Chapter 4 Developing Chemical Equations Unit 1 Chemistry















- Slides: 18

Chapter 4: Developing Chemical Equations Unit 1: Chemistry (5. 2) SNC 2 DP Chapter 5: Classifying Chemical Reactions Chapter 6: Acids and Bases

Remember. . . There are five main types of chemical reactions: 1) Synthesis Reaction 2) Decomposition Reaction 3) Single Displacement Reaction 4) Double Displacement reaction 5) Combustion reaction

Synthesis Reaction. . . A synthesis reaction is a chemical reaction in which two or more reactants combine to produce a new product.

Decomposition Reaction. . . A decomposition reaction is a chemical reaction in which a compound breaks down (decomposes) into two or more simpler compounds or elements.

Single Displacement Reaction Single displacement reaction: A + BC AC + B Where A is a metal A + BC BA + C Where A is a non-metal

Examples of Single Displacement Reaction Displacing silver (Ag) from silver nitrate (Ag. NO 3) Cu(s) + 2 Ag. NO 3(aq) Cu(NO 3)2(aq) + 2 Ag(s) Producing copper (Cu) metal Mg(s) + Cu. SO 4(aq) Mg. SO 4 (aq) + Cu (s)

Activity Series Activity series is a list of elements organized according to their chemical reactivity. Metals higher up on the activity series list will replace metals lower on the list during a single displacement reaction.

Solving a Single Displacement Reaction Complete the following chemical reaction: Sn. Cl 4 (aq) + Al (s) STEP 1: Determine which element is the metal and which is the non-metal. Reference the activity series. (only switch the metal elements if it is higher on the activity series). STEP 2: Combine the singe metal with the non metal and do the cross over method. (make sure you determine the charge of each element first. )

Solving a Single Displacement Reaction Complete the following chemical reaction: Sn. Cl 4 (aq) + Al (s) STEP 3: Write the skeletal equation. STEP 4: Write a balanced chemical equation. (make sure to double check that both the reactants and products are balanced)

LET’S PRACTICE ! Complete and balance the following single displacement reactions. Make sure to refer to the activity series. 1)Cu. F 2 (aq) + Mg (s) 2) Cu (s) + HCl (aq) 3) Au(NO 3)3 (aq) + Ag (s) 4) Al (s) + Fe 2 O 3

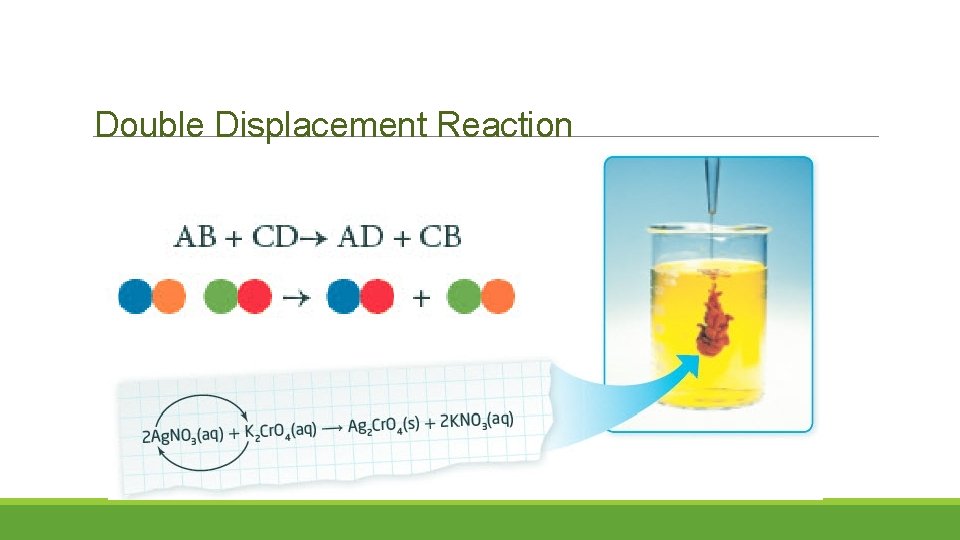
Double Displacement Reaction Double displacement reaction:

Double Displacement Reaction

Double Displacement Reaction Sulfur dioxide (SO 2) is used to preserve the colour of dried fruit. A double displacement reaction followed by a decomposition reaction releases the sulfur dioxide gas required for the process. The double replacement reaction Na 2 SO 3(aq) + 2 HCl(aq) 2 Na. Cl (aq) + H 2 SO 3(aq) The decomposition reaction H 2 SO 3(aq) H 2 O(l) + SO 2(g)

Solving a Double Displacement Reaction Complete the following chemical reaction: Pb(NO 3)2 (aq) + KI (s) STEP 1: Do the reverse cross-over method to determine the charge of each element. (reference the periodic table as well). STEP 2: Switch the non-metals and metals with one another (cations and anions) and write the skeletal equation.

Solving a Double Displacement Reaction Complete the following chemical reaction: Pb(NO 3)2 (aq) + KI (s) STEP 3: Write the balanced chemical equation and double check that the reactants and products are balanced.

LET’S PRACTICE ! Complete and balance the following double displacement reactions. 1) Sr. Cl 2 (aq) + Pb(NO 3)2 (aq) 2) Al. Cl 3 (aq) + Cu. NO 3 (aq) 3) KCl (aq) + Ag. NO 3 (aq) 4) Ca. I 2 (aq) + Na 2 CO 3(aq)

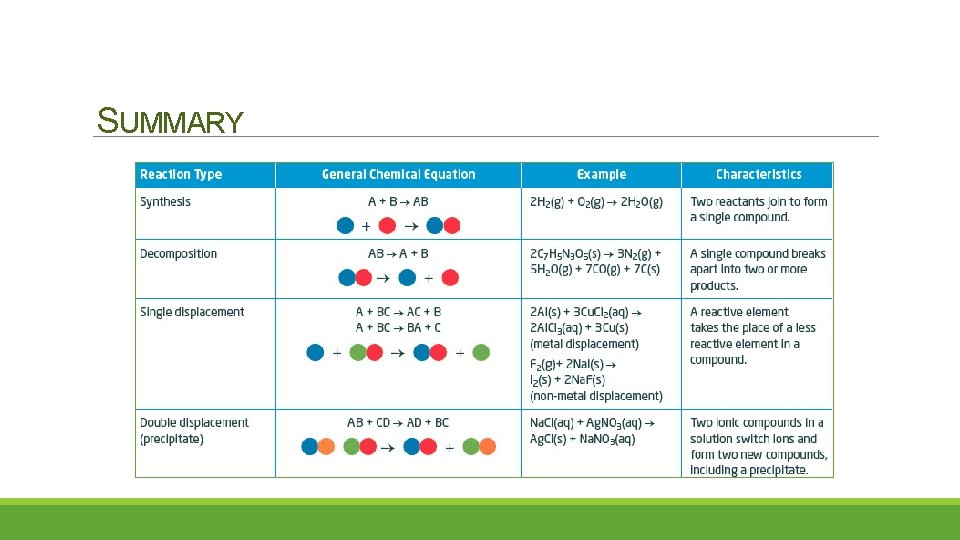
SUMMARY

HOMEWORK • Complete the worksheets given in class. • p. 198 # 2, 3, 5, 6 & 8