Chapter 3 The cycling of matter in ecosystems





















- Slides: 36

Chapter 3 - The cycling of matter in ecosystems • Biogeochemical cycles: circular pathways involving biological, geological, and chemical processes • Biogeochemical cycles include: – Hydrologic Cycle/Water Cycle – Carbon Cycle – Phosphorus Cycle – Nitrogen cycle – Sulfur Cycle

Objectives: • Identify and describe the flow of nutrients in each biogeochemical cycle. • Explain the impact that humans have on the biogeochemical cycles. • Be able to diagram each cycle.

What Sustains Life on Earth? • Solar energy, the cycling of matter, and gravity sustain the earth’s life. Figure 3 -7

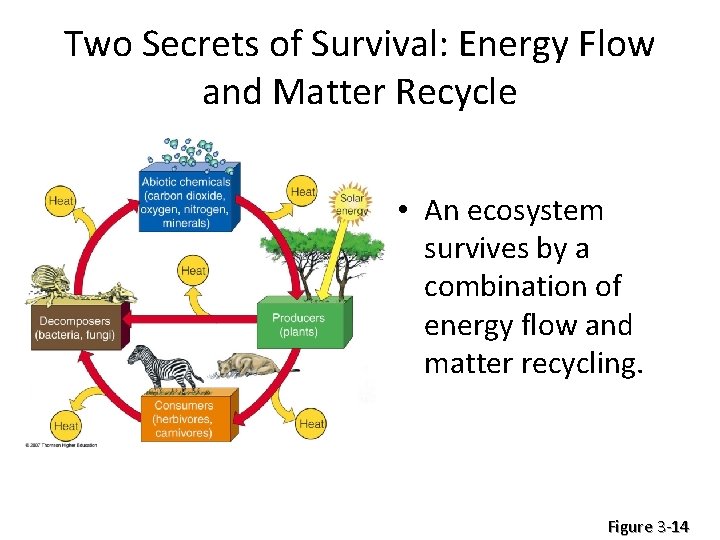
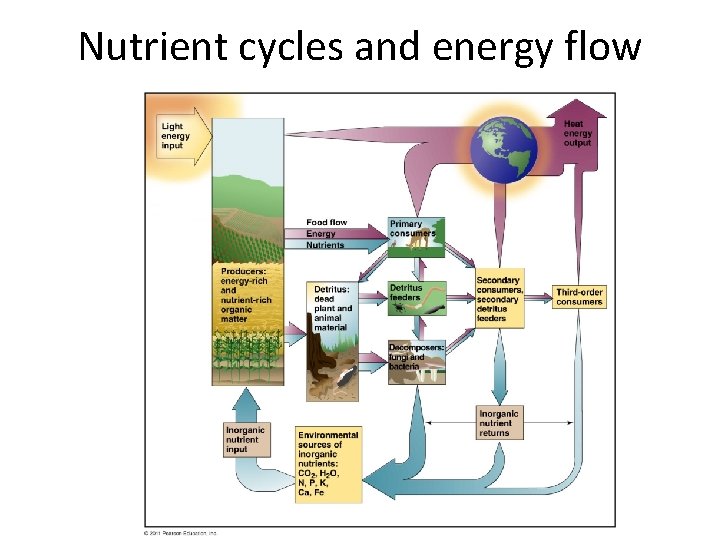
Two Secrets of Survival: Energy Flow and Matter Recycle • An ecosystem survives by a combination of energy flow and matter recycling. Figure 3 -14

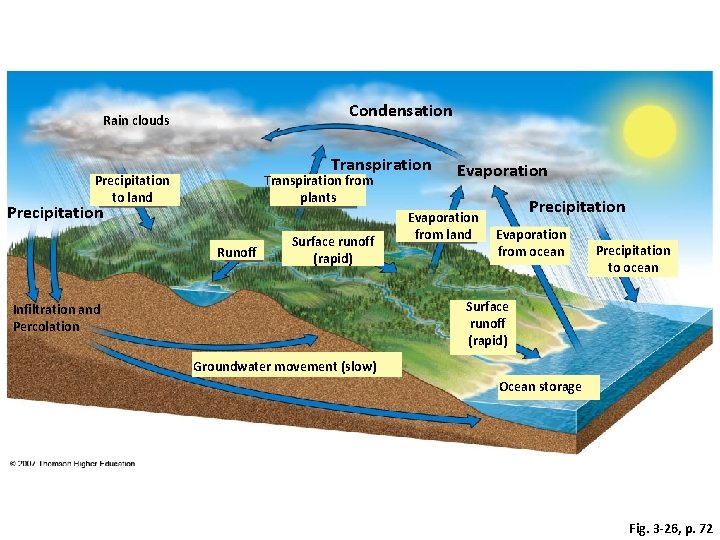
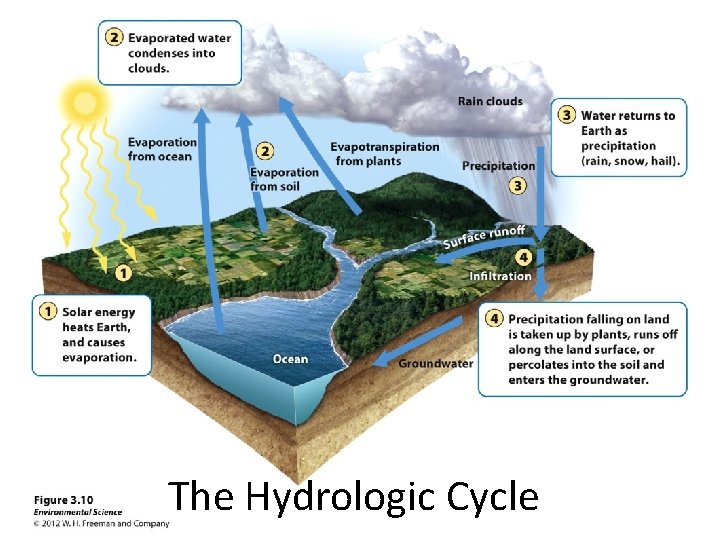
The Hydrologic Cycle The movement of water through the biosphere Water is essential to life; >50% of the typical mammal’s body weight Allows essential molecules to move within and between cells, draws nutrients into the leaves of trees, dissolves and removes toxic materials and performs many other critical biological functions Water is the primary agent responsible for dissolving and transporting the chemical elements necessary for living organisms

Condensation Rain clouds Transpiration Precipitation to land Transpiration from plants Precipitation Runoff Surface runoff (rapid) Evaporation from land Precipitation Evaporation from ocean Precipitation to ocean Surface runoff (rapid) Infiltration and Percolation Groundwater movement (slow) Ocean storage Fig. 3 -26, p. 72

The Hydrologic Cycle

Effects of Human Activities on Water Cycle • We alter the water cycle by: – Withdrawing large amounts of freshwater. – Clearing vegetation and eroding soils. – Polluting surface and underground water. – Contributing to climate change.

The Hydrologic Cycle Human Activities and the Hydrologic Cycle Remember: Earth is a closed system in regards to matter Humans can alter the hydrologic cycle in many ways: • Harvesting trees reduces evapotranspiration • Paving surfaces reduces infiltration/percolation Both Increase runoff and decrease groundwater recharge • Diverting water from one area to another can also impact the regional water cycle (drinking, irrigation, industrial uses)

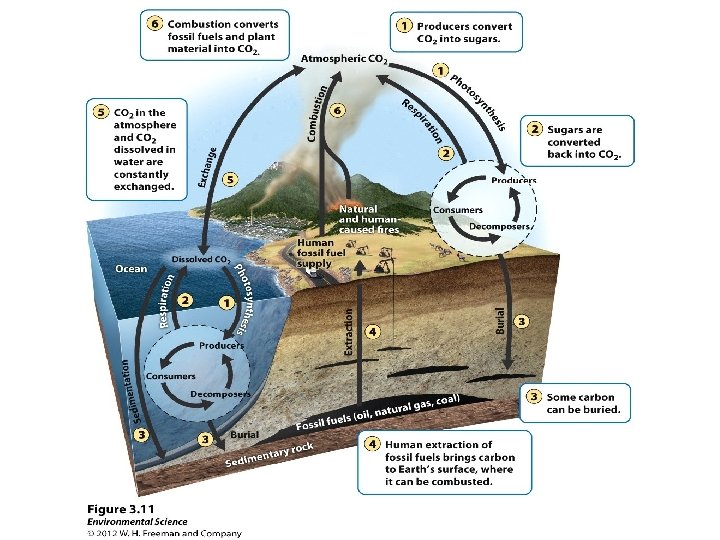
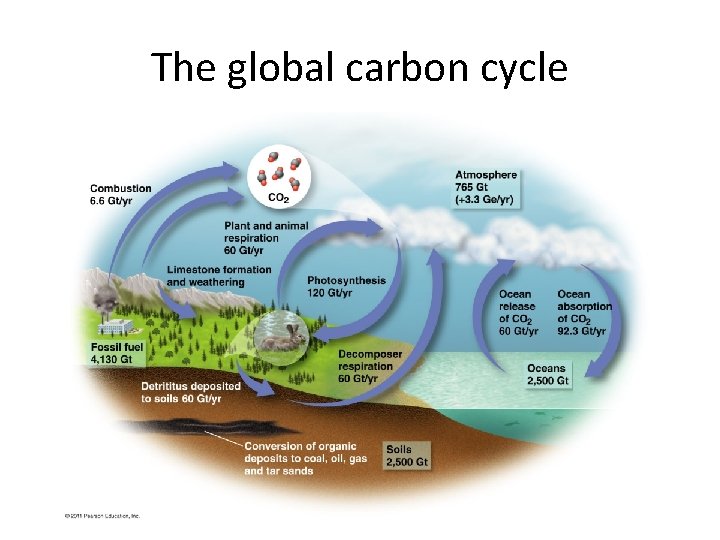
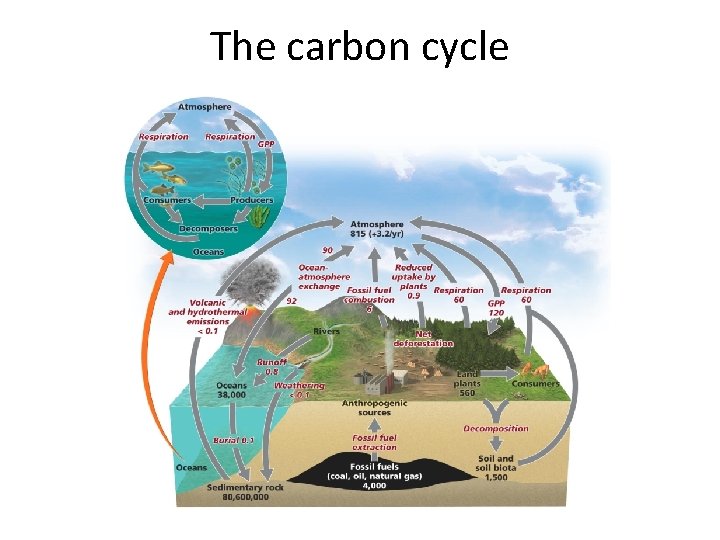
The Carbon Cycle How carbon (C) moves through our environment • Producers pull carbon dioxide (CO 2) from the air and use it in photosynthesis. • Consumers eat producers and return CO 2 to the air by respiration. • Decomposition of dead organisms, plus pressure underground, forms sedimentary rock and fossil fuels. This buried carbon is returned to the air when rocks are uplifted and eroded.

Carbon cycle: other ways to transfer carbon • Ocean water also absorbs carbon from multiple sources, eventually storing it in sedimentary rock or providing it to aquatic plants. • Limestone keeps carbon out of circulation – Weathering of exposed limestone releases carbon • A carbon atom cycles about every six years


The global carbon cycle

The carbon cycle

Human impacts on the carbon cycle • We have increased CO 2 in the atmosphere by burning fossil fuels and deforesting forests. • Atmospheric CO 2 concentrations may be the highest now than in 420, 000 years. • This is driving global warming and climate change.

Human impacts on the carbon cycle • Human intrusion into the cycle is significant • We are diverting or removing 40% of the photosynthetic effect of land plants • Burning fossil fuels has increased atmospheric CO 2 by 35% • Deforestation and soil degradation release significant amounts of CO 2 to the atmosphere – Recent reforestation and changed agricultural practices have improved this somewhat

Summary of Carbon Cycle • Acid rain: from the atmosphere delivers CO 2 dissolved in water vapor to earth. • Photosynthesis: turns CO 2 into plant biomass • Respiration: converts sugars and starches into energy for organisms and releases CO 2. • Decomposition: converts dead plants and animals into CO 2 and other inorganic nutrients. http: //www. youtube. com/watch? v=irw. Rz. QNS-GI

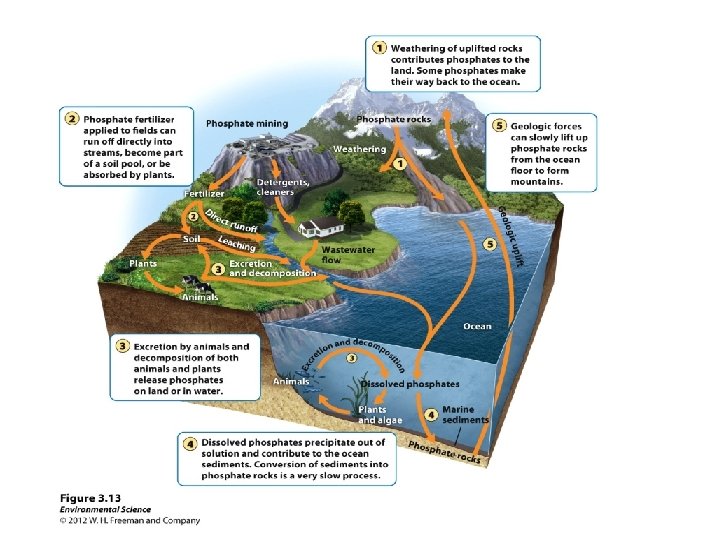
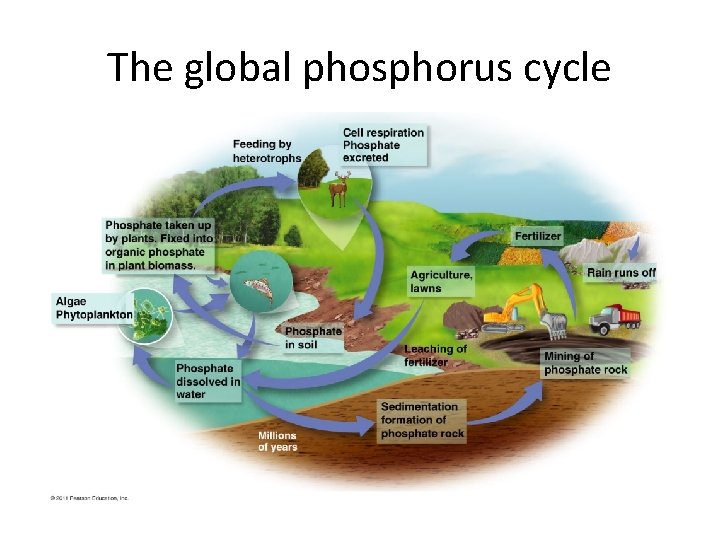
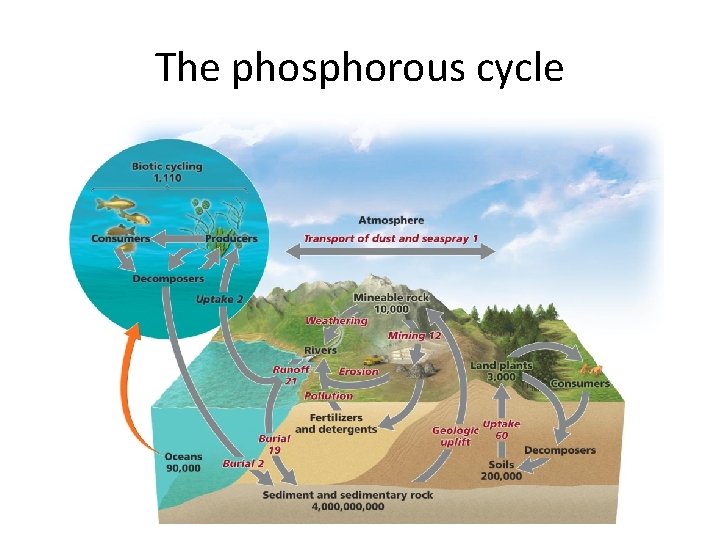
The Phosphorous Cycle How phosphorus (P) flows through our environment • P is most abundant in rocks. Weathering releases phosphate (PO 43–) ions from rocks into water. • As rock breaks down, phosphate is released – Replenishes phosphate lost through leaching or runoff • Plants take up phosphates in water, pass it on to consumers, who return it to the soil when they die. • Phosphates dissolved in lakes and oceans precipitate, settle, and can become sedimentary rock.


The global phosphorus cycle

The phosphorous cycle

Human impacts on the phosphorus cycle • The most serious intrusion comes from fertilizers • Phosphorus is mined and made into fertilizers, animal feeds, detergents, etc. • When added to soil, it can stimulate production – Human applications have tripled the amount reaching the oceans, accelerating the cycle – It can’t be returned to the soil • Excess phosphorus in water leads to severe pollution – Can cause too many bacteria and fish kills

Summary of Phosphorus Cycle • Weathering: of phosphate from rocks delivers phosphorus to soil. • Decomposition: of plant and animal wastes and remains delivers phosphorus to soil. • Humans make phosphates through mining and chemical weathering. http: //www. youtube. com/watch? v=3 iw. L 24 o. Vp. H 4

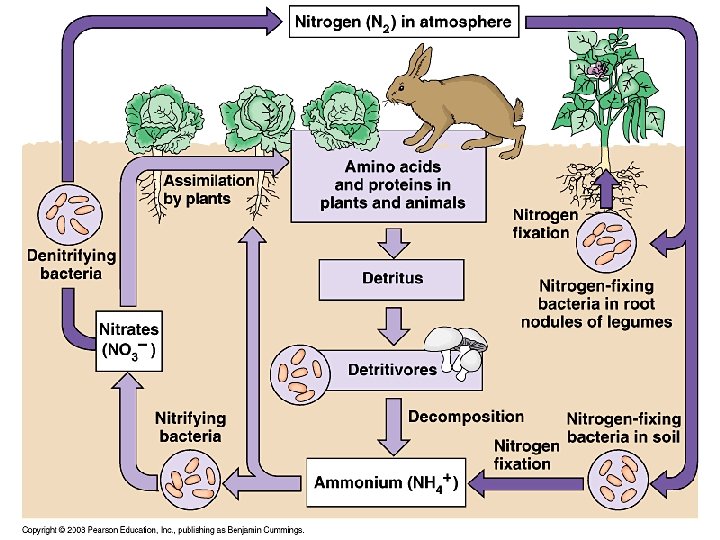
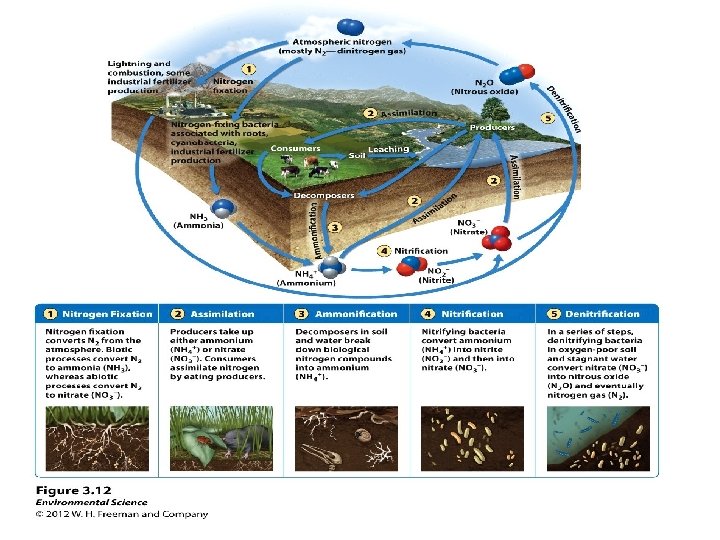
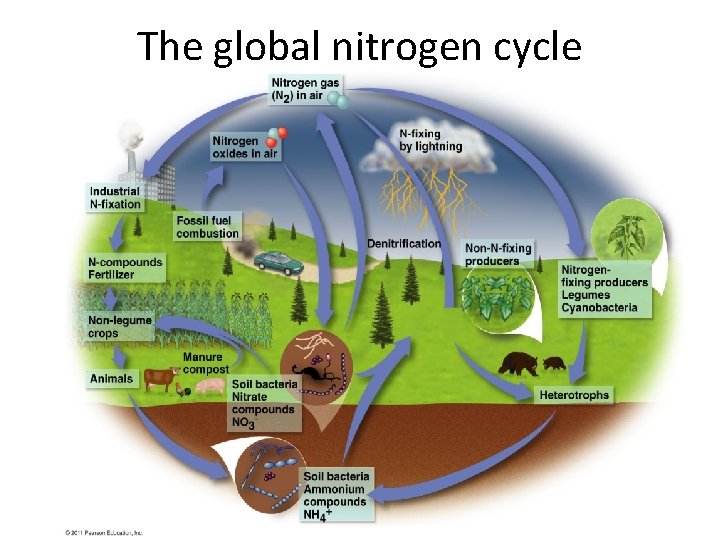
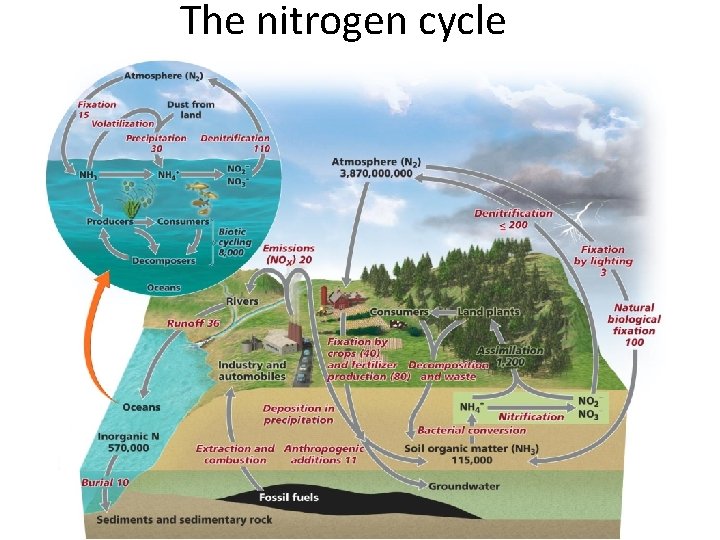
The Nitrogen Cycle • Is a unique cycle – Bacteria in soils, water, and sediments perform many steps of the cycle – Nitrogen is in high demand by aquatic and terrestrial plants • Air is the main reservoir of nitrogen (N) – Nonreactive nitrogen: most organisms can not use it • Reactive nitrogen (Nr): other forms of nitrogen that can be used by organisms

Plants take up nitrogen • Plants in terrestrial ecosystems (“non-N-fixing producers”) – Take up( N) as ammonium (NH 4) and incorporate it into proteins and nucleic acid compounds – The nitrogen moves through the food chain to decomposers, releasing nitrogen wastes • Soil bacteria (nitrifying bacteria) convert ammonium to nitrate to obtain energy – Nitrate is available for plant uptake • Nitrogen fixation: bacteria and cyanobacteria can use nonreactive N

Means of nitrogen fixation • Bacteria (genus Rhizobium) live in legume root nodules • The legume provides the bacteria a place to live and food – It receives a source of nitrogen in return – Nitrogen enters the food chain from the legumes • Three other processes “fix” nitrogen – Atmospheric nitrogen fixation: lightning – Industrial fixation: in fertilizer manufacturing – Combustion of fossil fuels: oxidizes nitrogen • Industrial fixation and fossil fuels release nitrogen oxides, which are converted to nitric acid (acid precipitation) – Denitrification: Microbes use nitrate as a substitute for oxygen

Nitrogen Cycle Summary • Nitrification: by bacteria convert NH 4 into NO 2 and then into NO 3 • Denitrification: Conversion of biological nitrogen (proteins-No 3 - and NH 4) into atmospheric nitrogen. • Biological Nitrogen Fixation: bacteria in the roots of legumes fix nitrogen. • Atmospheric Fixation: lightning converts N 2 to NO 3 - and NH 4 • Industrial Fixation: factories make fertilizer from atmospheric nitrogen. http: //www. youtube. com/watch? v=Ia. GNSm. QQm. Bc



The global nitrogen cycle

The nitrogen cycle

Human impacts on the nitrogen cycle • Human involvement in the nitrogen cycle is substantial • Many crops are legumes and draw N from the air – Peas, beans, soybeans, alfalfa – Increases the rate of nitrogen fixation • Nonleguminous crops (corn, wheat, potatoes) are heavily fertilized with nitrogen from industrial fixation • We are more than doubling the rate of nitrogen moving from air to land

Serious consequences of fertilization • Nitric acid has destroyed lakes, ponds, and forests • Atmospheric nitrogen oxides adds to ozone pollution, climate change, and stratospheric ozone depletion • Abundant nitrates are not incorporated into organisms – They are released into the soil, where they leach calcium and magnesium • Eutrophication of waterways • Nitrogen cascade: complex of ecological effects as Nr moves through the environment

Comparing the cycles • Carbon is mainly found in the atmosphere – Directly taken in by plants • Nitrogen and phosphorus are limiting factors • All three cycles have been sped up by human actions – Acid rain, greenhouse gases, eutrophication • Other cycles exist for other elements (e. g. , water) – All go on simultaneously – All come together in tissues of living things

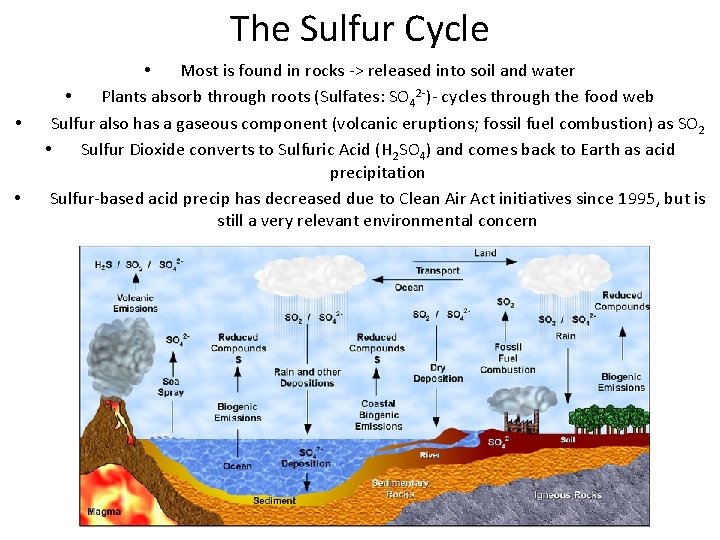
The Sulfur Cycle • • • Most is found in rocks -> released into soil and water • Plants absorb through roots (Sulfates: SO 42 -)- cycles through the food web Sulfur also has a gaseous component (volcanic eruptions; fossil fuel combustion) as SO 2 • Sulfur Dioxide converts to Sulfuric Acid (H 2 SO 4) and comes back to Earth as acid precipitation Sulfur-based acid precip has decreased due to Clean Air Act initiatives since 1995, but is still a very relevant environmental concern

Nutrient cycles and energy flow