Chapter 4 Forces Between Particles 4 3 Ionic

Chapter 4 Forces Between Particles 4. 3 Ionic Compounds Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings 1

Ionic Compounds Ionic compounds • consist of positive and negative ions. • have attractions called ionic bonds between positively and negatively charged ions. • have high melting and boiling points. • are solid at room temperature. 2

Salt is An Ionic Compound Sodium chloride or “table salt” is an example of an ionic compound. Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings 3

Ionic Formulas An ionic formula • consists of positively and negatively charged ions. • is neutral. • has charge balance. total positive charge = total negative charge The symbol of the metal is written first followed by the symbol of the nonmetal. 4

Charge Balance for Na. Cl, “Salt” In Na. Cl, • a Na atom loses its valence electron. • a Cl atom gains an electron. • the symbol of the metal is written first followed by the symbol of the nonmetal. Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings 5
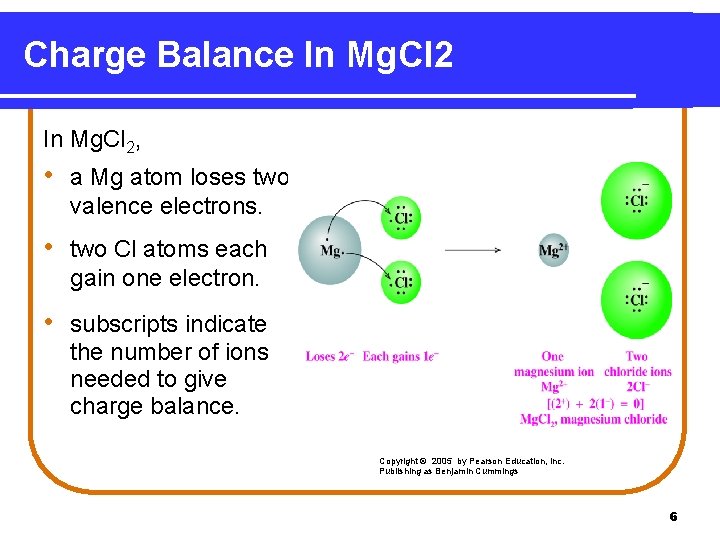
Charge Balance In Mg. Cl 2, • a Mg atom loses two valence electrons. • two Cl atoms each gain one electron. • subscripts indicate the number of ions needed to give charge balance. Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings 6
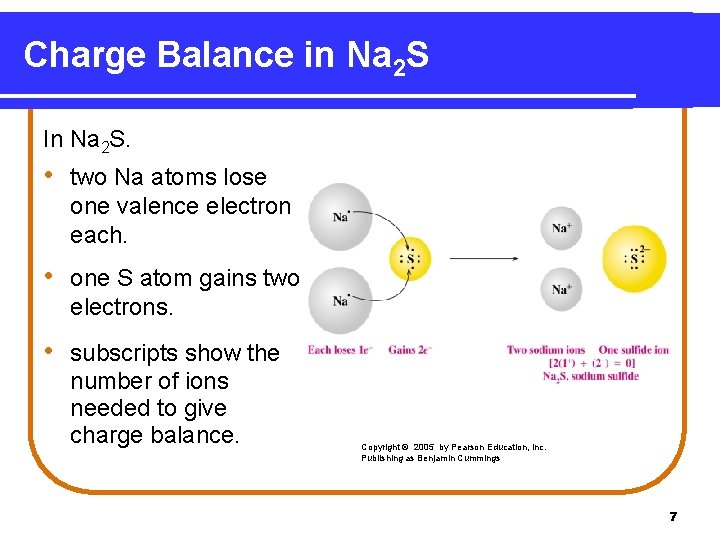
Charge Balance in Na 2 S In Na 2 S. • two Na atoms lose one valence electron each. • one S atom gains two electrons. • subscripts show the number of ions needed to give charge balance. Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings 7

Writing Ionic Formulas from Charges Charge balance is used to write the formula for sodium nitride, a compound containing Na+ and N 3−. 3 Na+ + N 3− Na+ = 3(+1) + 1(3 -) = Na 3 N 0 8

The balance of charges in Al 2 O 3 9

Formula from Ionic Charges Write the ionic formula of the compound with Ba 2+ and Cl. • Write the symbols of the ions. • • Ba 2+ Cl Balance the charges. Ba 2+ Cl two Cl- needed Cl Write the ionic formula using a subscript 2 for two chloride ions that give charge balance. Ba. Cl 2 10

Learning Check Select the correct formula for each of the following ionic compounds. A. Na+ and S 21) Na. S 2) Na 2 S 3) Na. S 2 B. Al 3+ and Cl 1) Al. Cl 3 2) Al. Cl 3) Al 3 Cl C. Mg 2+ and N 31) Mg. N 2) Mg 2 N 3 3) Mg 3 N 2 11

Solution A. Na+ and S 22) Na 2 S check: 2 Na+ + S 2 - = 2(1+) + 1(2 -) = 0 B. Al 3+ and Cl 1) Al. Cl 3 check: Al 3+ + Cl- = (3+) + 3(1 -) = 0 C. Mg 2+ and N 33) Mg 3 N 2 check: 2 Mg 2+ +2 N 3 - = 2(3+) + 2(3 -) = 0 12
- Slides: 12