Chapter 4 Carbon and the Molecular Diversity of

Chapter 4 Carbon and the Molecular Diversity of Life Power. Point Lectures for Biology, Seventh Edition Neil Campbell and Jane Reece Lectures by Chris Romero Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Overview: Carbon—The Backbone of Biological Molecules • All living organisms – Are made up of chemicals based mostly on the element carbon Figure 4. 1 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Concept 4. 1: Organic chemistry is the study of carbon compounds • Organic compounds – Range from simple molecules to colossal ones Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Concept 4. 2: Carbon atoms can form diverse molecules by bonding to four other atoms Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

The Formation of Bonds with Carbon • Carbon has four valence electrons • This allows it to form four covalent bonds with a variety of atoms Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• The bonding versatility of carbon – Allows it to form many diverse molecules, including carbon skeletons Name and Comments Molecular Structural Formula H (a) Methane CH 4 H C H H (b) Ethane H H C 2 H 6 H C C H H H (c) Ethene Figure 4. 3 A-C (ethylene) C 2 H 4 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings H H C C H H Ball-and. Stick Model Space. Filling Model
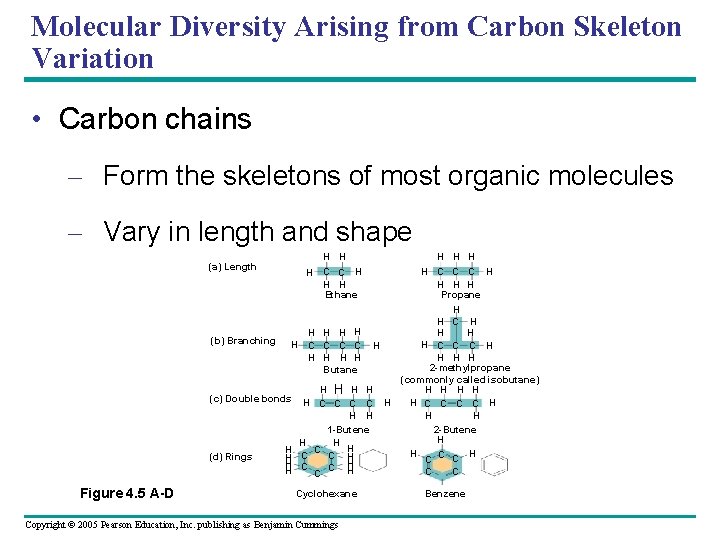
Molecular Diversity Arising from Carbon Skeleton Variation • Carbon chains – Form the skeletons of most organic molecules – Vary in length and shape (a) Length H H H C C H H H Ethane H H C C C H H Propane H H C H H H H (b) Branching H C C H H C C C H H H H 2 -methylpropane Butane (commonly called isobutane) H H H H (c) Double bonds H H C C C C H H H 1 -Butene 2 -Butene H H C C H (d) Rings H C C H H C C C Figure 4. 5 A-D Cyclohexane Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Benzene

Hydrocarbons • Hydrocarbons – Are molecules consisting of only carbon and hydrogen Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Hydrocarbons – Are found in many of a cell’s organic molecules Fat droplets (stained red) Figure 4. 6 A, B (a) A fat molecule Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings 100 µm (b) Mammalian adipose cells

Isomers • Isomers – Are molecules with the same molecular formula but different structures and properties Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
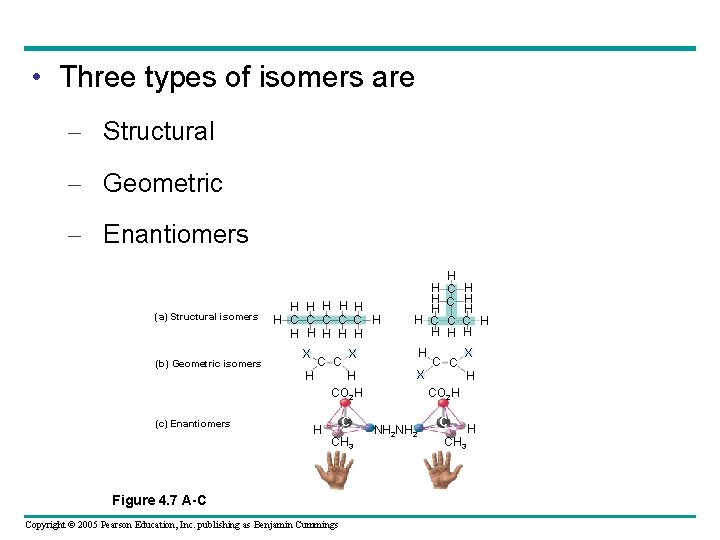
• Three types of isomers are – Structural – Geometric – Enantiomers (a) Structural isomers (b) Geometric isomers H H H C C C H H H X C C H (c) Enantiomers H H H C C C H H H X X H CO 2 H C CH 3 Figure 4. 7 A-C Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings C C X H CO 2 H NH 2 C CH 3 H
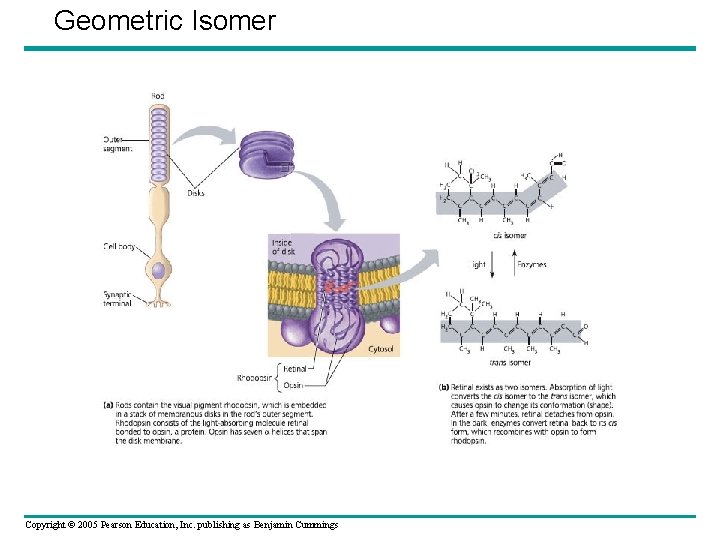
Geometric Isomer Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
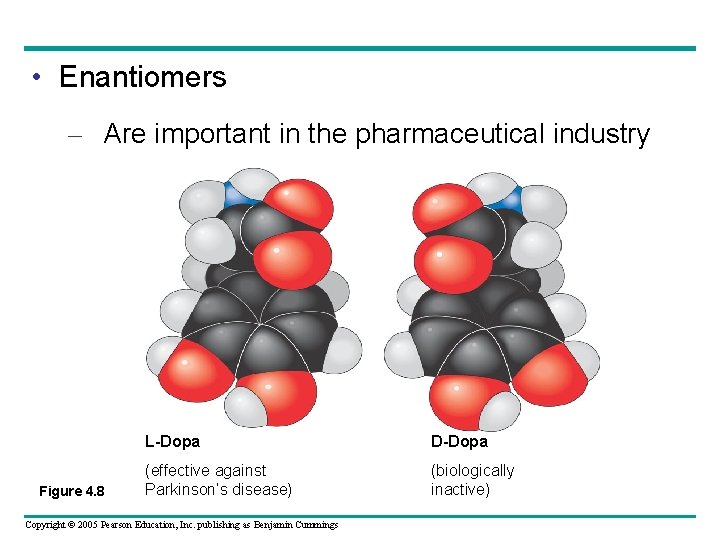
• Enantiomers – Are important in the pharmaceutical industry Figure 4. 8 L-Dopa D-Dopa (effective against Parkinson’s disease) (biologically inactive) Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

• Concept 4. 3: Functional groups are the parts of molecules involved in chemical reactions Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

The Functional Groups Most Important in the Chemistry of Life • Functional groups – Are the chemically reactive groups of atoms within an organic molecule Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

– Give organic molecules distinctive chemical properties Estradiol OH CH 3 HO Female lion OH CH 3 O Figure 4. 9 Male lion Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Testosterone

• Six functional groups are important in the chemistry of life – Hydroxyl – Carbonyl – Carboxyl – Amino – Sulfhydryl – Phosphate Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
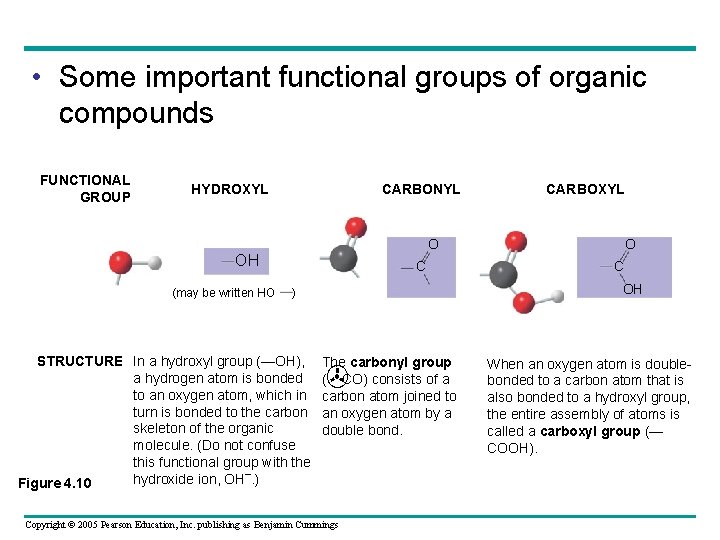
• Some important functional groups of organic compounds FUNCTIONAL GROUP HYDROXYL CARBONYL CARBOXYL O OH (may be written HO C C OH ) STRUCTURE In a hydroxyl group (—OH), a hydrogen atom is bonded to an oxygen atom, which in turn is bonded to the carbon skeleton of the organic molecule. (Do not confuse this functional group with the hydroxide ion, OH–. ) Figure 4. 10 O The carbonyl group ( CO) consists of a carbon atom joined to an oxygen atom by a double bond. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings When an oxygen atom is doublebonded to a carbon atom that is also bonded to a hydroxyl group, the entire assembly of atoms is called a carboxyl group (— COOH).
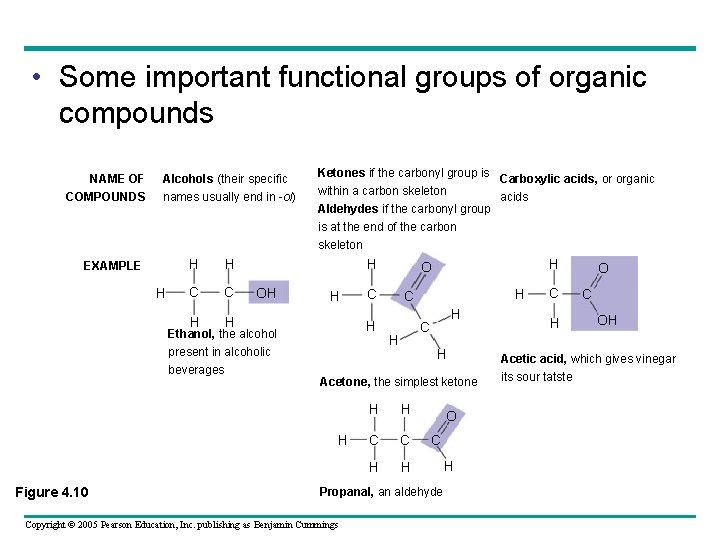
• Some important functional groups of organic compounds NAME OF COMPOUNDS Alcohols (their specific names usually end in -ol) Ketones if the carbonyl group is Carboxylic acids, or organic within a carbon skeleton acids Aldehydes if the carbonyl group is at the end of the carbon skeleton EXAMPLE H H H C C H H H OH H Ethanol, the alcohol present in alcoholic beverages H C H H Acetone, the simplest ketone H Figure 4. 10 C H H O H H C C H H O C Propanal, an aldehyde Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings H C H O C OH Acetic acid, which gives vinegar its sour tatste

• Some important functional groups of organic compounds FUNCTIONAL Is polar as a result of the PROPERTIES electronegative oxygen atom drawing electrons toward itself. Attracts water molecules, Has acidic properties because it is a source of hydrogen ions. The covalent bond between oxygen and hydrogen is so polar that hydrogen ions (H+) tend to dissociate reversibly; for example, A ketone and an aldehyde may be structural isomers with different properties, as is the case for acetone and propanal. helping dissolve organic compounds such as sugars (see Figure 5. 3). H H C H Figure 4. 10 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings H O C OH H C H O + H+ C O In cells, found in the ionic form, which is called a carboxylate group.
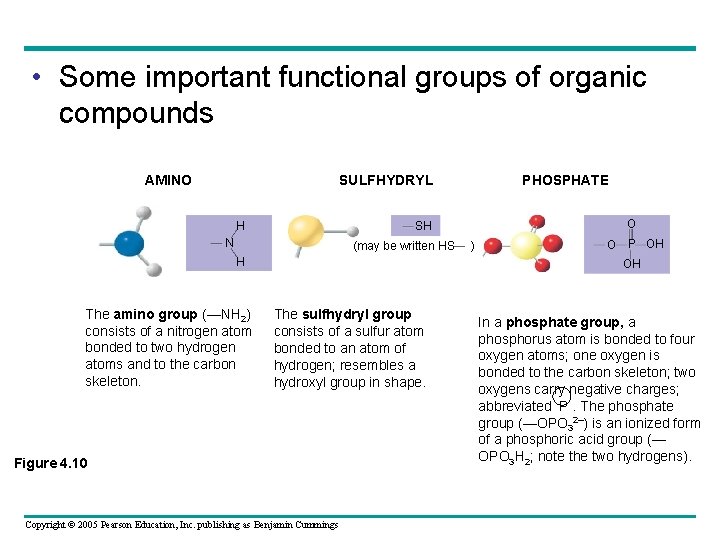
• Some important functional groups of organic compounds AMINO SULFHYDRYL H O SH N (may be written HS H The amino group (—NH 2) consists of a nitrogen atom bonded to two hydrogen atoms and to the carbon skeleton. PHOSPHATE ) O P OH OH The sulfhydryl group consists of a sulfur atom bonded to an atom of hydrogen; resembles a hydroxyl group in shape. Figure 4. 10 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings In a phosphate group, a phosphorus atom is bonded to four oxygen atoms; one oxygen is bonded to the carbon skeleton; two oxygens carry negative charges; abbreviated P. The phosphate group (—OPO 32–) is an ionized form of a phosphoric acid group (— OPO 3 H 2; note the two hydrogens).
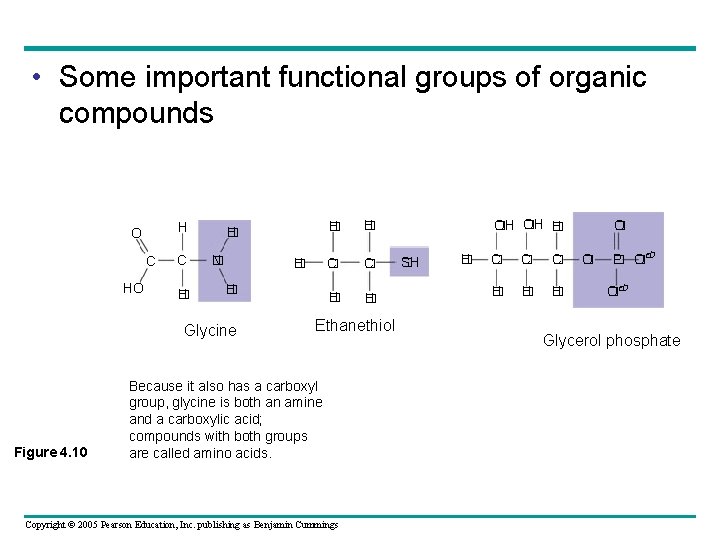
• Some important functional groups of organic compounds H O C H � H � N � H � Glycine Figure 4. 10 H � C � H � Ethanethiol Because it also has a carboxyl group, glycine is both an amine and a carboxylic acid; compounds with both groups are called amino acids. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings OH � � H SH � C � C � H � H � O � P O � � � O Glycerol phosphate
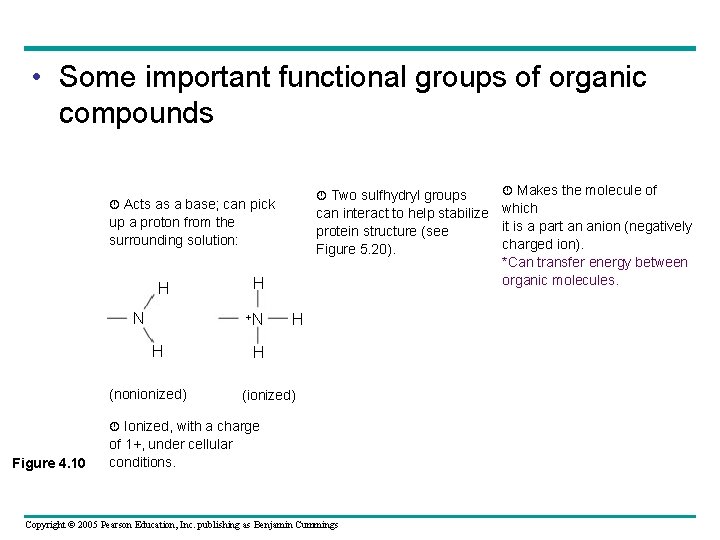
• Some important functional groups of organic compounds Two sulfhydryl groups Acts as a base; can pick can interact to help stabilize which it is a part an anion (negatively protein structure (see charged ion). Figure 5. 20). *Can transfer energy between organic molecules. up a proton from the surrounding solution: H N H +N H (nonionized) H H (ionized) Ionized, with a charge Figure 4. 10 Makes the molecule of of 1+, under cellular conditions. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

The Chemical Elements of Life: A Review • The versatility of carbon – Makes possible the great diversity of organic molecules Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
- Slides: 24