Chapter 4 Lecture Basic Chemistry Fourth Edition Chapter

Chapter 4 Lecture Basic Chemistry Fourth Edition Chapter 4 Atoms and Elements 4. 2 The Periodic Table Learning Goal Use the periodic table to identify the group and the period of an element; identify the element as a metal, a nonmetal, or a metalloid. © 2014 Pearson Education, Inc.

The Periodic Table In 1872, Dmitri Mendeleev • created the periodic table • arranged elements by increasing atomic mass • arranged elements into groups with similar properties © 2014 Pearson Education, Inc.
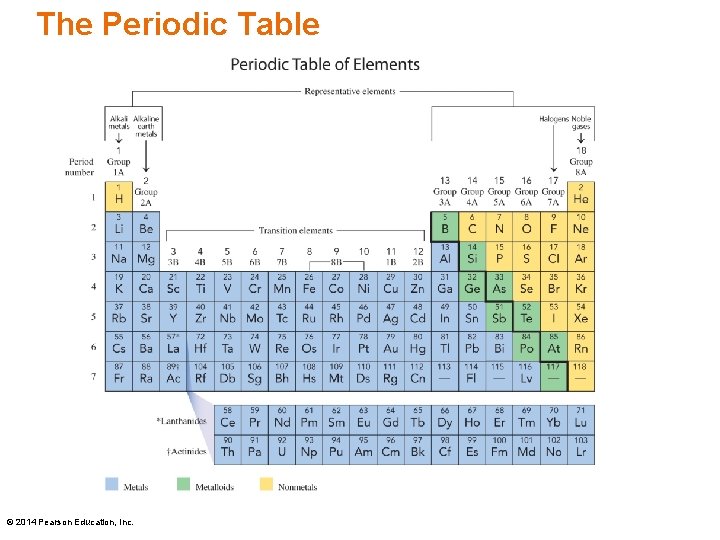
The Periodic Table © 2014 Pearson Education, Inc.

Periods and Groups On the periodic table • elements are arranged according to similar properties in vertical columns called groups • periods are horizontal rows of elements Periods 1 contains two elements: H and He Periods 2 and 3 each contain eight elements: Periods 2 – Li, Be, B, C, N, O, F, He Periods 3 – Na, Mg, Al, Si, P, S, Cl, Ar © 2014 Pearson Education, Inc.
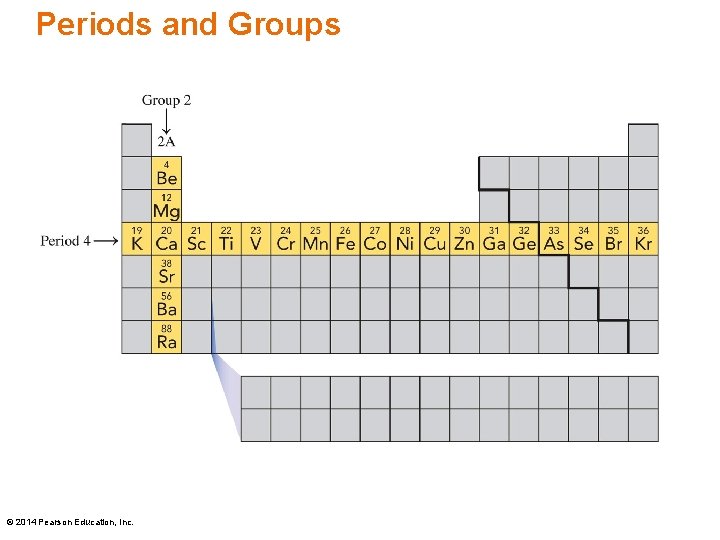
Periods and Groups © 2014 Pearson Education, Inc.

Group Numbers, written at the top of each vertical column, are written two ways: • The letter A is used for the representative elements 1 A to 8 A and the letter B for the transition elements. • The numbers 1– 18 are used for the columns from left to right. © 2014 Pearson Education, Inc.
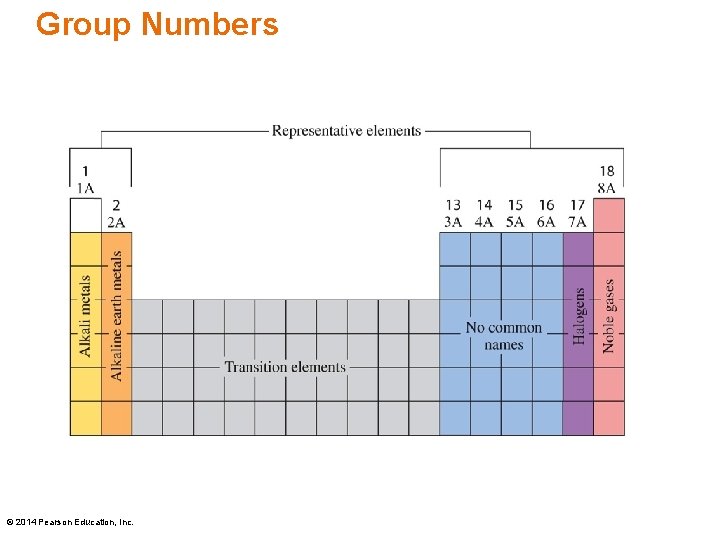
Group Numbers © 2014 Pearson Education, Inc.

Representative Elements – Groups and Names Representative elements include • Group 1 A (1), called Alkali metals Li, Na, K, Rb, Cs, and Fr • Group 2 A (2) called Alkaline earth metals Be, Mg, Ca, Sr, Ba, and Ra • Groups 3 A (13), 4 A (14), 5 A (15), and 6 A (16) • Group 7 A (17), called Halogens F, Cl, Br, I, and At • Group 8 A (18), called Noble gases He, Ne, Ar, Kr, Xe, and Rn © 2014 Pearson Education, Inc.
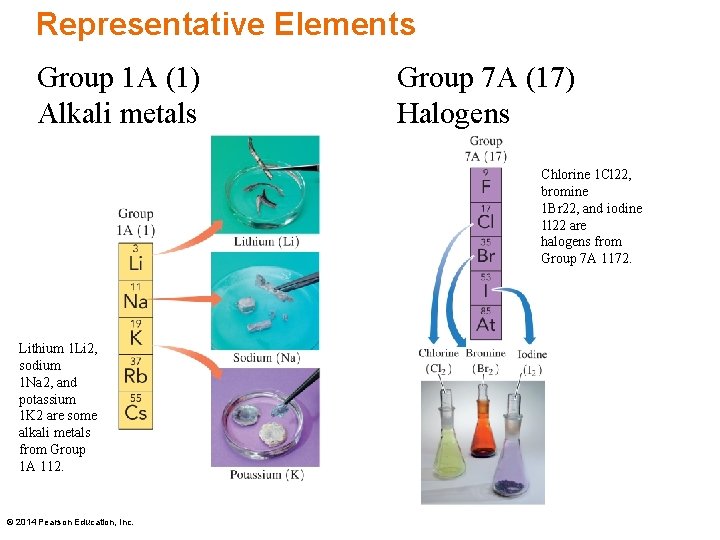
Representative Elements Group 1 A (1) Alkali metals Group 7 A (17) Halogens Chlorine 1 Cl 22, bromine 1 Br 22, and iodine 1 l 22 are halogens from Group 7 A 1172. Lithium 1 Li 2, sodium 1 Na 2, and potassium 1 K 2 are some alkali metals from Group 1 A 112. © 2014 Pearson Education, Inc.

Metals, Nonmetals, and Metalloids The heavy zigzag line on the periodic table separates the metals from nonmetals. Metalloids border the zigzag line. © 2014 Pearson Education, Inc.

Metals, Nonmetals, and Metalloids Most metals are shiny solids, such as copper (Cu), gold (Au), and silver (Ag). Metals are • ductile, can be shaped into wires • malleable, can be hammered into a flat sheet • good conductors of electricity • melted at much higher temperatures than nonmetals • solids at room temperature, except for mercury (Hg) © 2014 Pearson Education, Inc.

Metals, Nonmetals, and Metalloids Except for aluminum (Al), the elements along the zigzag line are metalloids. Metalloids • include B, Si, Ge, As, Sb, Te, Po, and At • exhibit some properties typical of metals and some typical of nonmetals • are better conductors of heat and electricity than nonmetals, but not as good as metals • are semiconductors because they can be modified to function as conductors or insulators © 2014 Pearson Education, Inc.

Metals, Nonmetals, and Metalloids Nonmetals include elements such as hydrogen (H), carbon (C), nitrogen (N), oxygen (O), chlorine (Cl), and sulfur (S). Nonmetals • not especially shiny, malleable, or ductile, and are often poor conductors of heat and electricity • typically have low melting points and low densities © 2014 Pearson Education, Inc.

Metals, Nonmetals, and Metalloids © 2014 Pearson Education, Inc.

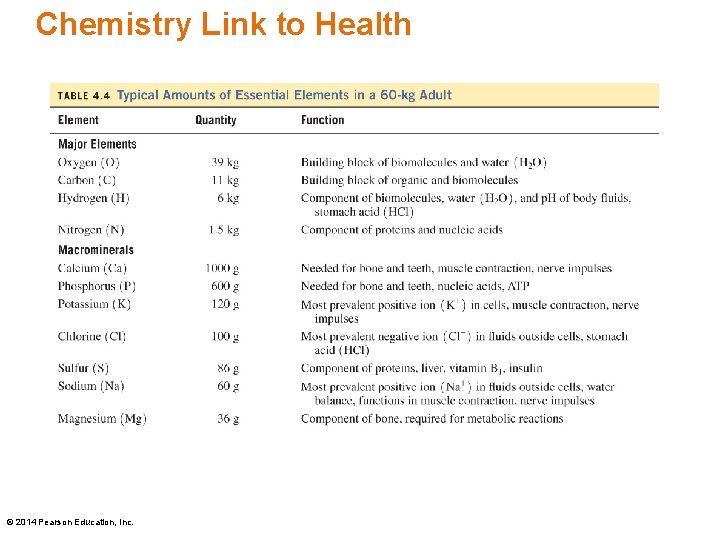
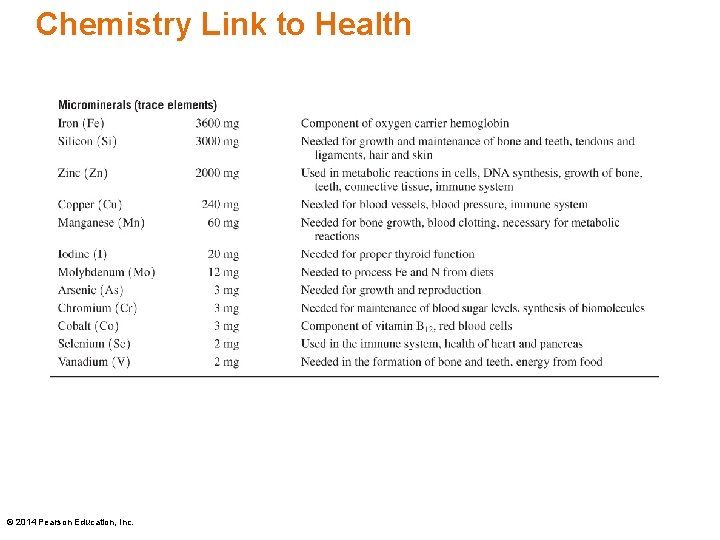
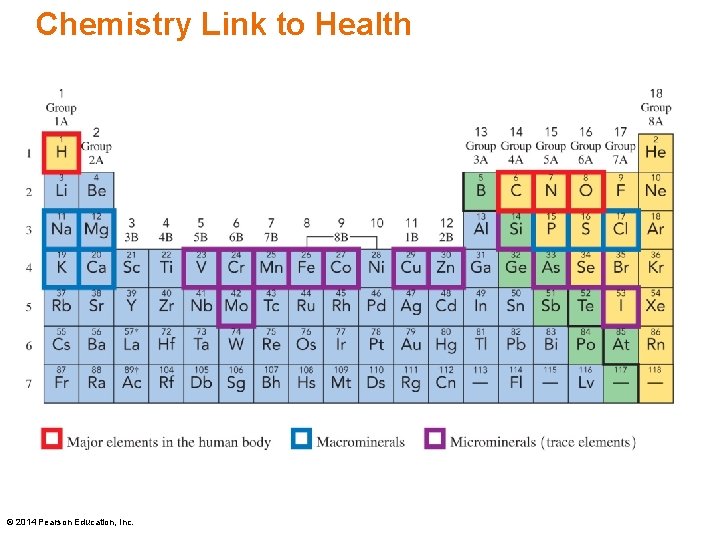
Chemistry Link to Health • Only 20 elements are essential for the well-being and survival of the human body. • Only four elements, oxygen (O), carbon (C), hydrogen (H) and nitrogen (N) make up 96 % of our body mass. • The macrominerals Ca, P, K, Cl, S, Na, and Mg are involved in the formation of bones and teeth, and the maintenance of essential body functions. © 2014 Pearson Education, Inc.
Chemistry Link to Health © 2014 Pearson Education, Inc.
Chemistry Link to Health © 2014 Pearson Education, Inc.
Chemistry Link to Health © 2014 Pearson Education, Inc.

Learning Check Match the elements to the description: A. Metals in Group 4 A (14) (1) Sn, Pb (2) C, Si (3) C, Si, Ge, Sn B. Nonmetals in Group 5 A (15) (1) As, Sb, Bi (2) N, P As, Sb C. (3) N, P, Metalloids in Group 4 A (14) (1) C, Si, Ge (2) Si, Ge (3) Si, Ge, Sn, Pb © 2014 Pearson Education, Inc.

Solution Match the elements to the description: A. Metals in Group 4 A (14) (1) Sn, Pb B. Nonmetals in Group 5 A (15) (2) N, P C. Metalloids in Group 4 A (14) (2) Si, Ge © 2014 Pearson Education, Inc.
- Slides: 20