Chapter 4 Lecture General Organic and Biological Chemistry

Chapter 4 Lecture General, Organic, and Biological Chemistry: An Integrated Approach Chapter 4 Laura Frost, Todd Deal and Karen Timberlake Introduction to Organic Compounds by Richard Triplett © 2011 Pearson Education, Inc.

Chapter Outline 4. 1 Alkanes: The Simplest Organic Compounds 4. 2 Representing the Structures of Organic Compounds 4. 3 Fatty Acids—Biological “Hydrocarbons” 4. 4 Families of Organic Compounds—Functional Groups 4. 5 Isomerism in Organic Compounds, Part 1 4. 6 Isomerism in Organic Compounds, Part 2

Introduction to Organic Compounds Introduction • Organic compounds are composed primarily of carbon and hydrogen, but may also include oxygen, nitrogen, sulfur, phosphorus, and a few other elements. • Biomolecules are proteins, carbohydrates, lipids, and DNA, and belong to a class of organic compounds.

Introduction to Organic Compounds, Continued • Inorganic compounds are compounds that do not contain carbon and hydrogen. • Organic compounds occur naturally and are also synthesized in the laboratory. • Carbon is unique in that it form bonds with other carbons and can form long chains, branched chains, and rings of various sizes and shapes. • More than 25 million organic compounds are known.

Introduction to Organic Compounds, Continued • Organic compounds are grouped in families based on their molecular structure and composition. Members in the same family behave similarly in chemical reactions. • An increased interest in the study of organic chemistry is due to the variety of organic compounds and the ability to manipulate them in the laboratory.

4. 1 Alkanes: The Simplest Organic Compounds • Alkanes make up the fossil fuels used for heating, transportation, and generating electricity. They are simple organic compounds made up solely of carbon and hydrogen. • Saturated hydrocarbons is a term used to refer to alkanes and means that there are only carbon –carbon single bonds in the molecules.

4. 1 Alkanes: The Simplest Organic Compounds, Continued • Straight-chain alkanes are alkanes made up of carbon atoms joined to one another to form a continuous, unbranched chain of various lengths. • Names of alkanes are based on the number of carbon atoms in the chain. For example, methane, the simplest alkane, contains one carbon atom. Ethane contains two carbon atoms, propane contains three carbon atoms, and butane contains four carbon atoms.

4. 1 Alkanes: The Simplest Organic Compounds, Continued • Alkanes of five or more carbon atoms are named with a numerical prefix (for example, pent- = 5, hex- = 6, hept- = 7) followed by the ending –ane (indicating the alkane family). • It is important to recognize the name of the first ten alkanes since they become the root names to other families of organic compounds.
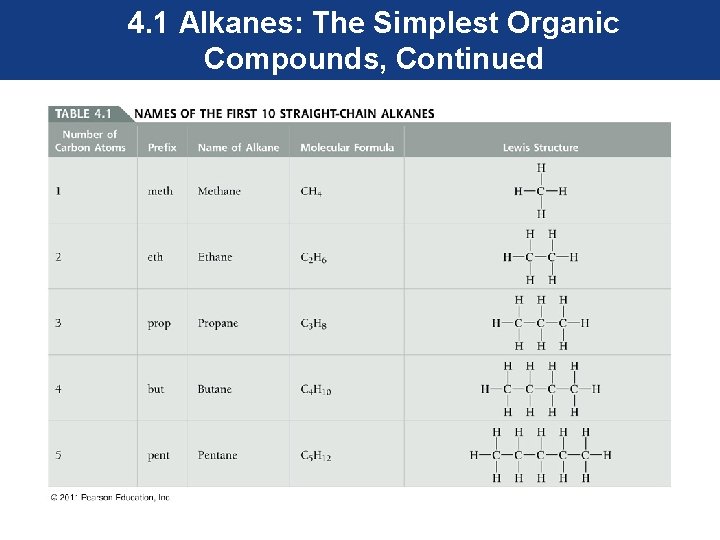
4. 1 Alkanes: The Simplest Organic Compounds, Continued
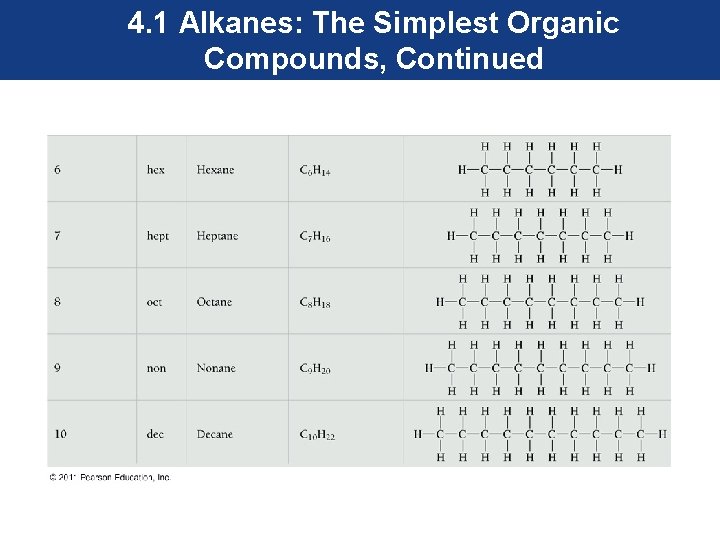
4. 1 Alkanes: The Simplest Organic Compounds, Continued

4. 1 Alkanes: The Simplest Organic Compounds, Continued Cycloalkanes • Carbon can form rings as well as long chains. • Ring structures made from carbon are called cycloalkanes and they are saturated. • The word cyclo is added in front of the name of the alkane when these compounds are named.
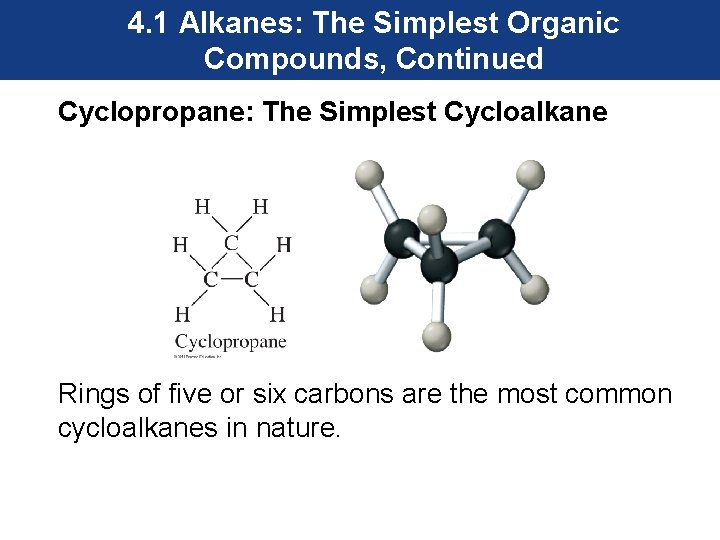
4. 1 Alkanes: The Simplest Organic Compounds, Continued Cyclopropane: The Simplest Cycloalkane Rings of five or six carbons are the most common cycloalkanes in nature.
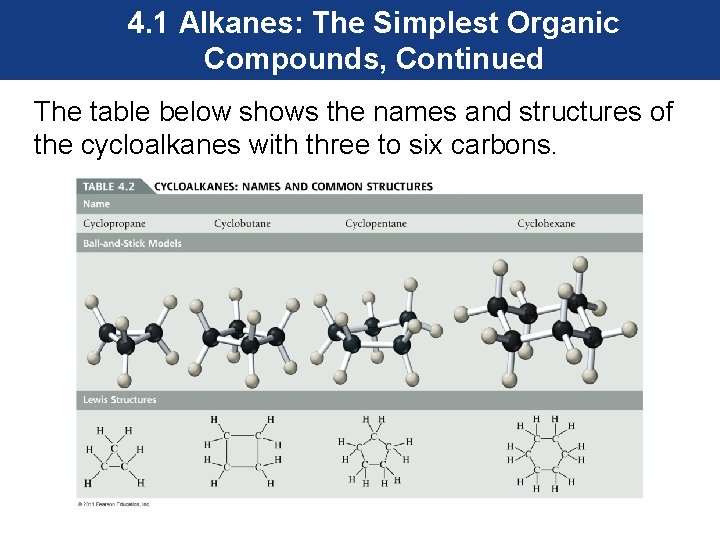
4. 1 Alkanes: The Simplest Organic Compounds, Continued The table below shows the names and structures of the cycloalkanes with three to six carbons.

4. 1 Alkanes: The Simplest Organic Compounds, Continued Alkanes Are Nonpolar Compounds • Electronegativities of carbon and hydrogen are similar enough that the bonding electrons between them are shared equally. • Equal sharing of electrons in the carbon– hydrogen bonds make these compounds nonpolar, regardless of their shape.

4. 2 Representing the Structures of Organic Compounds • Lewis structures show the connectivity of the atoms in a molecule, showing all atoms, bonds, and lone pairs of electrons. • As molecules get larger, chemists use simplified representations for the structures of organic molecules that show the entire structure, but omit some of the details. • For example, chemists omit the lone pair of electrons on oxygen when it is present in an organic compound.

4. 2 Representing the Structures of Organic Compounds, Continued Condensed Structural Formulas • A condensed structural formula, also known as condensed structure, shows all the atoms in a molecule, but show as few bonds as possible. This may or may not show lone pairs. It is not useful in drawing cycloalkanes. • Recall that carbon always forms four bonds and hydrogen forms one bond. This aids in interpreting condensed structures.
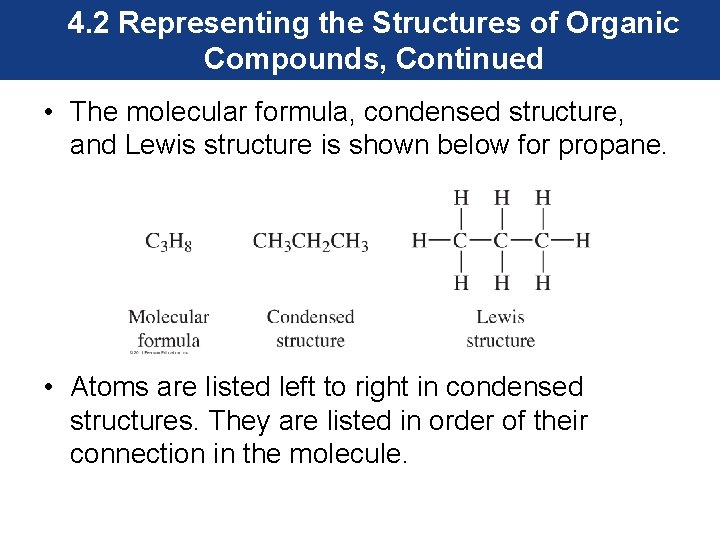
4. 2 Representing the Structures of Organic Compounds, Continued • The molecular formula, condensed structure, and Lewis structure is shown below for propane. • Atoms are listed left to right in condensed structures. They are listed in order of their connection in the molecule.
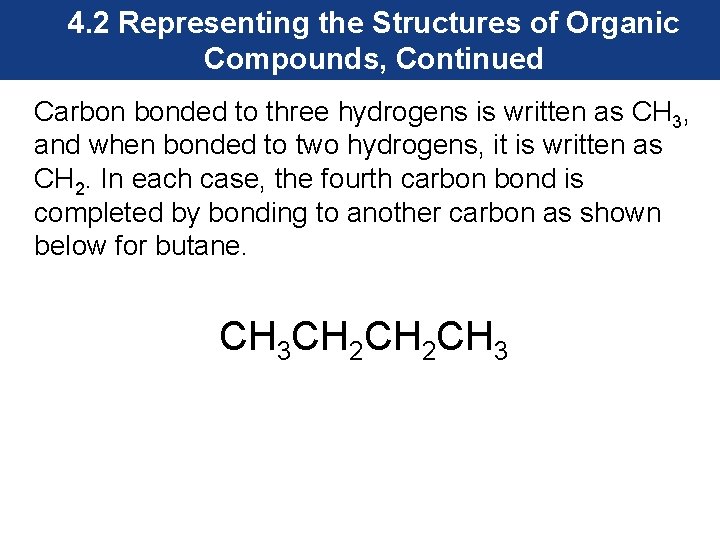
4. 2 Representing the Structures of Organic Compounds, Continued Carbon bonded to three hydrogens is written as CH 3, and when bonded to two hydrogens, it is written as CH 2. In each case, the fourth carbon bond is completed by bonding to another carbon as shown below for butane. CH 3 CH 2 CH 3
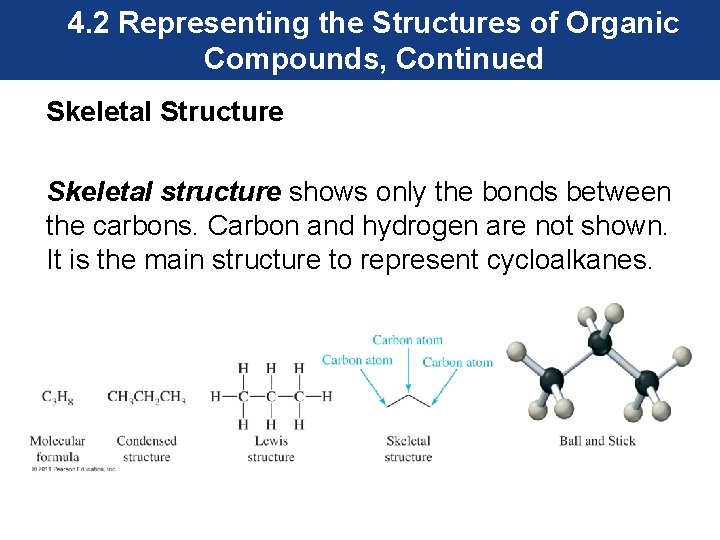
4. 2 Representing the Structures of Organic Compounds, Continued Skeletal Structure Skeletal structure shows only the bonds between the carbons. Carbon and hydrogen are not shown. It is the main structure to represent cycloalkanes.

4. 2 Representing the Structures of Organic Compounds, Continued • In the skeletal structure, carbon atoms are understood to be at the corner/angle created by the intersection of two bonds or at the termination of a bond. • Skeletal structures are used to represent cycloalkanes.

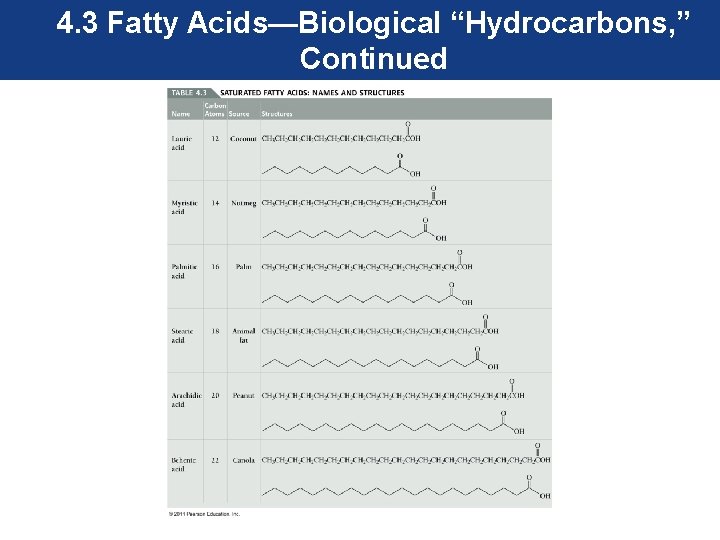
4. 3 Fatty Acids—Biological “Hydrocarbons” • Fatty acids have structures and properties similar to alkanes. As a result, they are called biological hydrocarbons. • Fatty acids belong to a class of biological compounds called lipids. Lipids are nonpolar biomolecules. • Saturated fatty acids contain carbon–carbon single bonds and are the main components of saturated fats.

4. 3 Fatty Acids—Biological “Hydrocarbons, ” Continued Structure and Polarity of Fatty Acid Molecules • Fatty acids are alkane-like compounds. Most common fatty acids are composed of 12 to 22 carbon atoms. Most naturally fatty acids contain an even number of carbon atoms. • Alkanes and fatty acids contain straight chains of carbon atoms. The difference is that fatty acids contain a functional group consisting of two oxygens and a hydrogen known as a carboxylic acid group.
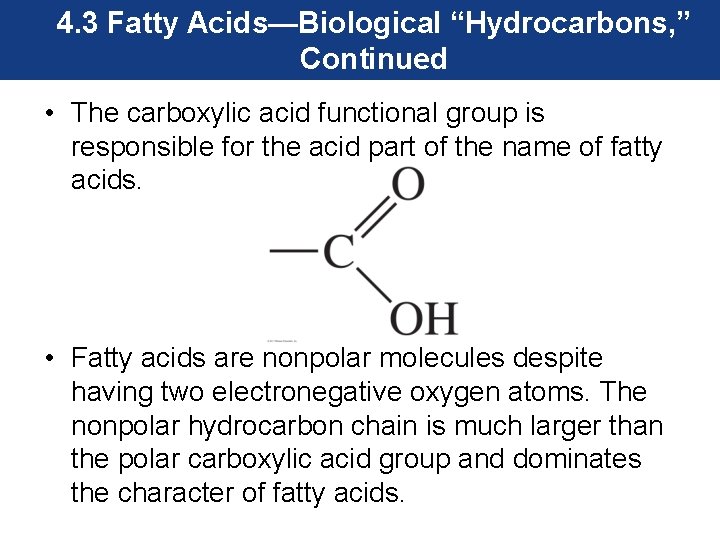
4. 3 Fatty Acids—Biological “Hydrocarbons, ” Continued • The carboxylic acid functional group is responsible for the acid part of the name of fatty acids. • Fatty acids are nonpolar molecules despite having two electronegative oxygen atoms. The nonpolar hydrocarbon chain is much larger than the polar carboxylic acid group and dominates the character of fatty acids.
4. 3 Fatty Acids—Biological “Hydrocarbons, ” Continued
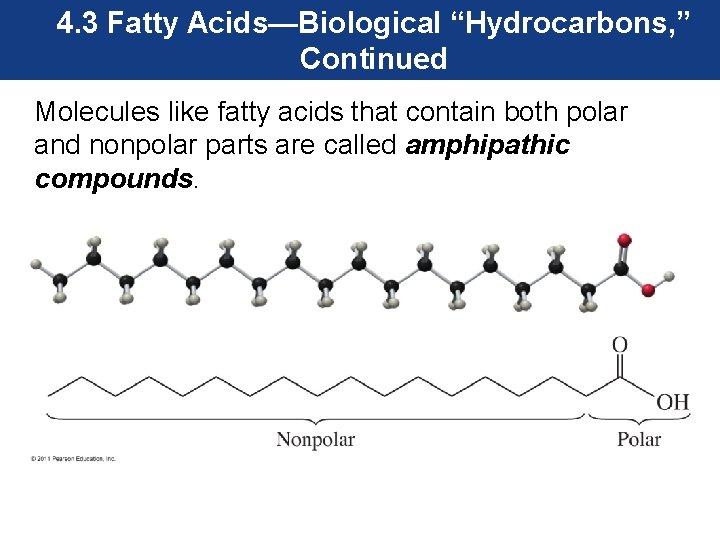
4. 3 Fatty Acids—Biological “Hydrocarbons, ” Continued Molecules like fatty acids that contain both polar and nonpolar parts are called amphipathic compounds.

4. 3 Fatty Acids—Biological “Hydrocarbons, ” Continued Unsaturated Fatty Acids • Unsaturated fatty acids are fatty acids that contain less than the maximum number of hydrogen atoms per carbon atom. • In these fatty acids, a carbon–carbon double bond exists for each missing pair of hydrogen atoms. • The double bond is another functional group found in organic compounds known as alkenes.

4. 3 Fatty Acids—Biological “Hydrocarbons, ” Continued • Fatty acids with one double bond are called monounsaturated, and those with two or more double bonds are called polyunsaturated. A double bond causes a U-shaped molecule. • The double bond does not affect the nonpolar nature of the unsaturated fatty acid.
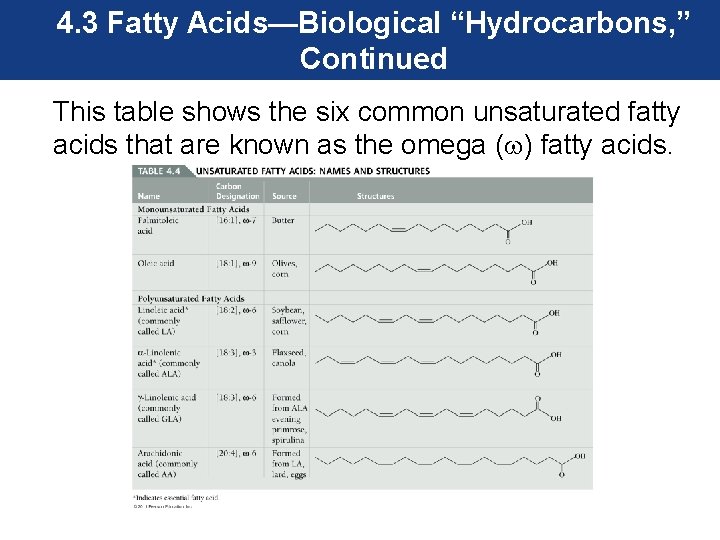
4. 3 Fatty Acids—Biological “Hydrocarbons, ” Continued This table shows the six common unsaturated fatty acids that are known as the omega ( ) fatty acids.

4. 3 Fatty Acids—Biological “Hydrocarbons, ” Continued Fatty Acids in Our Diets • Diets low in fats have been shown to be healthier. • Fats are needed in our diet because they play an important roles as insulation and protective coverings for our internal organs and nerve fibers. • A maximum of 30% of the calories in a normal diet should come from fatty acids.

4. 3 Fatty Acids—Biological “Hydrocarbons, ” Continued The polyunsaturated fatty acids linoleic and α-linoleic are considered essential fatty acids because our bodies cannot produce these compounds, so they must be obtained from our diet.

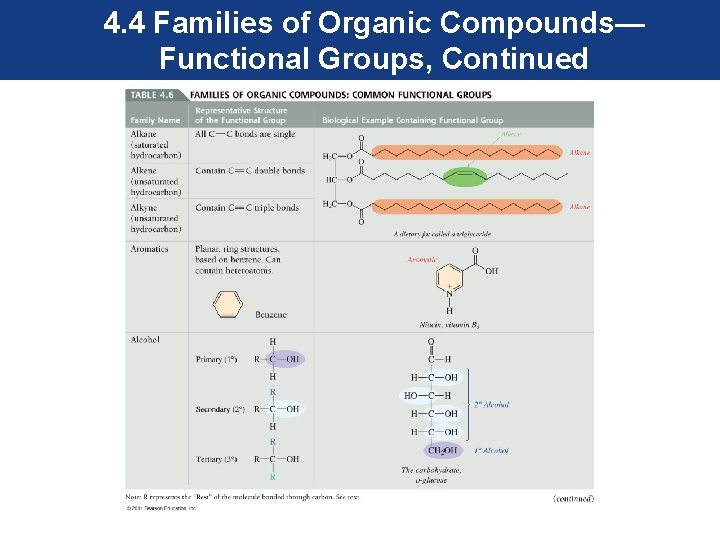
4. 4 Families of Organic Compounds— Functional Groups • Organic compounds are classified into families based on common functional groups. • Each functional group has specific properties and reactivity, and organic compounds that contain the same functional group behave similarly. • To understand the properties and reactivity of the many organic compounds, we need only to identify the functional group they contain.

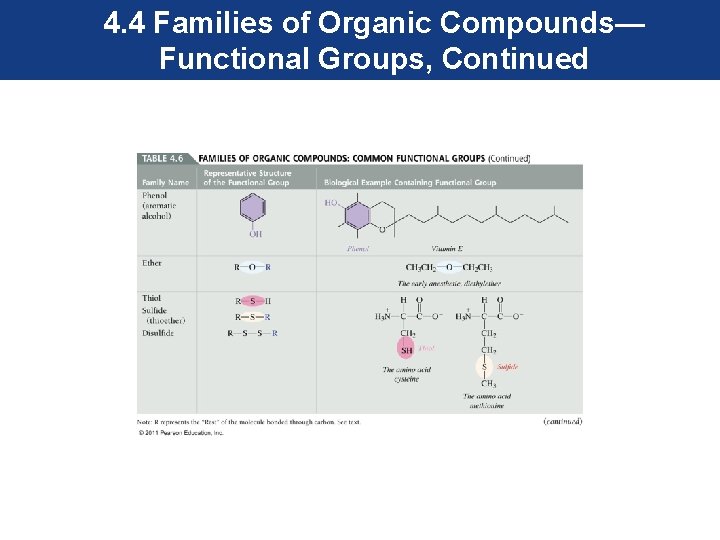
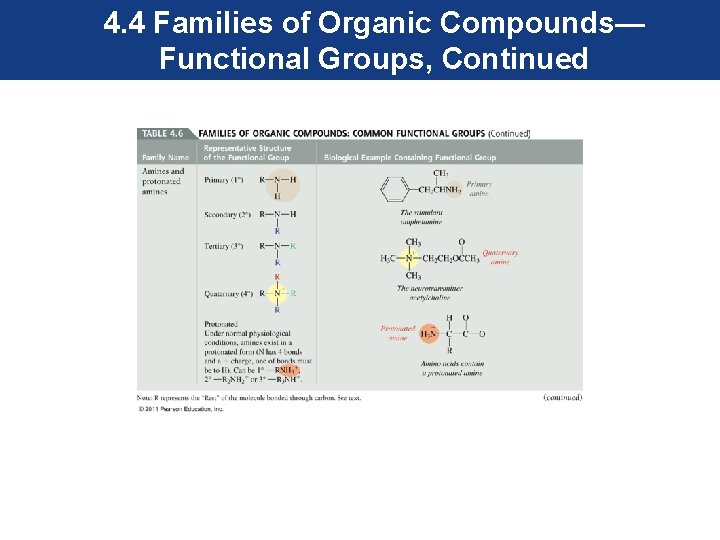
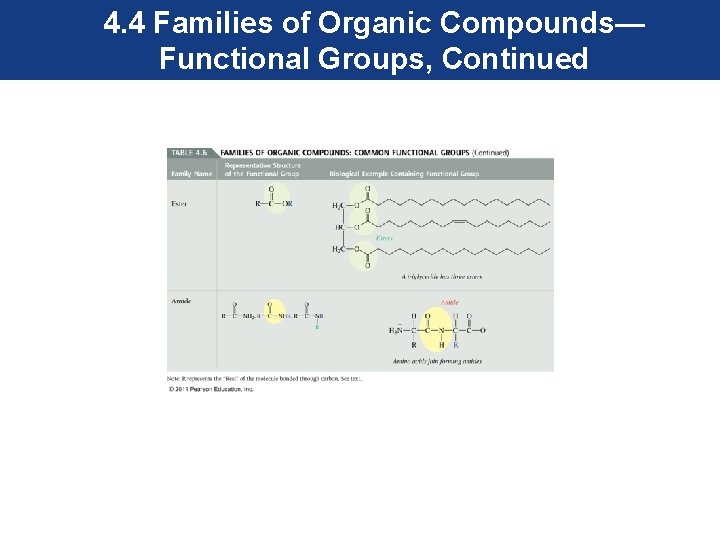
4. 4 Families of Organic Compounds— Functional Groups, Continued • Most functional groups contain atoms other than carbon and hydrogen. These atoms in their structure are called heteroatoms. • Most common heteroatoms are oxygen, nitrogen, sulfur, and the halogens.

4. 4 Families of Organic Compounds— Functional Groups, Continued • The first four functional groups—alkanes, alkenes, alkynes, and aromatics—will be considered in this chapter. • Those functional groups containing heteroatoms will be considered when they appear in the structure of biomolecules as they are studied. • The functional group is the reactive part of an organic molecule.
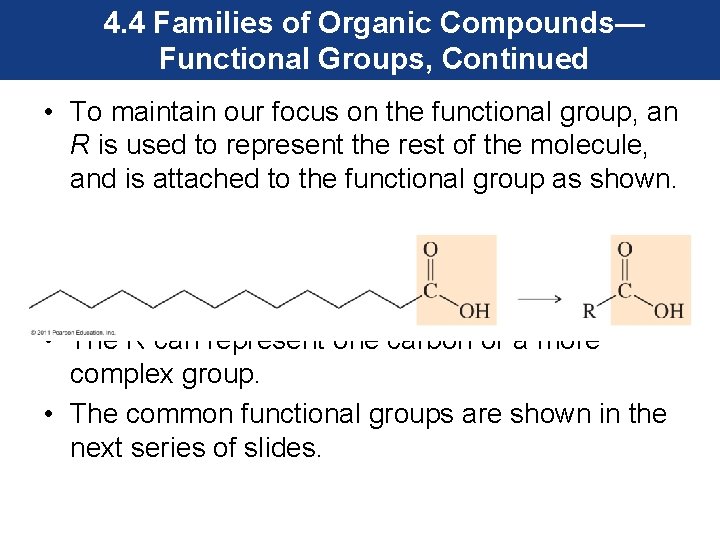
4. 4 Families of Organic Compounds— Functional Groups, Continued • To maintain our focus on the functional group, an R is used to represent the rest of the molecule, and is attached to the functional group as shown. • The R can represent one carbon or a more complex group. • The common functional groups are shown in the next series of slides.
4. 4 Families of Organic Compounds— Functional Groups, Continued
4. 4 Families of Organic Compounds— Functional Groups, Continued
4. 4 Families of Organic Compounds— Functional Groups, Continued

4. 4 Families of Organic Compounds— Functional Groups, Continued
4. 4 Families of Organic Compounds— Functional Groups, Continued

4. 4 Families of Organic Compounds— Functional Groups, Continued Unsaturated Hydrocarbons—Alkenes, Alkynes, and Aromatics Alkenes • Alkenes are hydrocarbons that contain carbon– carbon double bond(s). • Ethene, known as ethylene, a compound produced by ripening fruits, is the simplest alkene.

4. 4 Families of Organic Compounds— Functional Groups, Continued Alkenes • The double bond of alkenes is shorter and stronger than the single bond of alkanes. • The double bond makes alkenes more reactive than alkanes. • Many naturally occurring biological compounds contain the double bond found in alkenes.

4. 4 Families of Organic Compounds— Functional Groups, Continued Naturally Occurring Alkenes Alkene β-Carotene Function Orange pigment in carrots and sweet potatoes Limonene Scent of citrus fruits α-Pinene Scent of pine trees Fatty acids, cholesterol Biologically important compounds

4. 4 Families of Organic Compounds— Functional Groups, Continued

4. 4 Families of Organic Compounds— Functional Groups, Continued Alkynes • Alkynes are organic compounds that contain a carbon –carbon triple bond. • Ethyne (known as acetylene), a fuel used in welding torches, is the simplest alkyne. • The triple bond is shorter and stronger than the alkene’s double bond. This bond is more reactive than the double bond and is less stable.

4. 4 Families of Organic Compounds— Functional Groups, Continued Alkynes • Naturally occurring alkynes in nature are rare. • Alkynes occur mainly in short-lived compounds. • Histrionicotoxin, a poison secreted by the poison dart frog as a defense against its predators, contains two alkyne functional groups.

4. 4 Families of Organic Compounds— Functional Groups, Continued Aromatics • Aromatic compounds are six-carbon member rings with alternating double and single bonds. The simplest aromatic compound is benzene. • These compounds get their name, aromatic, because the first ones discovered have pleasant aromas. Compounds such as oil of spearmint and peppermint are compounds in the aromatic family.

4. 4 Families of Organic Compounds— Functional Groups, Continued Aromatics • When the benzene ring is part of a larger molecule it is called a phenyl group. • The aromatic ring is unreactive and is very stable. • Benzene contains three double bonds, but it is very stable and is resistant to reactions that would break double bonds.
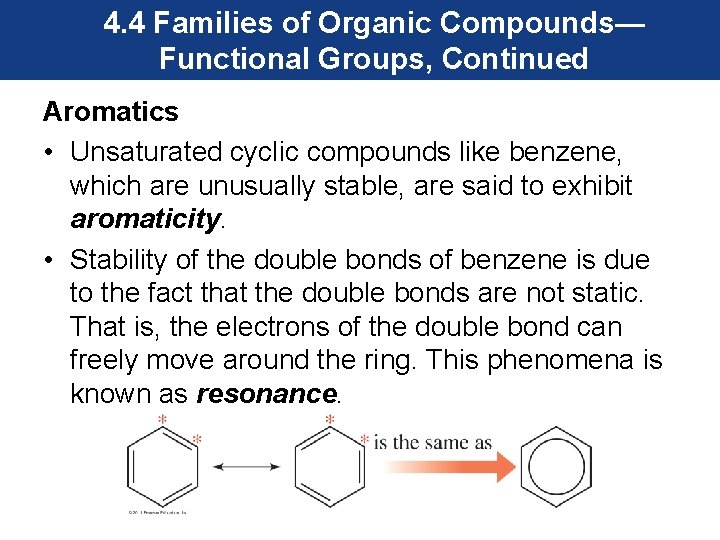
4. 4 Families of Organic Compounds— Functional Groups, Continued Aromatics • Unsaturated cyclic compounds like benzene, which are unusually stable, are said to exhibit aromaticity. • Stability of the double bonds of benzene is due to the fact that the double bonds are not static. That is, the electrons of the double bond can freely move around the ring. This phenomena is known as resonance.

4. 4 Families of Organic Compounds— Functional Groups, Continued Aromatics • Because electrons are equally shared with all the bonds of benzene, they are much less reactive. • Decreased reactivity means more stability of aromatic compounds. • Many compounds in tobacco smoke contain two or more benzene rings attached to each other.

4. 4 Families of Organic Compounds— Functional Groups, Continued Aromatics • These compounds are called polycyclic aromatic hydrocarbons or PAHs. Phenanthrene and benzo[α]pyrene are examples. Many have been shown to be carcinogenic (cancer causing). • Aromatic compounds are present in many plastics and pharmaceuticals.

4. 5 Isomerism in Organic Compounds, Part 1 Structural Isomers • Structural isomers are compounds with the same molecular formula but a different connectivity. • Butane, a four-carbon molecule, is the simplest alkane that has two structural isomers.
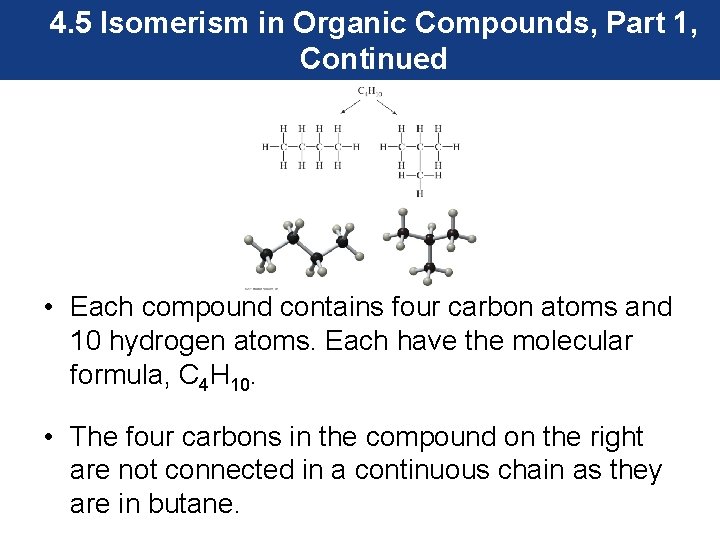
4. 5 Isomerism in Organic Compounds, Part 1, Continued • Each compound contains four carbon atoms and 10 hydrogen atoms. Each have the molecular formula, C 4 H 10. • The four carbons in the compound on the right are not connected in a continuous chain as they are in butane.

4. 5 Isomerism in Organic Compounds, Part 1, Continued • The second structural isomer of butane forms a branched alkane and is known as isobutane. • Branched-chain alkanes do not have all their carbon atoms connected in a single continuous chain. • Both compounds with the molecular formula of C 4 H 10 are structural isomers. That is, they have different connectivity of the atoms.

4. 5 Isomerism in Organic Compounds, Part 1, Continued Conformational Isomers • Conformational isomers are isomers that are not different compounds because they have different arrangements of the atoms of the compound. They are also known as conformers. • Consider butane. The structure of butane can be represented as shown on the next slide.
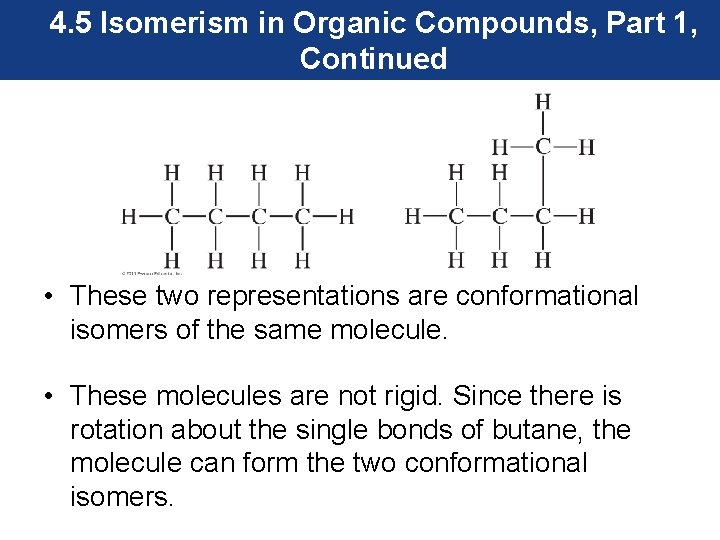
4. 5 Isomerism in Organic Compounds, Part 1, Continued • These two representations are conformational isomers of the same molecule. • These molecules are not rigid. Since there is rotation about the single bonds of butane, the molecule can form the two conformational isomers.
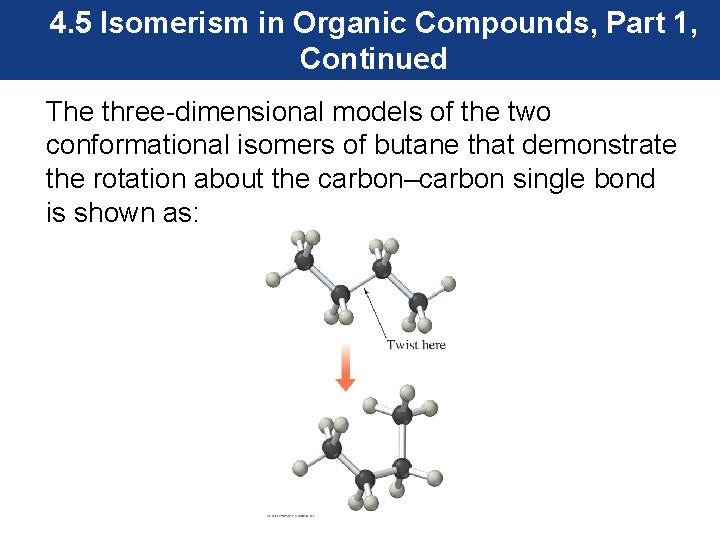
4. 5 Isomerism in Organic Compounds, Part 1, Continued The three-dimensional models of the two conformational isomers of butane that demonstrate the rotation about the carbon–carbon single bond is shown as:

4. 5 Isomerism in Organic Compounds, Part 1, Continued Nomenclature of Simple Organic Compounds • Systematically naming each organic compound is a useful method to determine whether two compounds are structural or conformational isomers. • Structural isomers will have different names, whereas conformational isomers will have the same name. • Naming of organic compounds was developed by the International Union of Pure and Applied Chemistry, or IUPAC.
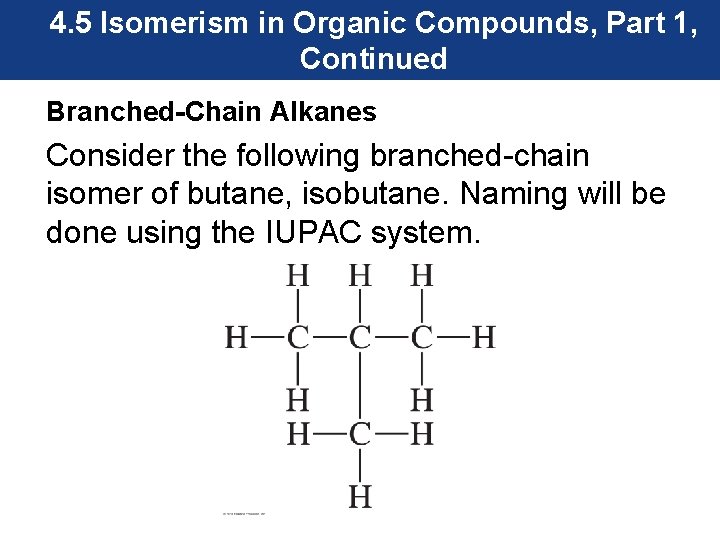
4. 5 Isomerism in Organic Compounds, Part 1, Continued Branched-Chain Alkanes Consider the following branched-chain isomer of butane, isobutane. Naming will be done using the IUPAC system.
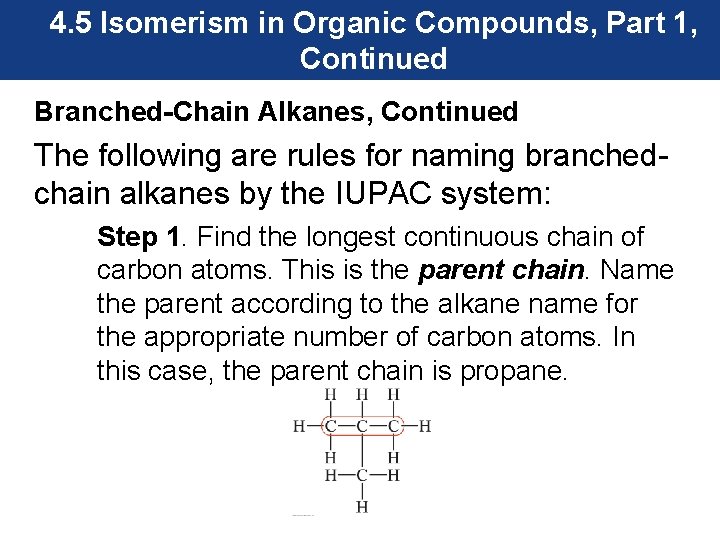
4. 5 Isomerism in Organic Compounds, Part 1, Continued Branched-Chain Alkanes, Continued The following are rules for naming branchedchain alkanes by the IUPAC system: Step 1. Find the longest continuous chain of carbon atoms. This is the parent chain. Name the parent according to the alkane name for the appropriate number of carbon atoms. In this case, the parent chain is propane.

4. 5 Isomerism in Organic Compounds, Part 1, Continued Branched-Chain Alkanes, Continued Step 2. Identify the groups bonded to the parent chain and not included in the main chain. These groups are called substituents and in this case, called alkyl groups. The name of the alkyl group is derived from the alkane with the same number of carbon atoms by changing the -ane ending with -yl. For example, a one carbon alkyl group is named as a methyl group.
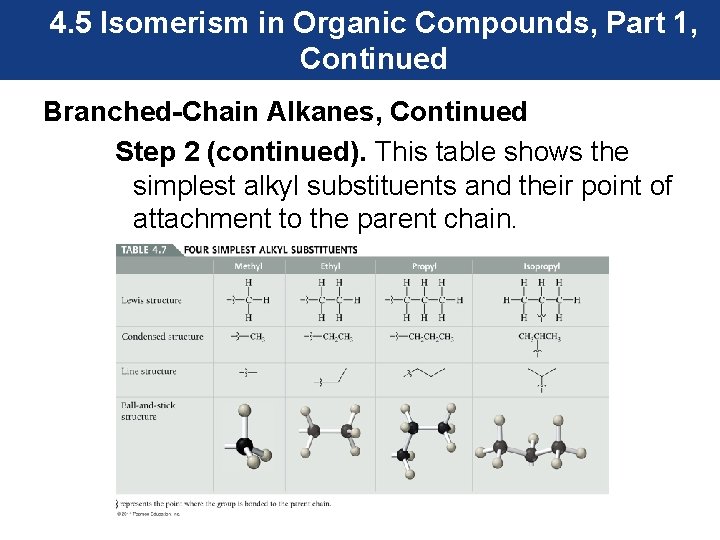
4. 5 Isomerism in Organic Compounds, Part 1, Continued Branched-Chain Alkanes, Continued Step 2 (continued). This table shows the simplest alkyl substituents and their point of attachment to the parent chain.
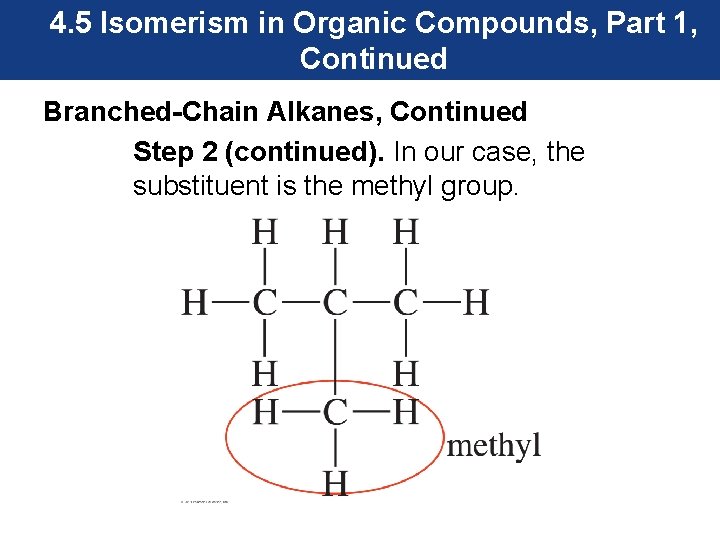
4. 5 Isomerism in Organic Compounds, Part 1, Continued Branched-Chain Alkanes, Continued Step 2 (continued). In our case, the substituent is the methyl group.
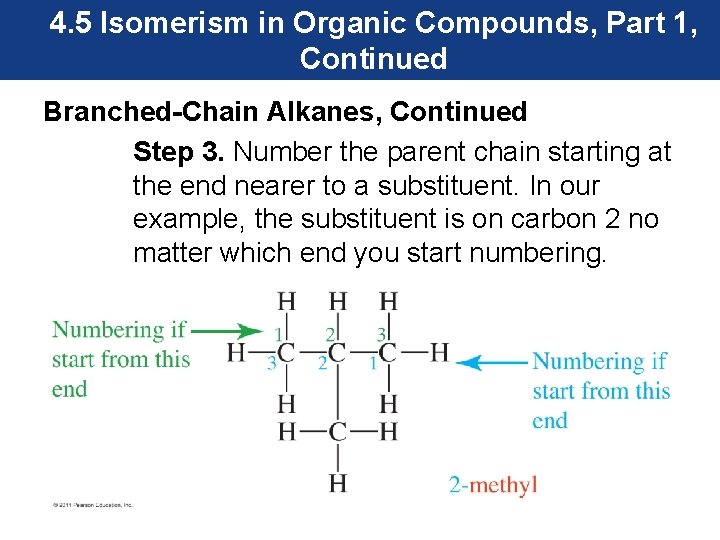
4. 5 Isomerism in Organic Compounds, Part 1, Continued Branched-Chain Alkanes, Continued Step 3. Number the parent chain starting at the end nearer to a substituent. In our example, the substituent is on carbon 2 no matter which end you start numbering.
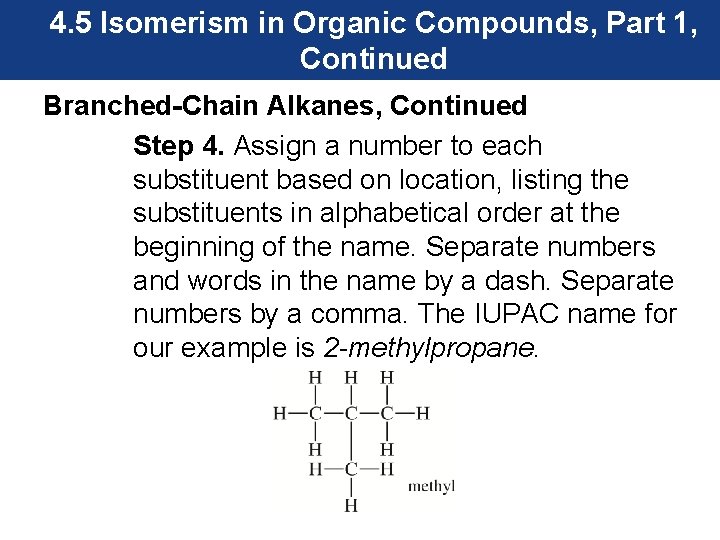
4. 5 Isomerism in Organic Compounds, Part 1, Continued Branched-Chain Alkanes, Continued Step 4. Assign a number to each substituent based on location, listing the substituents in alphabetical order at the beginning of the name. Separate numbers and words in the name by a dash. Separate numbers by a comma. The IUPAC name for our example is 2 -methylpropane.

4. 5 Isomerism in Organic Compounds, Part 1, Continued Branched-Chain Alkanes, Continued NOTE: If more than one substituent of the same type is present, indicate this by using the prefixes di -, tri-, and tetra-, but ignore these prefixes when alphabetizing. For example, two methyl groups on a parent chain would be named dimethyl.

4. 5 Isomerism in Organic Compounds, Part 1, Continued Haloalkanes • Halogens can serve as substituents on alkane chains and are known as haloalkanes, or alkyl halides. • When a halogen is present, the name of the halogen is changed by replacing the -ine ending with an -o. • Names of halogen substituents become fluoro, chloro, bromo, and iodo. • Rules for naming haloalkanes are the same for naming branched-chain alkanes.

4. 5 Isomerism in Organic Compounds, Part 1, Continued Cycloalkanes Rules for naming cylcoalkanes are as follows: Step 1. The ring serves as the parent. Step 2. As in branched-chain alkanes, identify the substituents. Step 3. Number the carbons giving the carbon with the substituent as carbon 1.

4. 5 Isomerism in Organic Compounds, Part 1, Continued Cycloalkanes, Continued Step 4. Assign numbers to the substituents. If only one substituent is present, it is assumed to be in position 1 and the 1 is implied and not listed. When more than one substituent is present, give all substituents the lowest possible numbers.

4. 6 Isomerism in Organic Compounds, Part 2 • Besides structural and conformational isomers, there is another type of isomer known as stereoisomers. • Stereoisomers differ from one another by the arrangement of their atoms in space. • Stereoisomers have different three-dimensional shapes.
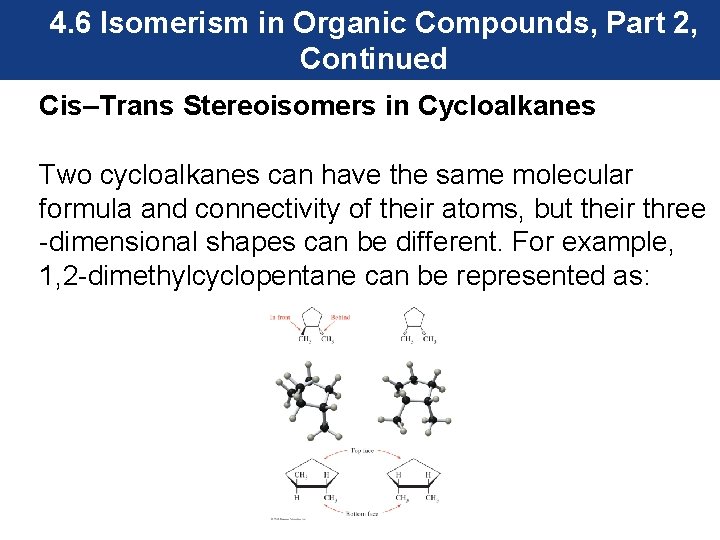
4. 6 Isomerism in Organic Compounds, Part 2, Continued Cis–Trans Stereoisomers in Cycloalkanes Two cycloalkanes can have the same molecular formula and connectivity of their atoms, but their three -dimensional shapes can be different. For example, 1, 2 -dimethylcyclopentane can be represented as:

4. 6 Isomerism in Organic Compounds, Part 2, Continued • The three-dimensional shapes of the two stereoisomers of 1, 2 -dimethylcylcopentane are different. • The methyl group bonded to the ring structure with a wedge-shaped bond is projecting out of the page toward the viewer. • The structure with the methyl groups bonded with a dashed bond are projecting behind the page away from the viewer.
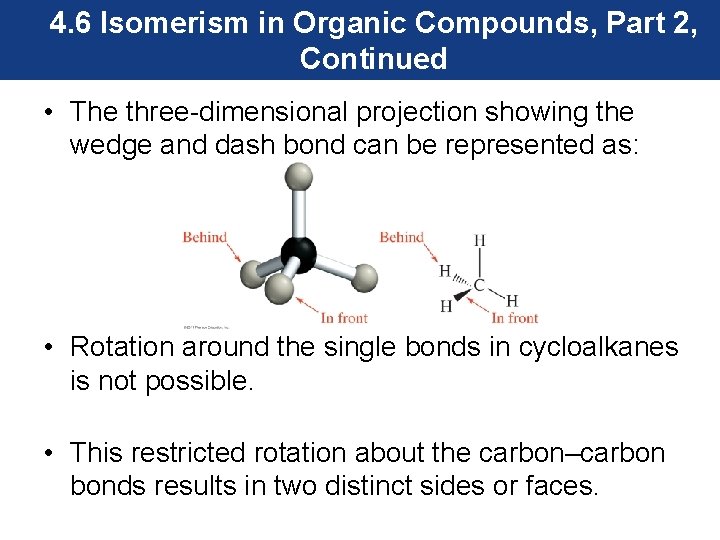
4. 6 Isomerism in Organic Compounds, Part 2, Continued • The three-dimensional projection showing the wedge and dash bond can be represented as: • Rotation around the single bonds in cycloalkanes is not possible. • This restricted rotation about the carbon–carbon bonds results in two distinct sides or faces.
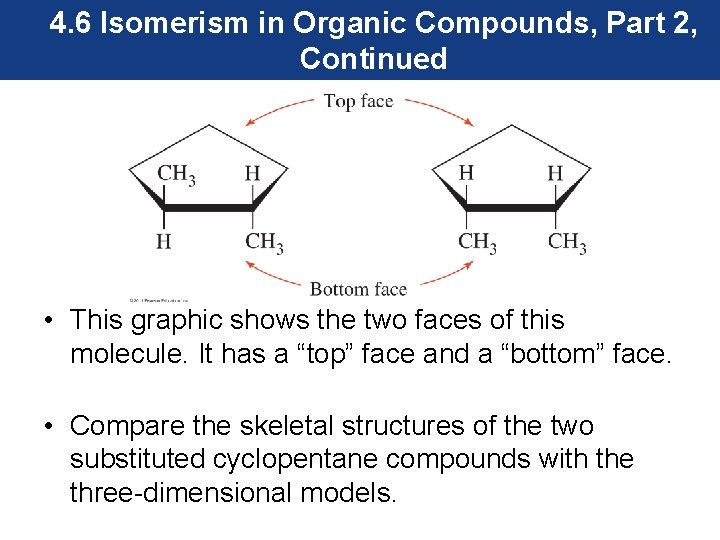
4. 6 Isomerism in Organic Compounds, Part 2, Continued • This graphic shows the two faces of this molecule. It has a “top” face and a “bottom” face. • Compare the skeletal structures of the two substituted cyclopentane compounds with the three-dimensional models.
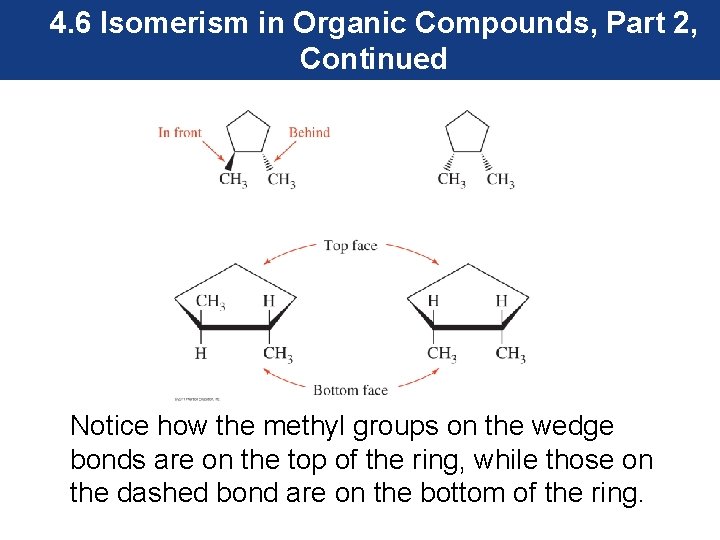
4. 6 Isomerism in Organic Compounds, Part 2, Continued Notice how the methyl groups on the wedge bonds are on the top of the ring, while those on the dashed bond are on the bottom of the ring.

4. 6 Isomerism in Organic Compounds, Part 2, Continued • How do we distinguish the two stereoisomers of 1, 2 -dimethylcyclopentane by name? • These two stereoisomers belong to a class called cis–trans stereoisomers. • The cis stereoisomer has the methyl groups on the same face of the cyclopentane ring. • The trans stereoisomer has the methyl groups on opposite sides of the ring from each other.
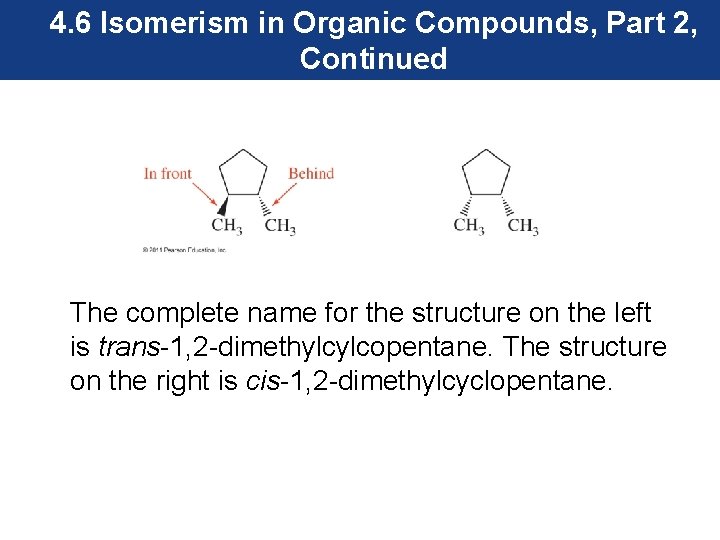
4. 6 Isomerism in Organic Compounds, Part 2, Continued The complete name for the structure on the left is trans-1, 2 -dimethylcylcopentane. The structure on the right is cis-1, 2 -dimethylcyclopentane.
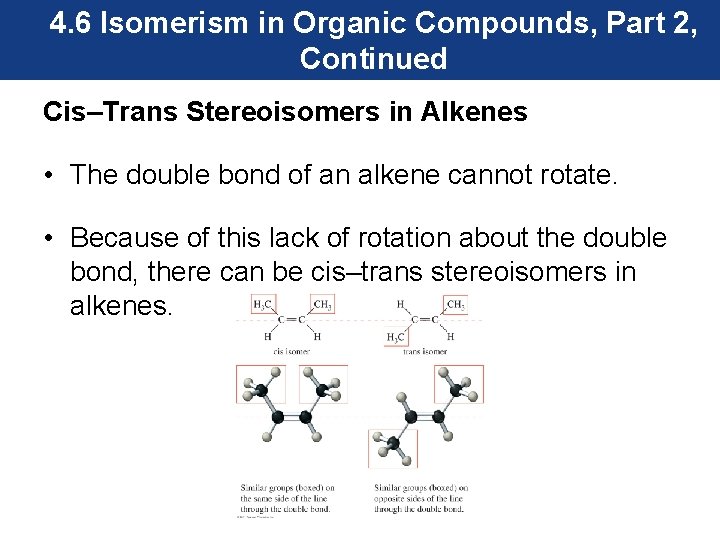
4. 6 Isomerism in Organic Compounds, Part 2, Continued Cis–Trans Stereoisomers in Alkenes • The double bond of an alkene cannot rotate. • Because of this lack of rotation about the double bond, there can be cis–trans stereoisomers in alkenes.

4. 6 Isomerism in Organic Compounds, Part 2, Continued • The compounds shown in the previous slide have a top and bottom face as represented by the dashed line. • The two compounds are not identical, and they are not conformers (they cannot be changed into the other by rotating around a bond). • The two compounds are cis–trans stereoisomers.
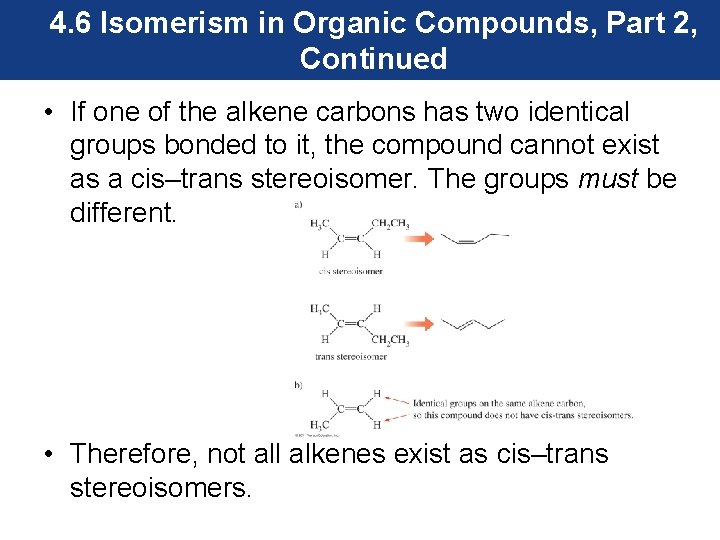
4. 6 Isomerism in Organic Compounds, Part 2, Continued • If one of the alkene carbons has two identical groups bonded to it, the compound cannot exist as a cis–trans stereoisomer. The groups must be different. • Therefore, not all alkenes exist as cis–trans stereoisomers.

4. 6 Isomerism in Organic Compounds, Part 2, Continued • Reconsider the carbon–carbon double bonds shown for unsaturated fatty acids. • These U-shaped skeletal structures in fatty acids are cis double bonds. All natural fatty acids are cis stereoisomers.

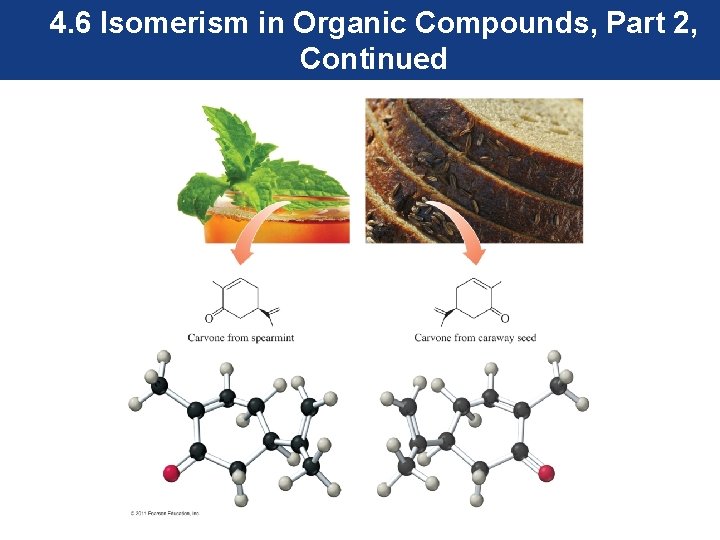
4. 6 Isomerism in Organic Compounds, Part 2, Continued Stereoisomers—Chiral Molecules and Enantiomers • The compounds in spearmint and caraway seeds that are responsible for their flavor are a pair of stereoisomers called carvones, and our taste buds can distinguish between them. • Stereoisomers like spearmint and caraway are identical except for the arrangement of groups in space.
4. 6 Isomerism in Organic Compounds, Part 2, Continued

4. 6 Isomerism in Organic Compounds, Part 2, Continued • The spearmint and caraway carvones have a molecular formula of C 10 H 16 O, so they are isomers. • However, they are not structural isomers; instead, they are stereoisomers. They are not cis –trans isomers. • If the carvone from spearmint is flipped over, we see that the ketone carbonyl group is behind the molecule, while the same group is in front of the molecule in the carvone from caraway.
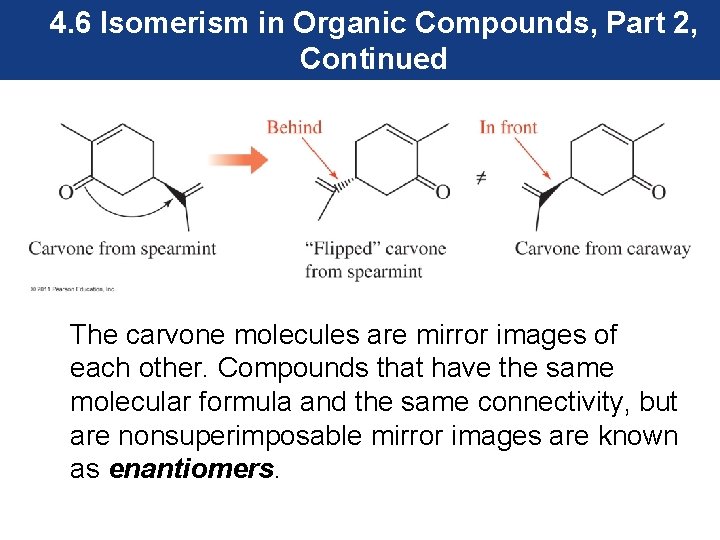
4. 6 Isomerism in Organic Compounds, Part 2, Continued The carvone molecules are mirror images of each other. Compounds that have the same molecular formula and the same connectivity, but are nonsuperimposable mirror images are known as enantiomers.
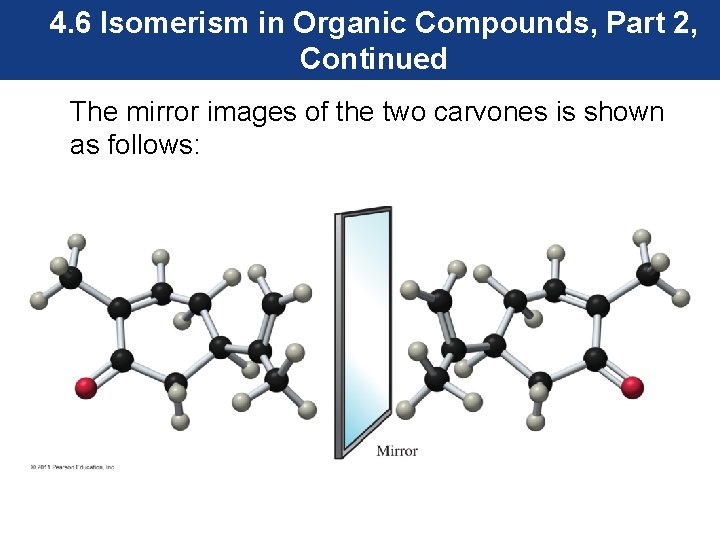
4. 6 Isomerism in Organic Compounds, Part 2, Continued The mirror images of the two carvones is shown as follows:

4. 6 Isomerism in Organic Compounds, Part 2, Continued • Common objects encountered in everyday life have nonsuperimposable mirror images. Shoes are nonsuperimposable mirror images. Your left hand is a nonsuperimposable mirror image of your right hand. • Objects that have nonsuperimposable images are termed chiral. • Molecules can also be chiral. Molecules that exist as enantiomers contain a chiral center.
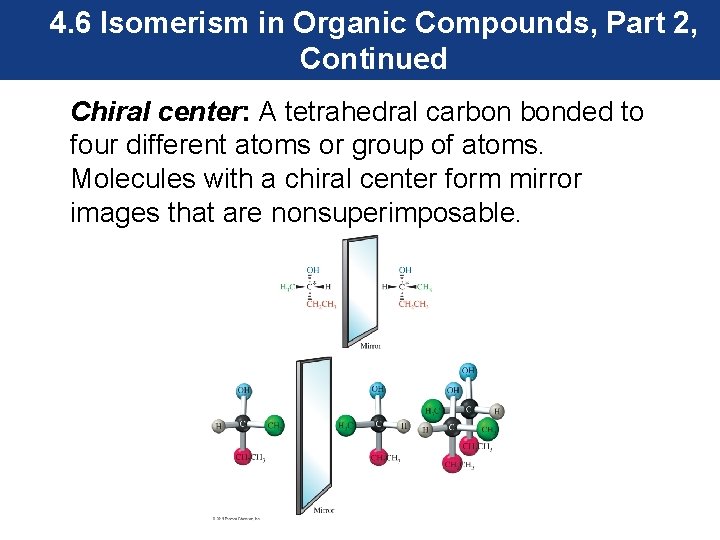
4. 6 Isomerism in Organic Compounds, Part 2, Continued Chiral center: A tetrahedral carbon bonded to four different atoms or group of atoms. Molecules with a chiral center form mirror images that are nonsuperimposable.

4. 6 Isomerism in Organic Compounds, Part 2, Continued The Consequences of Chirality • The cells of your tongue are also “handed, ” that is they have receptors that will only accept spearmint and other receptors that will only accept caraway. This is what gives these compounds their unique taste. • Pharmaceuticals are the same way. One shape may have biological activity, while the other may have no biological activity.

4. 6 Isomerism in Organic Compounds, Part 2, Continued • In some cases with enantiomers of a drug, one can be beneficial and the other harmful. The drug thalidomide is an example. One form was effective in treating morning sickness and the other form caused severe birth defects. • L-Dopa is effective in treating Parkinson’s disease, while its mirror image has no biological effect. • Ibuprofen, found in Advil, has one active enantiomer.

Chapter Summary 4. 1 Alkanes: The Simplest Organic Compounds • Alkanes contain only carbon atoms and hydrogen atoms. • The bonds between carbon are single bonds. • Alkanes are saturated hydrocarbons.

Chapter Summary, Continued 4. 2 Representing the Structures of Organic Compounds • Organic compounds can be represented with Lewis structures, showing all atoms, bonds, and lone pairs of electrons. • Condensed structural formulas show all atoms, but show bonds only when necessary. Lone pairs of electrons are not shown. • Skeletal structures show the bonding structure by showing all carbon–carbon bonds. They are useful in representing cycloalkanes.

Chapter Summary, Continued 4. 3 Fatty Acids—Biological “Hydrocarbons” • Fatty acids are alkane-like biomolecules that are components of dietary fats. • Fatty acids are nonpolar compounds, despite the fact that they contain a polar carboxylic acid group on one end of the molecule. • Fatty acids containing double bonds are known as unsaturated fatty acids. • Naturally occurring fatty acids contain an even number of carbon atoms and have a cis configuration around the double bonds.

Chapter Summary, Continued 4. 4 Families of Organic Compounds—Functional Groups • Organic compounds are grouped into families based on the type of functional group present. • Functional groups have specific properties and reactivity. • Compounds with the same functional group behave similarly. • The functional group is of the most interest in a molecule, so the rest of the molecule is represented as R.

Chapter Summary, Continued 4. 5 Isomerism in Organic Compounds, Part 1 • Organic compounds with the same molecular formula but different connectivity of atoms are known as structural isomers. • Straight-chain and branched-chain alkanes are examples. • Conformational isomers can be interconverted by rotating about carbon–carbon single bonds. • Naming organic compounds is useful in distinguishing between structural and conformational isomers.

Chapter Summary, Continued 4. 6 Isomerism in Organic Compounds, Part 2 • Stereoisomers are two molecules with the same molecular formula and connectivity, but a different arrangement of the atoms in space. • Cycloalkanes with two substituents on different ring carbons can exist as cis–trans stereoisomers. • Cycloalkanes have a top and bottom face, so they can have substituents on the same ring arranged differently on the faces.

Chapter Summary, Continued 4. 6 Isomerism in Organic Compounds, Part 2, Continued • Cis stereoisomers have both substituents on the same face of the ring. Trans stereoisomers have the substituents on opposite faces. • Alkenes can have cis–trans stereoisomers because of a lack of rotation around the double bond. • Organic compounds can exist as stereoisomers if they contain a chiral center, which is a carbon atom bonded to four different atoms or group of atoms. • Compounds with a single chiral center exist as enantiomers. They are nonsuperimposable mirror images.
- Slides: 96