Chapter 4 Carbon the Molecular Diversity of Life

Chapter 4~ Carbon & the Molecular Diversity of Life Slide shows combined and modified from: http: //gbs. glenbrook. k 12. il. us/Academics/gbssci/bio/apbio/Lecture/lecture. htm; http: //www. explorebiology. com/


• Organic chemistry is the study of CARBON compounds • Can form FOUR stable covalent bonds at same time (=tetravalence) • Common partners = O, H, N
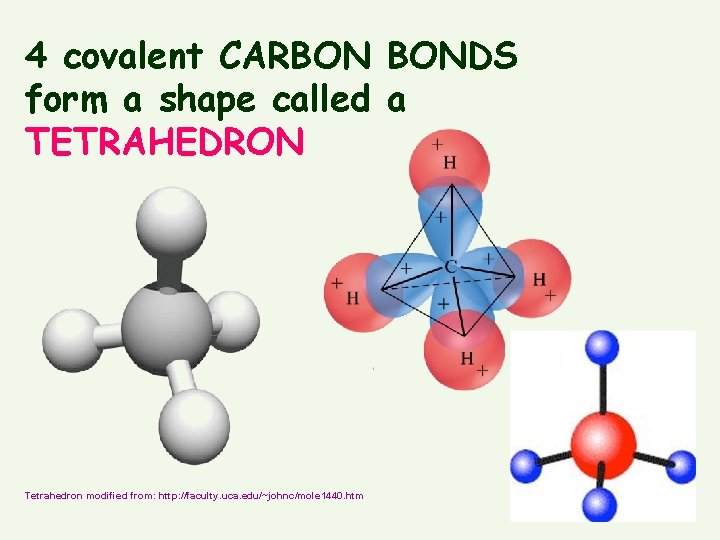
4 covalent CARBON BONDS form a shape called a TETRAHEDRON Tetrahedron modified from: http: //faculty. uca. edu/~johnc/mole 1440. htm
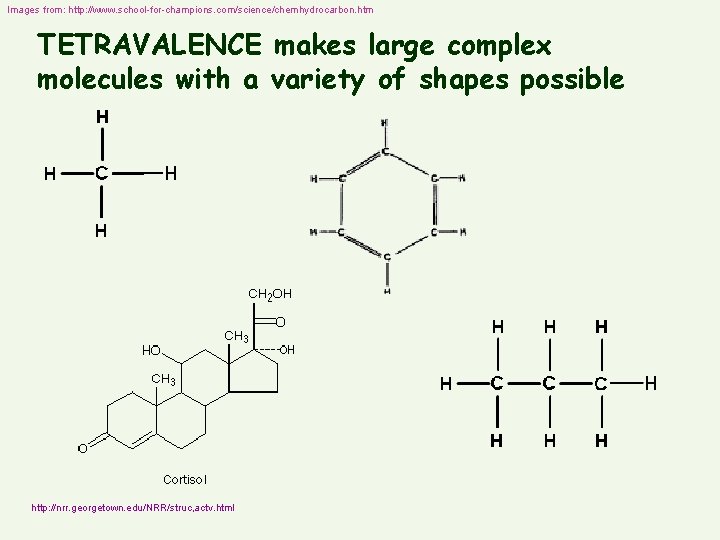
Images from: http: //www. school-for-champions. com/science/chemhydrocarbon. htm TETRAVALENCE makes large complex molecules with a variety of shapes possible http: //nrr. georgetown. edu/NRR/struc, actv. html
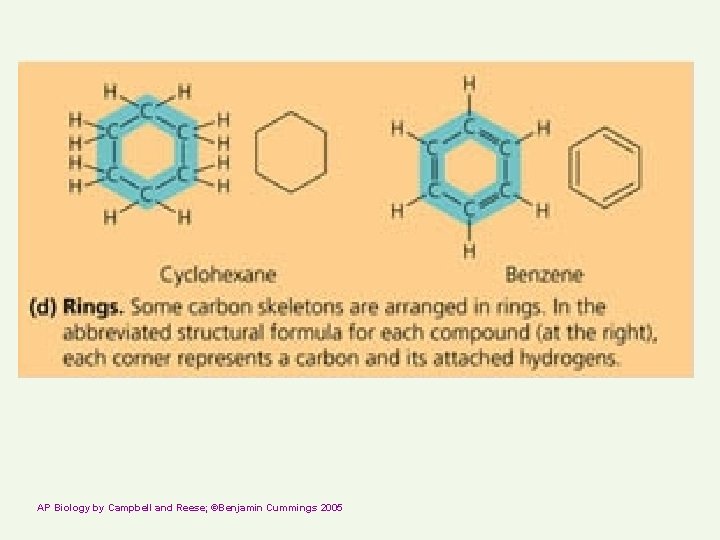
AP Biology by Campbell and Reese; ©Benjamin Cummings 2005

Hydrocarbons • Only carbon & hydrogen (Ex: petroleum; lipid ‘tails’) • Covalent bonding; • nonpolar • High energy storage http: //www. world-petroleum. org/education/petref/index. html

Carbon compounds Skeleton may have single or double bonds http: //telstar. ote. cmu. edu/Hughes/tutorial/cellmembranes/
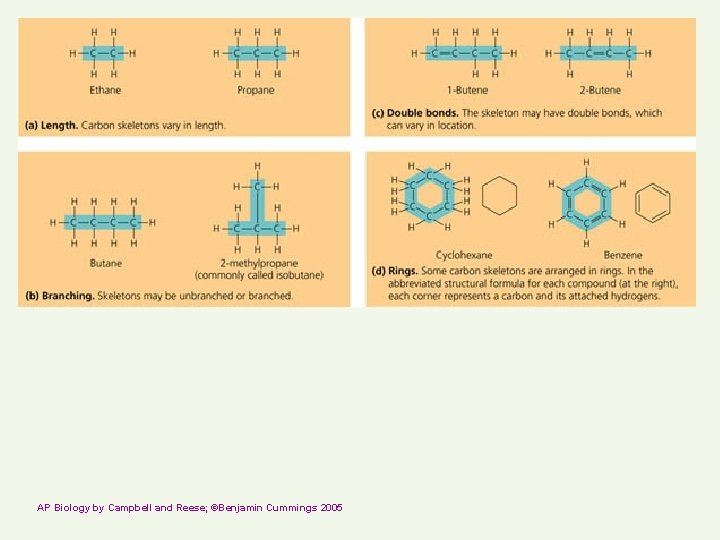
AP Biology by Campbell and Reese; ©Benjamin Cummings 2005

ISOMERScompounds that have the same number of atoms but different structures
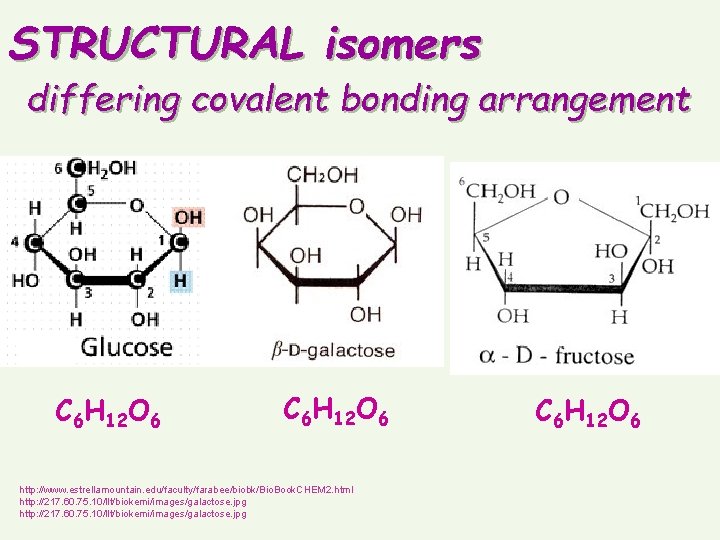
STRUCTURAL isomers differing covalent bonding arrangement C 6 H 12 O 6 http: //www. estrellamountain. edu/faculty/farabee/biobk/Bio. Book. CHEM 2. html http: //217. 60. 75. 10/llt/biokemi/images/galactose. jpg C 6 H 12 O 6
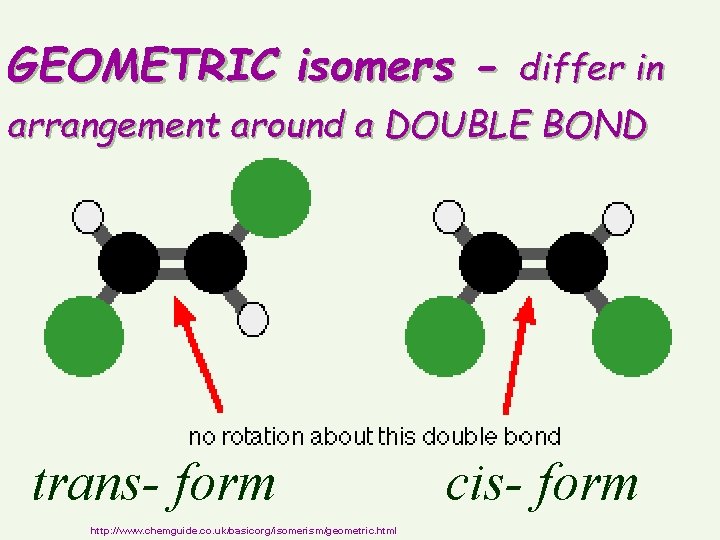
GEOMETRIC isomers - differ in arrangement around a DOUBLE BOND trans- form http: //www. chemguide. co. uk/basicorg/isomerism/geometric. html cis- form
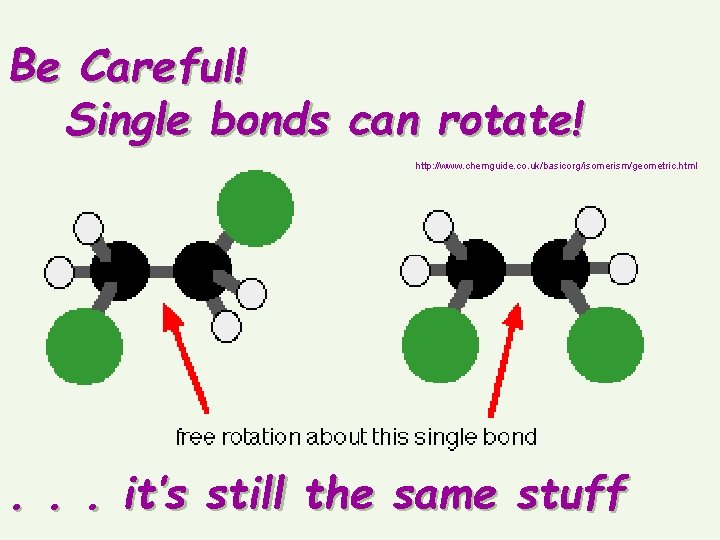
Be Careful! Single bonds can rotate! http: //www. chemguide. co. uk/basicorg/isomerism/geometric. html . . . it’s still the same stuff
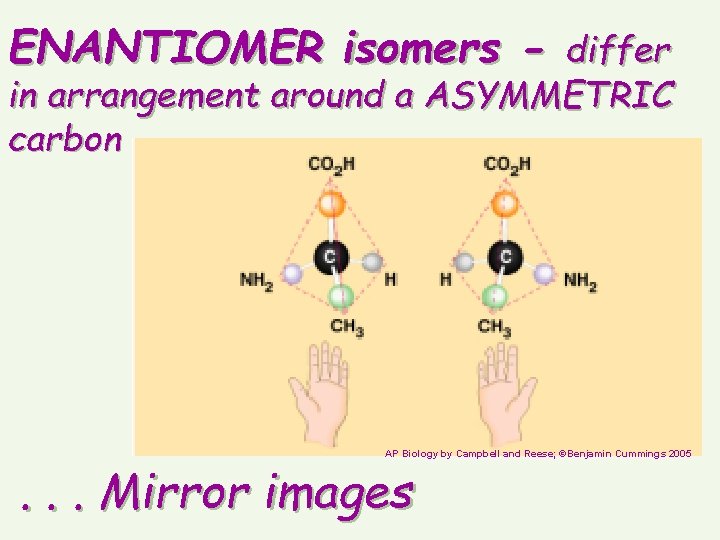
ENANTIOMER isomers - differ in arrangement around a ASYMMETRIC carbon AP Biology by Campbell and Reese; ©Benjamin Cummings 2005 . . . Mirror images




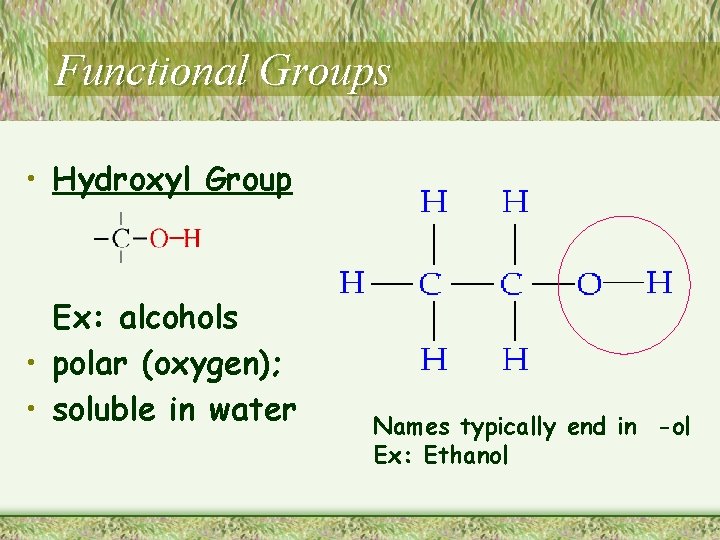
Functional Groups • Hydroxyl Group Ex: alcohols • polar (oxygen); • soluble in water Names typically end in -ol Ex: Ethanol

Functional Groups • Carbonyl Group KETONE: within carbon skeleton ALDEHYDE: at end of carbon skeleton
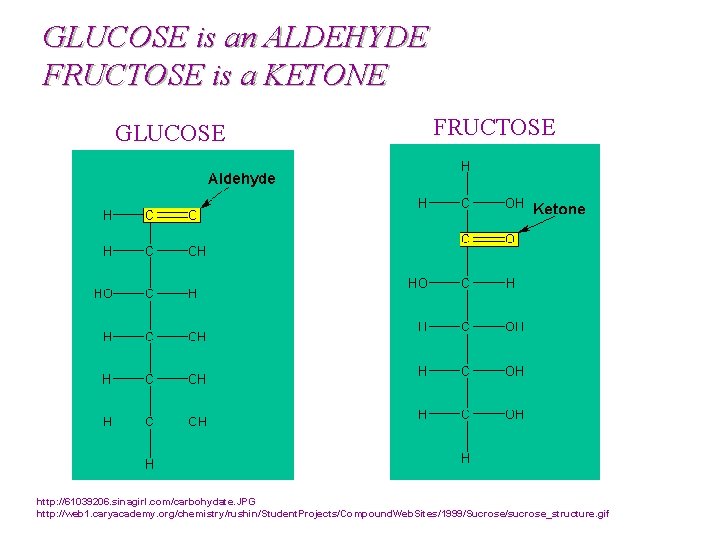
GLUCOSE is an ALDEHYDE FRUCTOSE is a KETONE GLUCOSE FRUCTOSE http: //61039206. sinagirl. com/carbohydate. JPG http: //web 1. caryacademy. org/chemistry/rushin/Student. Projects/Compound. Web. Sites/1999/Sucrose/sucrose_structure. gif
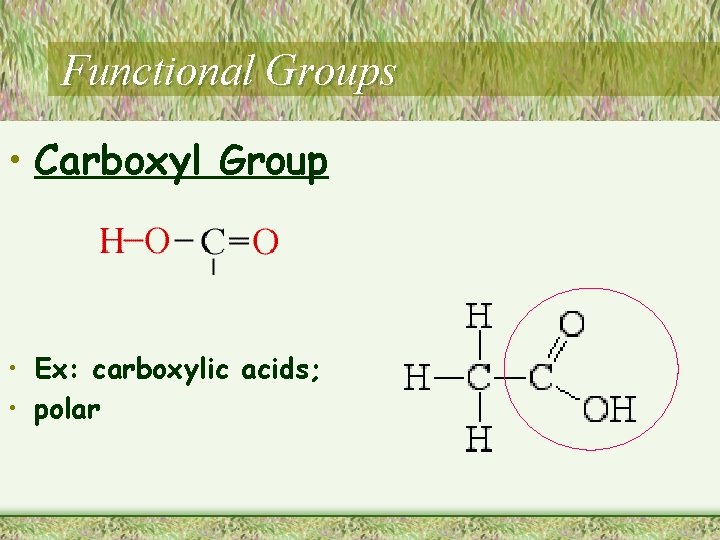
Functional Groups • Carboxyl Group • Ex: carboxylic acids; • polar
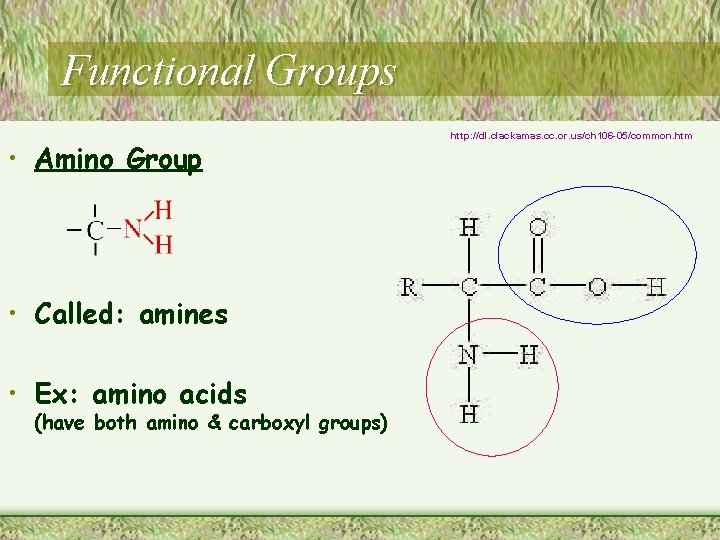
Functional Groups • Amino Group • Called: amines • Ex: amino acids (have both amino & carboxyl groups) http: //dl. clackamas. cc. or. us/ch 106 -05/common. htm

Functional Groups • Amino Group Can act as a base and pick up a H+ ion • Carboxyl Group can act as an acid and give up a H+ ion H | R -C -COO| +NH 3
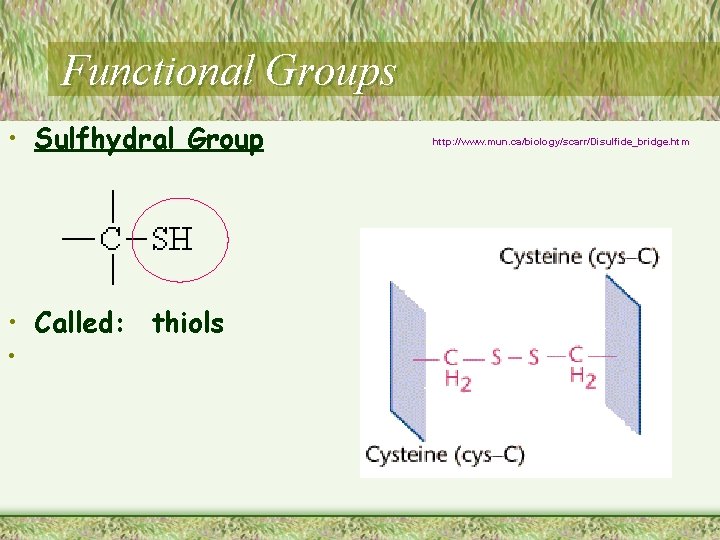
Functional Groups • Sulfhydral Group • Called: thiols • http: //www. mun. ca/biology/scarr/Disulfide_bridge. htm
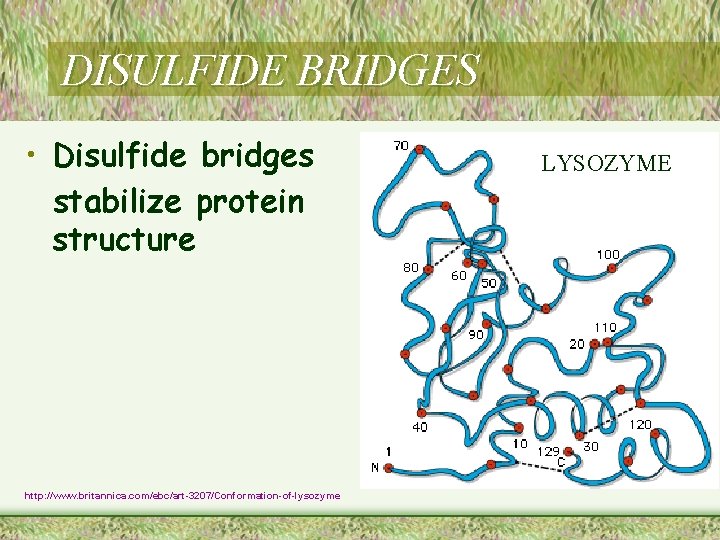
DISULFIDE BRIDGES • Disulfide bridges stabilize protein structure http: //www. britannica. com/ebc/art-3207/Conformation-of-lysozyme LYSOZYME
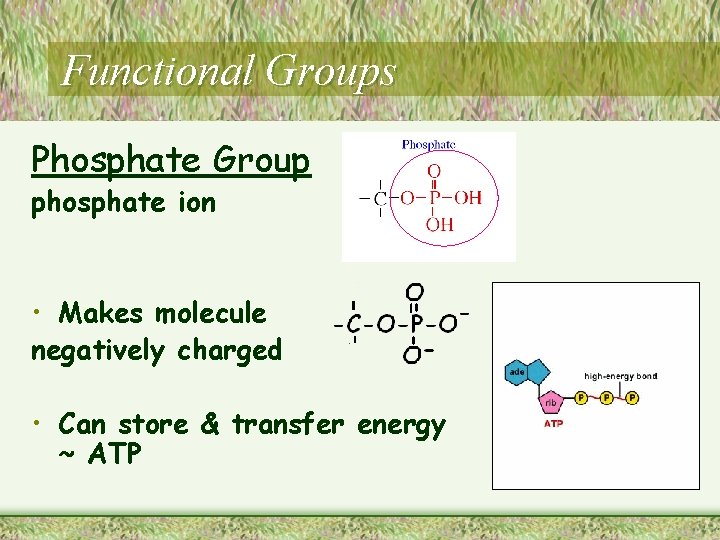
Functional Groups Phosphate Group phosphate ion • Makes molecule negatively charged • Can store & transfer energy ~ ATP
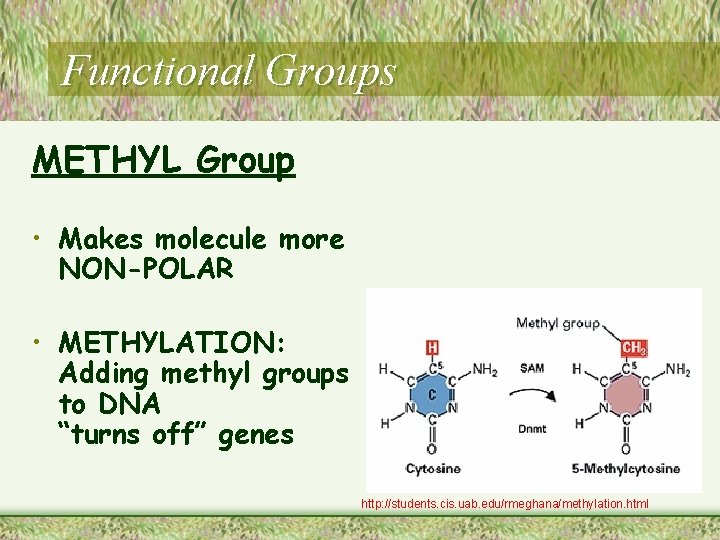
Functional Groups METHYL Group • Makes molecule more NON-POLAR • METHYLATION: Adding methyl groups to DNA “turns off” genes http: //students. cis. uab. edu/rmeghana/methylation. html
- Slides: 28