Chapter 38 B Quantum Physics A Power Point

Chapter 38 B - Quantum Physics A Power. Point Presentation by Paul E. Tippens, Professor of Physics Southern Polytechnic State University © 2007

Objectives: After completing this module, you should be able to: • Discuss the meaning of quantum physics and Planck’s constant for the description of matter in terms of waves or particles. • Demonstrate your understanding of the photoelectric effect, the stopping potential, and the de. Broglie wavelength. • Explain and solve problems similar to those presented in this unit.

Plank’s Constant In his studies of black-body radiation, Maxwell Planck discovered that electromagnetic energy is emitted or absorbed in discrete quantities. Planck’s Equation: E = hf (h = 6. 626 x 10 -34 J s) Apparently, light consists of tiny bundles of energy called photons, each having a welldefined quantum of energy. Photon E = hf

Energy in Electron-volts Photon energies are so small that the energy is better expressed in terms of the electron-volt. One electron-volt (e. V) is the energy of an electron when accelerated through a potential difference of one volt. 1 e. V = 1. 60 x 10 -19 J 1 ke. V = 1. 6 x 10 -16 J 1 Me. V = 1. 6 x 10 -13 J

Example 1: What is the energy of a photon of yellow-green light (λ = 555 nm)? First we find f from wave equation: c = fλ E = 3. 58 x 10 -19 J Or E = 2. 24 e. V Since 1 e. V = 1. 60 x 10 -19 J

Useful Energy Conversion Since light is often described by its wavelength in nanometers (nm) and its energy E is given in e. V, a conversion formula is useful. (1 nm = 1 x 10 -9 m) If λ is in nm, the energy in e. V is found from: Verify the answer in Example 1. . .

The Photo-Electric Effect Incident light Cathode C - Anode A + Ammeter A When light shines on the cathode C of a photocell, electrons are ejected from A and attracted by the positive potential due to battery. There is a certain threshold energy, called the work function W, that must be overcome before any electrons can be emitted.
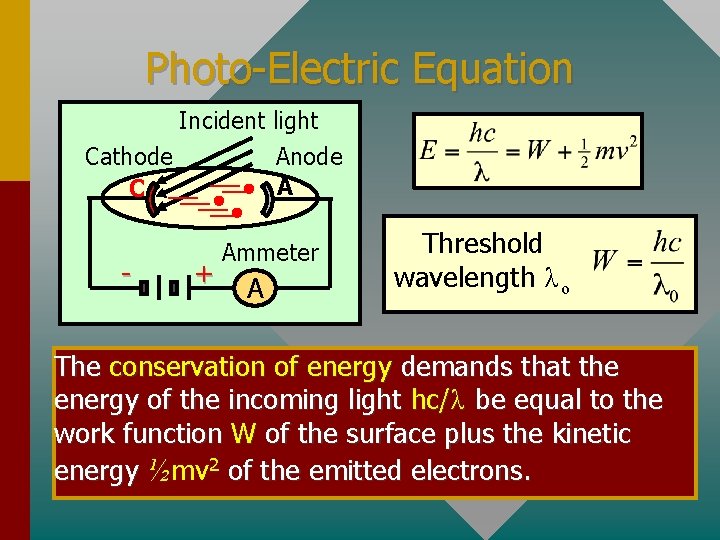
Photo-Electric Equation Incident light Cathode C - Anode A + Ammeter A Threshold wavelength λ ο The conservation of energy demands that the energy of the incoming light hc/λ be equal to the work function W of the surface plus the kinetic energy ½mv 2 of the emitted electrons.
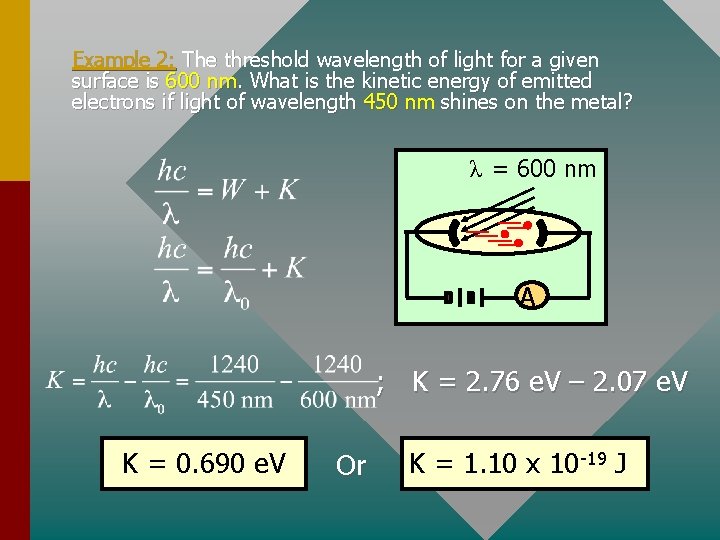
Example 2: The threshold wavelength of light for a given surface is 600 nm. What is the kinetic energy of emitted electrons if light of wavelength 450 nm shines on the metal? λ = 600 nm A ; K = 2. 76 e. V – 2. 07 e. V K = 0. 690 e. V Or K = 1. 10 x 10 -19 J
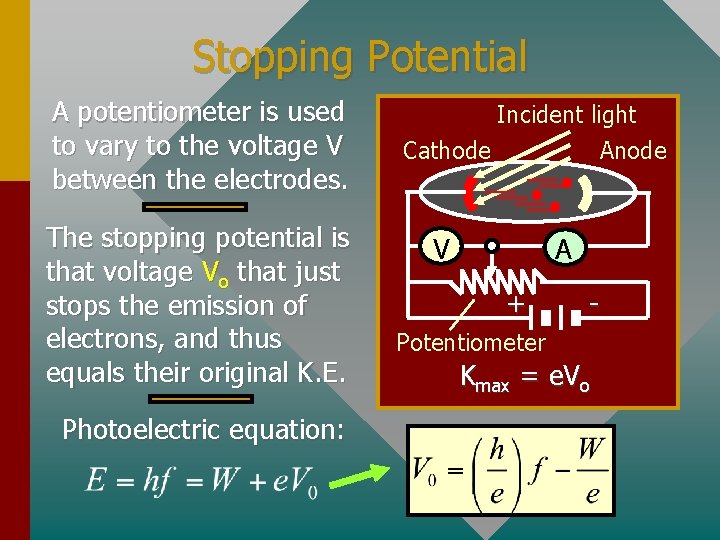
Stopping Potential A potentiometer is used to vary to the voltage V between the electrodes. The stopping potential is that voltage Vo that just stops the emission of electrons, and thus equals their original K. E. Photoelectric equation: Incident light Cathode Anode V A + - Potentiometer Kmax = e. Vo

Slope of a Straight Line (Review) The general equation for a straight line is: y = mx + b The x-intercept xo occurs when line crosses x axis or when y = 0. The slope of the line is the rise over the run: The slope of a line: y Slope y x xo x
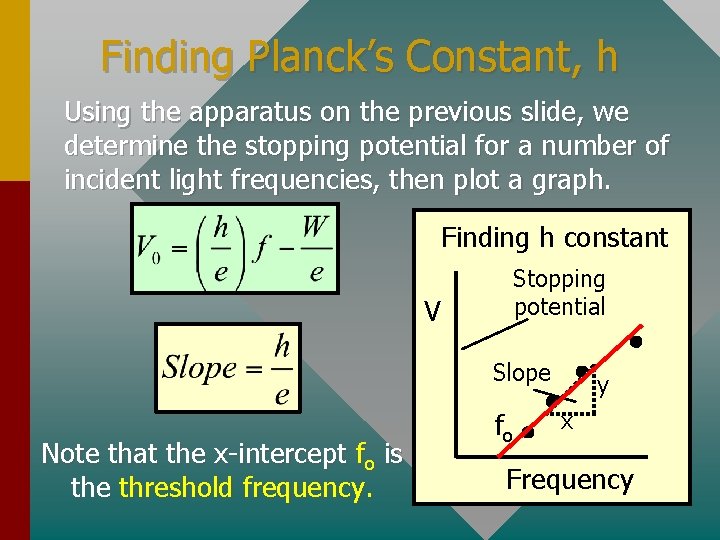
Finding Planck’s Constant, h Using the apparatus on the previous slide, we determine the stopping potential for a number of incident light frequencies, then plot a graph. Finding h constant V Stopping potential Slope Note that the x-intercept fo is the threshold frequency. fo y x Frequency

Example 3: In an experiment to determine Planck’s constant, a plot of stopping potential versus frequency is made. The slope of the curve is 4. 13 x 10 -15 V/Hz. What is Planck’s constant? Stopping potential V Slope fo y x Frequency h = e(slope) = (1. 6 x 10 -19 C)(4. 13 x 10 -15 V/Hz) Experimental Planck’s h = 6. 61 x 10 -34 J/Hz
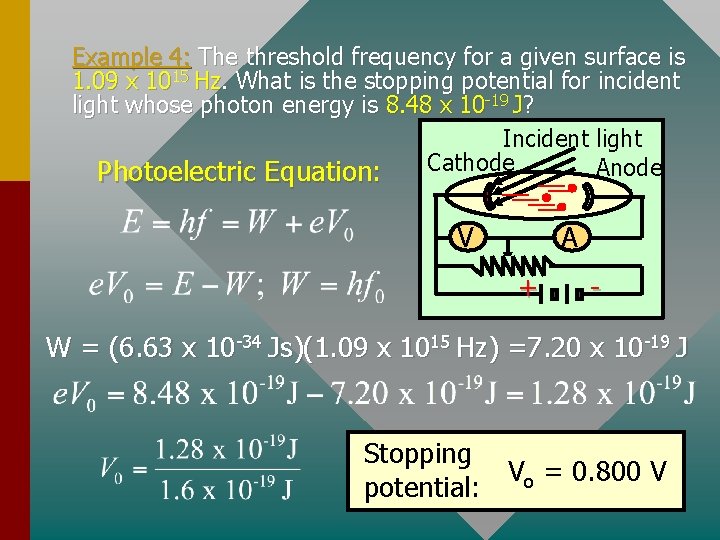
Example 4: The threshold frequency for a given surface is 1. 09 x 1015 Hz. What is the stopping potential for incident light whose photon energy is 8. 48 x 10 -19 J? Incident light Cathode Anode Photoelectric Equation: V A + - W = (6. 63 x 10 -34 Js)(1. 09 x 1015 Hz) =7. 20 x 10 -19 J Stopping Vo = 0. 800 V potential:

Total Relativistic Energy Recall that the formula for the relativistic total energy was given by: Total Energy, E For a particle with zero momentum p = 0: A light photon has mo = 0, but it does have momentum p: E = m o c 2 E = pc

Waves and Particles We know that light behaves as both a wave and a particle. The rest mass of a photon is zero, and its wavelength can be found from momentum. Wavelength of a photon: All objects, not just EM waves, have wavelengths which can be found from their momentum de Broglie Wavelength:
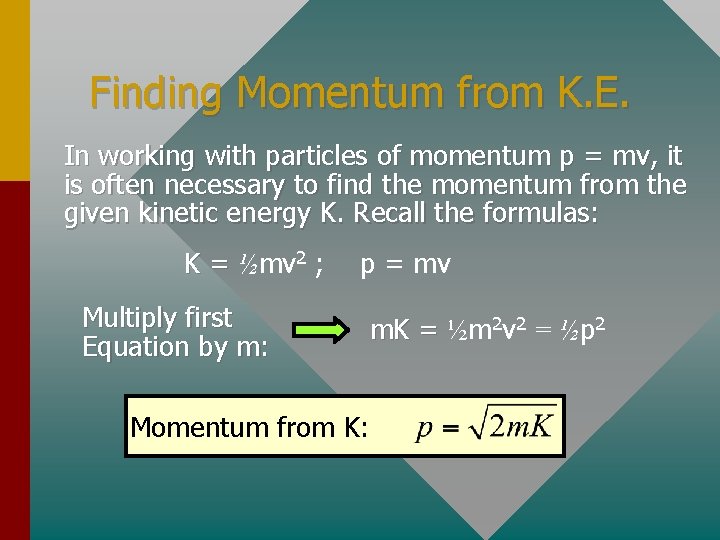
Finding Momentum from K. E. In working with particles of momentum p = mv, it is often necessary to find the momentum from the given kinetic energy K. Recall the formulas: K = ½mv 2 ; Multiply first Equation by m: p = mv m. K = ½m 2 v 2 = ½p 2 Momentum from K:

Example 5: What is the de Broglie wavelength of a 90 -e. V electron? (me = 9. 1 x 10 -31 kg. ) e- 90 e. V Next, we find momentum from the kinetic energy: p = 5. 12 x 10 -24 kg m/s λ = 0. 122 nm

Summary Apparently, light consists of tiny bundles of energy called photons, each having a welldefined quantum of energy. Planck’s Equation: E = hf Photon E = hf (h = 6. 626 x 10 -34 J s) The Electron-volt: 1 e. V = 1. 60 x 10 -19 J 1 ke. V = 1. 6 x 10 -16 J 1 Me. V = 1. 6 x 10 -13 J
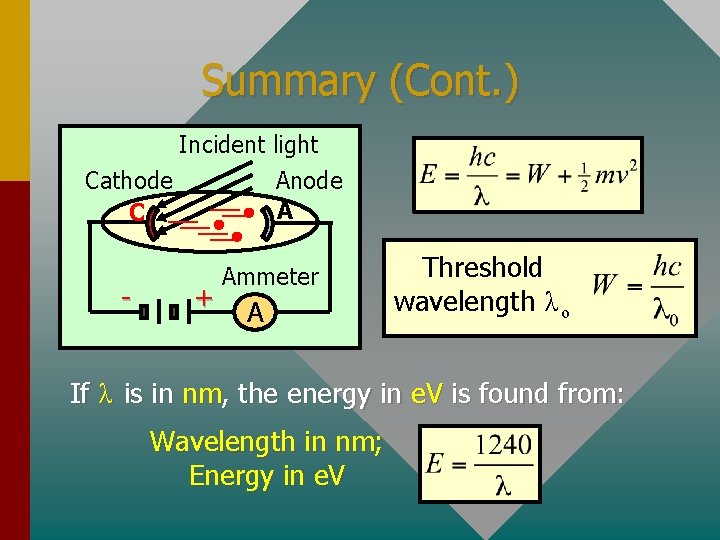
Summary (Cont. ) Incident light Cathode C - Anode A + Ammeter A Threshold wavelength λ ο If λ is in nm, the energy in e. V is found from: Wavelength in nm; Energy in e. V
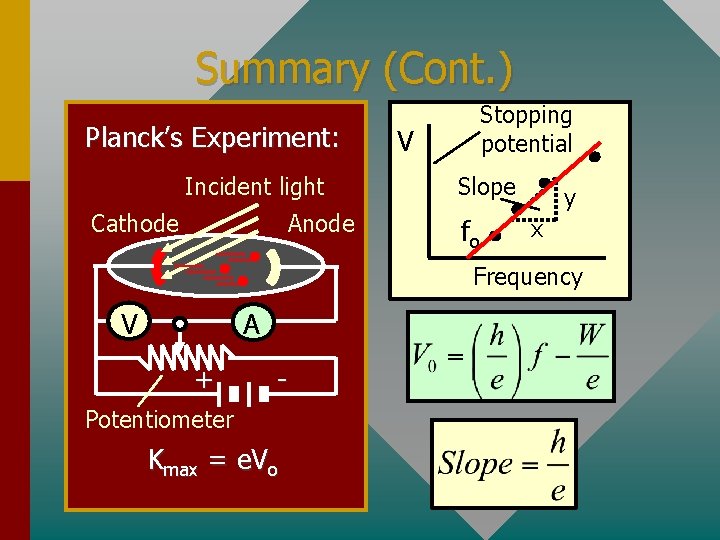
Summary (Cont. ) Planck’s Experiment: Incident light Cathode Anode Stopping potential V Slope fo y x Frequency V A + - Potentiometer Kmax = e. Vo

Summary (Cont. ) Quantum physics works for waves or particles: For a particle with zero momentum p = 0: A light photon has mo = 0, but it does have momentum p: Wavelength of a photon: E = m o c 2 E = pc de Broglie Wavelength:

CONCLUSION: Chapter 38 B Quantum Physics
- Slides: 23