Chapter 4 Cosmogenic Nuclides Table 4 1 Data

Chapter 4

Cosmogenic Nuclides Table 4. 1. Data on Cosmogenic Nuclides Nuclide Half life, yrs λ, yr 1 14 C 5730 1. 21 x 10 3 3 H 12. 33 5. 62 x 10 2 10 Be 1. 50 × 106 4. 64 x 10 7 26 Al 7. 16 × 105 9. 68 x 10 5 36 Cl 3. 08 × 105 2. 25 x 10 6 32 Si 276 2. 51 x 10 2

Cosmic Rays Cosmic rays are high energy (several Ge. V up to 1019 e. V) atomic nuclei, mainly H &, but nuclei of all the elements have been recognized. CERN’s Large Hadron Collider, mankind’s most powerful accelerator, produces energies of ~1013 e. V (10 Ge. V) A significant fraction of cosmic rays originates in he Sun, although these are mainly of energies too low to generate cosmogenic nuclides. The origin of the remainder is unclear; most likely originate in supernovae or similar high energy environments in the cosmos. The primary reaction that occurs when cosmic rays encounter the Earth is spallation, in which a nucleus struck by a high energy particle shatters into two or more pieces, including stable and unstable nuclei, as well as protons and neutrons. Short lived particles such as muons, pions, etc. are also created. The interaction of a cosmic ray with a nucleus sets of a chain reaction of sorts as the secondary particles and nuclear fragments, which themselves have very high energies, then strike other nuclei producing additional reactions of lower energy.

14 C Carbon 14 is by far the most familiar and useful of the cosmogenic dating schemes. Its usefulness results from its relatively short half life, a relatively high production rate, and the concentration of carbon in biological material. 14 C produced primarily by reactions with secondary particles, mainly by the 14 N(n, p)14 C reaction involving relatively slow neutrons. The high abundance of the target nuclide is part of the reason for the high production rate.

Analytical Techniques The traditional method of 14 C determination is counting of the β rays produced in its decay. Gas proportional counter. Carbon is converted to CO 2 and placed in a metal tube with a negatively charged wire (the anode) running through it. The emitted beta strips electrons from atoms which drift to the wire (the anode), producing a measurable pulse of current. In liquid scintillation counting, The carbon is converted into CO 2 and then to benzene (C 6 H 6) and mixed with a liquid scintillator (an organic liquid). β particle causes the scintillator to give off a photon, which is then detected with a photomultiplier, and the resulting electrical pulse sent to a counter. This technique is more sensitive and has largely replaced gas counters. Requires ~1 g carbon. Accelerator Mass Spectrometry. The low abundance of 14 C compared to stable C isotopes (14 C/12 C ~ 10– 12) requires very high resolution to avoid interference with those isotopes as well as others such as 14 N. That requires the ions be accelerated to high energies (Me. V rather than ke. V of conventional mass spectrometers). In the last few decades, dedicated instruments have been built for this purpose. Requires only a few milligrams C.

14 C Dating Reported in units of Beq/g C (counts per minute), known as specific activity. Equation of interest is: where N 0 is the atmospheric value of 13. 56 Beq/g. By historical convention, “radiocarbon ages” are reported in years before 1950 (the year of the first 14 C age determination; the abbreviation BP thus means not before present, but before 1950) and assuming a half life of 5568 years (instead of the currently accepted value of 5730 years). Partly, this is a consequence of atmospheric nuclear weapons tests in the ‘ 50’s, which contaminated the atmosphere with 14 C.

Cosmic Ray Flux Cosmogenic nuclide production affected by geomagnetic field, which deflects cosmic rays. Cosmogenic nuclide production increases toward the poles (due to magnetic field influence on cosmic rays), but atmosphere is well mixed for CO 2. Does affect other nuclides. Field and production rate has varied with time. The solar wind modulates the solar magnetosphere, which deflects galactic cosmic rays at the outer edge of the solar system (the heliopause). Varies with the sunspot cycle and perhaps on longer time scales: so called de. Vries Effects.
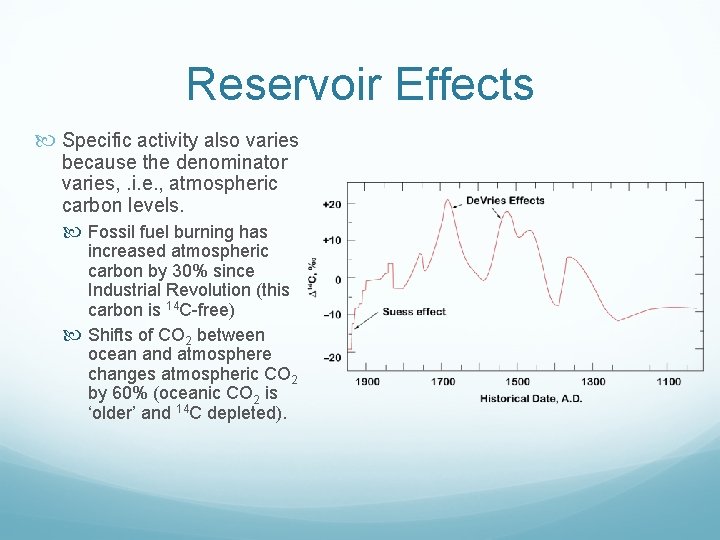
Reservoir Effects Specific activity also varies because the denominator varies, . i. e. , atmospheric carbon levels. Fossil fuel burning has increased atmospheric carbon by 30% since Industrial Revolution (this carbon is 14 C free) Shifts of CO 2 between ocean and atmosphere changes atmospheric CO 2 by 60% (oceanic CO 2 is ‘older’ and 14 C depleted).
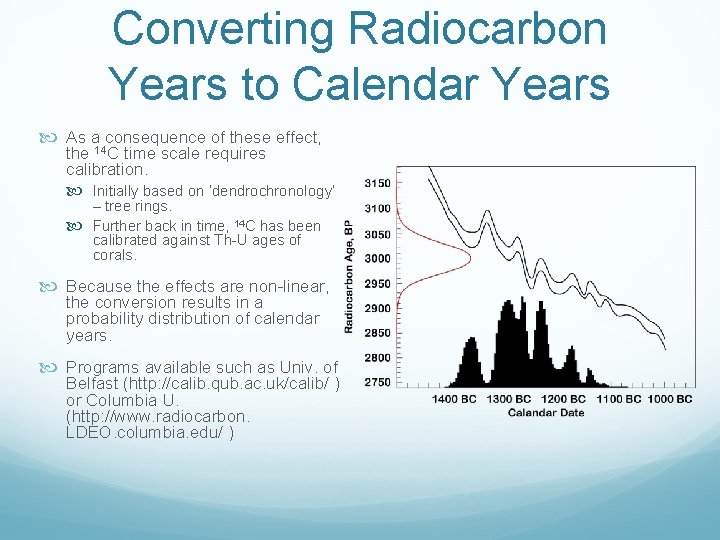
Converting Radiocarbon Years to Calendar Years As a consequence of these effect, the 14 C time scale requires calibration. Initially based on ‘dendrochronology’ – tree rings. Further back in time, 14 C has been calibrated against Th U ages of corals. Because the effects are non linear, the conversion results in a probability distribution of calendar years. Programs available such as Univ. of Belfast (http: //calib. qub. ac. uk/calib/ ) or Columbia U. (http: //www. radiocarbon. LDEO. columbia. edu/ )
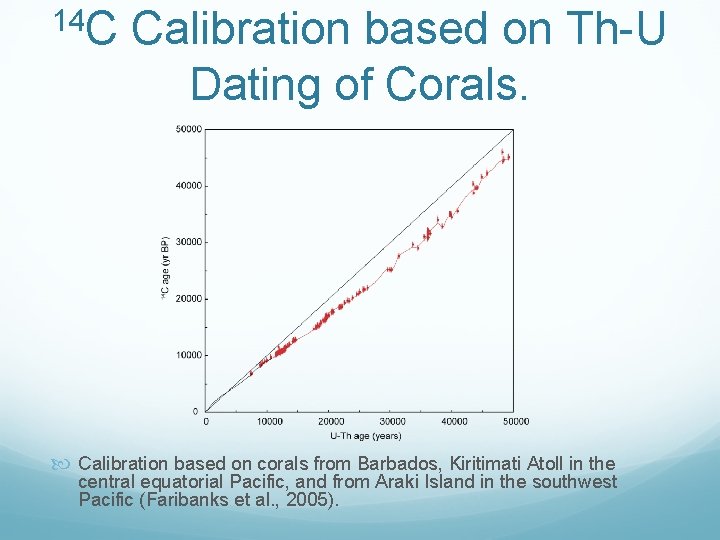
14 C Calibration based on Th U Dating of Corals. Calibration based on corals from Barbados, Kiritimati Atoll in the central equatorial Pacific, and from Araki Island in the southwest Pacific (Faribanks et al. , 2005).

10 Be, 26 Al, 36 Cl These nuclides have much longer half lives than 14 C and thus are be applied to longer time scale problems, such as Pleistocene chronology and dating of groundwater. 10 Be 26 Al is produced by spallation of 40 Ar 36 Cl is produced by 40 Ar(p, α)36 Cl reactions (probably mainly with secondary protons). is created by spallation reactions between cosmic rays and N and O nuclei. Production Rates 10 Be 10 2 – 10 3 atoms/cm 2/sec 26 Al and 36 Cl 10 5– 10 6 atoms/cm 2/sec Unlike 14 C, all are quickly washed out of the atmosphere. There are latitudinal variations in production and abundance. Abundance of 10 Be in marine sedimentation is, however, uniform due to mixing in oceans.

10 Be Dating 10 Be mainly used for dating sediments (some tectonic/high T application as well later). Begin with the equation: assuming a constant flux, ƒ, to the sediment: where brackets denote concentration Sedimentation rate, s, however, may vary, so: sedimentation rate is related to time and depth as z = s ×t
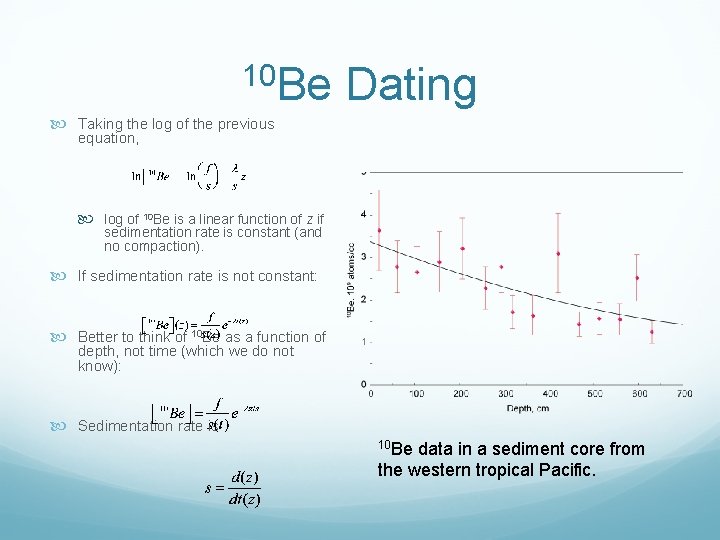
10 Be Dating Taking the log of the previous equation, log of 10 Be is a linear function of z if sedimentation rate is constant (and no compaction). If sedimentation rate is not constant: Better to think of 10 Be as a function of depth, not time (which we do not know): Sedimentation rate is 10 Be data in a sediment core from the western tropical Pacific.

10 Be Dating substituting for s and integrating: Solving for t(z): The integral term is simply the sum of all 10 Be above depth z. (Some similarity to constant flux model of 210 Pb) This assumes a constant production rate (it is not constant). If we know the production rate has varied in time, we can substitute ƒ(t) for ƒ in the above equation.

36 Cl 14 C 36 Cl Dating in Hydrology can be used for groundwater dating, but C is not conservative (respiration, dissolution, precipitation of carbonate). by contrast is nearly conservative in groundwater. Stable isotopes of Cl are 35 Cl and 37 Cl. 36 Cl produced by cosmogenic reactions on Ar in the atmosphere, hence there is a latitudinal dependence. Quickly washed out by rain, unlike C. Activity usually ratioed to total Cl.

Nucleogenic Production Complicating matters, there is some production of 36 Cl in the Earth by 35 Cl capture of fission derived neutrons. This nucleogenic 36 Cl will build up over time: where Φn is the neutron flux, ƒ is the capture cross section and [35 Cl] is the concentration of 35 Cl. So how will 36 Cl vary with time according to this equation? Will eventually reach secular equilibrium:

36 Cl Dating “Age” of groundwater can be determined from But groundwater in a given location could have multiple sources of multiple ages. So we need to know the initial ratio and secular equilibrium ratios as well. Evaporation, mixing with formation brines, dissolution of salt, etc. can all complicate age.
Great Artesian Aquifer of Australia
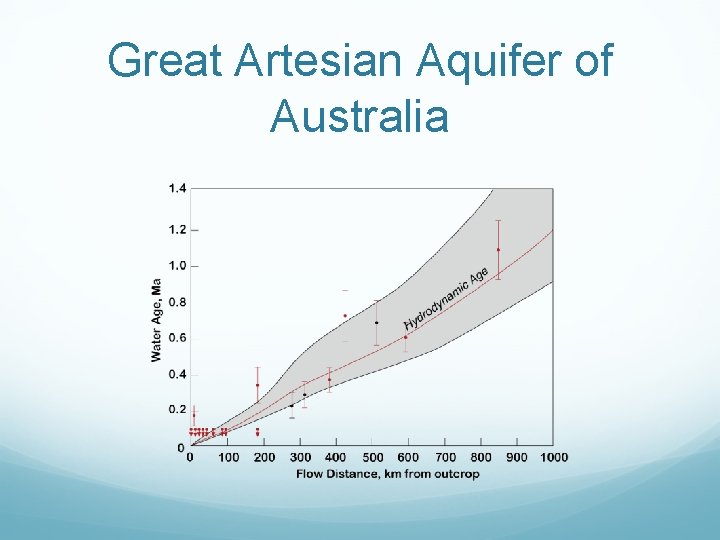
Great Artesian Aquifer of Australia

4 He dating of groundwater produced by α decay in rock Assuming water flows by piston flow, accumulation of 4 He will be: where Rα is the production rate, and ϕ is porosity. Complication: high diffusivity of He
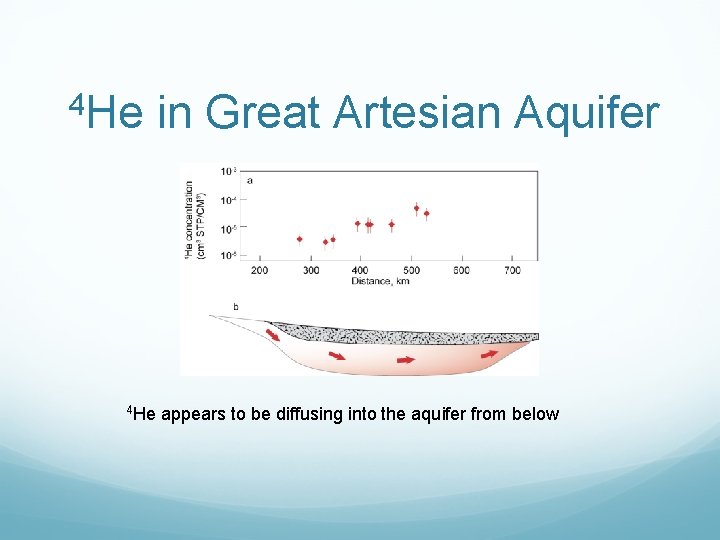
4 He in Great Artesian Aquifer appears to be diffusing into the aquifer from below

In Situ Produced Cosmogenic Nuclides Cosmic ray spallation produces both stable and unstable nuclides. Of the stable ones, only those that are very rare of interest

In Situ Production Some cosmic rays manage to penetrate to the Earth’s the surface. Flux then decreases exponentially as: where z is depth, ρ is density and l is a constant that depends on the nature of the particles, their energy, and nature (composition) of materials. l is ~160 g/cm 2, most rock has ρ~2. 5 g/cc, so flux would decrease to 1/e or 0. 36 at a depth of 64 cm. Muons penetrate more effectively (l~1000 g/cm 2, but interact weakly with matter).

3 He dating of surfaces and erosion rates Production of a stable nuclide at the surface will be N = Pt (Abundance increases linearly with time, assuming constant production rate). 3 He accumulation has been used to date lava flow surfaces. P is a function of both latitude and altitude. The concentration as a function of time and depth will be Of course if depth is constant, this integrates simply to: Now suppose that depth varies as a result of erosion occurring at rate ε. Depth is then a function of time:

3 He dating of surfaces and erosion rates Substituting for z and integrating: Now substituting z 0 = z + εt, at the surface (z=0) this reduces to Cannotbe solved directly. However, if the age of the rock, t, is known from some other method, one can measure a series of values, C, as function of depth and make a series of guesses of the erosion rate until a curve based on this equation fits the data. Using this procedure, Kurz estimated an erosion rate of 10 m/Ma for Haleakala (for higher erosion rates, it would be necessary to take account of the muon produced 3 He).

Erosion Ages from Unstable Cosmogenic Nuclides For a nuclide such as 26 Al or 36 Cl, need to take account of its decay. The concentration as a function of time and depth will be: After many half lives the concentration at the surface of eroding rock will be: While the concentration is steady state, it does depend on the erosion rate, which we can easily solve for.

Exposure Ages with Cosmogenic Nuclides Concentration of unstable cosmogenic nuclide at the surface will change with time as: Integrating What happens eventually?

36 Cl Dating of Moraine Boulders in the Sierra Nevada
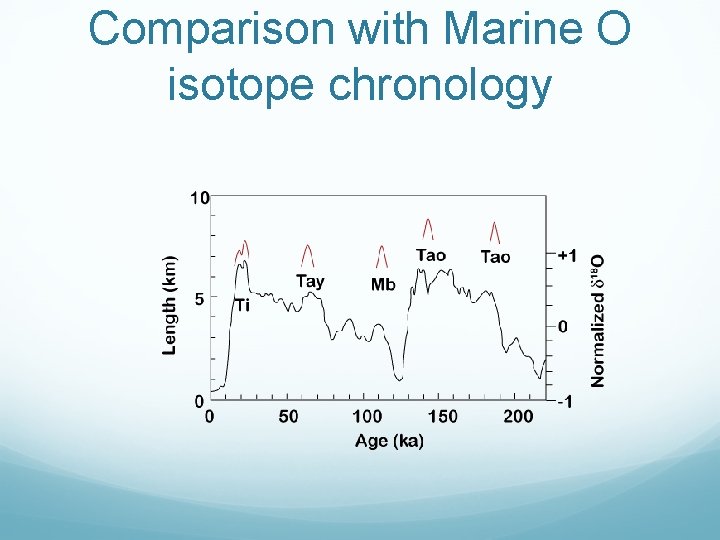
Comparison with Marine O isotope chronology

Fission Track Dating

Fission Track Dating A small fraction of 238 U decay through fission rather than α decay. λƒ = 8. 46 x 10 17 yr 1. Fission produces two unequal nuclear fragments (plus some neutrons). Some of the decay energy converted to kinetic energy of the fragments, which then fly through the crystal lattice, damaging it. The lattice damage can be enhanced by acid leaching so it is visible under the microscope. Number fission tracks is a function of time: where λƒ/λα = 5 x 10– 7. Note that λƒ+λα ≈ λα Tracks anneal at a temperature dependent rate, with “closure T” being fairly low, depending on mineral.

Analytical Tracks must intersect surface to be counted so the track density, so density will be: q is fraction intersecting surface U concentration is typically determined by irradiating the sample with neutrons and inducing fission of 235 U; number of induced tracks is where ϕ is thermal neutron dose and σ is reaction cross section.

Zeta Method In practice, sample is etched, tracks counted, then a “detector” placed on sample and package is irradiated with neutrons. Induced tracks counted in detector. Age is related to track density as: Neutrons induce fission of 235 U, producing additional tracks. Where f is the neutron “dose” and s is the reaction cross section. In practice, we irradiate a standard of known age, and pile the various constants into the zeta factor, ζ: where ρd is the track density in a U doped standard. similar to 40 Ar/39 Ar, zeta determined by analyzing standards. Rearranging and solving for t:
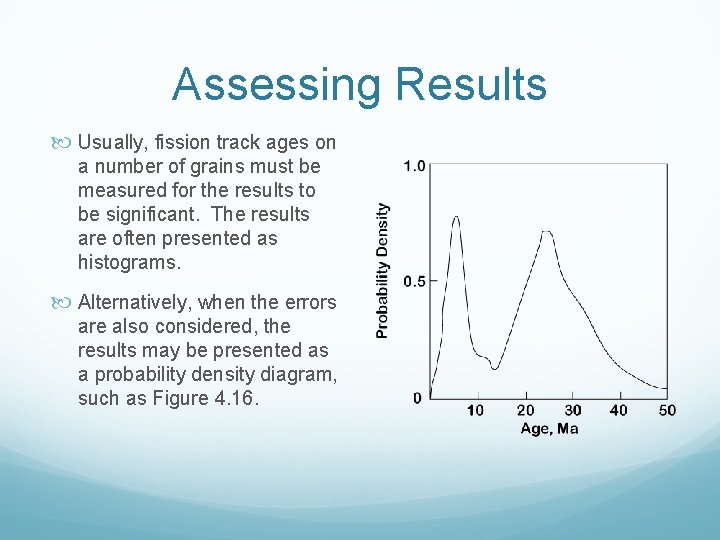
Assessing Results Usually, fission track ages on a number of grains must be measured for the results to be significant. The results are often presented as histograms. Alternatively, when the errors are also considered, the results may be presented as a probability density diagram, such as Figure 4. 16.
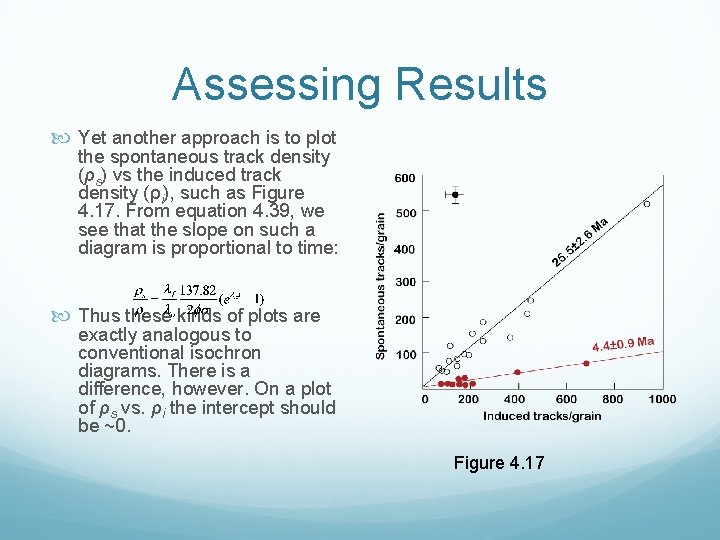
Assessing Results Yet another approach is to plot the spontaneous track density (ρs) vs the induced track density (ρi), such as Figure 4. 17. From equation 4. 39, we see that the slope on such a diagram is proportional to time: Thus these kinds of plots are exactly analogous to conventional isochron diagrams. There is a difference, however. On a plot of ρs vs. ρi the intercept should be ~0. Figure 4. 17
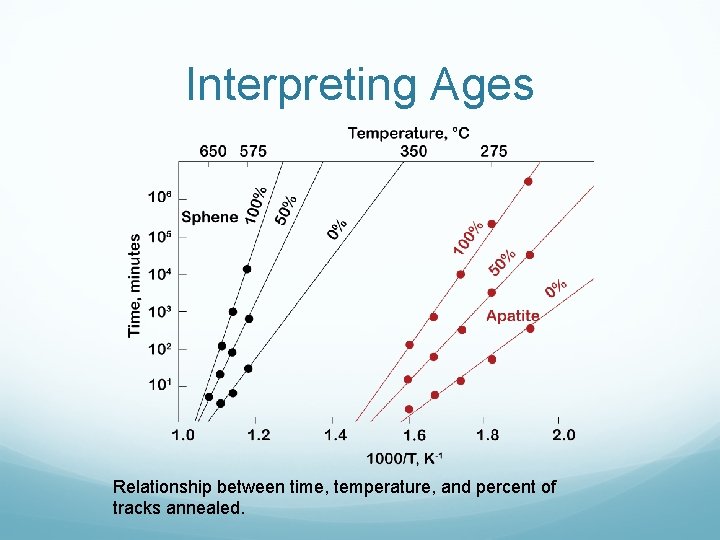
Interpreting Ages Relationship between time, temperature, and percent of tracks annealed.
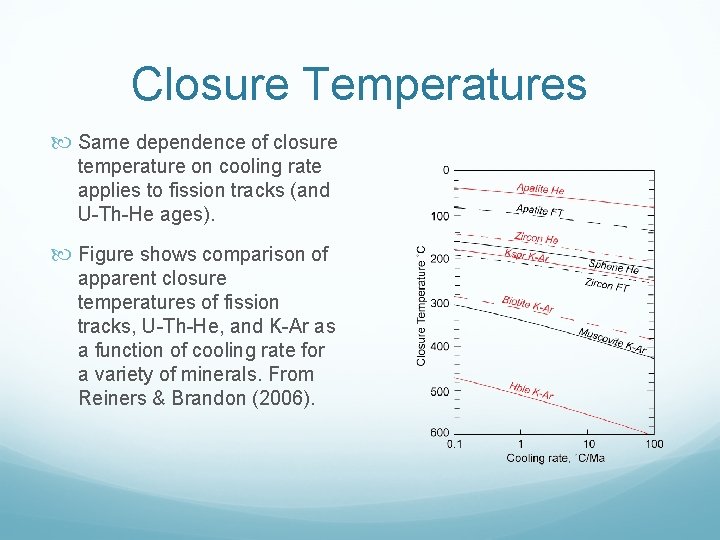
Closure Temperatures Same dependence of closure temperature on cooling rate applies to fission tracks (and U Th He ages). Figure shows comparison of apparent closure temperatures of fission tracks, U Th He, and K Ar as a function of cooling rate for a variety of minerals. From Reiners & Brandon (2006).

Uplift Rates As mountains respond to erosion by uplift, underlying rock cools as it approaches the surface so that temperature at depth z will be: where Ts is the surface temperature and TL is the temperature of the base of a layer of thickness L, HT is the radiogenic heating (˚C/Ma), k is thermal conductivity and ε is erosion rate. If erosion rates are high, geothermal gradient can be non linear.
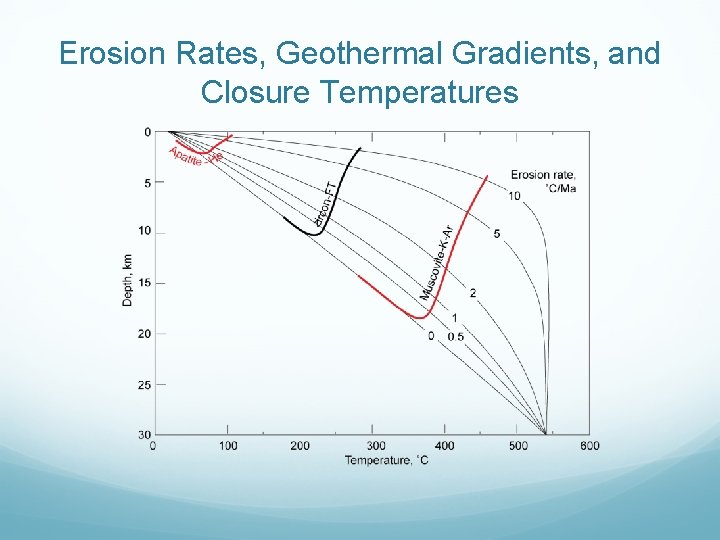
Erosion Rates, Geothermal Gradients, and Closure Temperatures
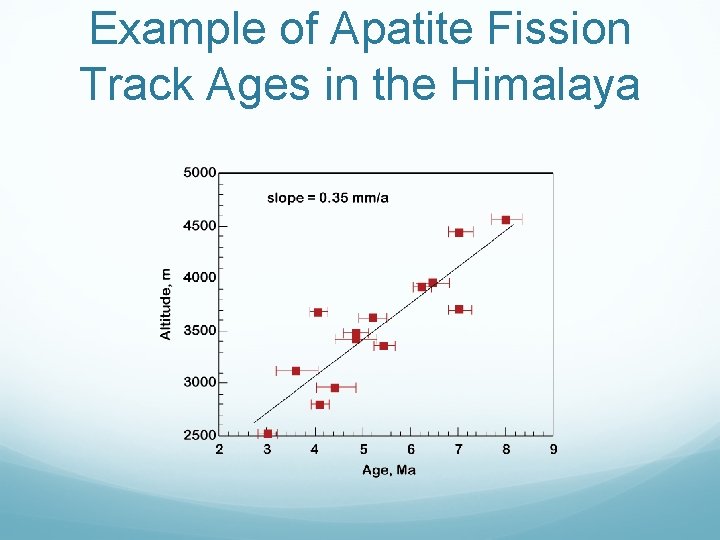
Example of Apatite Fission Track Ages in the Himalaya
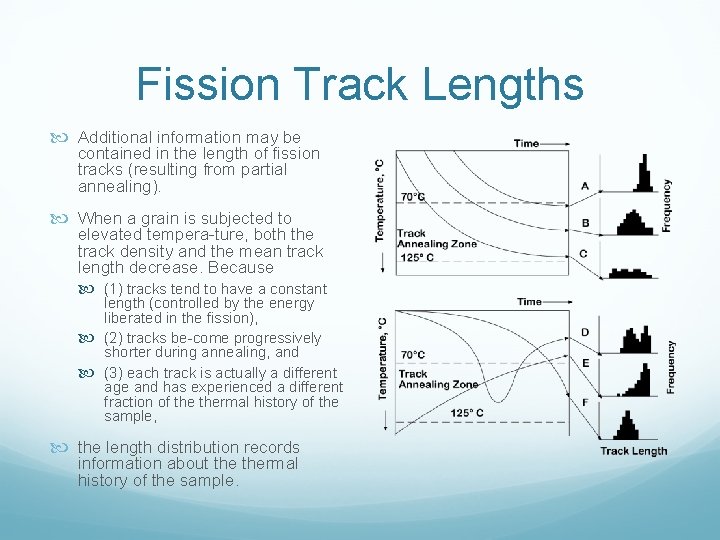
Fission Track Lengths Additional information may be contained in the length of fission tracks (resulting from partial annealing). When a grain is subjected to elevated tempera ture, both the track density and the mean track length decrease. Because (1) tracks tend to have a constant length (controlled by the energy liberated in the fission), (2) tracks be come progressively shorter during annealing, and (3) each track is actually a different age and has experienced a different fraction of thermal history of the sample, the length distribution records information about thermal history of the sample.

Mass Spectrometry Quick Overview
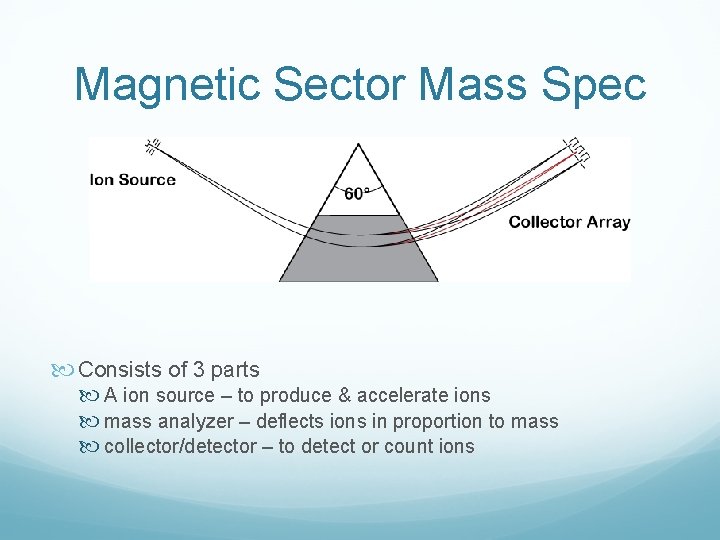
Magnetic Sector Mass Spec Consists of 3 parts A ion source – to produce & accelerate ions mass analyzer – deflects ions in proportion to mass collector/detector – to detect or count ions

Ion Source Ions can be produced by thermal ionization collisions with electrons produced by a hot filament plasma secondary ionization laser ionization

Mass Analyzer A charged particle moving in a magnetic experiences a force F = qv × B A. 1 Since the force is always di rect ed perpendicular to the direction of motion, the particle begins to move in a circular path. The motion is thus much like swinging a ball at the end of a string, and we can use equation for a centripetal force Equating: The velocity of the particle can be determined from its energy, which is the accelerating potential, V, times the charge: Substituting: We select for mass by adjusting B Typical value of r for a thermal ionization mass spec is 54 cm.
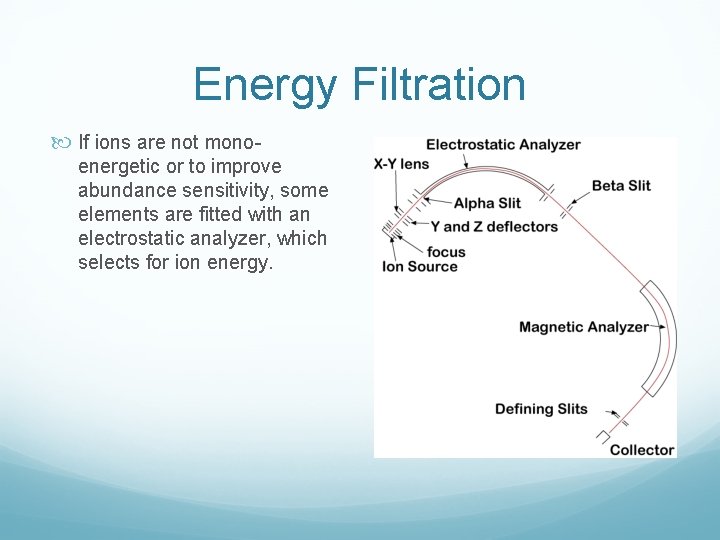
Energy Filtration If ions are not mono energetic or to improve abundance sensitivity, some elements are fitted with an electrostatic analyzer, which selects for ion energy.
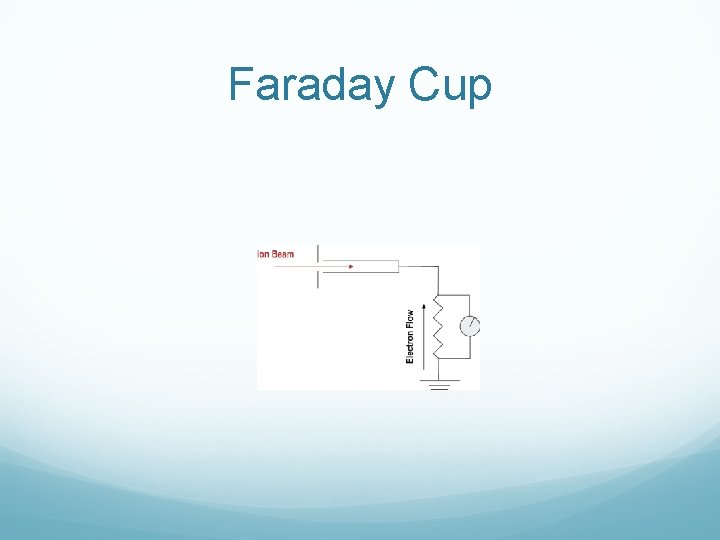
Faraday Cup

Electron Multiplier Ion beam strikes a charged surface producing a cascade of electrons. Can be used in either analog or ion counting modes.

Daly Detector
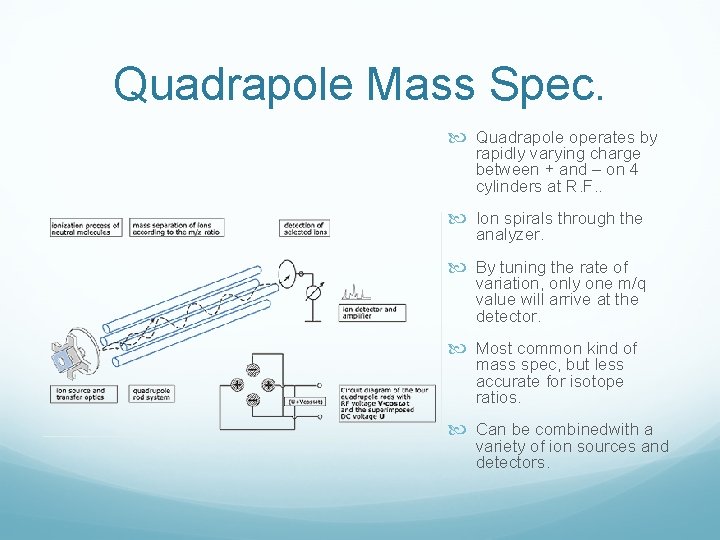
Quadrapole Mass Spec. Quadrapole operates by rapidly varying charge between + and – on 4 cylinders at R. F. . Ion spirals through the analyzer. By tuning the rate of variation, only one m/q value will arrive at the detector. Most common kind of mass spec, but less accurate for isotope ratios. Can be combinedwith a variety of ion sources and detectors.

Accelerator Mass Spec.

Isotope Dilution
- Slides: 52