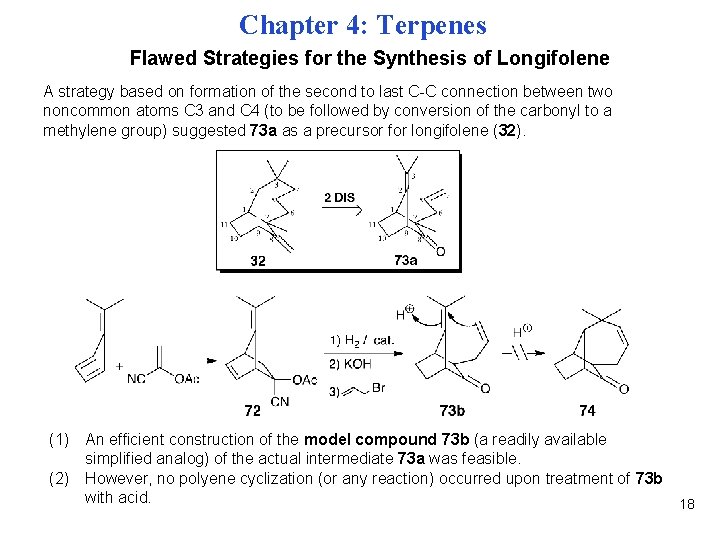
Chapter 4: Terpenes Flawed Strategies for the Synthesis of Longifolene A strategy based on formation of the second to last C-C connection between two noncommon atoms C 3 and C 4 (to be followed by conversion of the carbonyl to a methylene group) suggested 73 a as a precursor for longifolene (32). (1) An efficient construction of the model compound 73 b (a readily available simplified analog) of the actual intermediate 73 a was feasible. (2) However, no polyene cyclization (or any reaction) occurred upon treatment of 73 b with acid. 18
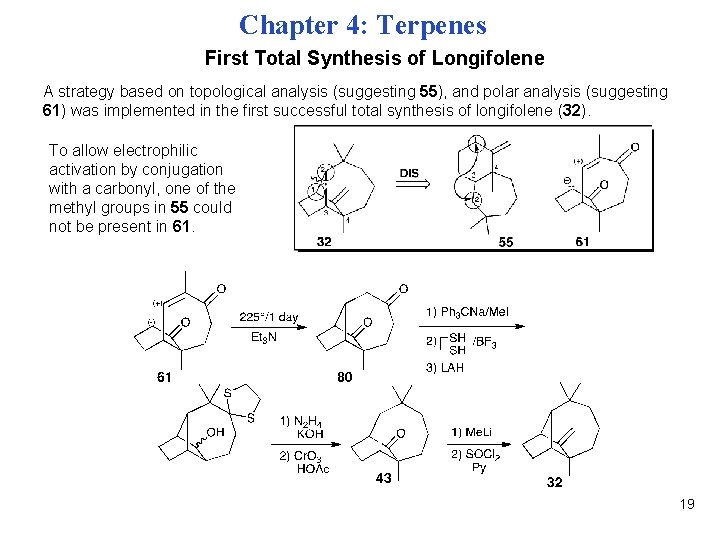
Chapter 4: Terpenes First Total Synthesis of Longifolene A strategy based on topological analysis (suggesting 55), and polar analysis (suggesting 61) was implemented in the first successful total synthesis of longifolene (32). To allow electrophilic activation by conjugation with a carbonyl, one of the methyl groups in 55 could not be present in 61. 19
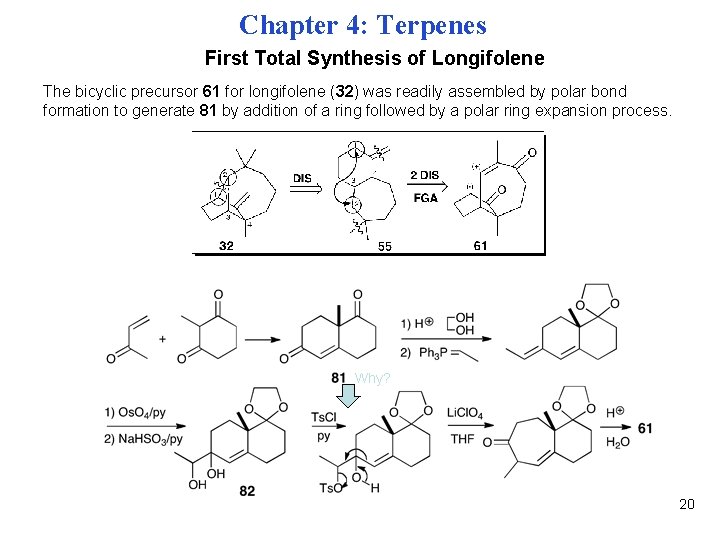
Chapter 4: Terpenes First Total Synthesis of Longifolene The bicyclic precursor 61 for longifolene (32) was readily assembled by polar bond formation to generate 81 by addition of a ring followed by a polar ring expansion process. Why? 20
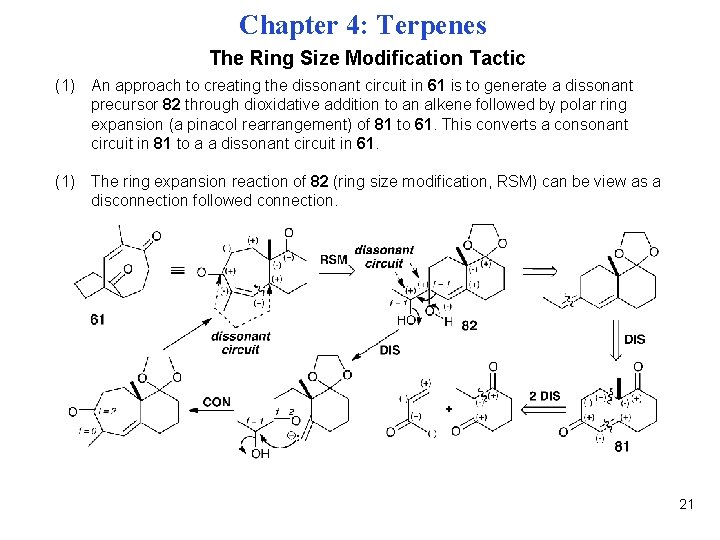
Chapter 4: Terpenes The Ring Size Modification Tactic (1) An approach to creating the dissonant circuit in 61 is to generate a dissonant precursor 82 through dioxidative addition to an alkene followed by polar ring expansion (a pinacol rearrangement) of 81 to 61. This converts a consonant circuit in 81 to a a dissonant circuit in 61. (1) The ring expansion reaction of 82 (ring size modification, RSM) can be view as a disconnection followed connection. 21
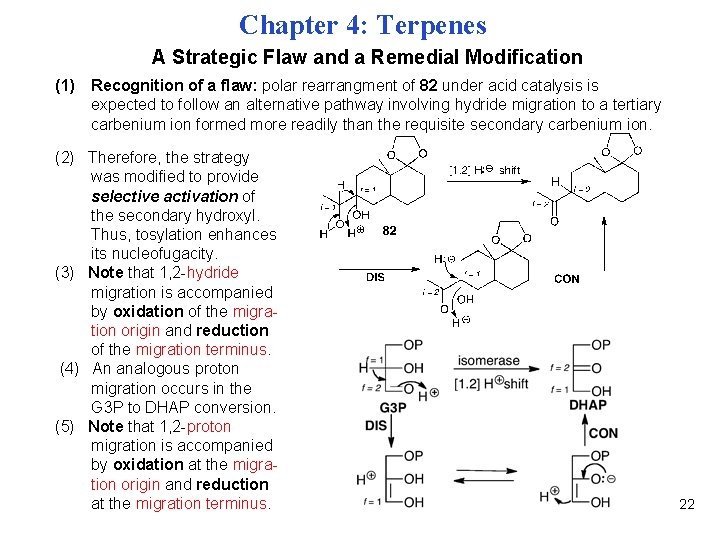
Chapter 4: Terpenes A Strategic Flaw and a Remedial Modification (1) Recognition of a flaw: polar rearrangment of 82 under acid catalysis is expected to follow an alternative pathway involving hydride migration to a tertiary carbenium ion formed more readily than the requisite secondary carbenium ion. (2) Therefore, the strategy was modified to provide selective activation of the secondary hydroxyl. Thus, tosylation enhances its nucleofugacity. (3) Note that 1, 2 -hydride migration is accompanied by oxidation of the migration origin and reduction of the migration terminus. (4) An analogous proton migration occurs in the G 3 P to DHAP conversion. (5) Note that 1, 2 -proton migration is accompanied by oxidation at the migration origin and reduction at the migration terminus. 22
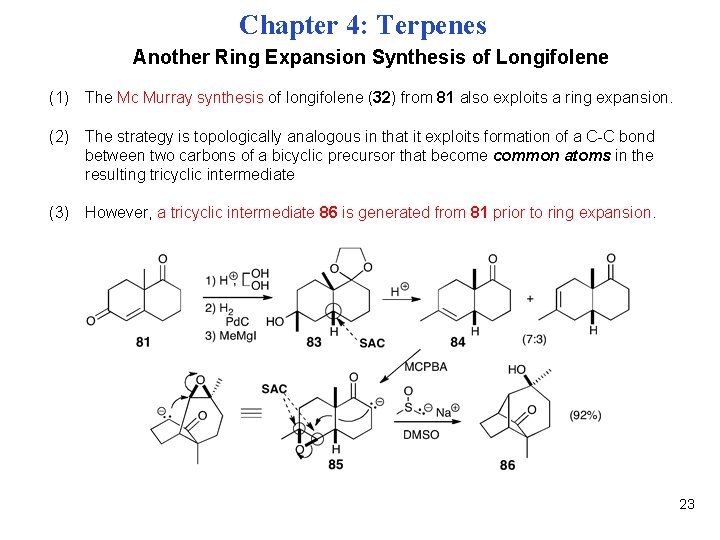
Chapter 4: Terpenes Another Ring Expansion Synthesis of Longifolene (1) The Mc Murray synthesis of longifolene (32) from 81 also exploits a ring expansion. (2) The strategy is topologically analogous in that it exploits formation of a C-C bond between two carbons of a bicyclic precursor that become common atoms in the resulting tricyclic intermediate (3) However, a tricyclic intermediate 86 is generated from 81 prior to ring expansion. 23
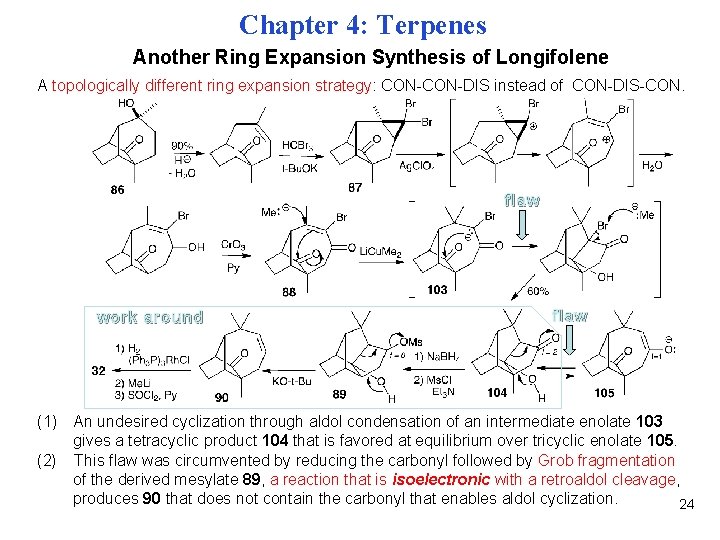
Chapter 4: Terpenes Another Ring Expansion Synthesis of Longifolene A topologically different ring expansion strategy: CON-DIS instead of CON-DIS-CON. flaw work around flaw (1) An undesired cyclization through aldol condensation of an intermediate enolate 103 gives a tetracyclic product 104 that is favored at equilibrium over tricyclic enolate 105. (2) This flaw was circumvented by reducing the carbonyl followed by Grob fragmentation of the derived mesylate 89, a reaction that is isoelectronic with a retroaldol cleavage, produces 90 that does not contain the carbonyl that enables aldol cyclization. 24
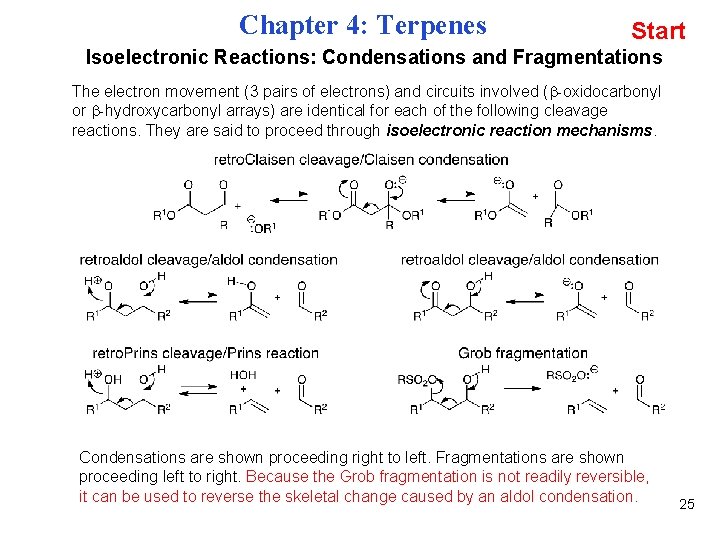
Chapter 4: Terpenes Start Isoelectronic Reactions: Condensations and Fragmentations The electron movement (3 pairs of electrons) and circuits involved ( -oxidocarbonyl or -hydroxycarbonyl arrays) are identical for each of the following cleavage reactions. They are said to proceed through isoelectronic reaction mechanisms. Condensations are shown proceeding right to left. Fragmentations are shown proceeding left to right. Because the Grob fragmentation is not readily reversible, it can be used to reverse the skeletal change caused by an aldol condensation. 25
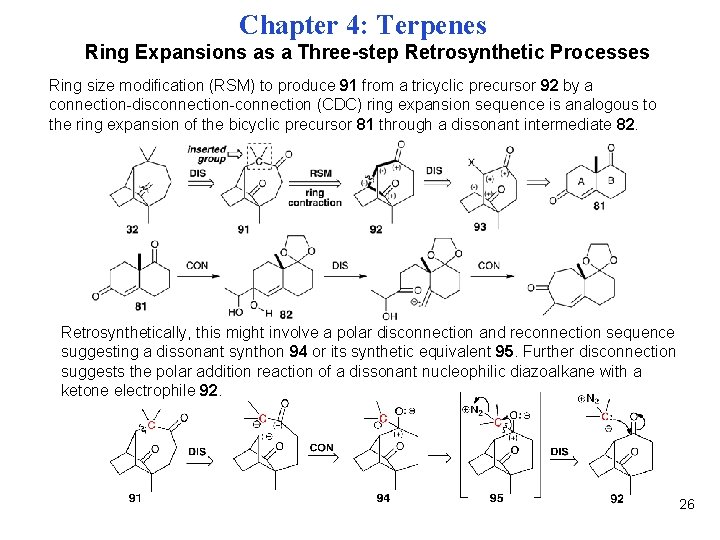
Chapter 4: Terpenes Ring Expansions as a Three-step Retrosynthetic Processes Ring size modification (RSM) to produce 91 from a tricyclic precursor 92 by a connection-disconnection-connection (CDC) ring expansion sequence is analogous to the ring expansion of the bicyclic precursor 81 through a dissonant intermediate 82. Retrosynthetically, this might involve a polar disconnection and reconnection sequence suggesting a dissonant synthon 94 or its synthetic equivalent 95. Further disconnection suggests the polar addition reaction of a dissonant nucleophilic diazoalkane with a ketone electrophile 92. 26
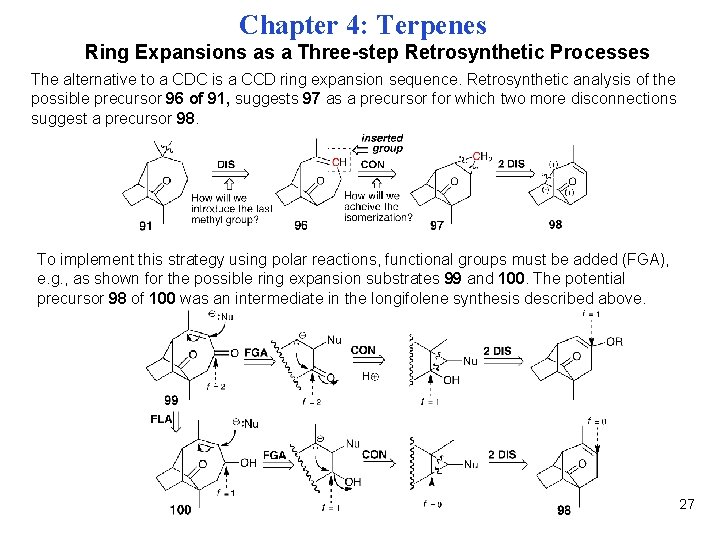
Chapter 4: Terpenes Ring Expansions as a Three-step Retrosynthetic Processes The alternative to a CDC is a CCD ring expansion sequence. Retrosynthetic analysis of the possible precursor 96 of 91, suggests 97 as a precursor for which two more disconnections suggest a precursor 98. To implement this strategy using polar reactions, functional groups must be added (FGA), e. g. , as shown for the possible ring expansion substrates 99 and 100. The potential precursor 98 of 100 was an intermediate in the longifolene synthesis described above. 27
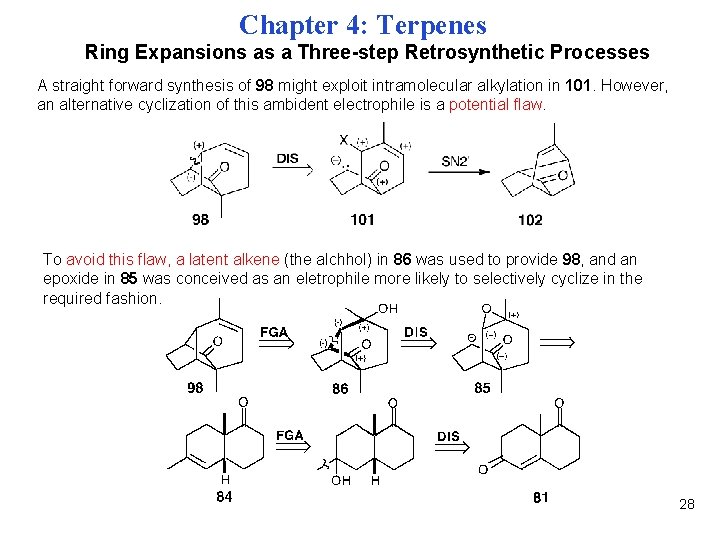
Chapter 4: Terpenes Ring Expansions as a Three-step Retrosynthetic Processes A straight forward synthesis of 98 might exploit intramolecular alkylation in 101. However, an alternative cyclization of this ambident electrophile is a potential flaw. To avoid this flaw, a latent alkene (the alchhol) in 86 was used to provide 98, and an epoxide in 85 was conceived as an eletrophile more likely to selectively cyclize in the required fashion. 28
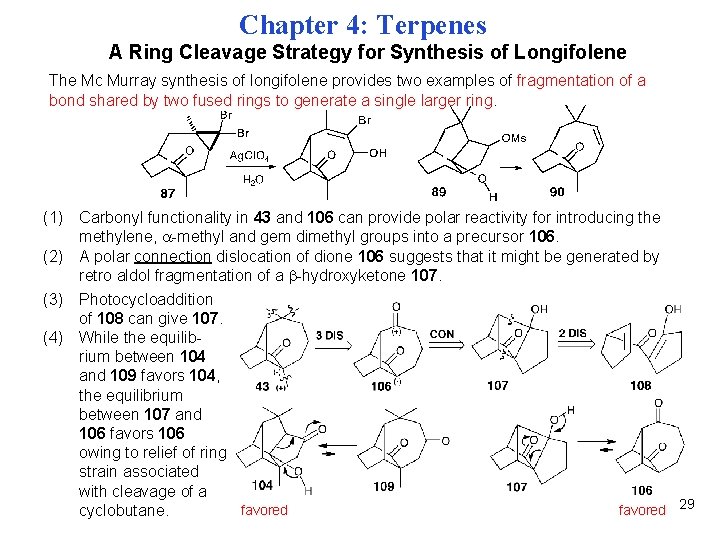
Chapter 4: Terpenes A Ring Cleavage Strategy for Synthesis of Longifolene The Mc Murray synthesis of longifolene provides two examples of fragmentation of a bond shared by two fused rings to generate a single larger ring. (1) Carbonyl functionality in 43 and 106 can provide polar reactivity for introducing the methylene, -methyl and gem dimethyl groups into a precursor 106. (2) A polar connection dislocation of dione 106 suggests that it might be generated by retro aldol fragmentation of a -hydroxyketone 107. (3) Photocycloaddition of 108 can give 107. (4) While the equilibrium between 104 and 109 favors 104, the equilibrium between 107 and 106 favors 106 owing to relief of ring strain associated with cleavage of a favored 29 cyclobutane.
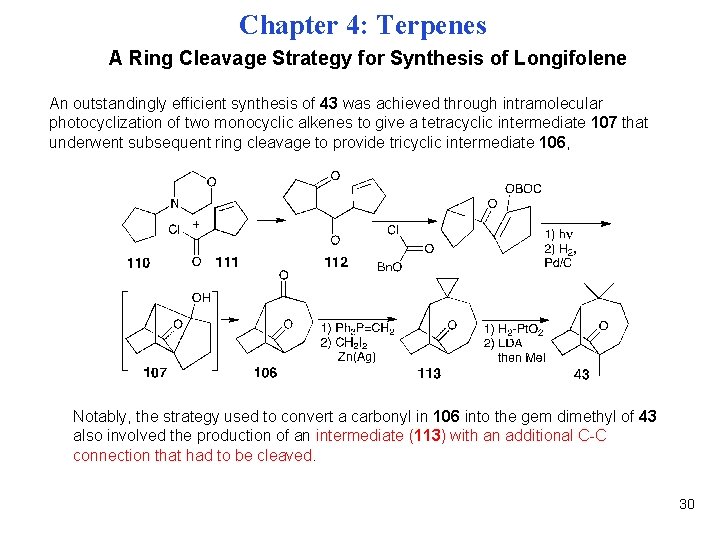
Chapter 4: Terpenes A Ring Cleavage Strategy for Synthesis of Longifolene An outstandingly efficient synthesis of 43 was achieved through intramolecular photocyclization of two monocyclic alkenes to give a tetracyclic intermediate 107 that underwent subsequent ring cleavage to provide tricyclic intermediate 106, Notably, the strategy used to convert a carbonyl in 106 into the gem dimethyl of 43 also involved the production of an intermediate (113) with an additional C-C connection that had to be cleaved. 30
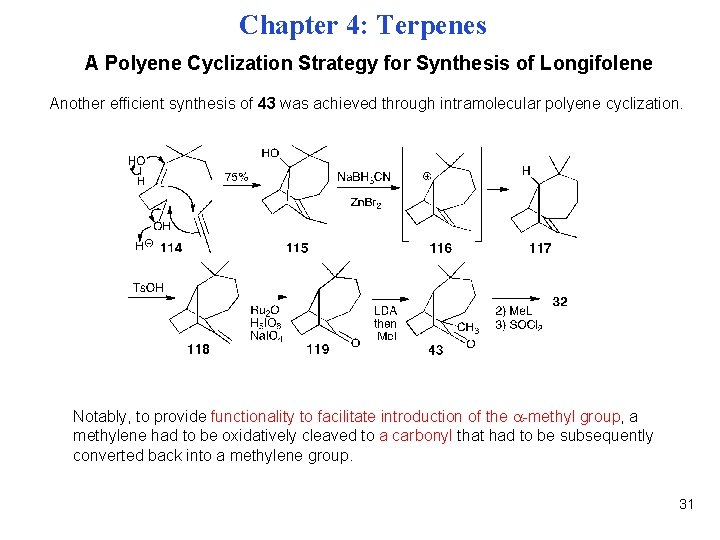
Chapter 4: Terpenes A Polyene Cyclization Strategy for Synthesis of Longifolene Another efficient synthesis of 43 was achieved through intramolecular polyene cyclization. Notably, to provide functionality to facilitate introduction of the -methyl group, a methylene had to be oxidatively cleaved to a carbonyl that had to be subsequently converted back into a methylene group. 31
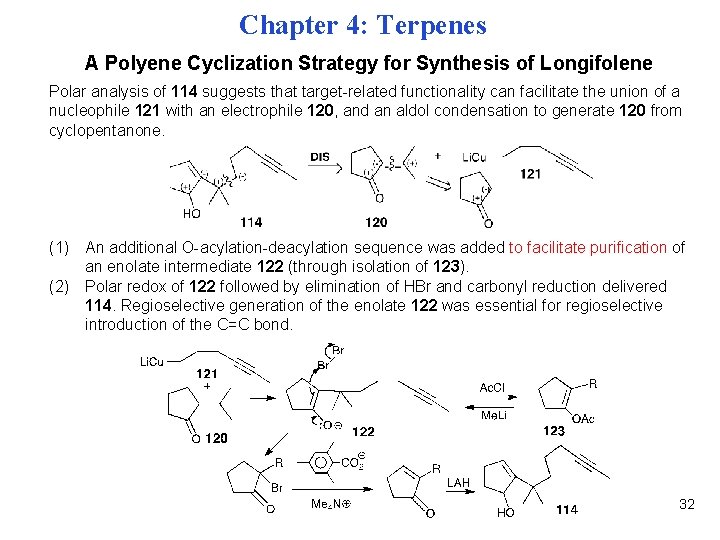
Chapter 4: Terpenes A Polyene Cyclization Strategy for Synthesis of Longifolene Polar analysis of 114 suggests that target-related functionality can facilitate the union of a nucleophile 121 with an electrophile 120, and an aldol condensation to generate 120 from cyclopentanone. (1) An additional O-acylation-deacylation sequence was added to facilitate purification of an enolate intermediate 122 (through isolation of 123). (2) Polar redox of 122 followed by elimination of HBr and carbonyl reduction delivered 114. Regioselective generation of the enolate 122 was essential for regioselective introduction of the C=C bond. 32
- Slides: 32