CHAPTER 4 Carbon and the Molecular Diversity of

CHAPTER 4 Carbon and the Molecular Diversity of Life
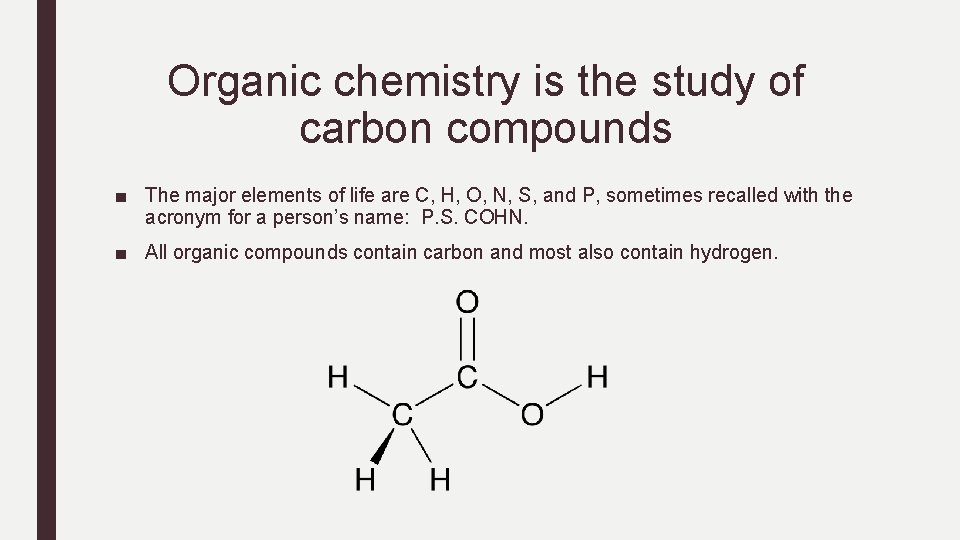
Organic chemistry is the study of carbon compounds ■ The major elements of life are C, H, O, N, S, and P, sometimes recalled with the acronym for a person’s name: P. S. COHN. ■ All organic compounds contain carbon and most also contain hydrogen.
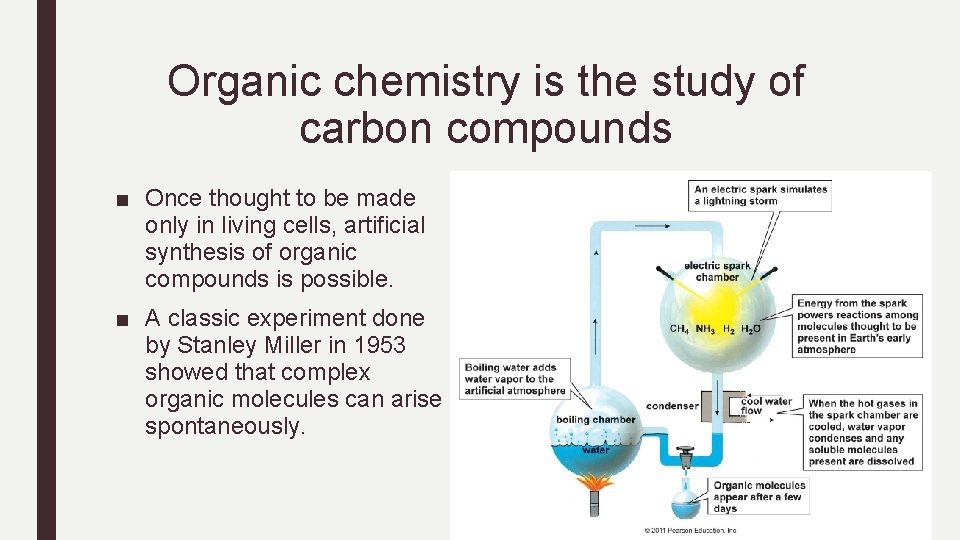
Organic chemistry is the study of carbon compounds ■ Once thought to be made only in living cells, artificial synthesis of organic compounds is possible. ■ A classic experiment done by Stanley Miller in 1953 showed that complex organic molecules can arise spontaneously.

Carbon atoms form diverse molecules by bonding to four other atoms ■ Carbon is unparalleled in its ability to form molecules that are large, complex, and diverse. – It has 4 valence electrons. – It can form up to 4 covalent bonds. – These can be single, double, or triple covalent bonds – It can form large molecules – These molecules can be chains, ring-shaped, or branched.

Carbon atoms form diverse molecules by bonding to four other atoms ■ Isomers are molecules that have the same molecular formula but differ in their arrangement of these atoms. These differences can result in molecules that are very different in their biological activities.

A few chemical groups are key to the functioning of biological molecules. ■ Functional groups attached to the carbon skeleton have diverse properties. The behavior of organic molecules is dependent on the identity of their functional groups. ■ Common Functional Groups: – Hydroxyl (-OH) – Carboxyl (-COOH) – Carbonyl (C=O) – Amino (-NH 2) – Phosphate (-OPO 3) – Sulfhydryl (-SH) – Methyl (-CH 3)
- Slides: 6