Chapter 4 Carbon and the Molecular Diversity of

Chapter 4 Carbon and the Molecular Diversity of Life Power. Point Lectures for Biology, Seventh Edition Neil Campbell and Jane Reece Lectures by Chris Romero Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Organic Compounds • All compounds discovered can be classified into two broad categories: inorganic and organic • "Organic" = • The compounds of life consist of primarily 6 elements: "CHONPS" There are four main groups of biologically important organic molecules: carbohydrates, proteins, lipids, and nucleic acids. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Carbon: The backbone of Biological Molecules • All living organisms are made up of chemicals based mostly on the element carbon – “What sets the carbon atom apart is that it is shamelessly promiscuous. It is the party animal of the atomic world, latching onto many other atoms (including itself) and holding tight, forming molecular conga lines of hearty robustness-the very trick of nature necessary to build proteins and DNA. ” -Bill Bryson, from “A Short History of Nearly Everything” Figure 4. 1 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Chemistry of CARBON is the chemistry of LIFE! • Carbon forms the “backbone” (framework) of all organic molecules, largely because of its versatility in bonding • C has four e- in its outermost energy level, but needs 8 to fill it, so it readily forms covalent bonds! fact: 98. 89% of carbon is C-12 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
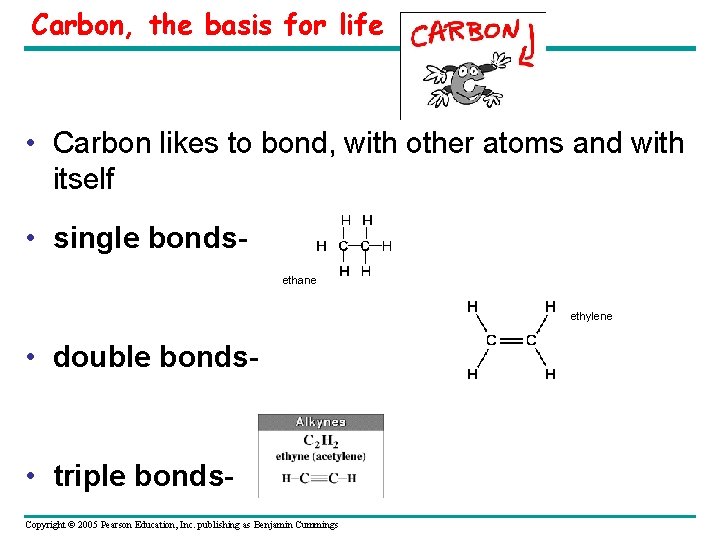
Carbon, the basis for life • Carbon likes to bond, with other atoms and with itself • single bonds- ethane ethylene • double bonds- • triple bonds- Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Carbon videos NPR: It’s All About Carbon Episode 2: Making Carbon Bonds Episode 3: Breaking Carbon Bonds Episode 4: Carbon In Love Episode 5: What Do We Do? Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Concept 4. 1: Organic chemistry is the study of carbon compounds • Organic compounds range from simple molecules to colossal ones – The concept of “vitalism” is the idea that organic compounds arise only within living organisms – Was disproved when chemists synthesized the compounds in the laboratory In 1953, Stanley Miller simulated what were thought to be environmental EXPERIMENT conditions on the lifeless, primordial Earth. As shown in this recreation, Miller used electrical discharges (simulated lightning) to trigger reactions in a primitive “atmosphere” of H 2 O, H 2, NH 3 (ammonia), and CH 4 (methane)—some of the gases released by volcanoes. RESULTS A variety of organic compounds that play key roles in living cells were synthesized in Miller’s apparatus. Organic compounds may have been synthesized abiotically on the Figure 4. 2 CONCLUSION early Earth, setting the stage for the origin of life. (We will explore this hypothesis in more detail in Chapter 26. ) Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

OTHER ELEMENTS IN ORGANIC COMPOUNDS: • Along with carbon and hydrogen, other elements are found in organic compounds. The most common of these elements are nitrogen, phosphorus, sulfur, and oxygen (the CHONPS elements). • Oxygen: Oxygen, as O 2 makes up 21% of the earth's atmosphere and is found in the great majority of organic compounds in living systems. • Nitrogen : Nitrogen is found in all proteins and nucleic acids. 79% of our atmosphere is N 2. The bond between the two nitrogen atoms is a triple bond and is a difficult bond to break. The only way for most organisms to get usable nitrogen is through nitrogen fixing bacteria. • Phosphorus and Phosphates: Phosphorus is found in living systems as phosphates, in ions such as HPO 42 - or H 2 PO 4 -. Phosphorus is covalently bonded to four oxygen atoms. When the oxygen-phosphate bond is broken, energy is released. Phosphorus is an important element in nucleic acids and ATP. . • Sulfur: Sulfur occurs in some proteins. The sulfur appears as part of sulfhydryl groups (-SH) in the amino acid cysteine. These groups allow parts of a proteins to bond together covalently via disulfide bridges Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
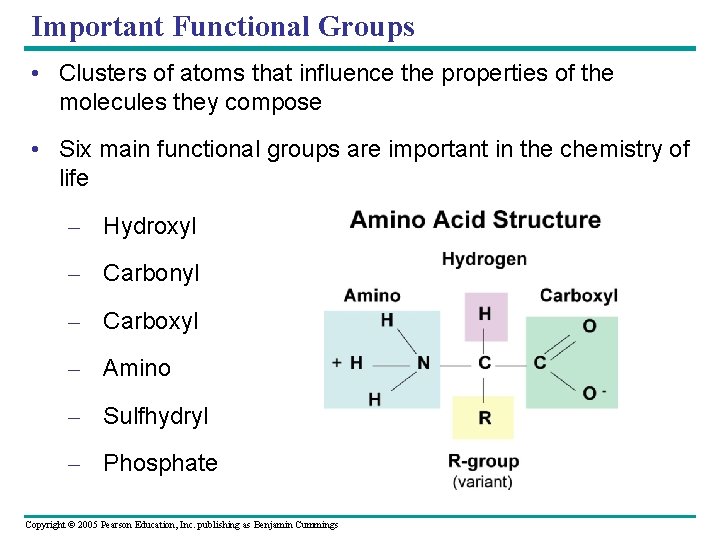
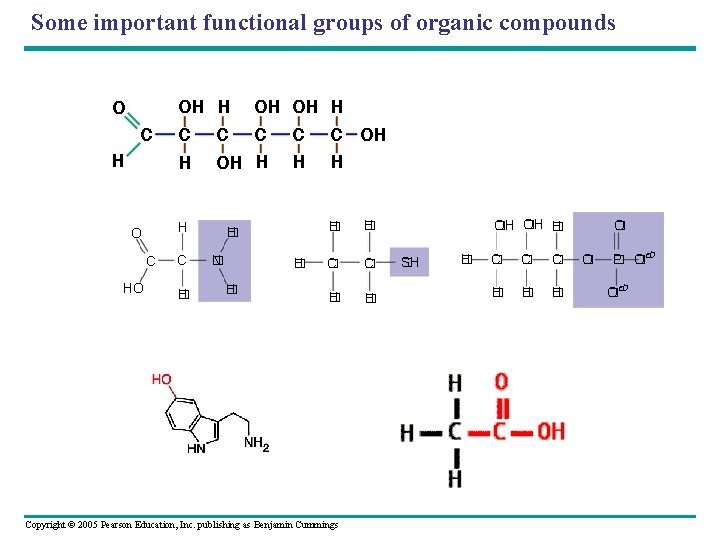
Important Functional Groups • Clusters of atoms that influence the properties of the molecules they compose • Six main functional groups are important in the chemistry of life – Hydroxyl – Carbonyl – Carboxyl – Amino – Sulfhydryl – Phosphate Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
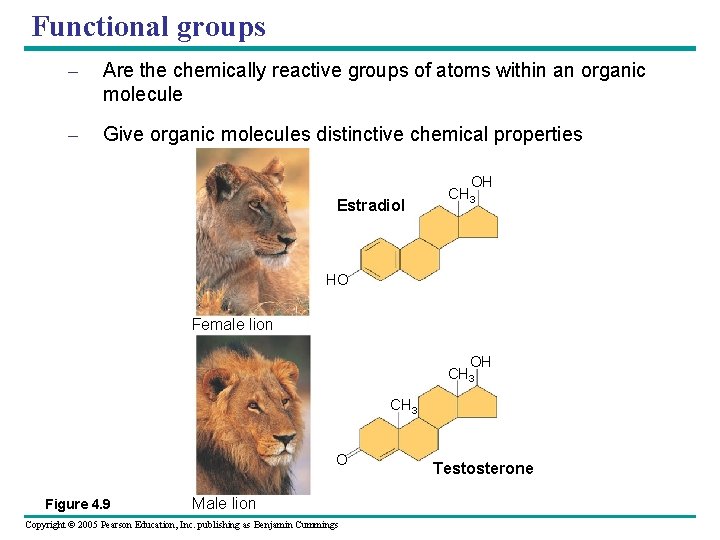
Functional groups – Are the chemically reactive groups of atoms within an organic molecule – Give organic molecules distinctive chemical properties Estradiol OH CH 3 HO Female lion OH CH 3 O Figure 4. 9 Male lion Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Testosterone

Hydroxyl group • Polar, thus water-soluble; forms hydrogen bonds FUNCTIONAL GROUP HYDROXYL Is polar as a result of the electronegative oxygen atom drawing electrons toward itself. Attracts water molecules, helping dissolve organic compounds such as sugars (see Figure 5. 3). O O OH (may be written HO ) STRUCTURE In a hydroxyl group (—OH), a hydrogen atom is bonded to an oxygen atom, which in turn is bonded to the carbon skeleton of the organic molecule. (Do not confuse this functional group with the hydroxide ion, OH–. ) Figure 4. 10 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings C C OH
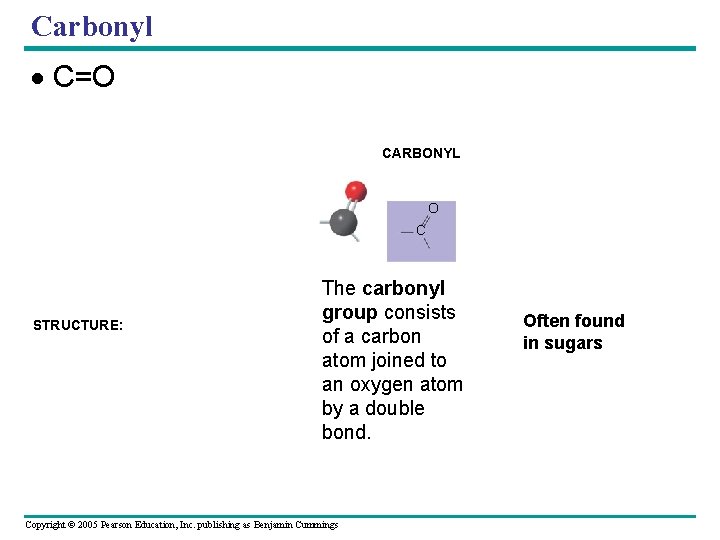
Carbonyl C=O FUNCTIONAL GROUP HYDROXYL CARBONYL CARBOXYL O OH C C OH (may be written HO ) STRUCTURE: O The carbonyl group consists of a carbon atom joined to an oxygen atom by a double bond. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Often found in sugars

Carbonyl group A ketone and an aldehyde may be structural isomers with different properties, as is the case with many monosaccharides. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Carboxyl • Weak acid (hydrogen donor); when it loses a hydrogen, it becomes negatively charged (polar) FUNCTIONAL GROUP HYDROXYL CARBONYL CARBOXYL O OH (may be written HO ) STRUCTURE Figure 4. 10 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings C OH When an oxygen atom is doublebonded to a carbon atom that is also bonded to a hydroxyl group, the entire assembly of atoms is called a carboxyl group (—COOH).

Carboxyl Has acidic properties because it is a source of hydrogen ions. The covalent bond between oxygen and hydrogen is so polar that hydrogen ions (H+) tend to dissociate reversibly; for example, H H C H Figure 4. 10 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings H O C OH H C H O + H+ C O In cells, found in the ionic form, which is called a carboxylate group.
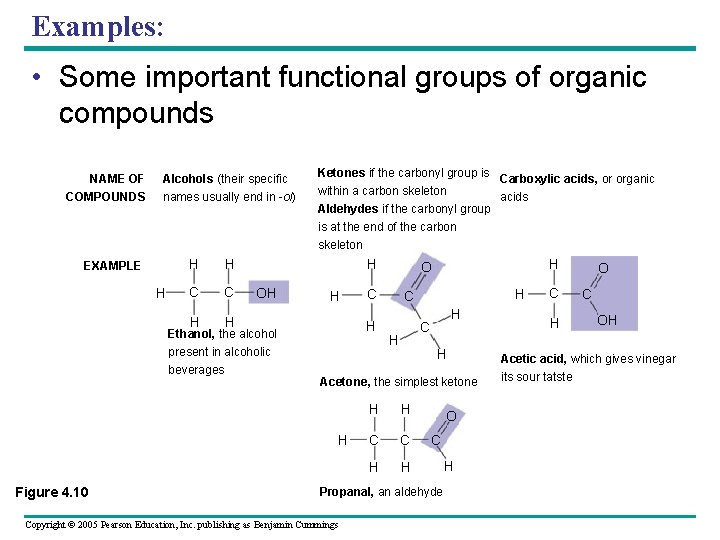
Examples: • Some important functional groups of organic compounds NAME OF COMPOUNDS Alcohols (their specific names usually end in -ol) Ketones if the carbonyl group is Carboxylic acids, or organic within a carbon skeleton acids Aldehydes if the carbonyl group is at the end of the carbon skeleton EXAMPLE H H H C C H H H OH H Ethanol, the alcohol present in alcoholic beverages H C H H Acetone, the simplest ketone H Figure 4. 10 C H H O H H C C H H O C Propanal, an aldehyde Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings H C H O C OH Acetic acid, which gives vinegar its sour tatste
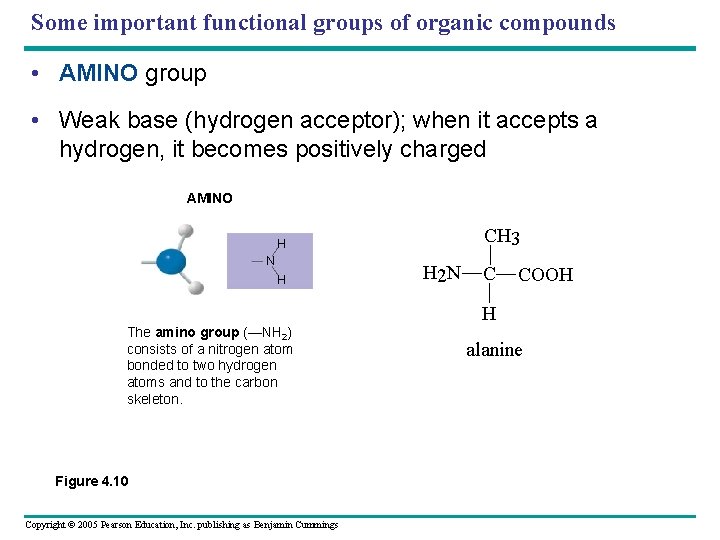
Some important functional groups of organic compounds • AMINO group • Weak base (hydrogen acceptor); when it accepts a hydrogen, it becomes positively charged AMINO SULFHYDRYL H N SH (may be written HS ) H The amino group (—NH 2) consists of a nitrogen atom bonded to two hydrogen atoms and to the carbon skeleton. Figure 4. 10 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings PHOSPHATE O O P OH OH
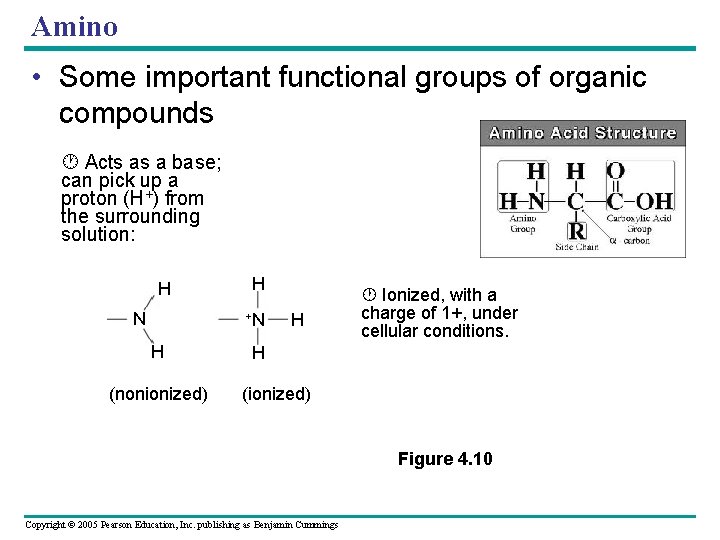
Amino • Some important functional groups of organic compounds Acts as a base; can pick up a proton (H+) from the surrounding solution: H N H +N H (nonionized) H Ionized, with a charge of 1+, under cellular conditions. H (ionized) Figure 4. 10 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
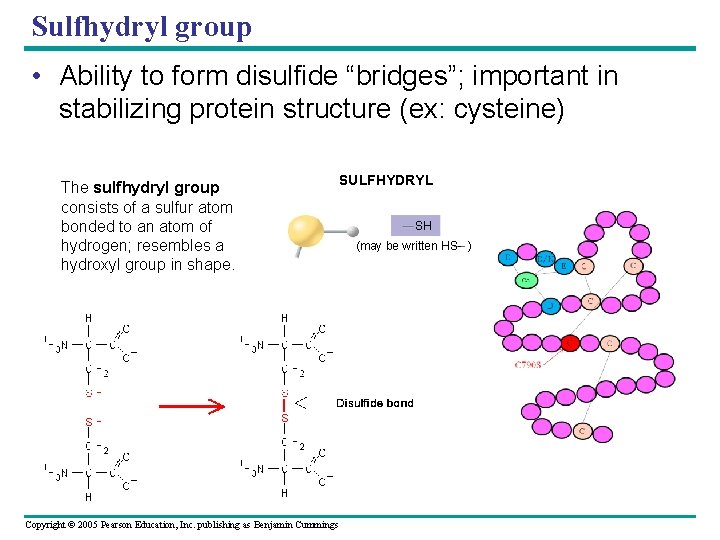
Sulfhydryl group • Ability to form disulfide “bridges”; important in stabilizing protein structure (ex: cysteine) AMINO The sulfhydryl group consists of a sulfur atom bonded to an atom of H hydrogen; resembles a N hydroxyl group in shape. H Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings SULFHYDRYL SH (may be written HS- ) PHOSPHATE O O P OH OH
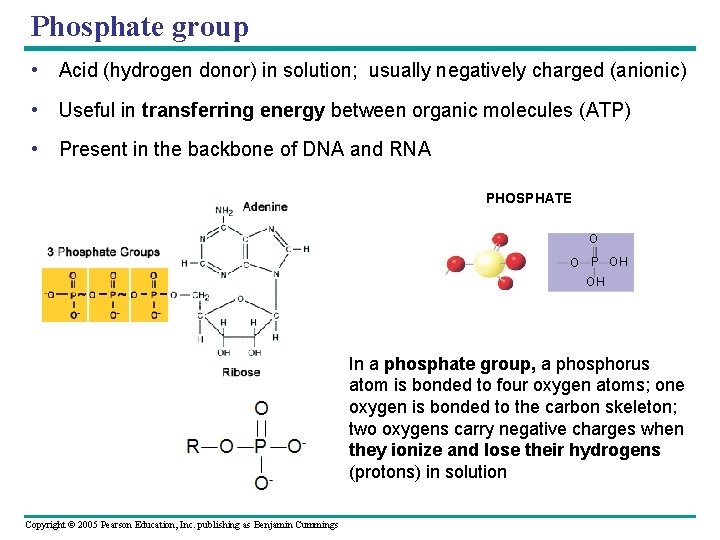
Phosphate group • Acid (hydrogen donor) in solution; usually negatively charged (anionic) • Useful in transferring energy between organic molecules (ATP) • Present in the backbone of DNA and RNA AMINO SULFHYDRYL H N SH (may be written HS ) H PHOSPHATE O O P OH OH In a phosphate group, a phosphorus atom is bonded to four oxygen atoms; one oxygen is bonded to the carbon skeleton; two oxygens carry negative charges when they ionize and lose their hydrogens (protons) in solution Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
Some important functional groups of organic compounds H O C H � H � N � H � H � Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings OH � � H SH � C � C � H � H � O � P O � � � O
- Slides: 21