Chapter 35 Serway Jewett 6 th Ed How













- Slides: 36

Chapter 35 Serway & Jewett 6 th Ed.

How to View Light As a Ray As a Wave As a Particle

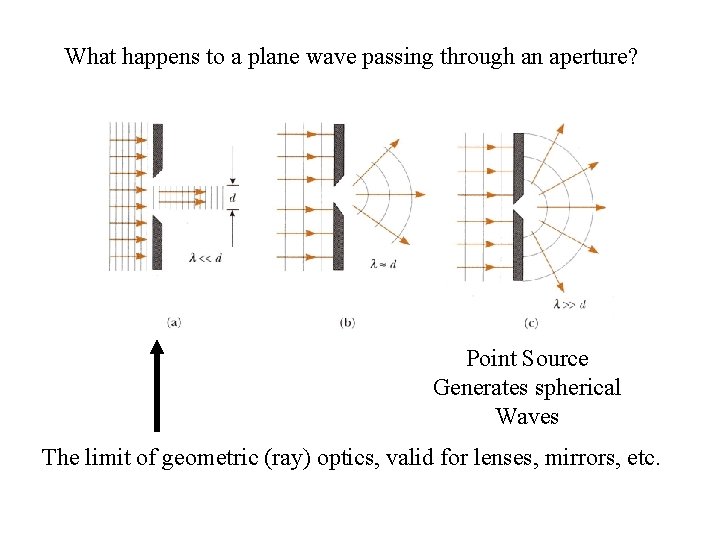
What happens to a plane wave passing through an aperture? Point Source Generates spherical Waves The limit of geometric (ray) optics, valid for lenses, mirrors, etc.

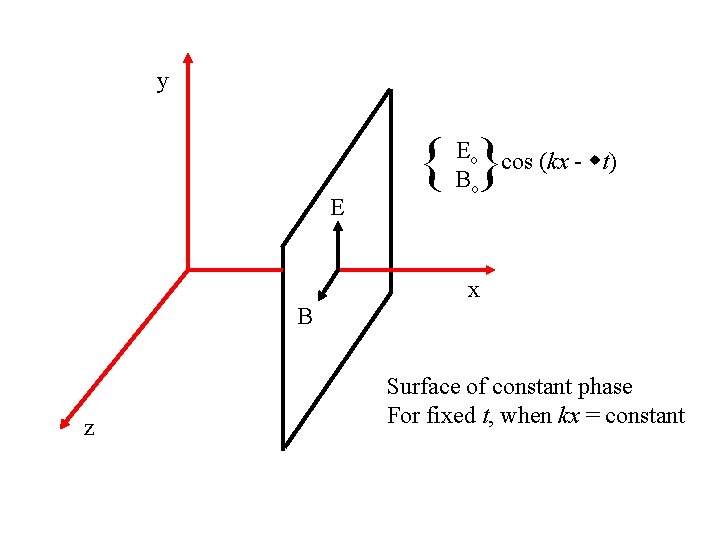
y { } E Eo cos (kx - t) Bo x B z Surface of constant phase For fixed t, when kx = constant

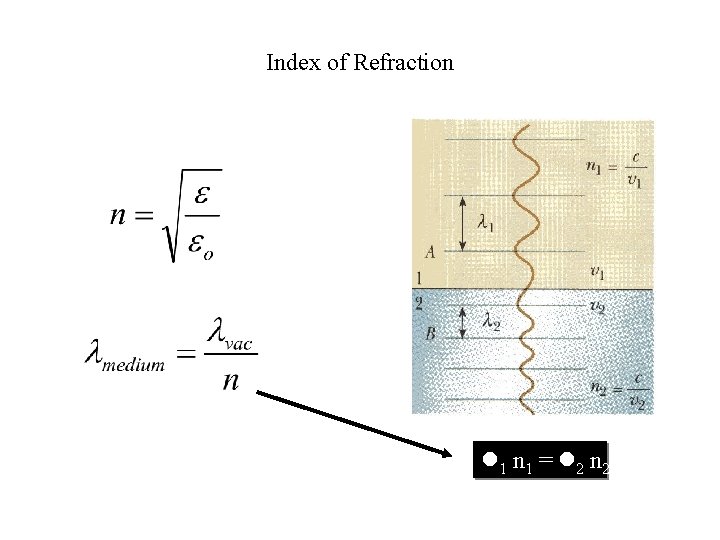
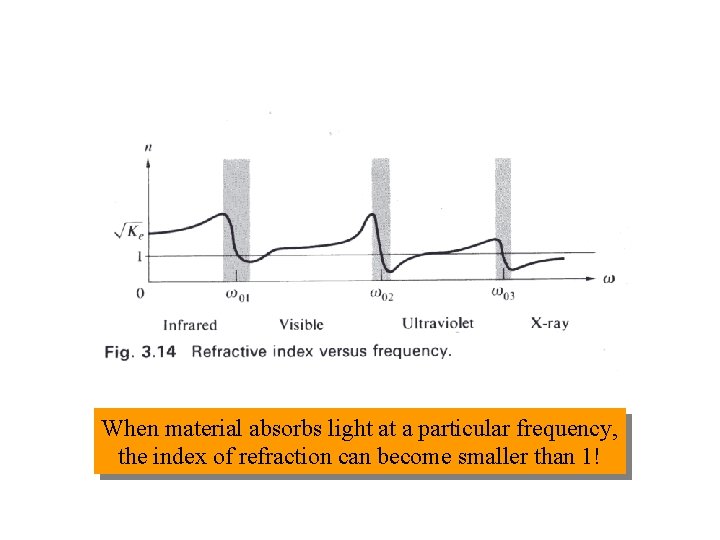
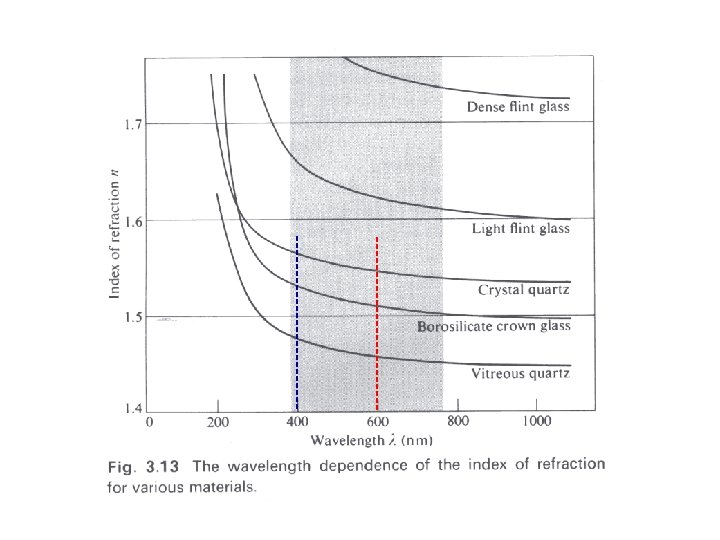
Index of Refraction 1 n 1 = 2 n 2


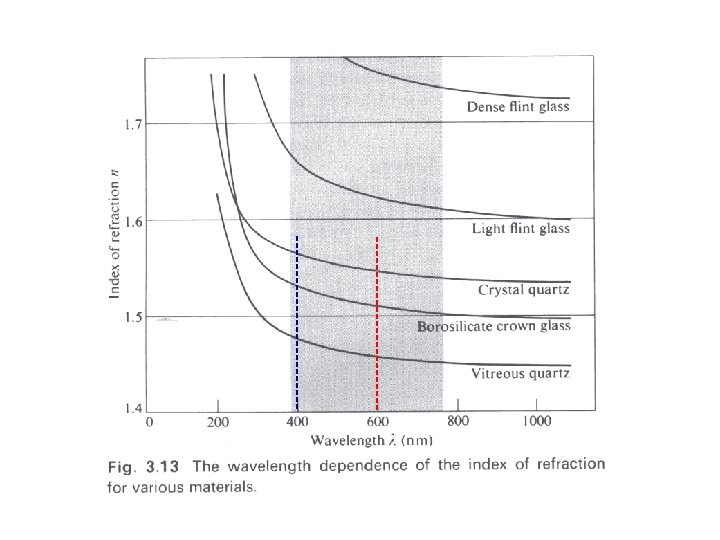
When material absorbs light at a particular frequency, the index of refraction can become smaller than 1!


Reflection and Refraction

Oct. 18, 2004

Fundamental Rules for Reflection and Refraction in the limit of Ray Optics 1. Huygens’s Principle 2. Fermat’s Principle 3. Electromagnetic Wave Boundary Conditions

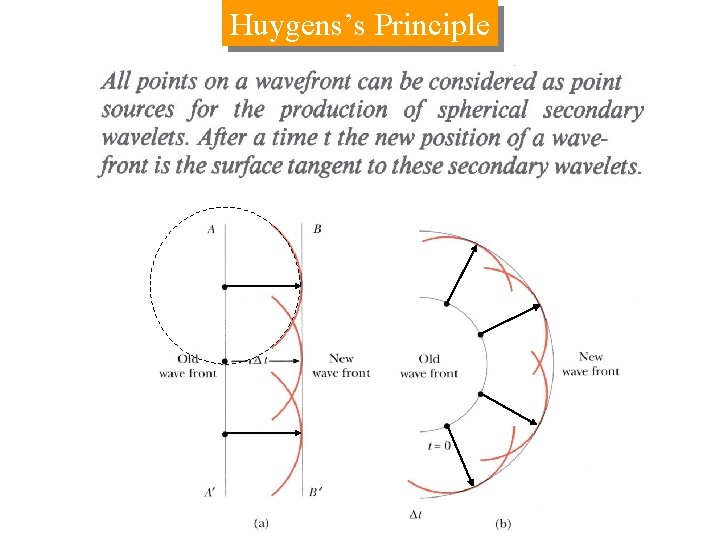
Huygens’s Principle

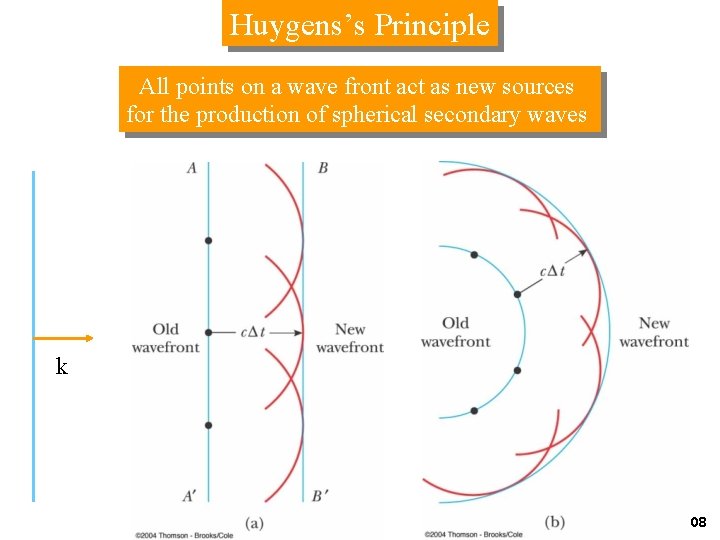
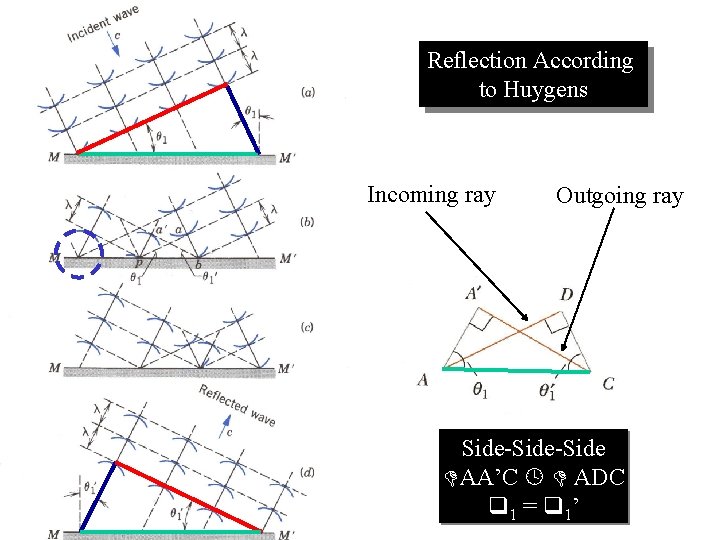
Huygens’s Principle All points on a wave front act as new sources for the production of spherical secondary waves k Fig 35 -17 a, p. 1108

Reflection According to Huygens Incoming ray Outgoing ray Side-Side AA’C ADC 1 = 1’

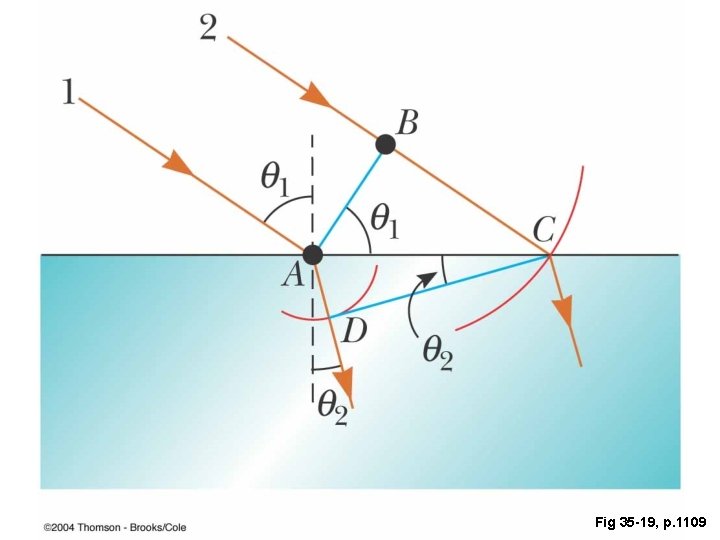
Refraction

Fig 35 -19, p. 1109

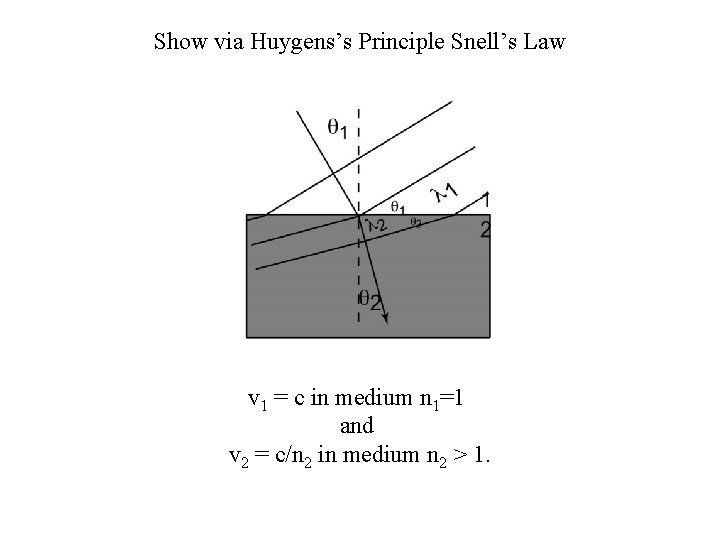
Show via Huygens’s Principle Snell’s Law v 1 = c in medium n 1=1 and v 2 = c/n 2 in medium n 2 > 1.

Fundamental Rules for Reflection and Refraction in the limit of Ray Optics ü Huygens’s Principle 2. Fermat’s Principle 3. Electromagnetic Wave Boundary Conditions

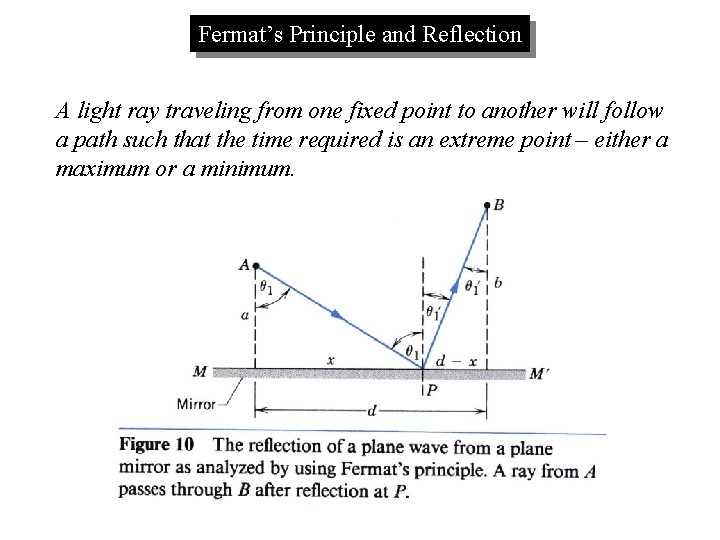
Fermat’s Principle and Reflection A light ray traveling from one fixed point to another will follow a path such that the time required is an extreme point – either a maximum or a minimum.

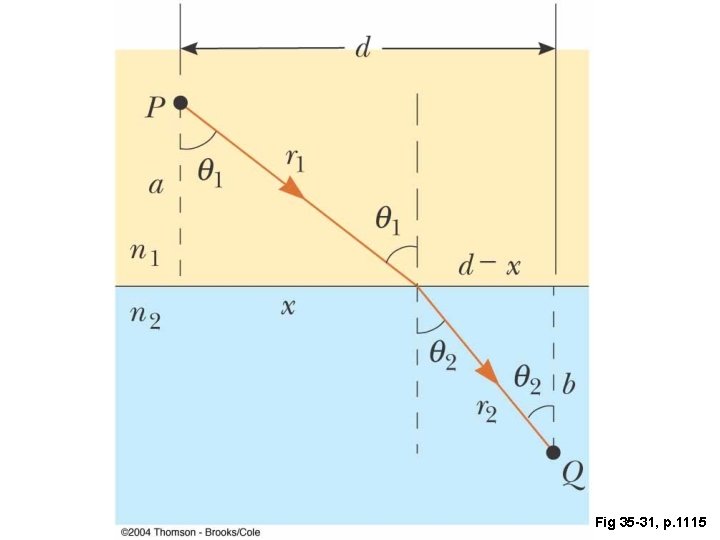
Fig 35 -31, p. 1115

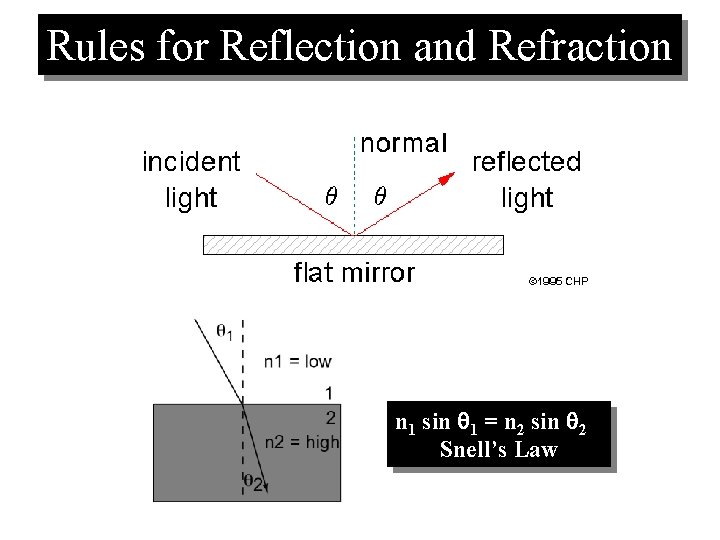
Rules for Reflection and Refraction n 1 sin 1 = n 2 sin 2 Snell’s Law

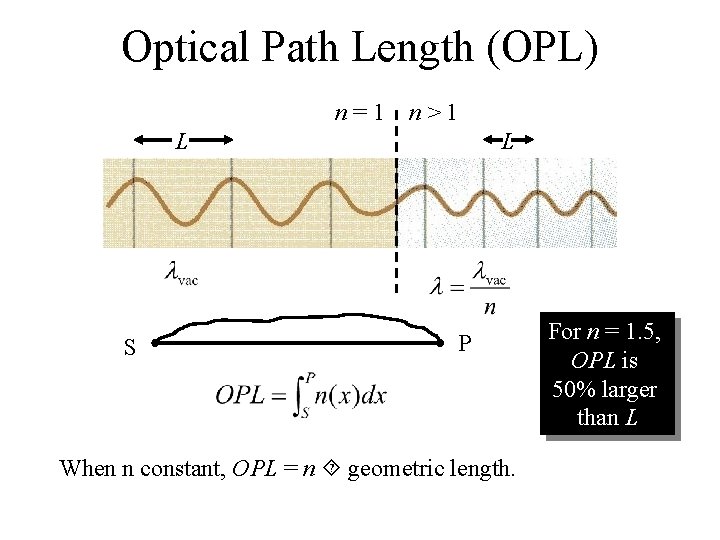
Optical Path Length (OPL) n=1 n>1 L L S P When n constant, OPL = n geometric length. For n = 1. 5, OPL is 50% larger than L

Fermat’s Principle, Revisited A ray of light in going from point S to point P will travel an optical path (OPL) that minimizes the OPL. That is, it is stationary with respect to variations in the OPL.

Fundamental Rules for Reflection and Refraction in the limit of Ray Optics ü Huygens’s Principle 1. Fermat’s Principle 3. Electromagnetic Wave Boundary Conditions

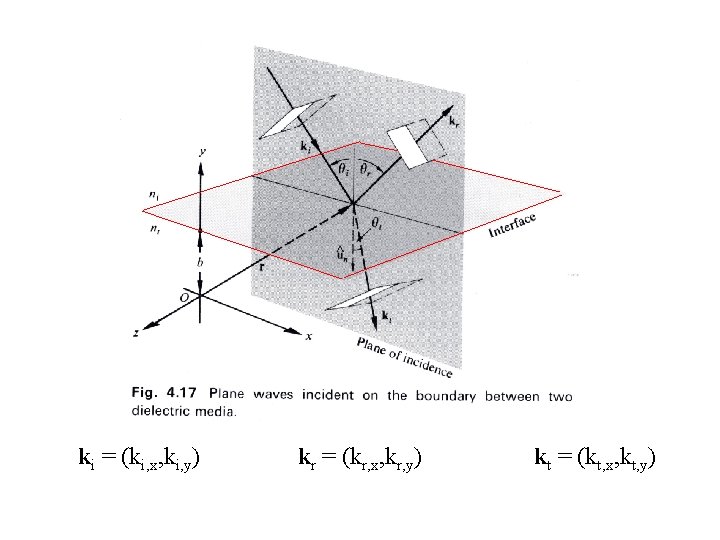
ki = (ki, x, ki, y) kr = (kr, x, kr, y) kt = (kt, x, kt, y)

Fig 35 -22, p. 1110


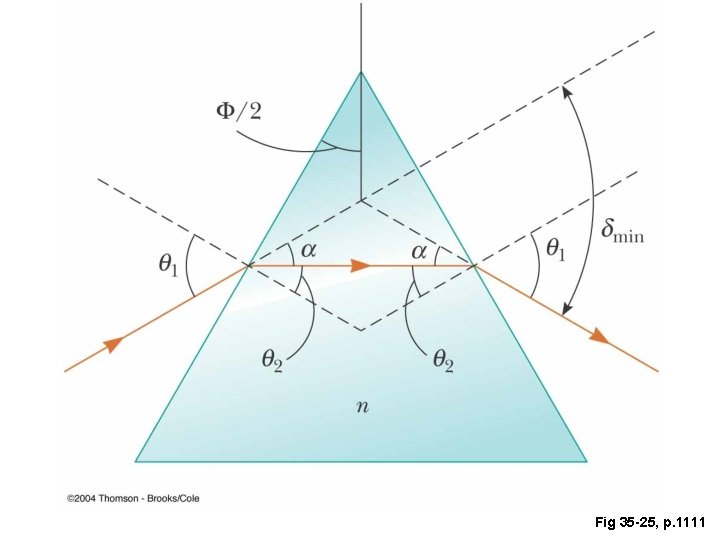
Fig 35 -25, p. 1111

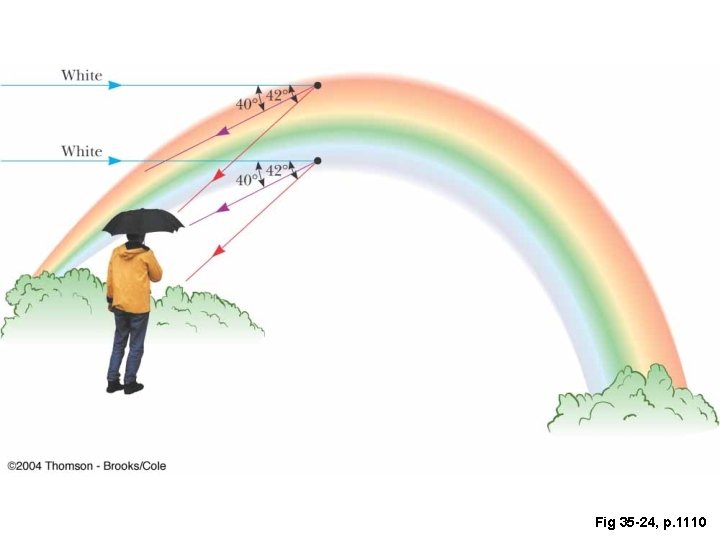
Fig 35 -24, p. 1110

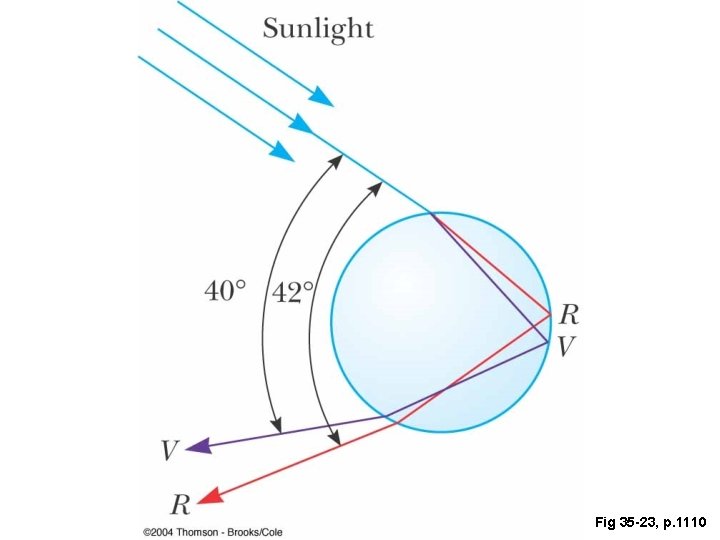
Fig 35 -23, p. 1110

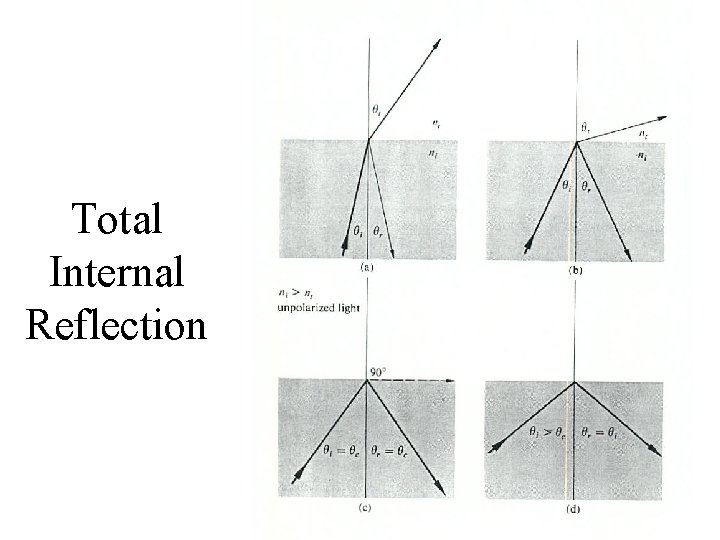
Total Internal Reflection

Total Internal Reflection

p. 1114

p. 1114

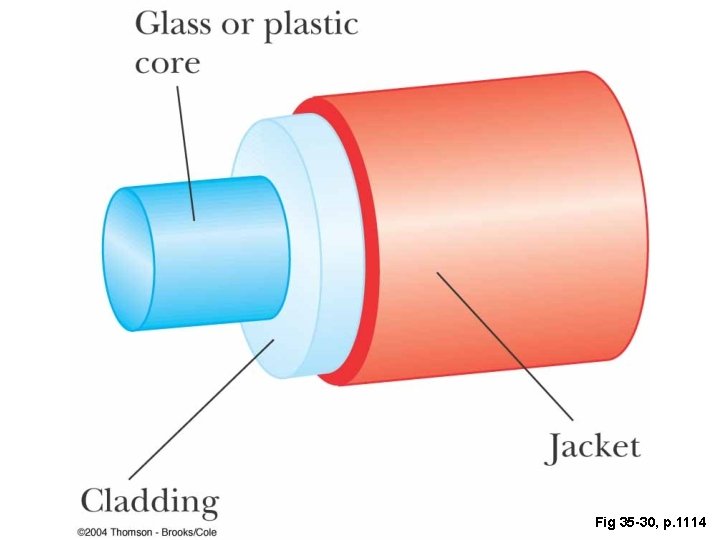
Fig 35 -30, p. 1114

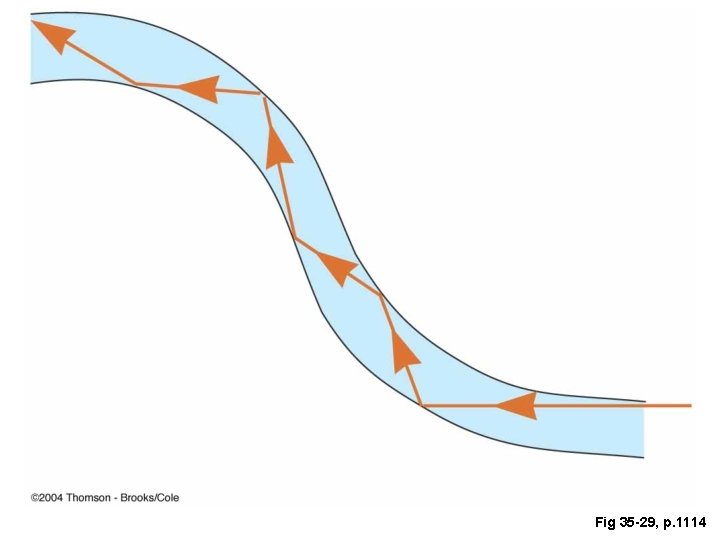
Fig 35 -29, p. 1114