Chapter 4 Protein Structure and Function Proteins l

























![Reaction Rates l KM – [S] that allows rxn to proceed at ½ it Reaction Rates l KM – [S] that allows rxn to proceed at ½ it](https://slidetodoc.com/presentation_image_h/bcc0f3d856b2cb133fdbbb11501101c4/image-44.jpg)



- Slides: 57

Chapter 4 Protein Structure and Function

Proteins l l Make up about 15% of the cell Have many functions in the cell l l l l Enzymes Structural Transport Motor Storage Signaling Receptors Gene regulation Special functions

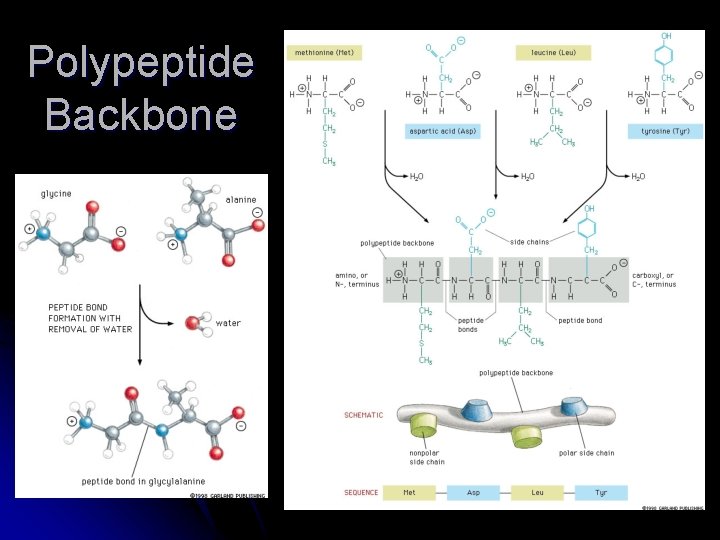
Shape = Amino Acid Sequence Proteins are made of 20 amino acids linked by peptide bonds l Polypeptide backbone is the repeating sequence of the N-C-C-N-C-C… in the peptide bond l The side chain or R group is not part of the backbone or the peptide bond l

Polypeptide Backbone

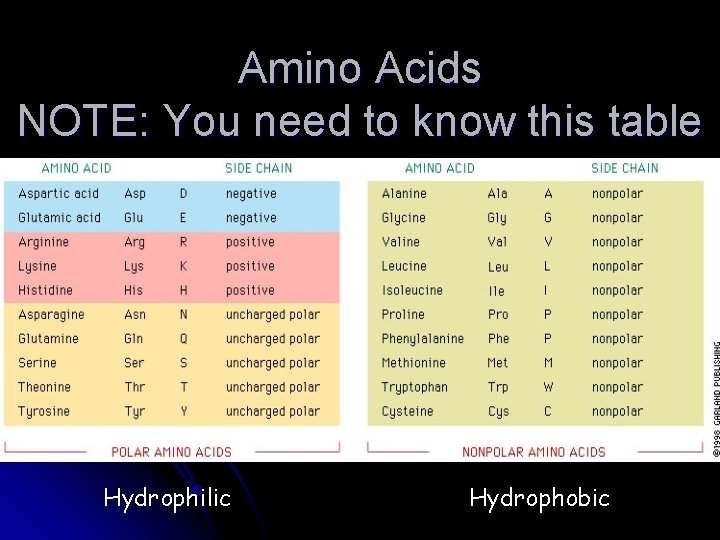
Amino Acids NOTE: You need to know this table Hydrophilic Hydrophobic

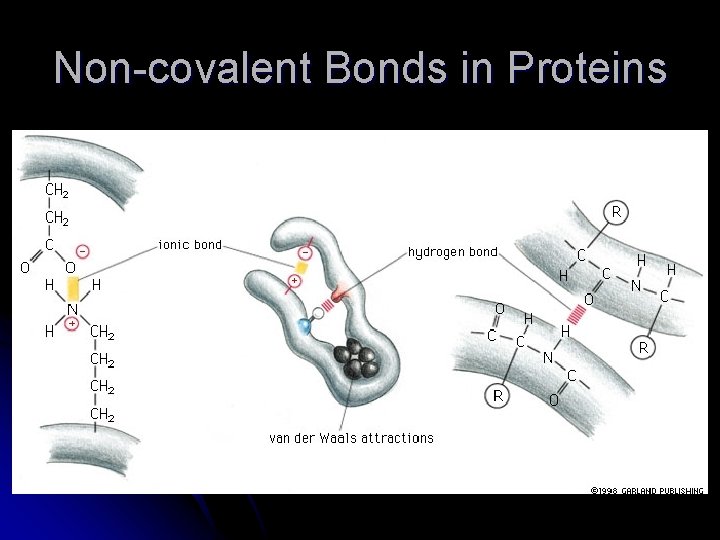
Protein Folding The peptide bond allows for rotation around it and therefore the protein can fold and orient the R groups in favorable positions l Weak non-covalent interactions will hold the protein in its functional shape – these are weak and will take many to hold the shape l

Non-covalent Bonds in Proteins

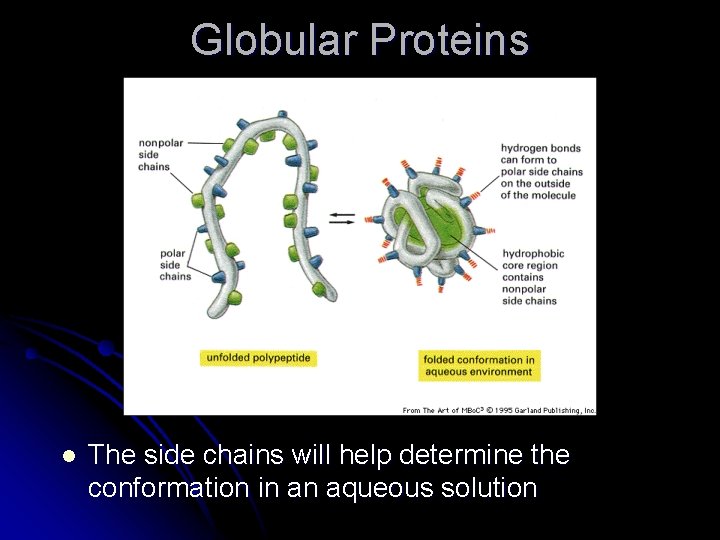
Globular Proteins l The side chains will help determine the conformation in an aqueous solution

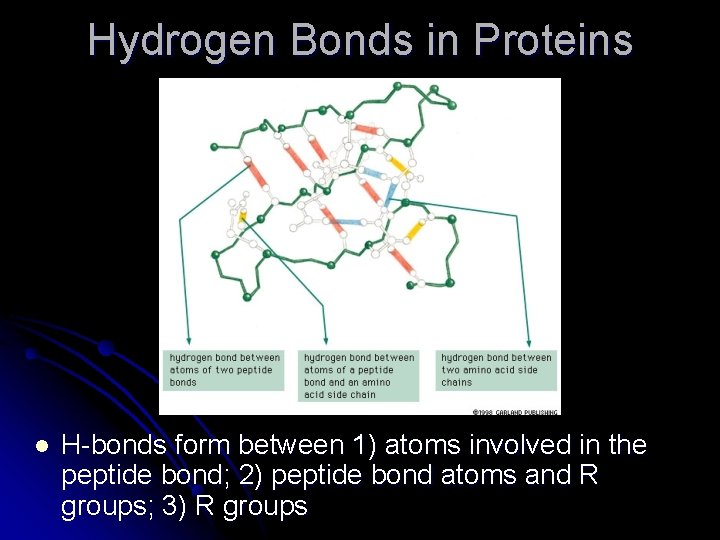
Hydrogen Bonds in Proteins l H-bonds form between 1) atoms involved in the peptide bond; 2) peptide bond atoms and R groups; 3) R groups

Protein Folding Proteins shape is determined by the sequence of the amino acids l The final shape is called the conformation and has the lowest free energy possible l Denaturation is the process of unfolding the protein l l Can be down with heat, p. H or chemical compounds l In the chemical compound, can remove and have the protein renature or refold

Folding@home l l l The Stanford Folding@home research goal is to understand protein folding, misfolding, and related diseases. Calculations to create models requires a supercomputer OR many smaller computers (distributed computing). You can participate by visiting: l l Fold@home web site: http: //folding. stanford. edu/ Article on Folding@home: http: //www. sciencedaily. com/releases/2002/10/02102 2070813. htm

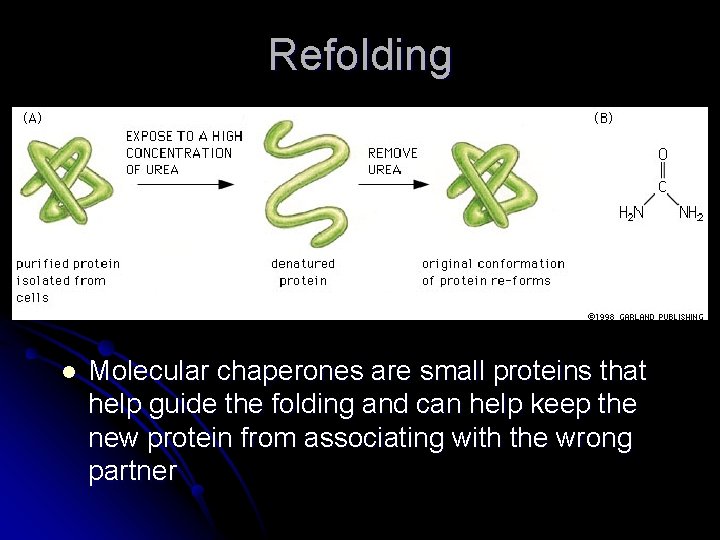
Refolding l Molecular chaperones are small proteins that help guide the folding and can help keep the new protein from associating with the wrong partner

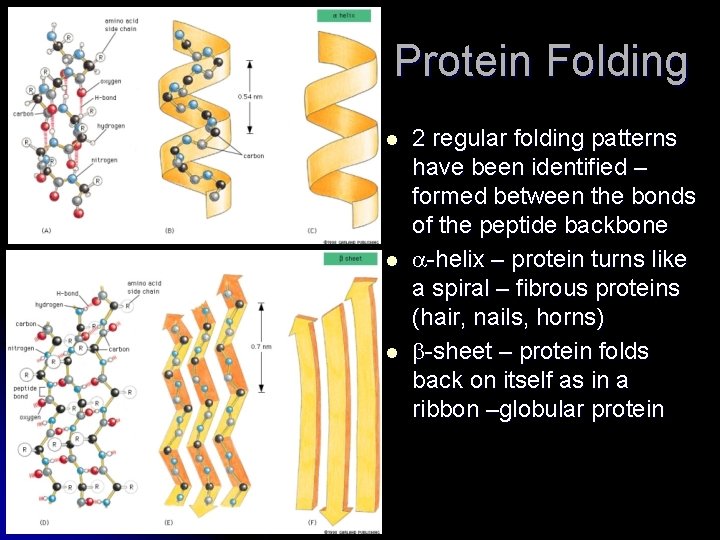
Protein Folding l l l 2 regular folding patterns have been identified – formed between the bonds of the peptide backbone -helix – protein turns like a spiral – fibrous proteins (hair, nails, horns) -sheet – protein folds back on itself as in a ribbon –globular protein

Sheets l l l Core of many proteins is the sheet Form rigid structures with the H-bond Can be of 2 types l l Anti-parallel – run in an opposite direction of its neighbor (A) Parallel – run in the same direction with longer looping sections between them (B)

Helix l l Formed by a H-bond between every 4 th peptide bond – C=O to N-H Usually in proteins that span a membrane The helix can either coil to the right or the left Can also coil around each other – coiled-coil shape – a framework for structural proteins such as nails and skin

CD from Text l The CD that is included on your textbook back cover has some video clips that will show the helix and sheets as well as other things in this chapter. You will want to look at them.

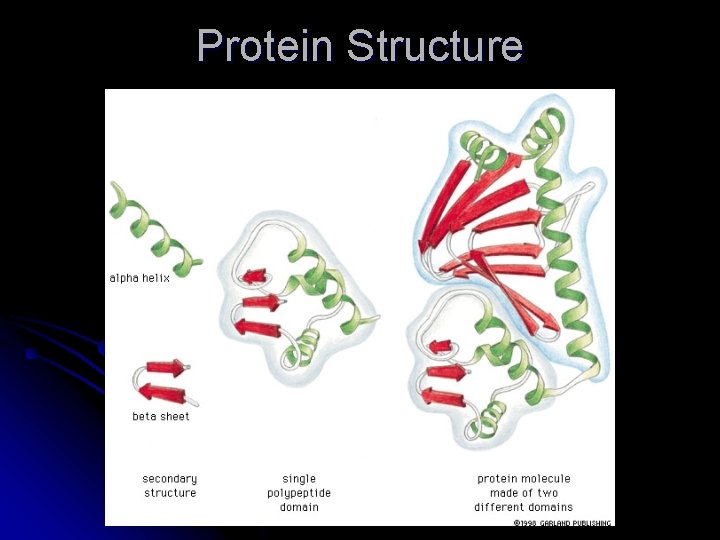
Levels of Organization l Primary structure l Amino l acid sequence of the protein Secondary structure l. H bonds in the peptide chain backbone l -helix l and -sheets Tertiary structure l Non-covalent interactions between the R groups within the protein l Quanternary structure l Interaction between 2 polypeptide chains

Protein Structure

Domains A domain is a basic structural unit of a protein structure – distinct from those that make up the conformations l Part of protein that can fold into a stable structure independently l Different domains can impart different functions to proteins l Proteins can have one to many domains depending on protein size l

Domains

Useful Proteins l l l There are thousands and thousands of different combinations of amino acids that can make up proteins and that would increase if each one had multiple shapes Proteins usually have only one useful conformation because otherwise it would not be efficient use of the energy available to the system Natural selection has eliminated proteins that do not perform a specific function in the cell

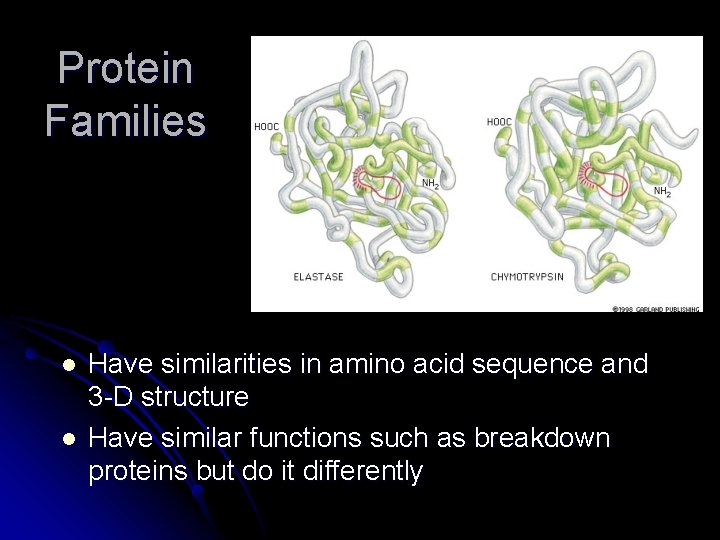
Protein Families l l Have similarities in amino acid sequence and 3 -D structure Have similar functions such as breakdown proteins but do it differently

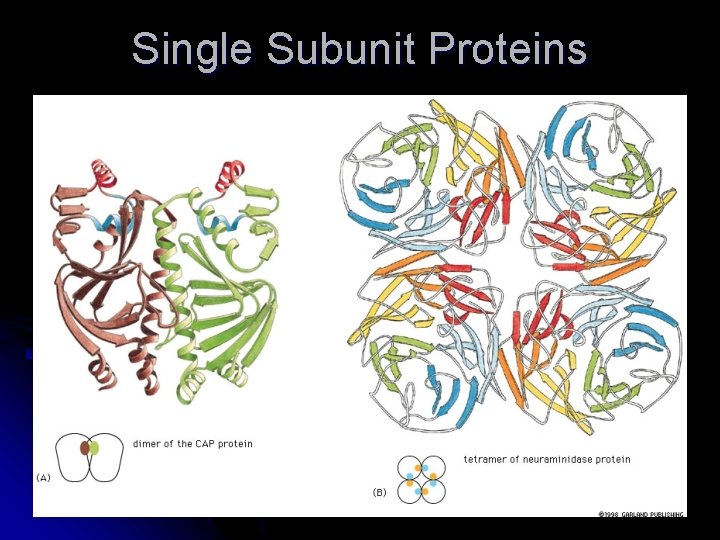
Proteins – Multiple Peptides l Non-covalent bonds can form interactions between individual polypeptide chains l Binding site – where proteins interact with one another l Subunit – each polypeptide chain of large protein l Dimer – protein made of 2 subunits l Can be same subunit or different subunits

Single Subunit Proteins

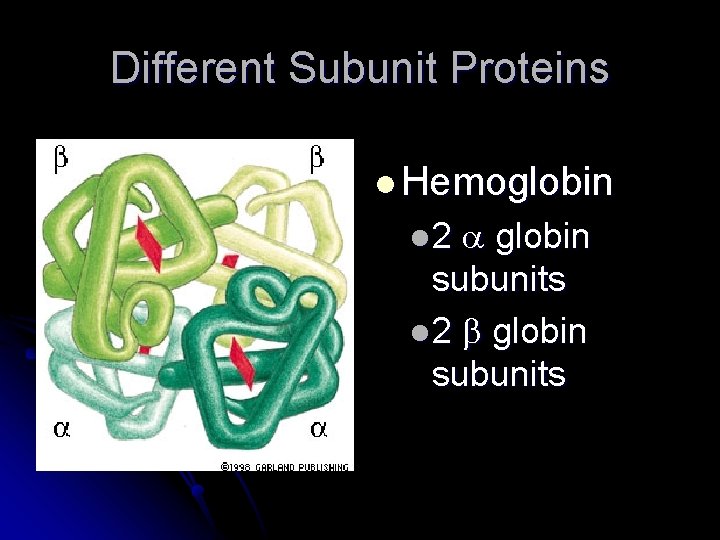
Different Subunit Proteins l Hemoglobin subunits l 2

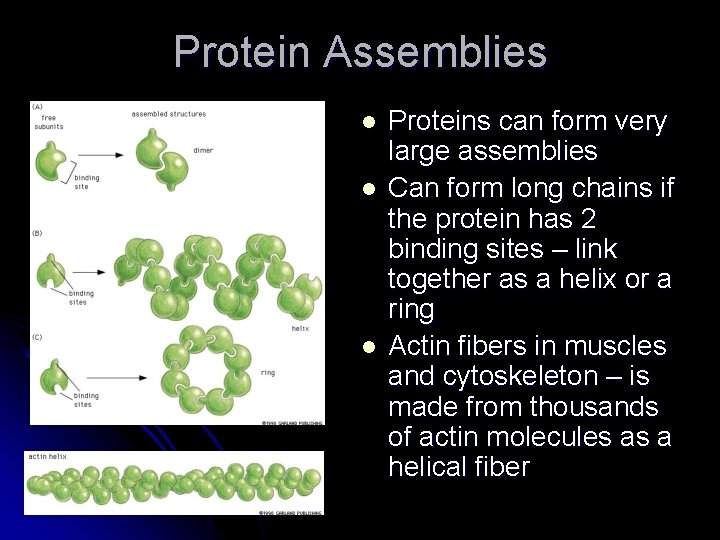
Protein Assemblies l l l Proteins can form very large assemblies Can form long chains if the protein has 2 binding sites – link together as a helix or a ring Actin fibers in muscles and cytoskeleton – is made from thousands of actin molecules as a helical fiber

Types of Proteins l Globular Proteins – most of what we have dealt with so far l Compact shape like a ball with irregular surfaces l Enzymes are globular l Fibrous Proteins – usually span a long distance in the cell l 3 -D structure is usually long and rod shaped

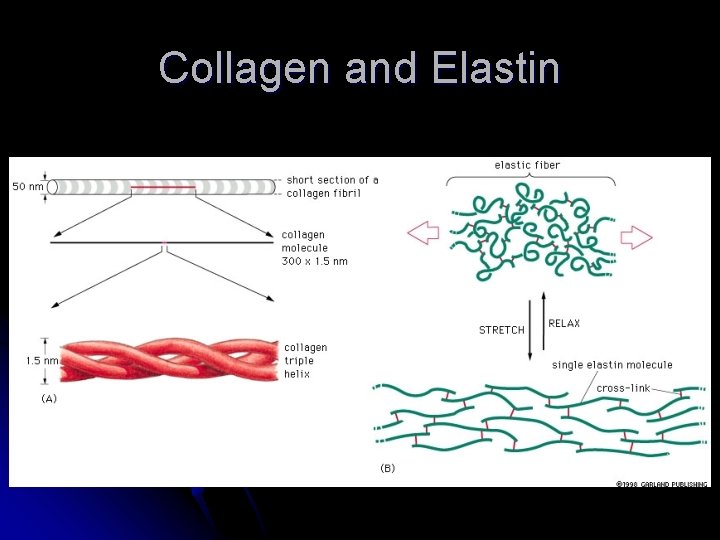
Important Fibrous Proteins l Intermediate filaments of the cytoskeleton l Structural l Keratin l scaffold inside the cell in hair, horns and nails Extracellular matrix l Bind cells together to make tissues l Secreted from cells and assemble in long fibers l Collagen – fiber with a glycine every third amino acid in the protein l Elastin – unstructured fibers that gives tissue an elastic characteristic

Collagen and Elastin

Stabilizing Cross-Links l l Cross linkages can be between 2 parts of a protein or between 2 subunits Disulfide bonds (S-S) form between adjacent -SH groups on the amino acid cysteine

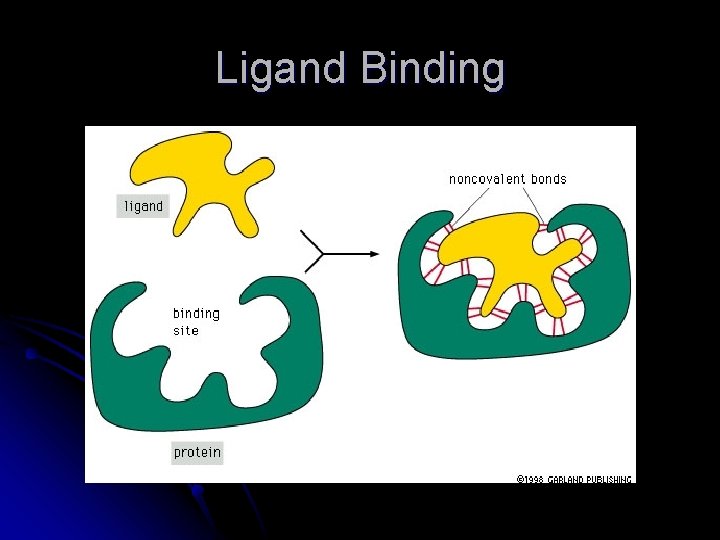
Proteins at Work l l The conformation of a protein gives it a unique function To work proteins must interact with other molecules, usually 1 or a few molecules from the thousands to 1 protein Ligand – the molecule that a protein can bind Binding site – part of the protein that interacts with the ligand l Consists of a cavity formed by a specific arrangement of amino acids

Ligand Binding

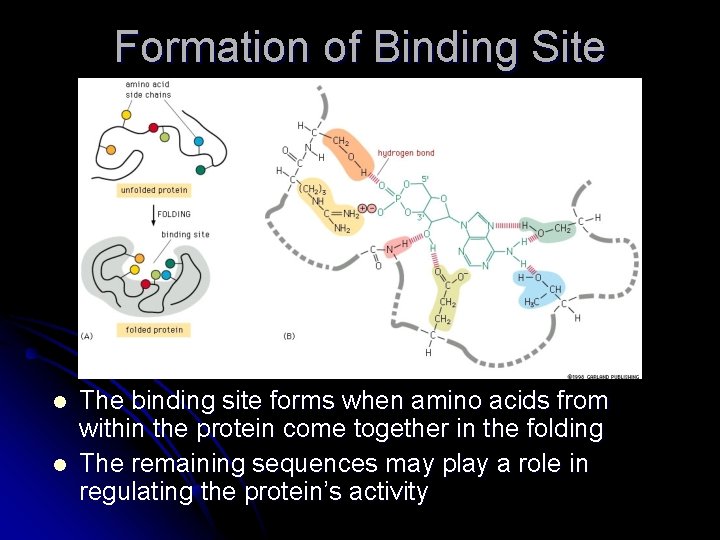
Formation of Binding Site l l The binding site forms when amino acids from within the protein come together in the folding The remaining sequences may play a role in regulating the protein’s activity

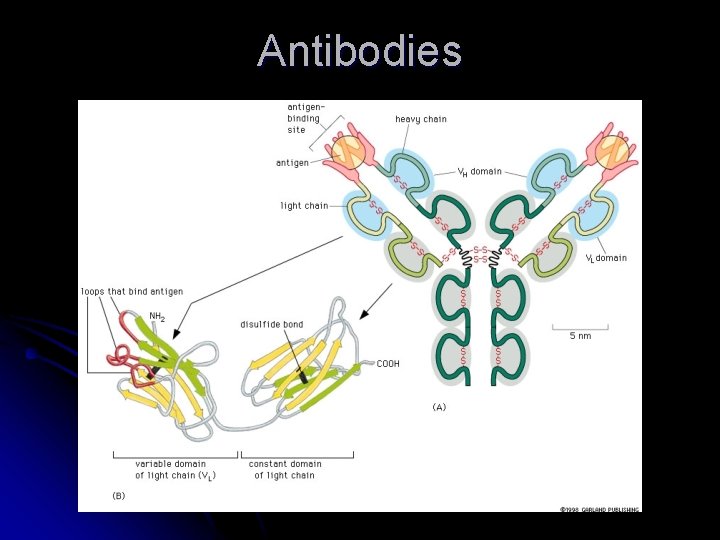
Antibody Family A family of proteins that can be created to bind to almost any molecule l Antibodies (immunoglobulins) are made in response to a foreign molecule ie. bacteria, virus, pollen… called the antigen l Bind together tightly and therefore inactivates the antigen or marks it for destruction l

Antibodies Y-shaped molecules with 2 binding sites at the upper ends of the Y l The loops of polypeptides on the end of the binding site are what imparts the recognition of the antigen l Changes in the sequence of the loops make the antibody recognize different antigens - specificity l

Antibodies

Binding Strength l l Can be measured directly Antibodies and antigens are mixing around in a solution, eventually they will bump into each other in a way that the antigen sticks to the antibody, eventually they will separate due to the motion in the molecules This process continues until the equilibrium is reached – number sticking is constant and number leaving is constant This can be determined for any protein and its ligand

Equilibrium Constant l l Concentration of antigen, antibody and antigen/antibody complex at equilibrium can be measured – equilibrium constant (K) Larger the K the tighter the binding or the more non -covalent bonds that hold the 2 together

Enzymes as Catalysts l l Enzymes are proteins that bind to their ligand as the 1 st step in a process An enzyme’s ligand is called a substrate l l May be 1 or more molecules Output of the reaction is called the product Enzymes can repeat these steps many times and rapidly, called catalysts Many different kinds – see table 5 -2, p 168

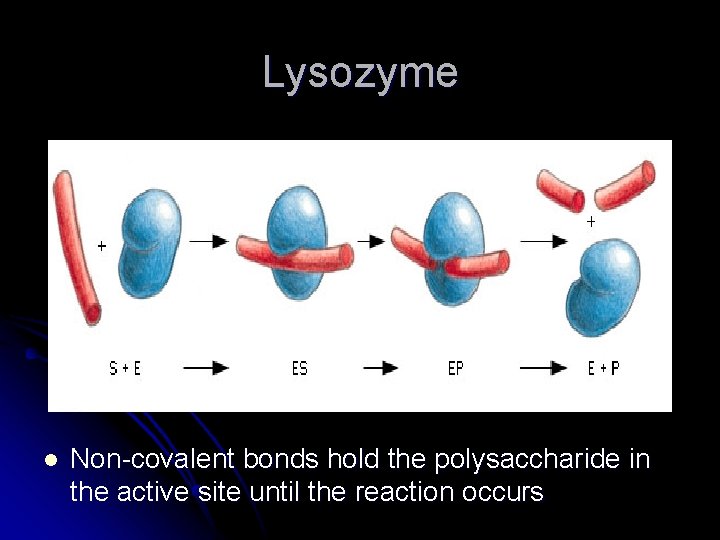
Enzymes at Work l l Lysozyme is an important enzyme that protects us from bacteria by making holes in the bacterial cell wall and causing it to break Lysozyme adds H 2 O to the glycosidic bond in the cell wall Lysozyme holds the polysaccharide in a position that allows the H 2 O to break the bond – this is the transition state – state between substrate and product Active site is a special binding site in enzymes where the chemical reaction takes place

Lysozyme l Non-covalent bonds hold the polysaccharide in the active site until the reaction occurs

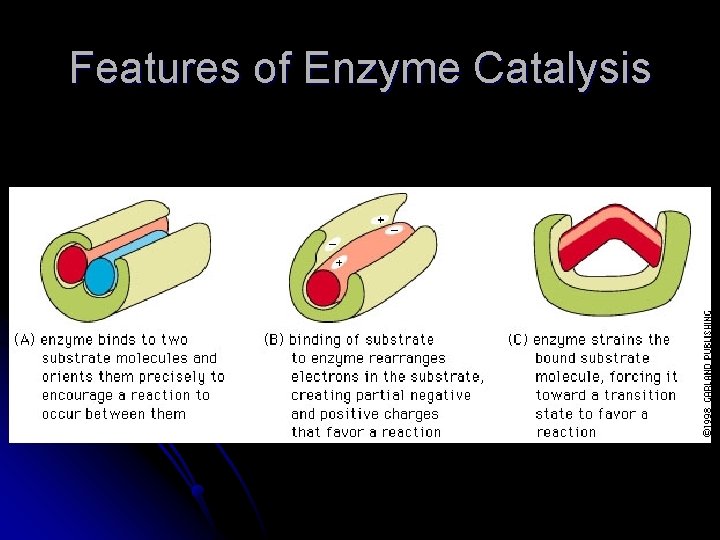
Features of Enzyme Catalysis

Enzyme Performance l E + S EP E + P Step 1 – binding of the substrate l l Limiting step depending on [S] and/or [E] Vmax – maximum rate of the reaction Turnover number determines how fast the substrate can be processed = rate of rxn [E] Step 2 – stabilize the transition state l l State of substrate prior to becoming product Enzymes lowers the energy of transition state and therefore accelerates the reaction
![Reaction Rates l KM S that allows rxn to proceed at ½ it Reaction Rates l KM – [S] that allows rxn to proceed at ½ it](https://slidetodoc.com/presentation_image_h/bcc0f3d856b2cb133fdbbb11501101c4/image-44.jpg)
Reaction Rates l KM – [S] that allows rxn to proceed at ½ it maximum rate

Prosthetic Groups l l Occasionally the sequence of the protein is not enough for the function of the protein Some proteins require a non-protein molecule to enhance the performance of the protein l l When a prosthetic group is required by an enzyme it is called a co-enzyme l l Hemoglobin requires heme (iron containing compound) to carry the O 2 Usually a metal or vitamin These groups may be covalently or noncovalently linked to the protein


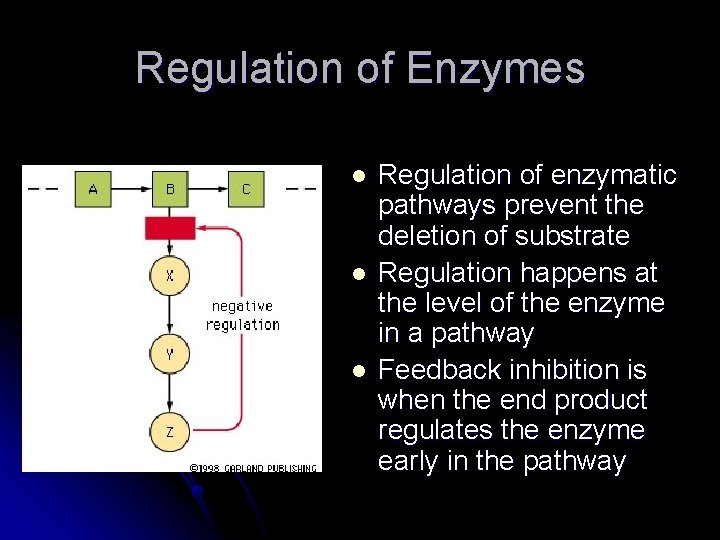
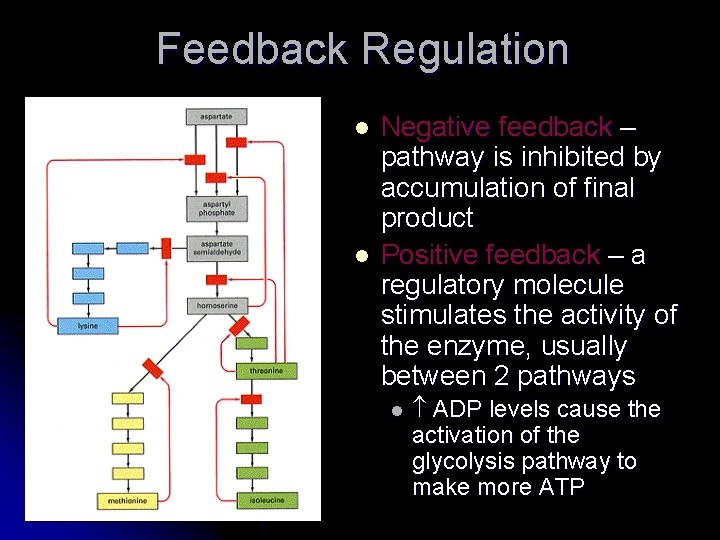
Regulation of Enzymes l l l Regulation of enzymatic pathways prevent the deletion of substrate Regulation happens at the level of the enzyme in a pathway Feedback inhibition is when the end product regulates the enzyme early in the pathway

Feedback Regulation l l Negative feedback – pathway is inhibited by accumulation of final product Positive feedback – a regulatory molecule stimulates the activity of the enzyme, usually between 2 pathways l ADP levels cause the activation of the glycolysis pathway to make more ATP

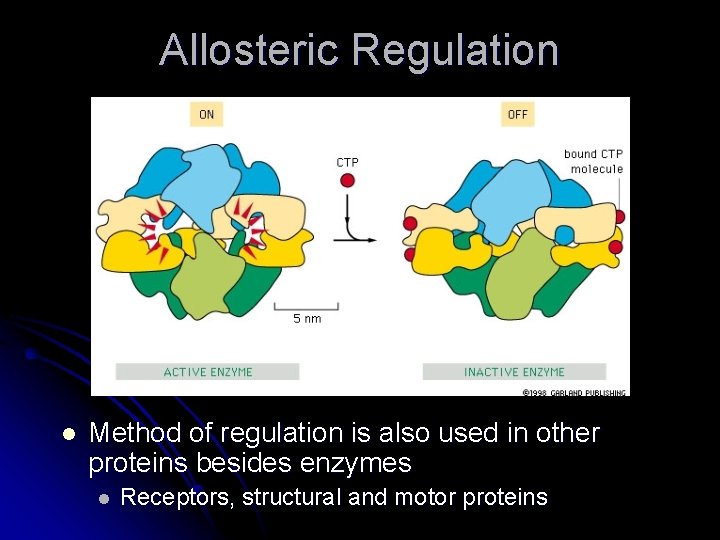
Allostery l l l Conformational coupling of 2 widely separated binding sites must be responsible for regulation – active site recognizes substrate and 2 nd site recognizes the regulatory molecule Protein regulated this way undergoes allosteric transition or a conformational change Protein regulated in this manner is an allosteric protein

Allosteric Regulation l Method of regulation is also used in other proteins besides enzymes l Receptors, structural and motor proteins

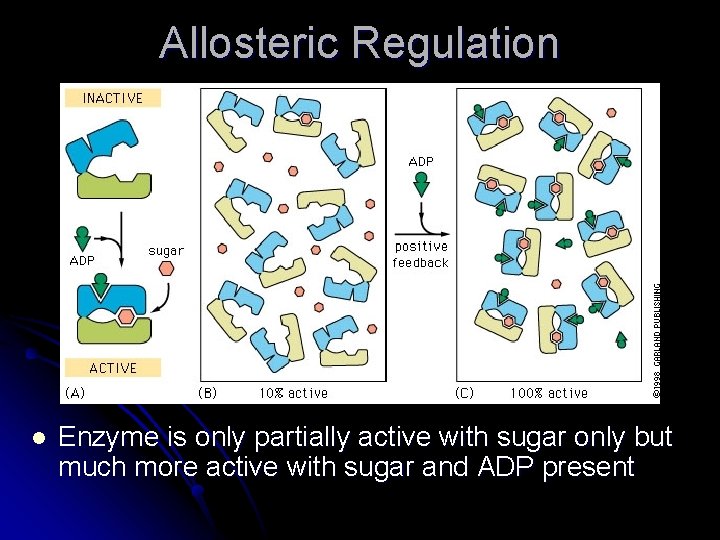
Allosteric Regulation l Enzyme is only partially active with sugar only but much more active with sugar and ADP present

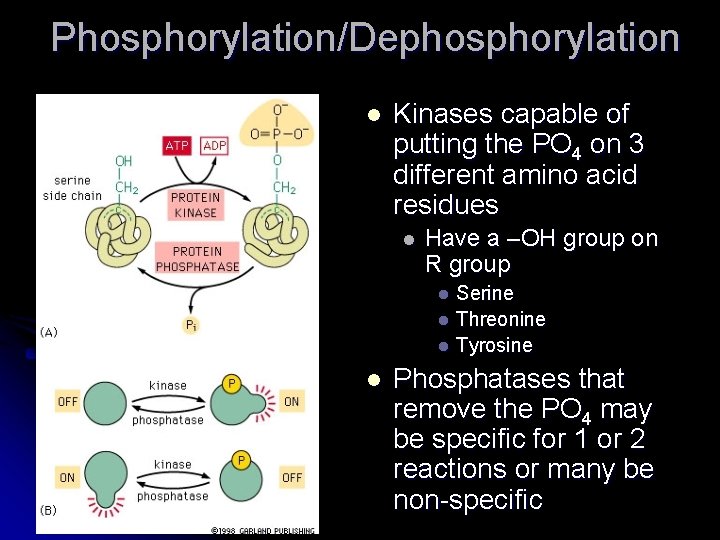
Phosphorylation Some proteins are regulated by the addition of a PO 4 group that allows for the attraction of + charged side chains causing a conformation change l Reversible protein phosphorylations regulate many eukaryotic cell functions turning things on and off l Protein kinases add the PO 4 and protein phosphatase remove them l

Phosphorylation/Dephosphorylation l Kinases capable of putting the PO 4 on 3 different amino acid residues l Have a –OH group on R group Serine l Threonine l Tyrosine l l Phosphatases that remove the PO 4 may be specific for 1 or 2 reactions or many be non-specific

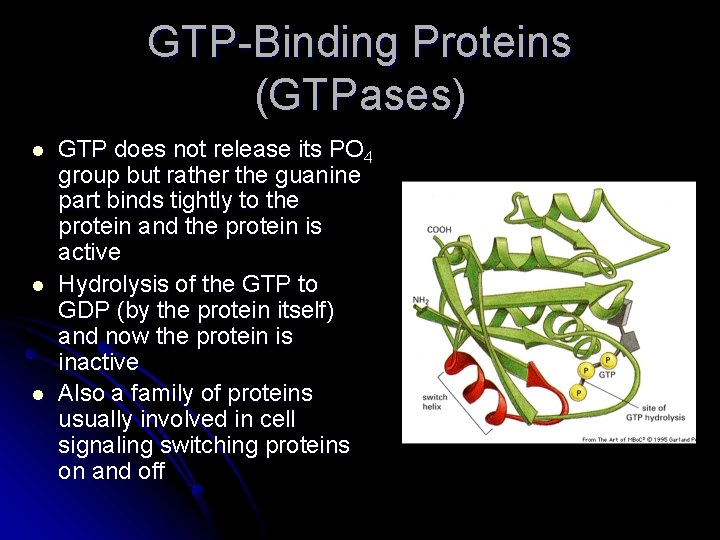
GTP-Binding Proteins (GTPases) l l l GTP does not release its PO 4 group but rather the guanine part binds tightly to the protein and the protein is active Hydrolysis of the GTP to GDP (by the protein itself) and now the protein is inactive Also a family of proteins usually involved in cell signaling switching proteins on and off

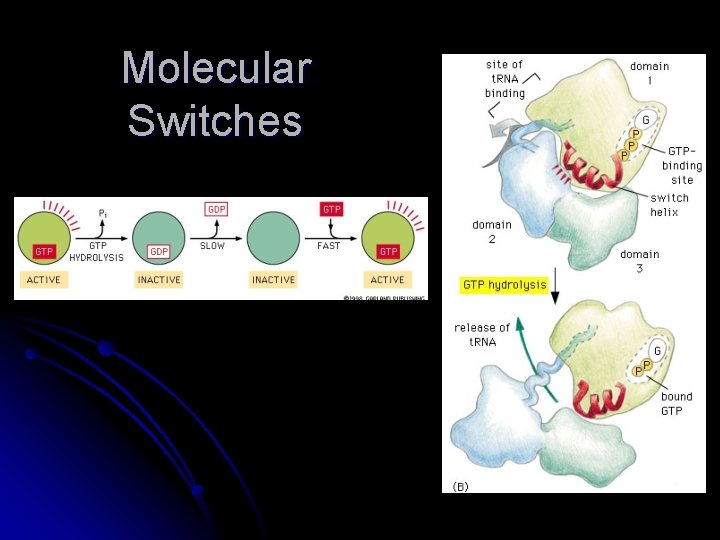
Molecular Switches

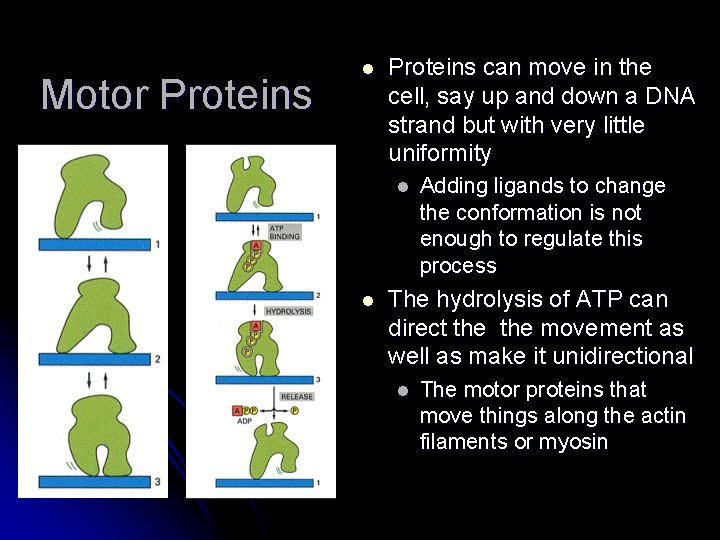
Motor Proteins l Proteins can move in the cell, say up and down a DNA strand but with very little uniformity l l Adding ligands to change the conformation is not enough to regulate this process The hydrolysis of ATP can direct the movement as well as make it unidirectional l The motor proteins that move things along the actin filaments or myosin

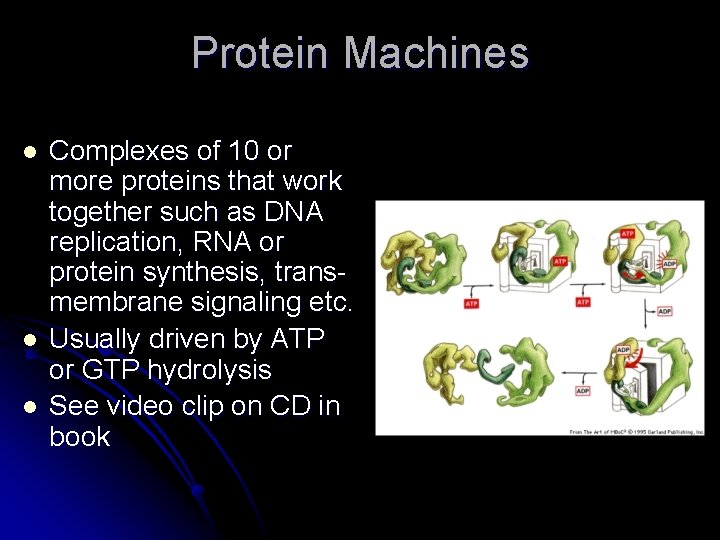
Protein Machines l l l Complexes of 10 or more proteins that work together such as DNA replication, RNA or protein synthesis, transmembrane signaling etc. Usually driven by ATP or GTP hydrolysis See video clip on CD in book