Chapter 4 Acidbase balance and acidbase disorders Department

Chapter 4 Acid-base balance and acid-base disorders Department of Pathophysiology, the School of Medicine, Shandong University 薛冰
internal environment homeostasis Water balance homeostasis acid-base balance electrolyte balance

Contents 1. Acid-Base Balance Acid-base Biochemistry l Regulation of p. H l Laboratory Tests 2. Simple Acid-base Disorders l Metabolic Acidosis l Respiratory Acidosis l Metabolic Alkalosis l Respiratory Alkalosis 3. Mixed Acid-base Disturbance l

Part I: Acid-base balance and its regulation l. The basic meaning of acid-base balance is the stable [H+] in the body fluid.

I、Concept of acid and base acid : An acid is a H+ donor, when it is dissolved in water. After the loss of H+, it becomes a base. HA (acid) → H+ + Aˉ(base) H 2 CO 3 → H+ +HCO 3ˉ H 2 SO 4, H 3 PO 4 base: A base is a H+ acceptor, when it is dissolved in water. After the combining of H+, it becomes an acid Aˉ(base)+ H+ → HA (acid) HCO 3ˉ +H+ → H 2 CO 3 OH-, HCO 3 -, SO 42 -, HPO 42 -, NH 3

II、Sources of acid and base The main origin of acid and base is the intracellular metabolism (catabolism of protein, carbohydrate and fat). (I) Source of acid v volatile acid v fixed acid
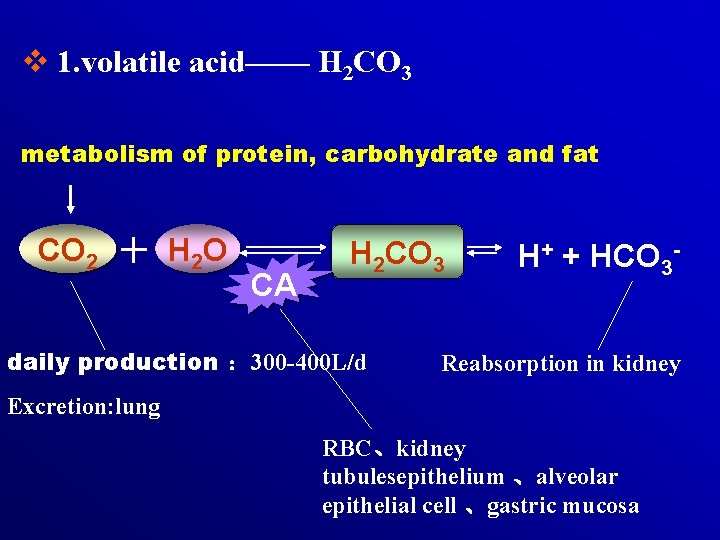
v 1. volatile acid—— H 2 CO 3 metabolism of protein, carbohydrate and fat CO 2 + H 2 O CA H 2 CO 3 daily production : 300 -400 L/d H+ + HCO 3 - Reabsorption in kidney Excretion: lung RBC、kidney tubulesepithelium 、alveolar epithelial cell 、gastric mucosa

2. unvolatile acid (fixed acid) : (50 -100 mmol/d) Uric acid, phosphoric acid (H 3 PO 4) and sulfuric acid (H 2 SO 4) are the products in the metabolic process of proteins and nuclear acids. Lactic acid and ketonic bodies (β-hydroxybutyric acid and acetoacetic acid) can be formed from the metabolic process of carbohydrate and fat as intermediate products, when the oxygen supply is not sufficiency. Exogenous acid(food and drug): Excretion through kidney

(II) Sources of base Origin of bases l Endogenous: deamination―>NH 3 Less than acid production l Exogenous input: vegetables, and fruits

III、 Regulation of acid-base balance

Henderson-Hasselbalch Equation Acid-base balance is mainly the balance between production and loss of acid and base. p. H=p. Ka+lg [HCO 3-] [H 2 CO 3] 20 =p. Ka+lg 1 = 6. 1 +1. 3 = 7. 4
![Acid-base balance : p. H∝ [HCO 3-] / [H 2 CO 3] or p. Acid-base balance : p. H∝ [HCO 3-] / [H 2 CO 3] or p.](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-12.jpg)
Acid-base balance : p. H∝ [HCO 3-] / [H 2 CO 3] or p. H∝ [HCO 3-] / Pa. CO 2 [H+]↑ Buffer ECF Neutralize H+ (Immediately) Respiratory ICF & bone pa. CO 2 ↓ Renal Excrete H+ Keep Na. HCO 3 e. H+ & i. K+ exchange ( 2~4 h) (1~3 min) (hs; 1~3 d)
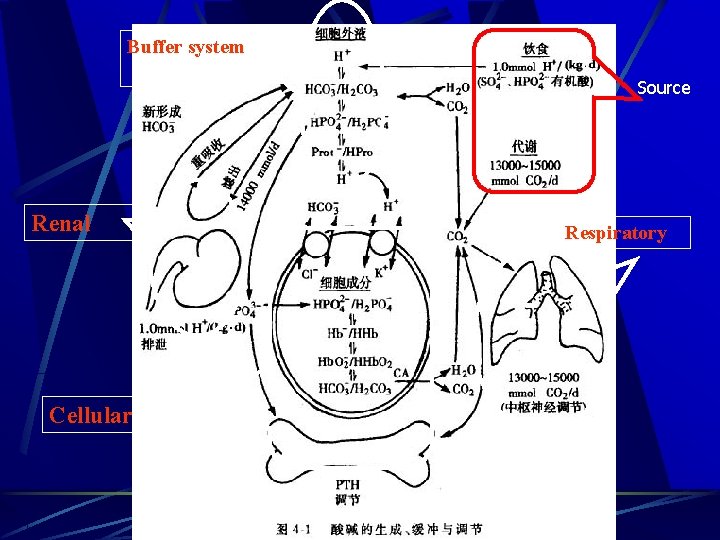
Buffer system Source Renal Cellular Respiratory

(I)blood buffer system Buffer systems :consists of a weak acid and its’ salt ※HCO 3 -/H 2 CO 3 is the most important buffer pair. 表 1 缓冲酸 全血五种缓冲系统 表 2 缓冲碱 全血中各缓冲体系的含量与分布 缓冲体系 占全血缓冲体系% H 2 CO 3 HCO 3 ¯ + H+ 血浆HCO 3 ¯ 35 H 2 PO 4 - HPO 42 ¯ + H+ 红细胞内Hb 18 HPr Pr ¯ + H+ Hb. O 2 -及Hb- 35 HHb Hb¯ + H+ 磷酸盐 5 HHb. O 2¯ + H+ 血浆蛋白 7

HCO 3 -/H 2 CO 3 buffer system v. Regulate CO 2 or HCO 3 - through kidney and lung, the most important buffer pair (50%)。 vfixed acid and base buffer system v. PH is dermatied by HCO 3 -/H 2 CO 3。
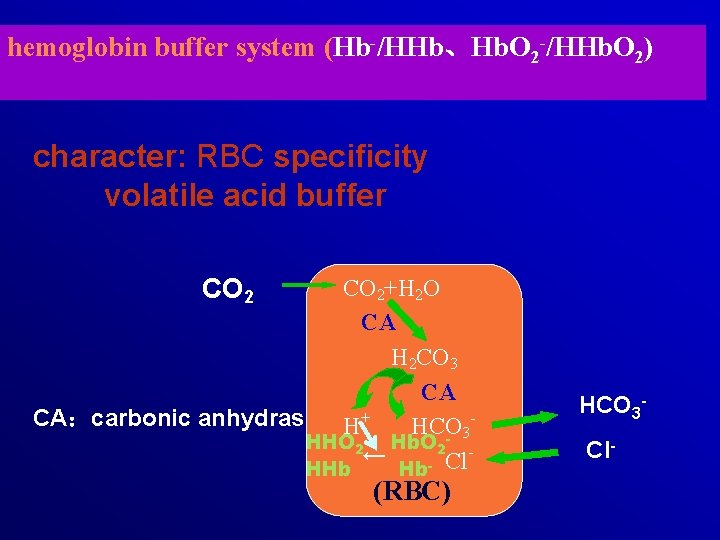
hemoglobin buffer system (Hb-/HHb、Hb. O 2 -/HHb. O 2) character: RBC specificity volatile acid buffer CO 2+H 2 O CA H 2 CO 3 CA CA:carbonic anhydrase H+ HCO 3 HHO 2 Hb. O 2 - ← Cl HHb Hb- (RBC) HCO 3 Cl-

phosphate buffer system HPO 42 -/H 2 PO 4 character: play a role in cell and kidney

protein buffer system Pr-/HPr Intracellular buffer
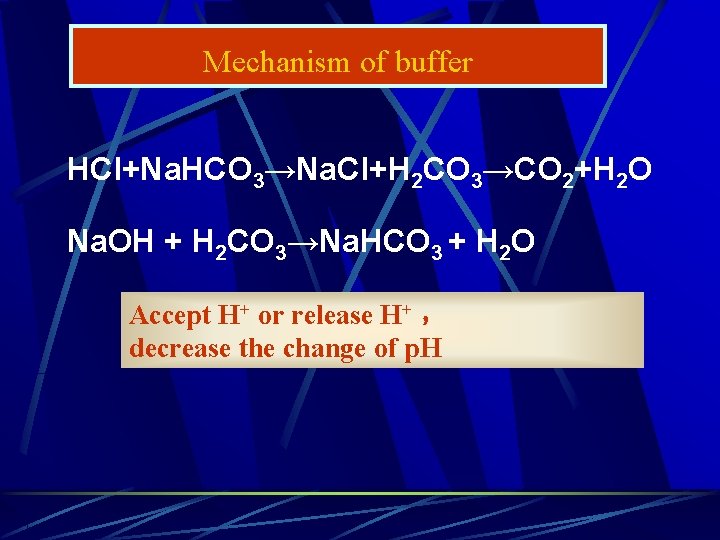
Mechanism of buffer HCl+Na. HCO 3→Na. Cl+H 2 CO 3→CO 2+H 2 O Na. OH + H 2 CO 3→Na. HCO 3 + H 2 O Accept H+ or release H+ , decrease the change of p. H

Character of Buffer Unvolatile acid: l HCO-3/H 2 CO 3 system: l ½ of the buffer capacity l Opened regulation: respiratory and renal Volatile acid : l Hb-/HHb、Hb. O 2 -/HHb. O 2

(II)Mechanisms of respiratory control change the depth or rate of respiration →change CO 2 elimination→ [HCO 3 -]/ Pa. CO 2 →Acid-base balance

1. central chemoreceptor Pa. CO 2 (N: 40 mm. Hg) ↑→ p. H of CSF↓ →to stimulate central chemoreceptor →☆ the respiratory center→Pulmonary ventilation volume ↑ Pa. CO 2 >60 mm. Hg (8 k. Pa) → Pulmonary ventilation volume ↑ 10 times but, Pa. CO 2 >80 mm. Hg (10. 7 k. Pa) →inhibit respiratory center,named as carbon dioxide narcosis

central chemoreceptor The central chemoreceptor is sensitive to the change of CO 2, which is easy to cross the blood-brain barrier. It takes time for the H+ to penetrate across the blood brain barrier into the interstitial fluid of the brain, the increase of [H+] in the brain is relatively slow, so the effect of H+ on the central chemoreceptor will be slow.

2. peripheral chemoreceptor Pa. O 2 ↓ 、p. H ↓ 、Pa. CO 2↑ →to stimulate peripheral chemoreceptor →☆ the respiratory center→ Pulmonary ventilation volume ↑ Pa. O 2 � 60 mm. Hg (8 k. Pa) → ☆ the respiratory center; but Pa. O 2 <30 mm. Hg →inhibit respirator center。 Less sensitive than central chemoreceptor

3. Characteristic of respiratory compensation (a) Timeliness. The respiratory response begins within several minutes. The respiratory response often takes 30 minutes for the respiratory compensation. 12~24 hours to get maximal compensation. (b) limited compensation

(III) Renal regulation of acid-base Balance Renal compensation begins from several hours after the addition of acid load, and it may take 3~5 days to reach the maximum of this compensatory capacity. Kidneys play a major role in the regulation of p. H in the body.
![Excrete the nonvolatile acid, reabsorb the bicarbonate ,“排酸保碱” →keep [HCO 3 -]→maintain acid-base balance。 Excrete the nonvolatile acid, reabsorb the bicarbonate ,“排酸保碱” →keep [HCO 3 -]→maintain acid-base balance。](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-27.jpg)
Excrete the nonvolatile acid, reabsorb the bicarbonate ,“排酸保碱” →keep [HCO 3 -]→maintain acid-base balance。 HCO 3 - filtrate through glomerulus freely(5000 mmol/d), 85%~90% is reabsorbed by proximal tubule,others are reabsorbed by distal convoluted tubule and collecting duct, 0. 1% is excreted→urine p. H 6. 0。 urine p. H vary from 4. 4 to 8. 0
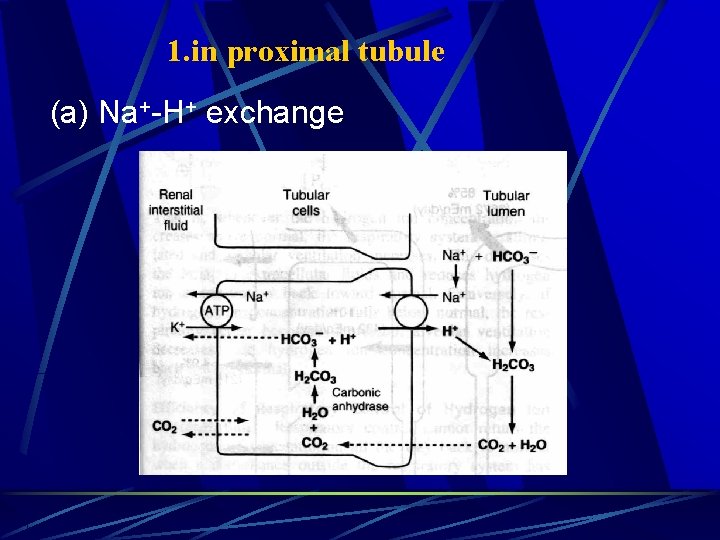
1. in proximal tubule (a) Na+-H+ exchange
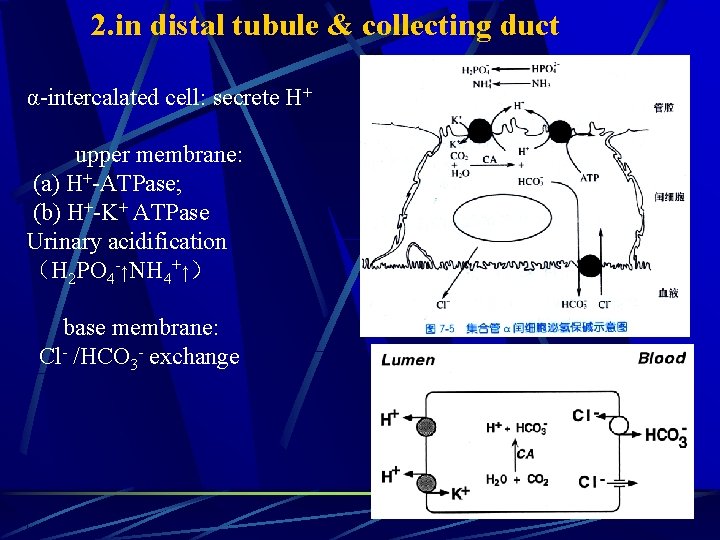
2. in distal tubule & collecting duct α-intercalated cell: secrete H+ upper membrane: (a) H+-ATPase; (b) H+-K+ ATPase Urinary acidification (H 2 PO 4 -↑NH 4+↑) base membrane: Cl- /HCO 3 - exchange
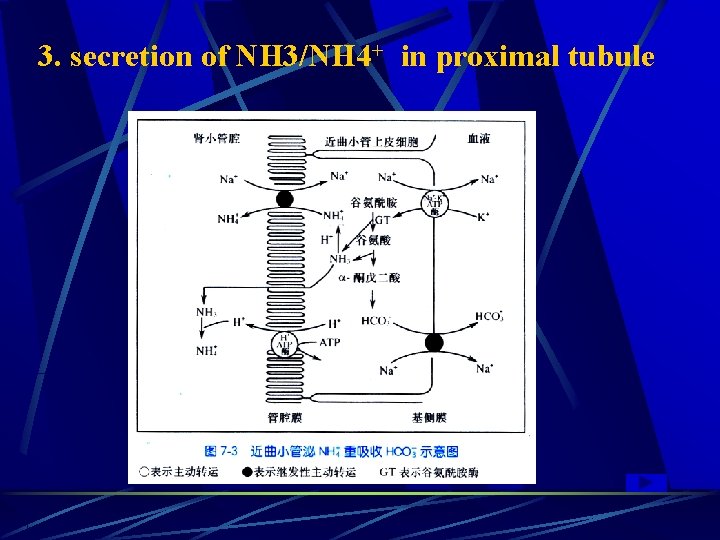
3. secretion of NH 3/NH 4+ in proximal tubule

4. competitive inhibition between K+-Na+ exchange and H+-Na+ in distal tubule K+-Na+ exchange: secrete K+, reabsorb Na+, H+-Na+ exchange: secrete H+, reabsorb Na+ acidosis, H+-Na+ exchange↑→ K+-Na+ exchange↓→hyperkalemia。
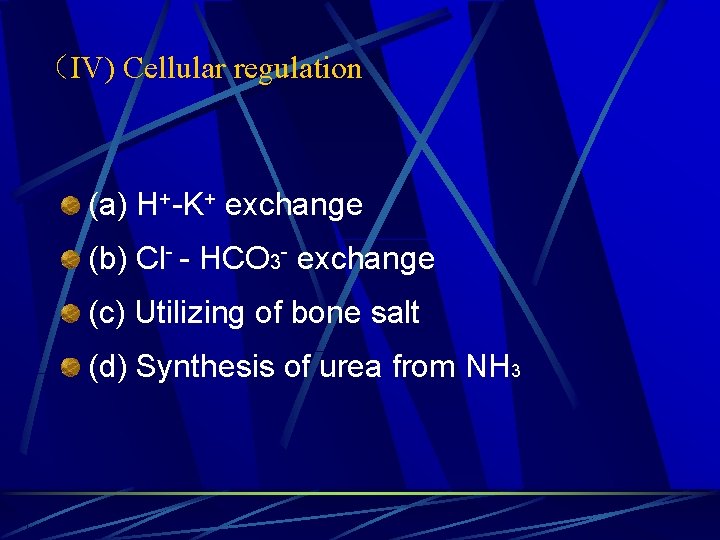
(IV) Cellular regulation (a) H+-K+ exchange (b) Cl- - HCO 3 - exchange (c) Utilizing of bone salt (d) Synthesis of urea from NH 3
![1. H+-K+ exchange When [H+] in ECF (serum) is increased, the H+ will move 1. H+-K+ exchange When [H+] in ECF (serum) is increased, the H+ will move](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-33.jpg)
1. H+-K+ exchange When [H+] in ECF (serum) is increased, the H+ will move into the cells, as a exchange for electrical neutrality, K+ will shift from ICF to the ECF. So the p. H of ECF (serum) will increase to normal, but hyperkalemia may occur.

2. Cl- - HCO 3 - exchange When CO 2 in ECF (serum) is increased, CO 2 will move into the cells, CO 2 combines H 2 O to form carbonic acid, then H 2 CO 3 dissociates to form H+ and HCO 3¯ , the HCO 3¯ moves out of the RBC, for neutrality, Cl ¯ moves into the cells.

3. Utilizing of bone salt In chronic metabolic acidosis, bone salt, Ca 3(PO 4)2, is also utilized as a buffer base, but the expense is decalcification of bone and osteoporosis (loose and soft bone). Ca 3(PO 4)2 + 4 H+ ←→ 3 Ca 2+ + 2 H 2 PO 4 ¯ It is not a good way of regulating acid-base balance by utilization of bone salt.
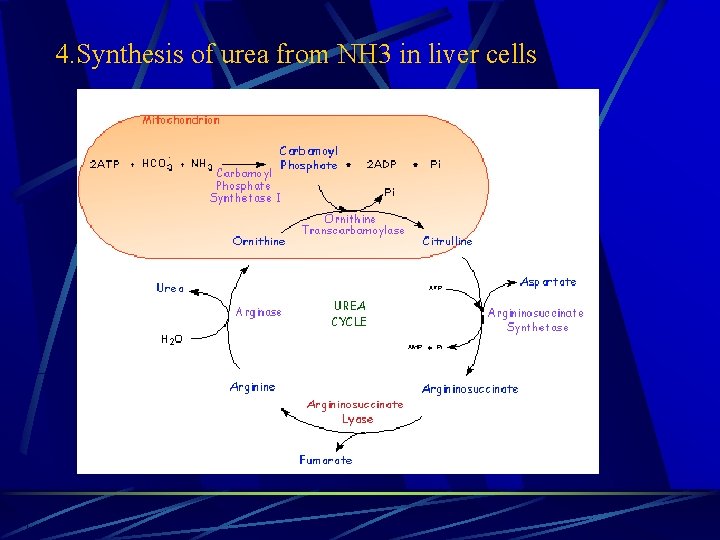
4. Synthesis of urea from NH 3 in liver cells
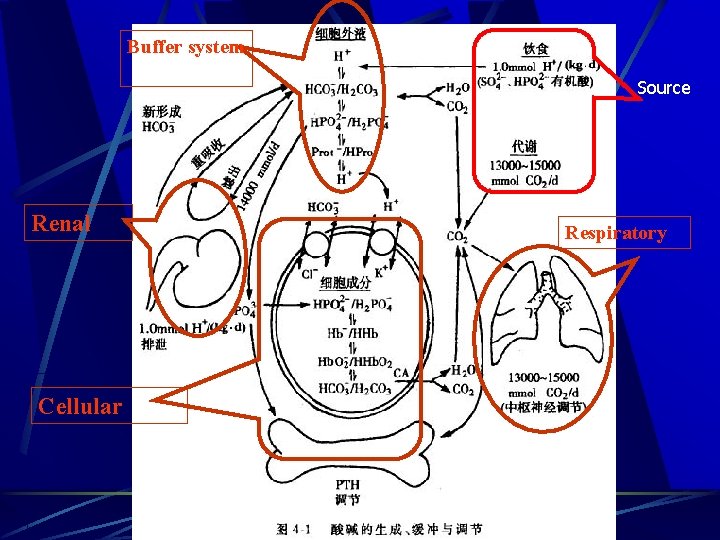
Buffer system Source Renal Cellular Respiratory

Part II laboratory tests of acid-base disturbances
![1. p. H is the negative logarithm (-log) of [H+] in a solution. [H+]=40 1. p. H is the negative logarithm (-log) of [H+] in a solution. [H+]=40](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-39.jpg)
1. p. H is the negative logarithm (-log) of [H+] in a solution. [H+]=40 nmol/L (p. H=7. 4) The normal range in artery blood =7. 35~7. 45 (7. 41) The survival range of p. H=6. 8~7. 8 According to the Henderson-Hasselbalch equation: The p. Ka is the dissociation constant of carbonic acid (=6. 1)
![24 [HCO 3 ¯ ] metabolic factor p. H =6. 1+ log -------------------- 24 [HCO 3 ¯ ] metabolic factor p. H =6. 1+ log --------------------](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-40.jpg)
24 [HCO 3 ¯ ] metabolic factor p. H =6. 1+ log -------------------- 1. 2 [H 2 CO 3] respiratory factors 20 = 6. 1+ log----- =6. 1+1. 3=7. 4 1 The p. H is determined by the ratio of [HCO 3¯ ] 20 -------=---- [H 2 CO 3] 1 No matter how the absolute amounts of HCO 3¯ and H 2 CO 3 change, once the ratio remains 20/1, the p. H would be 7. 4 (normal).
![24 [HCO 3 ¯ ] metabolic factor p. H =6. 1+ log ---------------------- 1. 24 [HCO 3 ¯ ] metabolic factor p. H =6. 1+ log ---------------------- 1.](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-41.jpg)
24 [HCO 3 ¯ ] metabolic factor p. H =6. 1+ log ---------------------- 1. 2 [H 2 CO 3] respiratory factors The primary changes determines the nature of the acid-base imbalance. The purpose of secondary change is to restore the p. H. According to the p. H: l compensatory acid-base disturbances l decompensatory acid-base disturbances

Clinical significance of PH (anticoagulant artery blood, insulation of air) A normal range of p. H may represent three different situations: ① acid-base balance; ② compensatory acidosis or alkalosis; ③ a mixed decompensatory acidosis and decompensatory alkalosis.

Clinical significance p. H<7. 35 decompensatory acidosis ( acidemia ) p. H>7. 45 decompensatory alkalosis (alkalemia)

2. Pa. CO 2 (partial pressure of carbon dioxide in arterial blood) CO 2 in blood: (a) 23% Hb. CO 2 in RBC (b) 70% HCO 3 - in plasma (c) 7% CO 2 molecule in plasma CO 2 is determined by the rate of CO 2 production and the rate of CO 2 elimination. Pa. CO 2 is the tension of CO 2 caused by CO 2 molecule movement. The normal range = 33~46(40) mm. Hg (4. 39~6. 25 k. Pa).

Pa. CO 2 The capability of normal lung to eliminate CO 2 is very good. CO 2 retention will not occur with normal ventilation. Generally speaking, the Pa. CO 2 is determined mainly by the respiration, so the Pa. CO 2 is called the “respiratory factor”. Higher Pa. CO 2 is due to the inhibition of respiration. Lower Pa. CO 2 is due to overventilation.

Significance Pa. CO 2>46 mm. Hg Primary increase: respiratory acidosis Secodary increase: metabolic alkalosis (compensated by lung) Pa. CO 2<33 mm. Hg Primary decrease: respiratory alkalosis Secodary decrease: metabolic acidosis (compensated by lung)
![3. [HCO 3 -] Actual bicarbonate (AB) The normal [HCO 3¯ ] is 22~27(24) 3. [HCO 3 -] Actual bicarbonate (AB) The normal [HCO 3¯ ] is 22~27(24)](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-47.jpg)
3. [HCO 3 -] Actual bicarbonate (AB) The normal [HCO 3¯ ] is 22~27(24) mmol/L. AB is measured under “actual condition” in which both respiratory factor and metabolic factor affected the [HCO 3¯ ]. CO 2 +H 2 O=H 2 CO 3=H++HCO 3 ¯

Standard bicarbonate (SB) SB is measured under “standard condition” (temperature 37~38℃, full oxygenation of hemoglobin, Pa. CO 2 = 40 mm. Hg). Standard condition means that the respiratory factor is eliminated, then the [HCO 3¯ ] is only affected by metabolic factor. l Higher SB means metabolic alkalosis or respiratory acidosis compensated by kidneys. l Low SB means metabolic acidosis or respiratory alkalosis compensated by kidneys.

Normally the AB=SB. CO 2 +H 2 O=H 2 CO 3=H++HCO 3 If AB>SB (CO 2 retention), the reason must be the effect of respiratory factor, which indicates respiratory acidosis or metabolic alkalosis compensated by lung. If AB<SB (CO 2 depletion), the reason must be the respiratory factor, which means respiratory alkalosis or the metabolic acidosis compensated by lung.

4. Buffer base (BB) Sum of all buffer basees in blood In plasma: HCO 3 ¯ =24 Protein¯ =17 In RBC: Hb¯ Hb. O 2¯ =6. 3 HPO 4 2¯ =1. 0 BB=45~55 mmol/L Determined by metabolic factors

Significance Normal BB: acid-base balance metabolic acidosis + metabolic alkalosis Increased BB: Primary increase: metabolic alkalosis Secodary increase: respiratory acidosis Decreased BB: Primary decrease: metabolic acidosis Secodary decrease: respiratory alkalosis

5. Base excess (BE) Under “standard condition” (temperature 37~38℃, full oxygenation of hemoglobin, Pa. CO 2 = 40 mm. Hg), titrate the whole blood to p. H 7. 4 with how much acid or base (mmol/L). If with acid, there is must more base (excess) in the blood, BE is expressed with positive value If with base, there is must more acid (deficit) in the blood, BE is expressed with negative value

Significance Normal BE= -3. 0~+3. 0 Only metabolic factor determines BE In metabolic alkalosis the positive BE increases. In metabolic acidosis the negative BE increases.
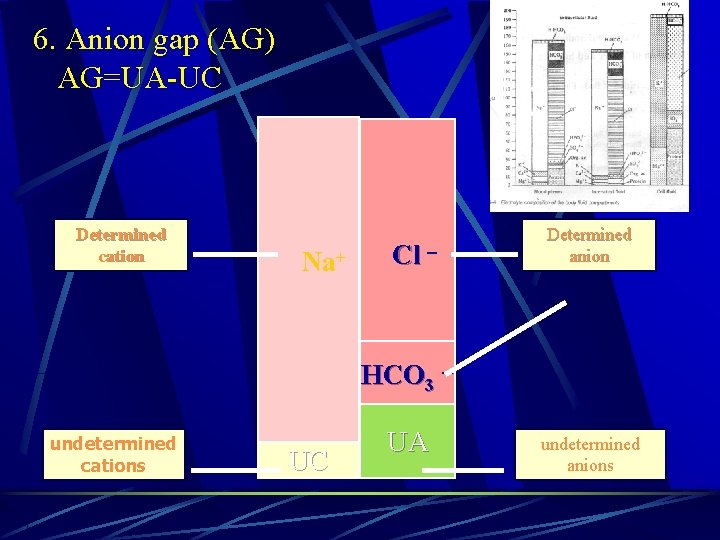
6. Anion gap (AG) AG=UA-UC Determined cation Na+ Cl - Determined anion HCO 3 - undetermined cations UC UA undetermined anions

The AG can be calculated by: UA+ HCO 3¯ + Cl¯ =UC+Na+ =Na+-(Cl¯ + HCO 3¯ ) The normal range is 10~14 mmol/L. Na+ AG indicates those anions, other than HCO 3¯ and Cl¯, which is required to counter-balance Na+. Cl - HCO 3 - UC UA AG

Significance(i) Actually the AG represents the proteins with negative charge, phosphate, sulfate and organic anions (lactic acid, keto-acid, etc. ). An increased AG is the same meaning as the accumulation of nonvolatile acids in the body and must be the metabolic acidosis.

Significance(ii) For the classification of metabolic acidosis a) metabolic acidosis with normal AG ( with increased Cl ¯ ) b) metabolic acidosis with high AG (with normal Cl ¯).


Part III Acid–Base Disturbance

Contents 1. Simple Acid-base Disorders l Metabolic Acidosis l Respiratory Acidosis l Metabolic Alkalosis l Respiratory Alkalosis 2. Mixed Acid-base Disturbance
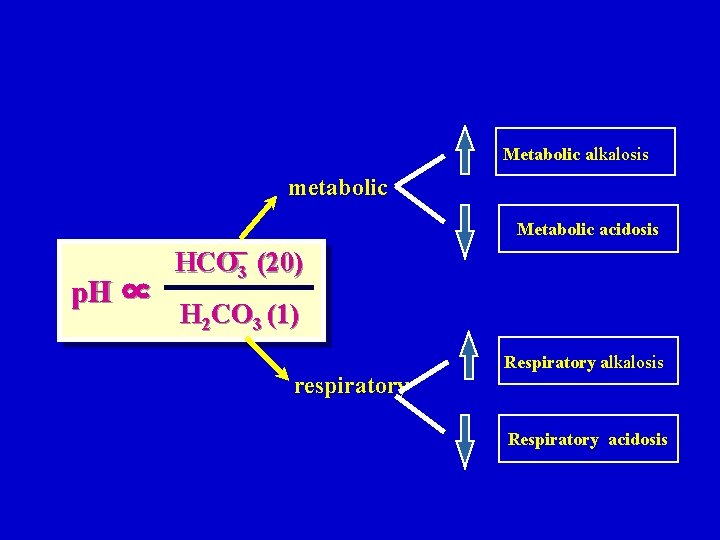
Metabolic alkalosis metabolic Metabolic acidosis p. H ∝ HCO-3 (20) H 2 CO 3 (1) respiratory Respiratory alkalosis Respiratory acidosis

I、 Metabolic acidosis It is defined as a primary decrease in plasma [HCO 3 -], the p. H tends to decrease. [HCO 3 ¯] in serum, p. H = p. Ka + lg -------- [H 2 CO 3] The meaning of “primary” indicates the change happened firstly compared with ”secondary” change of another parameter. The metabolic acidosis is the most common type of acidbase imbalance.

1. Classification Metabolic acidosis is classified into two types: (1) metabolic acidosis with normal AG (with increased Cl-) (2) metabolic acidosis with high AG (with normal Cl-). An increased AG means the accumulation of nonvolatile acids in the body.

2. Causes and Pathogenesis (1) Metabolic acidosis characterized by normal anion gap (AG) (2) Metabolic acidosis characterized by increased anion gap (AG)

(1)Metabolic acidosis characterized by normal AG Normal AG means: a) the glomerular filtration rate (GFR) is sufficient to excrete sulfate, phosphate and other nonvolatile acids, b) normal production of organic acids c) no accumulation of nonvolatile acids in the body. d) Decreased HCO 3 -:Cl- increase Normal Acidosis
![Metabolic acidosis characterized by normal AG The decreased [HCO 3 -] is caused by Metabolic acidosis characterized by normal AG The decreased [HCO 3 -] is caused by](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-66.jpg)
Metabolic acidosis characterized by normal AG The decreased [HCO 3 -] is caused by l a) increased loss of HCO 3 - from kidneys, l b) increased loss of HCO 3 - from intestinal tract, l c) excessive production of Cl-.
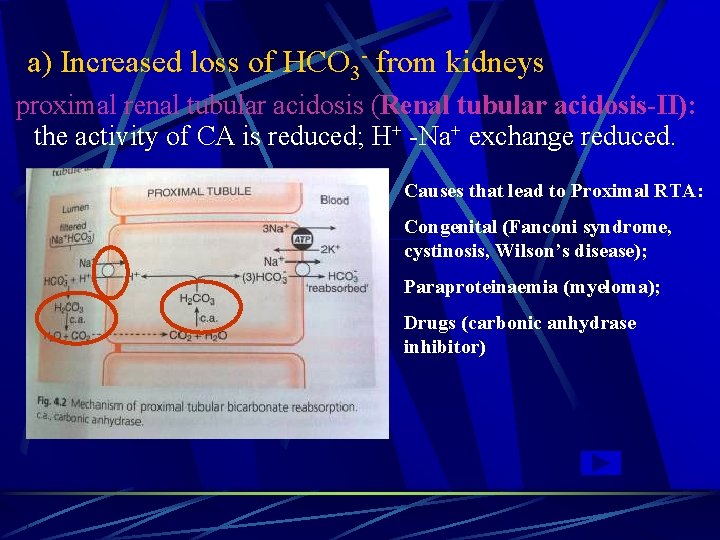
a) Increased loss of HCO 3 - from kidneys proximal renal tubular acidosis (Renal tubular acidosis-II): the activity of CA is reduced; H+ -Na+ exchange reduced. Causes that lead to Proximal RTA: Congenital (Fanconi syndrome, cystinosis, Wilson’s disease); Paraproteinaemia (myeloma); Drugs (carbonic anhydrase inhibitor)
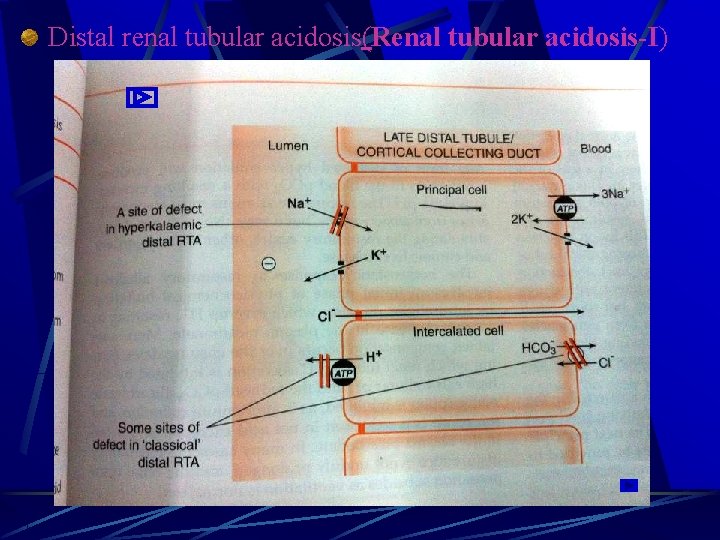
Distal renal tubular acidosis(Renal tubular acidosis-I)

Causes of distal RTA Classical type Congential Hyperglobulinaemia Authoimmune connective tissue disease(e. g systemic lupus erythematosus) Toxins and drugs(toluene, lithium, amphotericin) Hyperkalemic type Hypoaldosteronism Obstructive nephropathy renal transplant rejection Drugs(amiloride, spironolactone)

Different between proximal RTA and distal RTA Urine PH: Distal RTA: secretion of H+ in collecting duct dysfunction Urine PH Proximal RTA: early of mild: [HCO 3 -]reabsorption Urine PH Serious acidosis: plasma [HCO 3 -] filtration of [HCO 3 -] collecting duct secretion H+, Urine PH Effects on the other organ: Proximal RTA: amino aciduria, glycosuria, phosphaturia. Distal RTA: nephrocalcinosis(calcium releasing to buffer H+ in blood), stone, skeletal growth dysfunction Electrolyte disturbance: hypovolaemia, hypokalemia

b) Increased loss of HCO 3¯ from intestinal tract The main extrarenal loss of HCO 3¯ is from intestinal tract, like diarrhea and fistula in intestinal tract, because there is more HCO 3 ¯ in intestinal juice than in serum.

c) Excessive production of Cl¯ Infusion /ingestion HCl, NH 4 Cl 2 NH 4 Cl+CO 2 (NH 2)2 CO+2 HCl+H 2 O For electrical neutrality, the [HCO 3¯] is reduced after the [Cl¯] increased. d) Dilution of [HCO 3¯] e)hyperkalemia: paradoxical alkaline urine
![(2)Metabolic acidosis characterized by increased AG The reason of reduced [HCO 3¯] is the (2)Metabolic acidosis characterized by increased AG The reason of reduced [HCO 3¯] is the](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-74.jpg)
(2)Metabolic acidosis characterized by increased AG The reason of reduced [HCO 3¯] is the accumulation of organic acids (nonvolatile acid) in blood due to more acids: a) decreased excretion of organic acids b) overproduction of organic acids.

(a) Causes : Endogenous acid load increase a) acute and chronic renal failure Nonvolatile acids are filtered off through the glomerular membrane. In acute (less renal flow) and chronic renal failure (less permeability and area), the GFR is reduced, which results in the retention of nonvolatile acids in blood, so the AG is increased.

b) Incomplete catabolism of carbohydrates and fatty acids Normally the complete catabolism of carbohydrates and fatty acids produces CO 2 and water. The increased anaerobic glycolysis due to hypoxia results in lactic acidosis.

In insulin lack, the catabolism of carbohydrates is reduced, the lipolysis ( catabolism of fat) is increased, the ketogenesis is accelerated. If the production of keto-bodies is more than the catabolism and excretion of keto-bodies, accumulation of keto-bodies will result in diabetic keto-acidosis.

Alcoholic keto-acidosis occurs as the result of accelerated lipolysis due to reduced insulin secretion. Starvation causes metabolic acidosis due to the accelerated lipolysis, which leads to the overproduction of keto- bodies (accelerated ketogenesis). The accelerated production of lactic acid and ketone acid must exceed the excretion capability in kidneys, then the lactic acidosis and keto-acidosis will occur.

(b)Cause : Administration of exogenous acid Salicylate (for stop pain and anti-inflammation) can be converted to salicylic acid in the body. Salicylic acid is a kind of nonvolatile acid. methanol poisoning methanol formaldehyde formic acid Early: formic acid deposition Later: hypoxia, lactic acidosis
![3. Compensation of metabolic acidosis primary [HCO 3 -] ↓→secondary Pa. CO 2↓,regulate [HCO 3. Compensation of metabolic acidosis primary [HCO 3 -] ↓→secondary Pa. CO 2↓,regulate [HCO](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-80.jpg)
3. Compensation of metabolic acidosis primary [HCO 3 -] ↓→secondary Pa. CO 2↓,regulate [HCO 3 -] / Pa. CO 2,then p. H (1). Blood buffer:H+↑+ HCO 3 - H 2 CO 3

High Pa. CO 2 Low p. H nvia (2) Respiratory compensation chemoreceptors stimulate the respiratory center increase the rate and depth of respiration more carbon dioxide can be eliminated from lung normal Pa. CO 2 normal p. H Increased Pa. CO 2 and decreased p. H will stimulate the chemoreceptors located in respiratory center (central) and in carotic body (peripheral), and enhance the ventilation.

Kussmaul respiration (deep sighing respiration) is for increasing CO 2 excretion More CO 2 will be eliminated. The [H 2 CO 3] will decrease secondarily to the decrease of [HCO 3¯ ]. The ratio of [HCO 3¯ ]/[H 2 CO 3] will tend to normal. The p. H will tend to normal.

Deep, quick breathe is the main clinincal manifestation. Rapid, powerful:several minutes( respirator enhance), 30 min(compensation), 12 -24 h (maximal compensation ) 7. 4 , 4 L/min→ 7. 0 , 30 L/min.

Predicted compensatory formula ΔPa. CO 2(mm. Hg) = 1. 2 x ΔHCO 3 - ± 2 Secondary compensation primary change Or: Pa. CO 2=1. 5 x. HCO 3 -+8 ± 2 Pa. CO 2 can decline maximal to 10 mm. Hg. Value measured > value predicted: with respiratory acidosis Value measured < value predicted: with respiratory alkalosis Patient, diarrhoea, [HCO 3 -] 12 mmol/L, Pa. CO 2 28 mm. Hg. Type of acid-base disturbance? (18?, 38?)
![(3). intracellular buffering Patient with serious diarrhoea :p. H 7. 2,serum[K+] 5. 6 mmol/L (3). intracellular buffering Patient with serious diarrhoea :p. H 7. 2,serum[K+] 5. 6 mmol/L](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-85.jpg)
(3). intracellular buffering Patient with serious diarrhoea :p. H 7. 2,serum[K+] 5. 6 mmol/L Correct acidosis: p. H 7. 4,serum[K+] 2. 5 mmol/L Renal tubule lumen ECF [H+] serum[K+] + H++Pr-→HPr K H+ K+ Na+ acidosis → hyperkalemia

(4) Renal compensation begins from several hours after the addition of acid load, and it may take 3~5 days to reach the maximum of this compensatory capacity. The reabsorption of HCO 3¯ is increased. Net acid excretion with urine is increased.
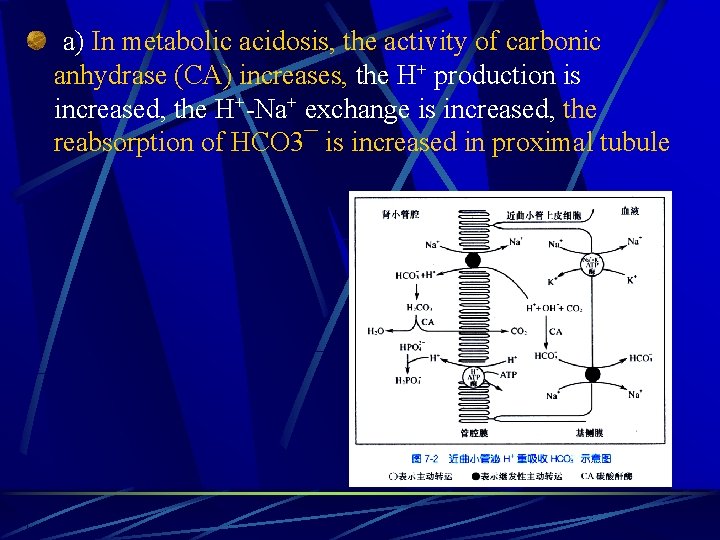
a) In metabolic acidosis, the activity of carbonic anhydrase (CA) increases, the H+ production is increased, the H+-Na+ exchange is increased, the reabsorption of HCO 3¯ is increased in proximal tubule
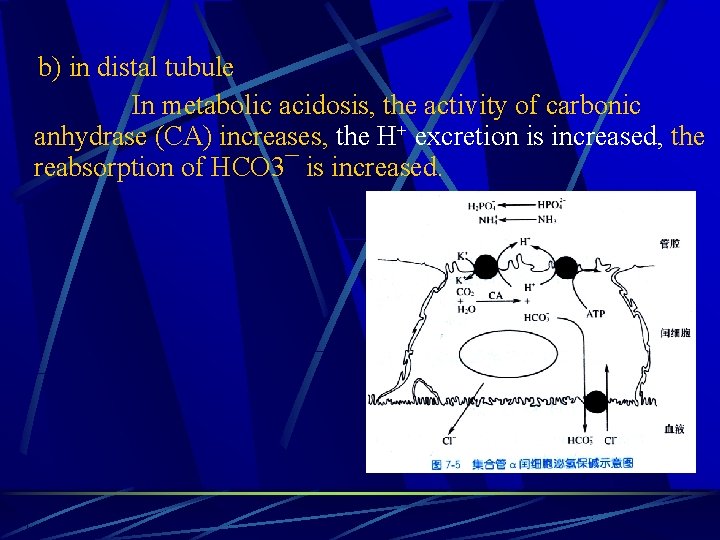
b) in distal tubule In metabolic acidosis, the activity of carbonic anhydrase (CA) increases, the H+ excretion is increased, the reabsorption of HCO 3¯ is increased.

c) In metabolic acidosis, the activity of glutaminase is increased, more glutamine will be decomposed into HCO 3¯ and NH 4+. More NH 4+ is excreted into tubular lumen. Thus more HCO 3¯ will be reabsorpted to the blood.
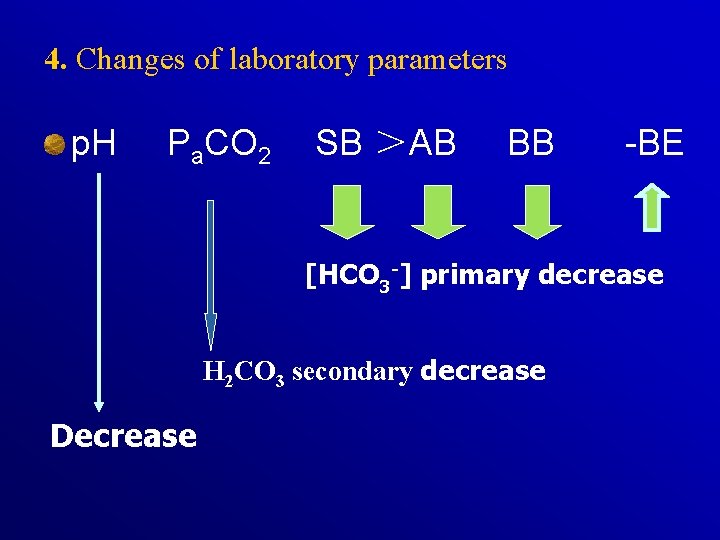
4. Changes of laboratory parameters p. H Pa. CO 2 SB >AB BB -BE [HCO 3 -] primary decrease H 2 CO 3 secondary decrease Decrease

5 Effects on the body The main manifestations are: l (A) effects on the cardiovascular system l (B) depression of mental activity l (C) hyperventilation
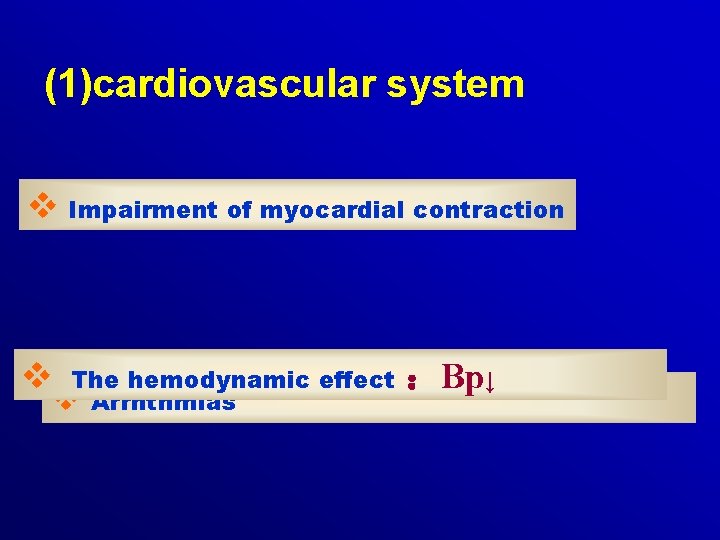
(1)cardiovascular system v Impairment of myocardial contraction v The hemodynamic effect :Bp↓ v Arrhthmias

(a) Impairment of myocardial contraction Ca 2+ combining with troponin will start the myocardial contraction. ①H+ is a competitive inhibitor for Ca 2+ combining with troponin. After H+ moves into the myocardial cells, the myocardial contraction is impaired. Severe acidosis may cause myocardial failure and low blood pressure because of the low cardiac output.

② The protein expression of L-type of voltagedependent calcium channel in myocardial cell membrane is reduced. Ca 2+ in-flow is reduced. ③ The protein expression of calcium channel in sarcoplasmic reticulum(SR) membrane is reduced. The release of Ca 2+ from SR is reduced.

(b) Arrhythmia due to hyperkalemia Causes of hyperkalemia: H+-K+ exchange of cell Decreased renal excretion of K+ Effect of hyperkalemia: Ventricular Arrhythmia

(c) The hemodynamic effect: Low BP due to arteriole dilation The reaction of arteriole to catecholamine (dopamine, adrenalin, noradrenalin) is decreased in acidosis, which leads to decrease of peripheral resistance and reduced venous return(precapillary sphincter dilate more obviously).

(2) Depression of mental activity (a) Manifestations: l slowness, tired, confused, coma, paralysis of the cardiovascular or respiratory centre (b) Mechanisms: l a) Increased [H+] causes cerebral vasodilatation. More blood supply will increase the CHP, then cause brain edema and high intracranial pressure. l b) High [H+] increases the permeability of cerebral blood vessels. Decreased plasma COP and increased interstitial COP can lead to brain edema. l c) Reduced ATP production.

Glutamic acid Glutamate decarboxylase r-GABA, r- gama aminobutyric acid gama γ-GABA transminase Succinic acid d) The production of GABA (gama aminobutyric acid, a inhibitory transmitter) is increased due to the activity of enzyme for the production is increased, and the activity of enzyme for the decomposition is decreased in low p. H (acidosis).

3. respiratory system 4. skeletal system

6. Principle of treatment for metabolic acidosis treatment of primary disease supplement of base: Na. HCO 3 , Sodium lactate Prevent electrolytic disorder(hypokalemia, hypocaicemia )
![Prevent the hypokalemia and hypocalcemia during treatment After the correction of acidosis, the [K+] Prevent the hypokalemia and hypocalcemia during treatment After the correction of acidosis, the [K+]](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-101.jpg)
Prevent the hypokalemia and hypocalcemia during treatment After the correction of acidosis, the [K+] will fall down rapidly by moving into the cells. In acidosis, [Ca 2+] increases, [Ca 2+] reduces during the correcting of acidosis. OH Ca 2+ ------→combining calcium Ca 2+ ←------combining calcium H+

II、 Respiratory acidosis 1. concept Characterized by a primarily increase in the Pa. CO 2 and a low p. H.

2. Causes and Pathogenesis The basic reasons: l (a) decreased ventilation, which leads to the decreased elimination of CO 2 from lung; l (b) increased inhalation of CO 2.

(1) Acute respiratory acidosis a) depression of respiratory center by cerebral diseases (trauma, infections) and drugs (over-dosage of anesthetics, sedatives) b) neuromuscular disorders (acute hypokalemia, periodic paralysis , myasthenia gravis , poliomyelitis, Guillain-Barre syndrome), c) cardiopulmonary arrest. d) obstruction of respiratory tract. e) mis-operating of respirator.

(2) Chronic respiratory acidosis Chronic obstructive pulmonary diseases (emphysema, chronic bronchitis with hypoventilation) cause the chronic respiratory acidosis. Chest wall diseases (fracture of rib) Brain tumors (affecting the respiratory center in which the ventilation is decreased)
![3. Compensation of respiratory acidosis (1) Non-[HCO 3¯ ]/[H 2 CO 3] buffering systems 3. Compensation of respiratory acidosis (1) Non-[HCO 3¯ ]/[H 2 CO 3] buffering systems](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-106.jpg)
3. Compensation of respiratory acidosis (1) Non-[HCO 3¯ ]/[H 2 CO 3] buffering systems (2) Cellular compensation H+ moves into the cell CO 2 moves into the cell (3)The renal compensation ( How about buffer pair: [HCO 3¯ ]/[H 2 CO 3] and respiratory compensation? )
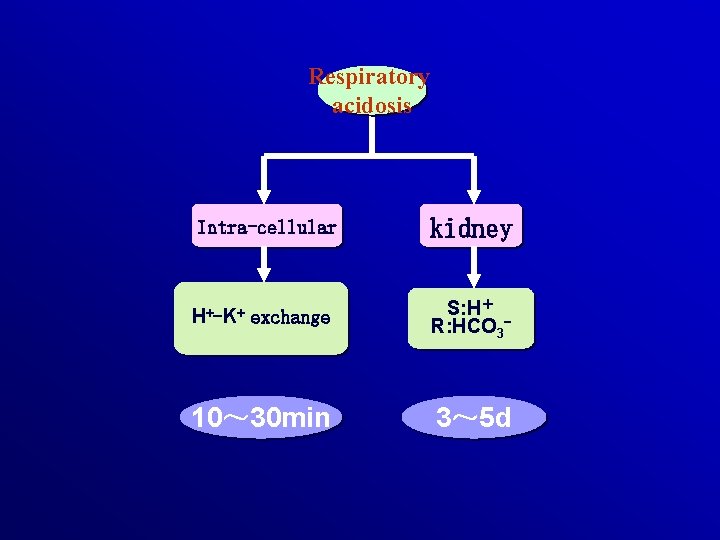
Respiratory acidosis Intra-cellular kidney H+-K+ exchange S: H+ R: HCO 3 - 10~ 30 min 3~ 5 d
![(1)intracellular buffering: acute v RBC CA CO 2 ↑ [HCO 3 - ] ↑ (1)intracellular buffering: acute v RBC CA CO 2 ↑ [HCO 3 - ] ↑](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-108.jpg)
(1)intracellular buffering: acute v RBC CA CO 2 ↑ [HCO 3 - ] ↑ CO 2+H 2 O→H 2 CO 3 HCO 3 - Cl- K+ CO 2+H 2 O→H 2 CO 3 H+ H+ +Hb. HHb [K+]↑ limited,△Pa. CO 210 mm. Hg/ △ [HCO 3 - ] 0. 7 -1 mmol/L
![(2)renal compensation: chronic v Pa. CO 2↑, [H+] ↑→CA activity↑→ secrete H+ in kidney (2)renal compensation: chronic v Pa. CO 2↑, [H+] ↑→CA activity↑→ secrete H+ in kidney](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-109.jpg)
(2)renal compensation: chronic v Pa. CO 2↑, [H+] ↑→CA activity↑→ secrete H+ in kidney v Pa. CO 2↑, [H+] ↑→ GT activity↑→ secrete NH 3 v HCO 3 - reabsorption v urine p. H↓ Powerful effect,△Pa. CO 210 mm. Hg/ △ [HCO 3 - ] 3. 54. 0 mmol/L。
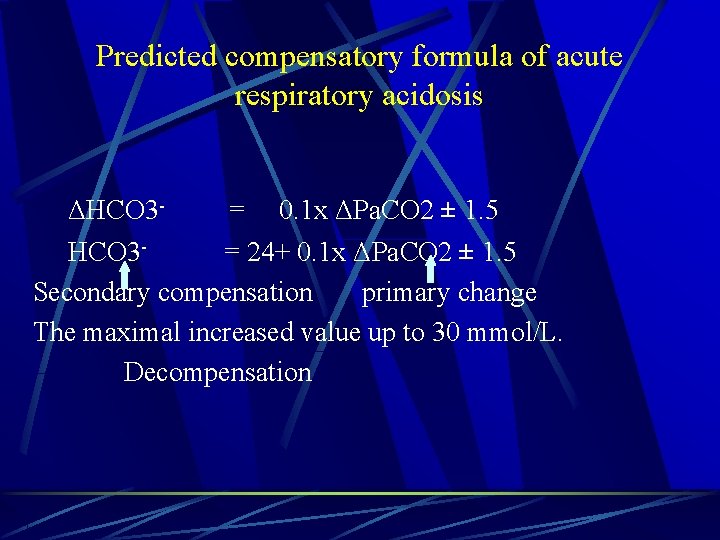
Predicted compensatory formula of acute respiratory acidosis ΔHCO 3 - = 0. 1 x ΔPa. CO 2 ± 1. 5 HCO 3 - = 24+ 0. 1 x ΔPa. CO 2 ± 1. 5 Secondary compensation primary change The maximal increased value up to 30 mmol/L. Decompensation

Predicted compensatory formula of chronic respiratory acidosis ΔHCO 3 - = 0. 4 x ΔPa. CO 2 ± 3 HCO 3 - = 24+0. 4 x ΔPa. CO 2 ± 3 Secondary compensation primary change Value measured > value predicted: with metabolic alkalosis Value measured < value predicted: with metabolic acidosis. Maximal compensatory value up to: 45 mmol/L
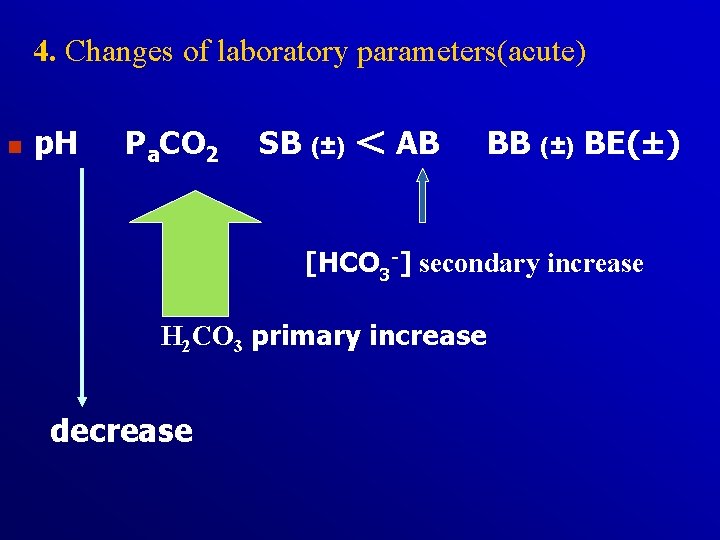
4. Changes of laboratory parameters(acute) n p. H Pa. CO 2 SB (±) < AB BB (±) BE(±) [HCO 3 -] secondary increase H 2 CO 3 primary increase decrease
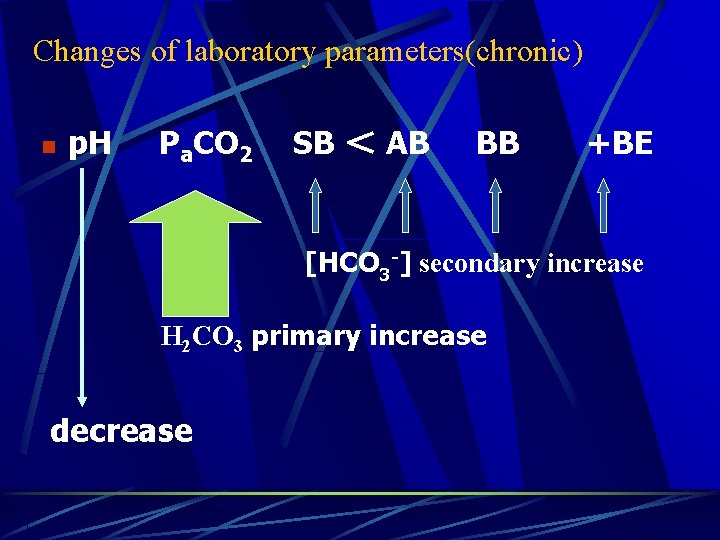
Changes of laboratory parameters(chronic) n p. H Pa. CO 2 SB < AB BB +BE [HCO 3 -] secondary increase H 2 CO 3 primary increase decrease

5. Effect of respiratory acidosis Same as metabolic acidosis,but CNS manefistation is more serious v cerebral blood flow increase(CO 2 dilate vessel;contract vessel viaαreceptor): No αreceptor on cerebral vessels → CO 2 dilate cerebral vessels→intracranial pressure ↑→headache vpulmonary encephalopathy : vcarbon dioxide narcosis: Pa. CO 2 > 80 mm. Hg vcardiovascular : like metabolic acidosis

6. treatment priciples (a) Treat the primary diseases which cause respiratory acidosis. (antibiotic, antispastic drugs) (b) Improve properly the ventilation. (c) Prevent from (respiratory alkalosis) over-ventilation during artificial respiration. (d) Be careful to alkaline drug(Na. HCO 3) THAM

III、 Metabolic alkalosis 1. concept Characterized by a primarily elevation in plasma HCO 3 - concentration and a high p. H.

2. Classification According to therapeutic effect of 0. 9% Na. Cl, (A) saline-responsive alkalosis (B) saline-resistant alkalosis

3. Pathogenesis (1) saline-responsive alkalosis (a) Increased loss of H+ (b) More administration of HCO 3¯ or precursors of bicarbonate

(a) Increased loss of H+ a) from stomach There is a lot of H+ in the gastric juice. Vomiting and gastric suction will lose H+ [HCO 3 - ] cl- is lost through gastric juice hypochloremic alkalosis HCO 3 - is absorbed into blood in stomach, then to intestinal juice to neutralize H+. Hypokalemia alkalosis Effective blood volumedecrease secondary aldosterone increase
b) Increased loss H+ from kidneys Some diuretics (e. g. furosemide) can inhibit the reabsorption of Cl¯ and Na+ in loop, more Na+ is reabsorpted with HCO 3¯ (without Cl ¯) in distal tubules; renal fluid folw rate increase

(b) More administration of HCO 3¯ or precursors of bicarbonate a) Patients with gastric ulcer may be orally given excessive Na. HCO 3 to neutralize gastric juice. b)Sharp correction of acidosis by excessive alkali administration can lead to metabolic alkalosis. c) Transfusion of anticoagulant blood with sodium citrate. .

(2) Chloride-resistant type Primary hyperaldosteronism Secondary hyperaldosteronism caused by: hypovolemia Cushing’s syndrome severe hypokalemia: Paradoxical acid urine

4. Compensation of metabolic alkalosis The compensation of metabolic alkalosis is the opposite direction of the compensation in metabolic acidosis.
![(1) Respiratory compensation :[H+] →pulmonary ventilation volume → CO 2 elimination → Pa. CO (1) Respiratory compensation :[H+] →pulmonary ventilation volume → CO 2 elimination → Pa. CO](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-124.jpg)
(1) Respiratory compensation :[H+] →pulmonary ventilation volume → CO 2 elimination → Pa. CO 2↑ →HCO 3 -/H 2 CO 3(quickly, limted) But 46<Pa. CO 2<60 mm. Hg, respiratory center is excited , Pa. CO 2 seldom higher than 55 mm. Hg △ Pa. CO 2=0. 7×△[ HCO 3- ]± 5 Value measured > value predicted: with respiratory acidosis Value measured < value predicted: with respiratory alkalosis
![(2) intracellular buffering [H+] Renal tubule lumen in ECF H+ 血K+ H++Pr- HPr H+ (2) intracellular buffering [H+] Renal tubule lumen in ECF H+ 血K+ H++Pr- HPr H+](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-125.jpg)
(2) intracellular buffering [H+] Renal tubule lumen in ECF H+ 血K+ H++Pr- HPr H+ K+ K+ Na+ alkalosis → hypokalemia

(3).renal compensation v secrte H+ ↓ v secrete NH 3↓ v reabsorb HCO 3 -↓ v urine p. H But hypokalemia-alkalosis excrete aciduria
![paradoxical aciduria Serum [K+]↓ K+ H+ cell K+ H+ Renal tubule lumen K+ H+ paradoxical aciduria Serum [K+]↓ K+ H+ cell K+ H+ Renal tubule lumen K+ H+](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-127.jpg)
paradoxical aciduria Serum [K+]↓ K+ H+ cell K+ H+ Renal tubule lumen K+ H+ Na+↓ Na+↑ Urine [H+ ] ↑ Hypokalemia alka

5. Effects on the body a) Effects on the central nervous system. b) The left-shift of oxygen-hemoglobin dissociation curve c) Decrease of ionized calcium (Ca 2+) in plasma d) Hypokalemia

(1) Effects on the central nervous system Manifestations: Excitability is increased. dysphoria (agitation), fatigue (very tired), malaise (discomfort), delirium ( mental disturbance with wild talk and wild excitement), confusion, stupor coma. The production of GABA (gama aminobutyric acid, a inhibitory transmitter), is decreased due to the activity of enzyme for the production is reduced in alkalosis. Hypoxia
![(2). increase in neuromuscular excitability p. H , [Ca 2+]↓ 手足搐搦 (Carpopedal Spasm) (2). increase in neuromuscular excitability p. H , [Ca 2+]↓ 手足搐搦 (Carpopedal Spasm)](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-130.jpg)
(2). increase in neuromuscular excitability p. H , [Ca 2+]↓ 手足搐搦 (Carpopedal Spasm)
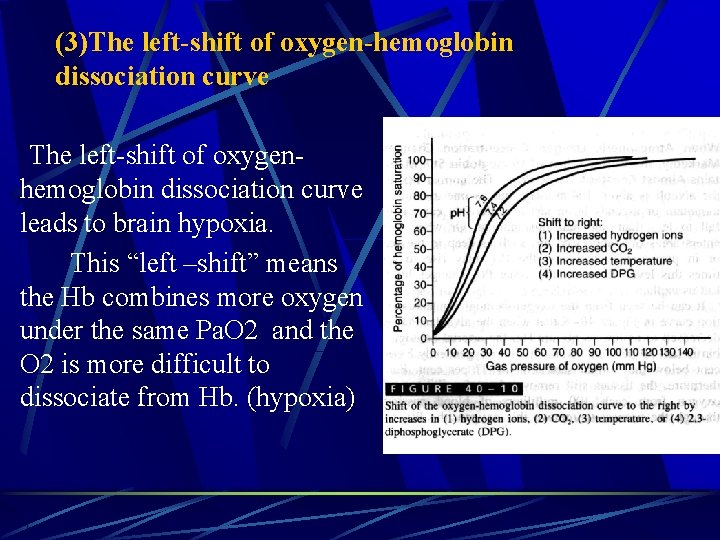
(3)The left-shift of oxygen-hemoglobin dissociation curve The left-shift of oxygen- hemoglobin dissociation curve leads to brain hypoxia. This “left –shift” means the Hb combines more oxygen under the same Pa. O 2 and the O 2 is more difficult to dissociate from Hb. (hypoxia)

(4)Hypokalemia Causes: (a)H+ shifts out of the cells as the compensation of alkalosis. Therefore the K+ moves into the cells as an exchange for electroequilibrium. (b) More K+ is in the cells including in the renal tubular cells, so the excretion of K+ from kidneys is increased. Manifestations: arrhythmias

Predicted compensatory formula ΔPa. CO 2(mm. Hg) = 0. 7 x ΔHCO 3 - ± 5 Secondary compensation primary change Or: Pa. CO 2=40+0. 7 x. HCO 3 -± 5 Pa. CO 2 can increase maximal to 55 mm. Hg. Value measured > value predicted: with respiratory acidosis Value measured < value predicted: with respiratory alkalosis

6. Changes of laboratory parameters n p. H Pa. CO 2 SB < AB BB BE [HCO 3 -] primary increase H 2 CO 3 secondary increase
![7. Principle of treatment. For chloride-sensitive type (A) Replenish 0. 9% Na. Cl [Na+] 7. Principle of treatment. For chloride-sensitive type (A) Replenish 0. 9% Na. Cl [Na+]](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-135.jpg)
7. Principle of treatment. For chloride-sensitive type (A) Replenish 0. 9% Na. Cl [Na+] [Cl-]( mmol/L) ----------------------------0. 9%Na. Cl 154 Plasma 140 104 ----------------------------a) Dilute the [HCO 3 -] b) Increase the blood volume, reduce the reabsorption of HCO 3 -. c) increased Cl- in distal tubule leads to increased excretion of HCO 3 - in collecting duct.
![(B) Replenish NH 4 Cl to increase the [Clˉ] and blood volume. (C) Replenish (B) Replenish NH 4 Cl to increase the [Clˉ] and blood volume. (C) Replenish](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-136.jpg)
(B) Replenish NH 4 Cl to increase the [Clˉ] and blood volume. (C) Replenish KCl for the patients with potassium deficiency. (D) secerious metabolic alkalosis:

For chloride-resistant type Treating of underlying disorders Antagonists of aldosterone Replenish KCl Acetazolamide (inhabit the CA activity) for the patients with edema with alkalosis.

IV Respiratory alkalosis 1. concept Characterized by a primarily reduction in the Pa. CO 2 and a high p. H.

2. Causes and Pathogenesis The only pathogenesis is the increased alveolar ventilation (hyperventilation). The basic reason of hyperventilation is the stimulation of respiratory center. Anxiety Hysteria Central nervous diseases Gram-negative septicemia Fever Salicylate intoxication Hypoxia due to high altitude, pulmonary disease Mis-operation of mechanical ventilator
![3. Compensation of respiratory alkalosis (1)intracellular buffering :acute alkalosis [H 2 CO 3]↓ v 3. Compensation of respiratory alkalosis (1)intracellular buffering :acute alkalosis [H 2 CO 3]↓ v](http://slidetodoc.com/presentation_image_h/1283ba97952aa8c8d1a3044b779e873b/image-140.jpg)
3. Compensation of respiratory alkalosis (1)intracellular buffering :acute alkalosis [H 2 CO 3]↓ v RBC H 2 CO 3 HCO 3 - + H+ [K+]↓ K+ CO 2 H+ K+ H 2 CO 3 HHb H++HCO 3 Cl- v plasma Cl- HCO 3

Predicted compensatory formula for acute respiratory alkalosis ΔHCO 3 - = 0. 2 x ΔPa. CO 2 ± 2. 5 HCO 3 - = 24+0. 2 x (Pa. CO 2 -40)± 2. 5 Secondary compensation primary change Value measured > value predicted: with metabolic alkalosis Value measured < value predicted: with metabolic acidosis. Maximal compensatory value up to: 18 mmol/L

(2)renal compensation: chronic alaklosis v v Secrete H+ ↓ Secrete NH 3↓ HCO 3 - reabsorption↓ Urine p. H Compensation △[ HCO 3- ] =0. 5 ×△ Pa. CO 2 ± 2. 5

Predicted compensatory formula for chronic respiratory alkalosis ΔHCO 3 - = 0. 5 x ΔPa. CO 2 ± 2. 5 HCO 3 - = 24+0. 5 x (Pa. CO 2 -40)± 2. 5 Secondary compensation primary change Value measured > value predicted: with metabolic alkalosis Value measured < value predicted: with metabolic acidosis. Maximal compensatory value up to: 12 mmol/L
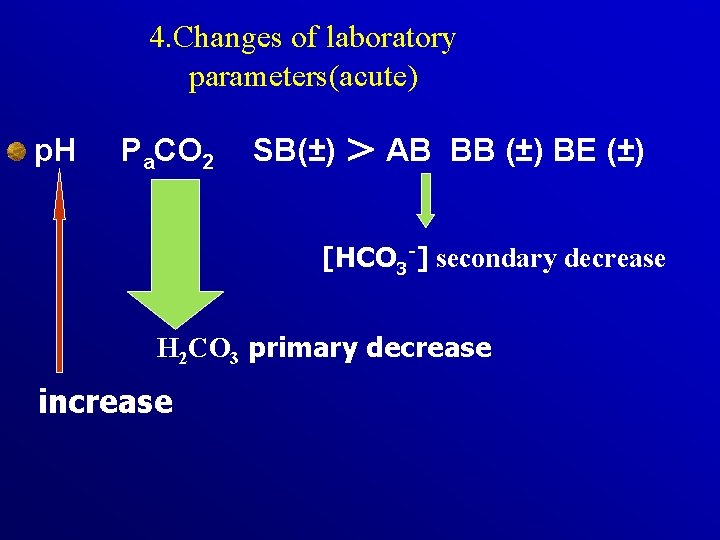
4. Changes of laboratory parameters(acute) p. H Pa. CO 2 SB(±) > AB BB (±) BE (±) [HCO 3 -] secondary decrease H 2 CO 3 primary decrease increase
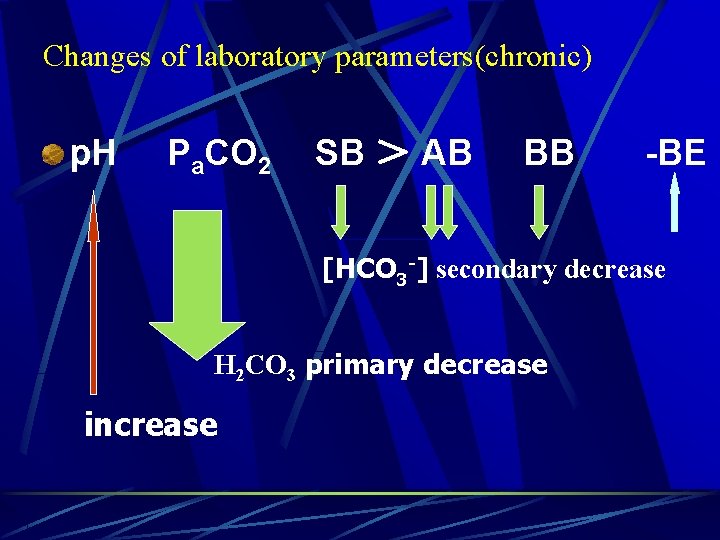
Changes of laboratory parameters(chronic) p. H Pa. CO 2 SB > AB BB -BE [HCO 3 -] secondary decrease H 2 CO 3 primary decrease increase
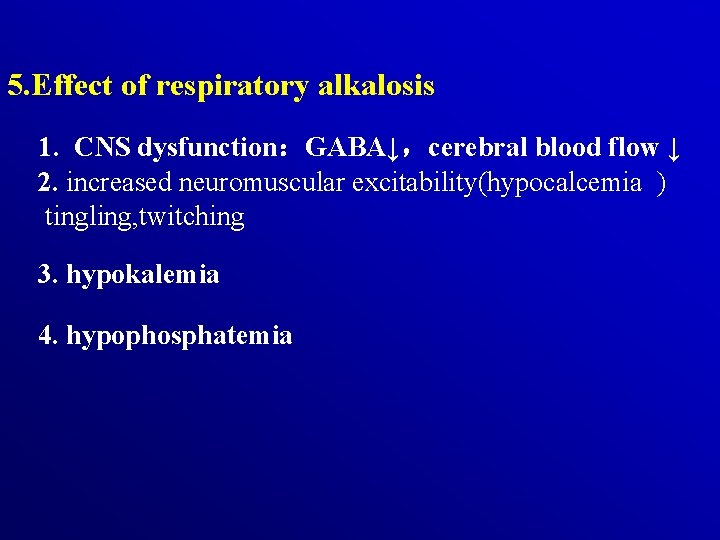
5. Effect of respiratory alkalosis 1. CNS dysfunction:GABA↓,cerebral blood flow ↓ 2. increased neuromuscular excitability(hypocalcemia ) tingling, twitching 3. hypokalemia 4. hypophosphatemia

6. treatment priciples Primary disease Prevent mis-operation of mechanical ventilator 5%CO 2 mixtrue gas inhalation or mask

V. Mixed Acid-base Disturbances Concept Metabolic acidosis, metabolic alkalosis, respiratory acidosis and respiratory alkalosis are four types of simple acid-base disturbance when these disturbances occur separately. A mixed acid-base disturbance is defined as the simultaneous co-existence of two or more simple disorders in the same patient. Double acid-base disorders Triple acid-base disorders


Mixed Acid-base Disturbances Any two or three simple acid-base disturbances can occur simultaneously in a patient except the respiratory acidosis and respiratory alkalosis, because one can never have hypoventilation and hyperventilation at the same time. Mixed acid-base disturbances occur frequently as a part of severe underlying illness with a high mortality.

Case discussion A 45 -year-old man had chronic cough for 20 years. He had a shortness of breath, orthopnea with edematous ankles for 1 month. The laboratory findings were: p. H = 7. 26 Pa. O 2=55 mm. Hg Pa. CO 2=60 mm. Hg AB = 22 mmol/L

Predicted: ΔHCO 3 - = 0. 4 x ΔPa. CO 2 ± 3 HCO 3 - = 24+0. 4 x 20 ± 3=29~35 Measured: 22 p. H reduced severely. No respiratory compensation for M. acidosis No renal compensation for R. acidosis Respiratory acidosis + metabolic acidosis
- Slides: 152