Chapter 4 3 Covalent structures Lewis electron dot



















- Slides: 24

Chapter 4. 3: Covalent structures Lewis (electron dot) structures show the electron domains in the valence shell and are used to predict molecular shape.

Important terms for this section �Lewis structure �Coordinate bond/dative bond �Electron deficient �VSEPR theory �Electron domain geometry � Linear � Trigonal planar � Bent � Tetrahedral �Molecular geometry � Linear � Tetrahedral � Bent � Trigonal pyramidal � Trigonal planar �Dipole moment �Delocalized electrons �Resonance �Giant molecular �Nanotechnology

Lewis structures �Lewis structures are used for covalent molecules only �Used to represent valence electrons �Steps: � 1. calculate total number of val. e- in molecule � 2. draw skeletal structure of molecule � 3. use crosses, dots or a line to show e- pairs � 4. add e- pairs to complete the octets (H only has 2) � 5. use double or triple bonds if necessary � 6. check that the total number of e- is equal to #1

�Notes: must add an e- for each negative charge (anion) or subtract one e- for each positive charge (cation) �Square brackets are needed for ions

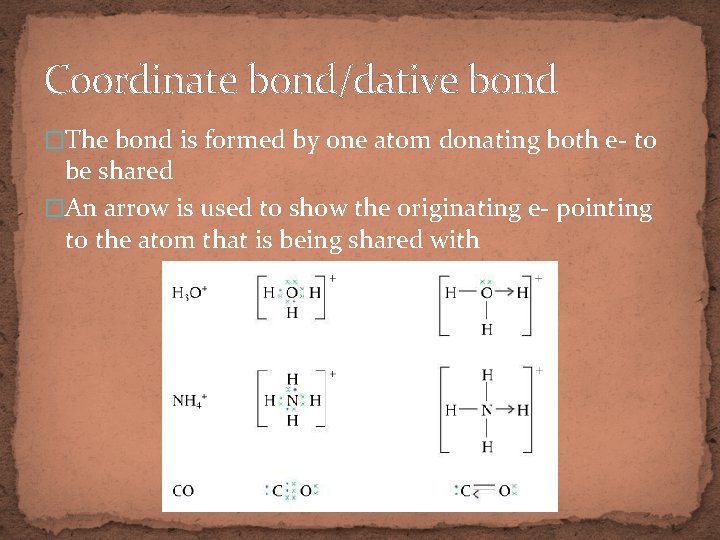
Coordinate bond/dative bond �The bond is formed by one atom donating both e- to be shared �An arrow is used to show the originating e- pointing to the atom that is being shared with

Incomplete octets (expanded in 14. 1) �Two primary exceptions: �Be and B �These are called electron deficient and are often the receivers of coordinate bonds

VSEPR theory �Valence Shell Electron Pair Repulsion theory �Electron pairs repel each other and will orient themselves as far away from each other as possible � This is lower energy and more stable �This is how the geometry is determined �Electron pairs and bonded pairs are referred to as electron domains: � Electron pair � Single bond � Double bond � Triple bond

VSEPR theory �Total number electron domains around central atom will determine geometric arrangement �Shape of molecule determined by angles between bonded atoms �Lone pairs have higher concentration of charge and therefore cause stronger repulsion than bonded pairs � Lone pair-lone pair > lone pair-bonding pair > bonding pair-bonding pair �Molecules with lone pairs have some distortions due to higher charge repulsion �Structures that do not have lone pairs ≠ Lewis structures

Two electron domains �Electron domain geometry: linear �Molecular geometry: linear

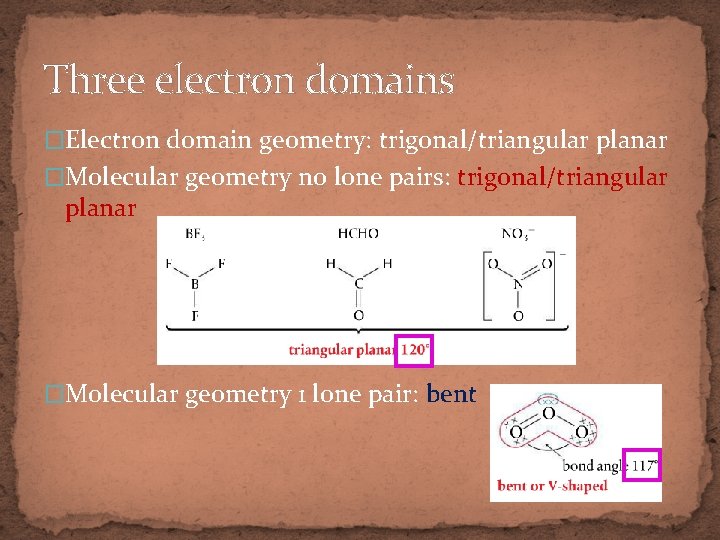
Three electron domains �Electron domain geometry: trigonal/triangular planar �Molecular geometry no lone pairs: trigonal/triangular planar �Molecular geometry 1 lone pair: bent

Four electron domains �Electron domain geometry: tetrahedral

Steps to determine shape of molecule � 1. draw Lewis structure � 2. count number of electron domains around central atom � 3. determine e- domain geometry: � 2 e- domains = linear � 3 e- domains = triangular planar � 4 e- domains = tetrahedral � 4. determine molecular geometry from the number of bonding e- domains � 5. consider extra repulsion caused by lone pairs and make adjustments

Polar bonds vs Polar molecules �Bonds can be polar while the molecule is still not considered polar… what? ? �Whether a molecule is polar depends on � Type of polar bonds � Orientation of bonds (shape) �If dipoles are equal in polarity AND symmetrically arranged: � Charge separation cancels out polar bonds = non-polar

Polar bonds vs Polar molecules �If bonds are different in polarity OR not symmetrically arranged: � Dipoles will not cancel = net dipole moment (overall dipole in molecule) � There is directionality in the bonds AND in the molecule!

Delocalized electrons & Resonance �Delocalized e- : electrons that are shared between more than 1 bonding position � Spread out e- increases stability of molecule � Occurs with molecules that have multiple bonds and the bond can be moved to another atom �Example: Ozone (O 3)

Delocalized e- & Resonance �O 3: how many bonds are on each oxygen? �Are they equal or different strength? �So the correct Lewis structure is: �Resonance: more than 1 valid Lewis structure can be drawn for the molecule � Resonance hybrid = liger � It is both a lion and tiger at the same time! Not switching!

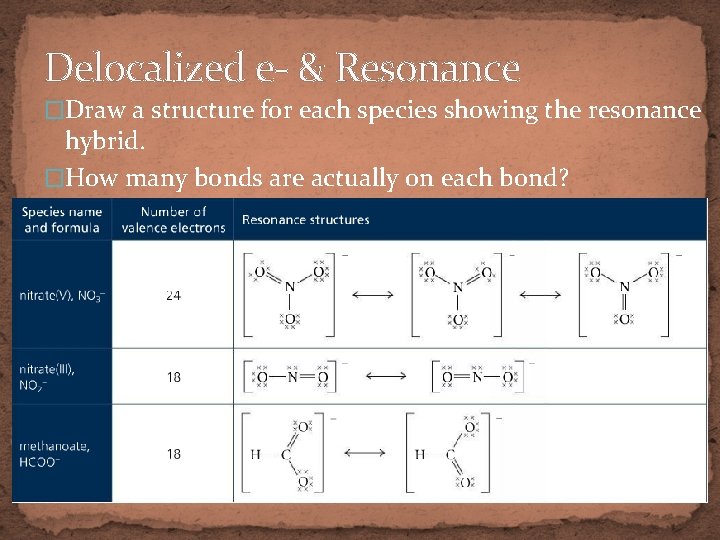
Delocalized e- & Resonance �Draw a structure for each species showing the resonance hybrid. �How many bonds are actually on each bond?

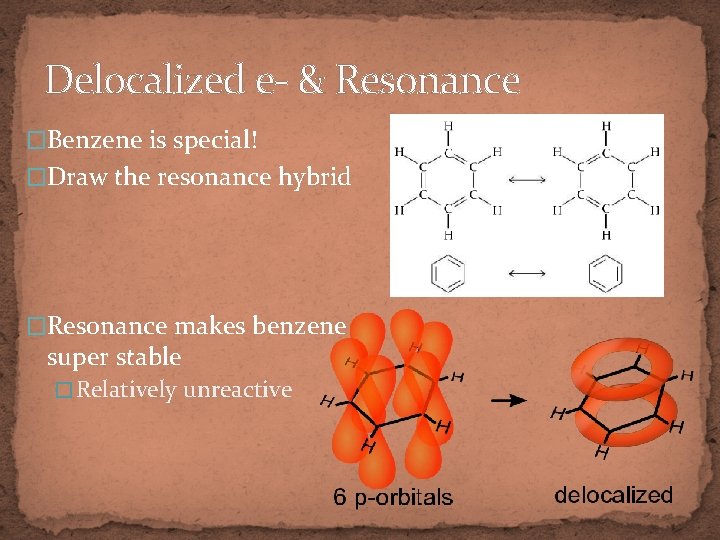
Delocalized e- & Resonance �Benzene is special! �Draw the resonance hybrid �Resonance makes benzene super stable � Relatively unreactive

Giant covalent crystalline solids �Giant molecular = network covalent = macromolecular structure �These are molecules of covalently bonded atoms that form giant structures (unlike the itty bitty methane CH 4, for example) �These have different properties than the smaller molecules

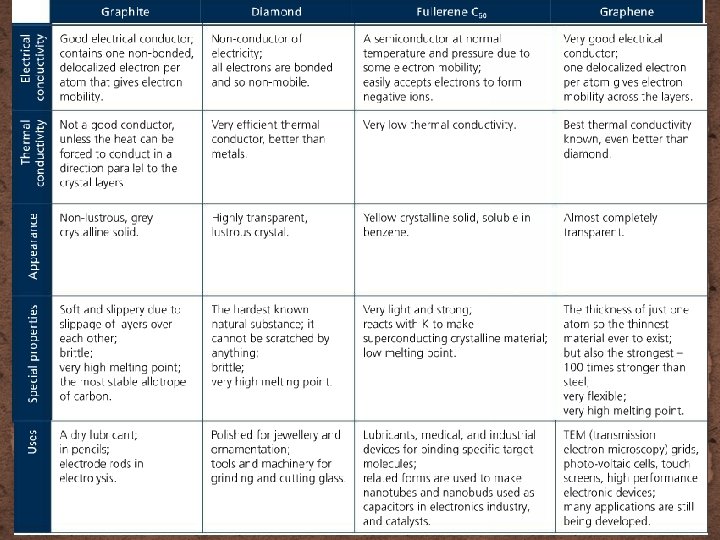
Carbon �Allotropes: different forms of an element in the same state of matter �Carbon solids can be in different forms � Graphite � Diamond � Fullerene C 60 � Graphene �All are made of only carbon atoms, but their structures are different and so have different properties

Carbon allotropes


Carbon allotropes – graphene and nanotechnology �Graphene is a relatively new form of carbon but expensive! � New conductive material: mix graphene with plastics � Transistors can be faster and smaller than Si transistors � Touch screens printed on plastic – light, flexible, almost unbreakable � Supercapacitors (store charge) replacing batteries, use in cars and mobile devices � Solar panels – graphene can be wrapped around surfaces such as clothing, furniture or vehicles �Nanotechnology: atomic scale manipulation of matter � Graphone: add H to graphene = variable magnetic properties � Flourographene: add F to graphene = rippled structure can make for an insulator

Silicon and Silicon dioxide �Silicon is right under carbon and forms 4 bonds � Second most abundant element in Earth’s crust (after O) �This can form a diamond-like structure for Si in elemental state �But in nature, tends to form silicon dioxide tetrahedral structures (sand/quartz/glass) �Here, Si. O 2 is actually only empirical �Properties: � Strong � Insoluble in H 2 O � High melting point � Non-conductor of electricity