Chapter 3 The Atom The Atom 450 B

Chapter 3 The Atom

The Atom • ______ (450 B. C. ) proposed that all matter is made up of tiny, indivisible particles. (atomos) 3 -2

Laws • Law of Conservation of Mass: mass is neither ______ during ordinary chemical rxns or ______ changes. 3 -3

Laws • Law of Definite Proportions: compounds contain the same ______ in exactly the same _______ by mass regardless of the _______ of the sample or the _____ of the compound. 3 -4

Laws • Law of Multiple Proportions: If two or more ________ are composed of the same ______, the _______ of the masses of the second element combined with a certain _____ of the first element is always a ratio of ___________. 3 -5

Daltons Atomic Theory In 1808 ______, an English schoolteacher, came up with an atomic theory to explain these laws. Many of the _____ of his theory still hold true today. 3 -6

Dalton’s Atomic Theory ~ Each ______ is composed of atoms. ~ Atoms of a given element are _____, and _____ than those of any other element. ~ A given compound forms by ______ of two or more different atoms, always in the same ____________ of atoms. ~ Atoms are neither ________ in any chemical reaction, only _______. 3 -7

The Structure of the Atom • ______: the smallest _____ of an element that ____ the chemical ____ of that element. 3 -8

The Discovery of the Electron • ______ (1897) an English physicist who discovered ____ using his famous ______ experiment. He determined the ________ ratio. 3 -9
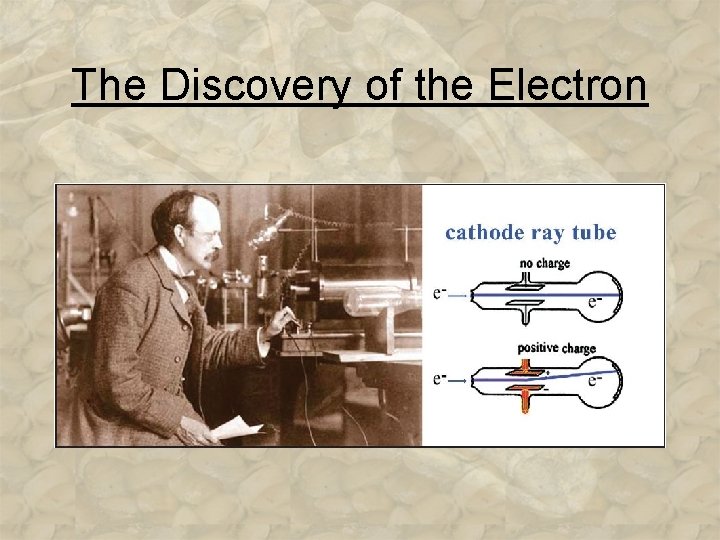
The Discovery of the Electron

The Discovery of the Electron • _____ (1909) an American _____ who determined the charge of electrons using his famous _____ experiment. The mass of an e- is approximately ____ the mass of an atom. 3 -11
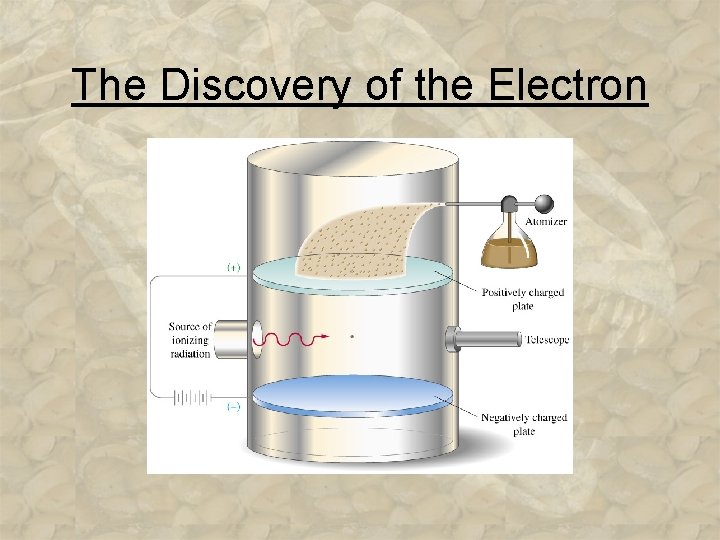
The Discovery of the Electron

The Discovery of the Electron Based on these discoveries, two inferences were made: 1. Because atoms are ______, they must contain a ____ charge to _____ the negative electrons. 2. Because e- are so light in mass _____ to atoms, atoms must contain other _______ that account for most of its mass. 3 -13
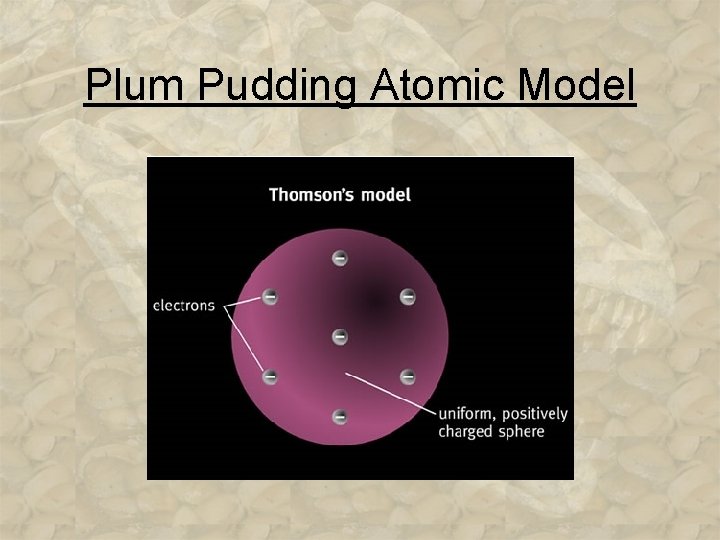
Plum Pudding Atomic Model

Discovery of the Nucleus 1911, Ernest ____ conducted experiments with _____ materials that released only _____ charged alpha particles…. 3 -15
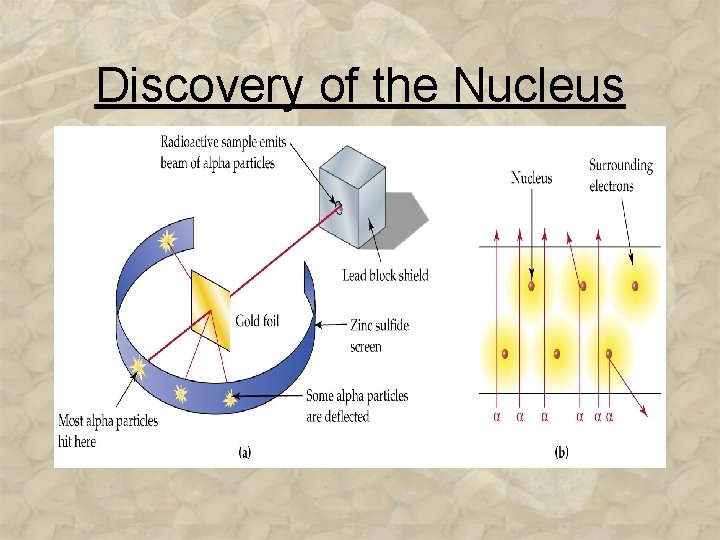
Discovery of the Nucleus
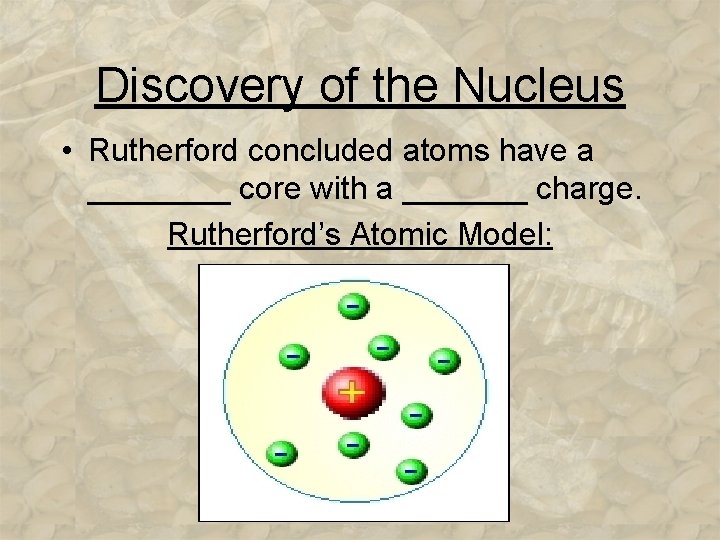
Discovery of the Nucleus • Rutherford concluded atoms have a ____ core with a _______ charge. Rutherford’s Atomic Model:
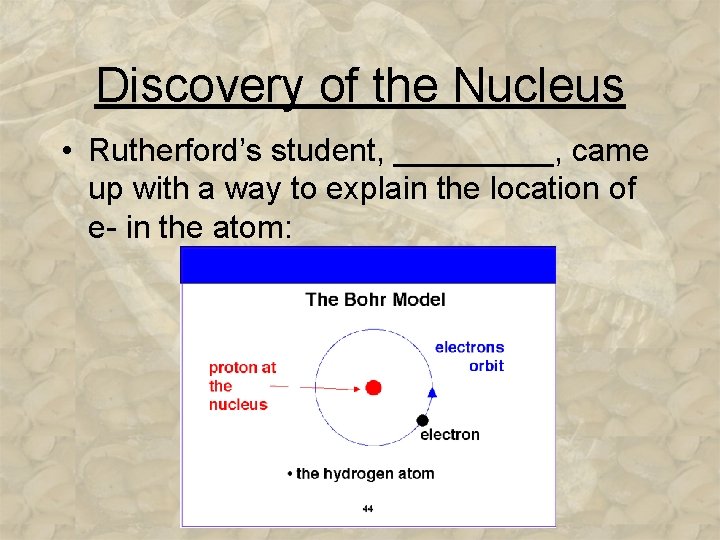
Discovery of the Nucleus • Rutherford’s student, _____, came up with a way to explain the location of e- in the atom:
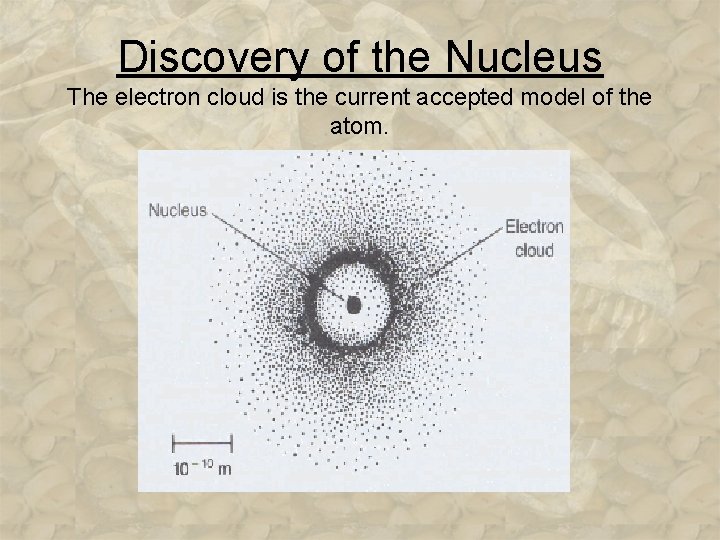
Discovery of the Nucleus The electron cloud is the current accepted model of the atom.

The Atom • Except for H, all ______ contain ___________. • A proton has a + charge _____________to the neg. charged e-. • Atoms contain ________ of p+ and e-. • Neutrons are _______ neutral. • P+ and no have almost ______ masses, electrons weigh 1836 times ______. 3 -20

The Atom • _______: short range p+ to p+, p+ to no and no to no forces hold the nuclear particles together. • __________ (Z): the number of protons in each atom of a particular element. ☺The atomic number _____ the element! 3 -21

Isotopes • _____: atoms of the same element that have ________ due to different numbers of _____. • _______: the total number of _______ and _____ that make up the nucleus of an isotope ~ Isotopes are written with the _______ written after the element name or symbol with a _______: ex. Uranium-235 or U-235 3 -22

Isotopes Uranium-235 or U-235 Mass number – atomic number = number of ____ 235 (protons + neutrons) – 92 protons = 143 neutrons This info could also be portrayed using a _______: 235 U 92

Isotopes ~ In nature, _____ are almost always found as a ______ of isotopes ~ Isotopes have ________ chemical properties ~Isotopes with _______ neutrons have a higher mass and are often described as “_______”. 3 -24
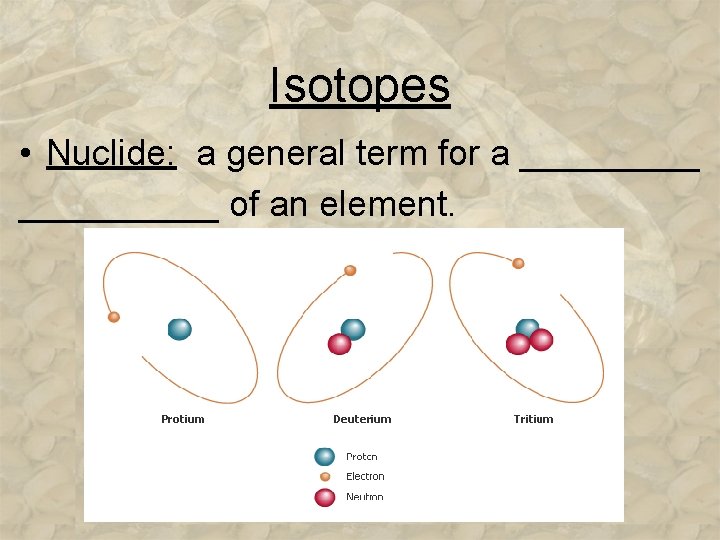
Isotopes • Nuclide: a general term for a __________ of an element.

Atomic Mass • Atomic Mass Unit (____): one amu is exactly _____ of the mass of a ____ atom. • ________: the weighted average of the atomic masses of the _________ isotopes of an element. 3 -26

Calculating the Average

Moles • _____: the amount of a substance that contains as many _______ as there atoms in exactly 12 g of ______.

Moles • _______: the number of particles in exactly one ______ of a pure substance ~ 6. 02214179 x 1023 (we’ll use _____)

Moles 602, 000, 000, 000 If you had 6. 02 x 1023 pennies and gave away 1 million a day to every person on earth, it would take you 3000 years to distribute all your money!! 3 -30

Molar Mass • ______: the mass of one mole of a pure substance. ~ the mass of _____ atoms or _____ is measured in _____. The mass of a mole of the same substance is _______ the same, with the units _______. Ex. H 20 = H x 2 = 1. 01 x 2 = 2. 02 + O x 1 = 16. 00 x 1 = +16. 00 18. 02 3 -31
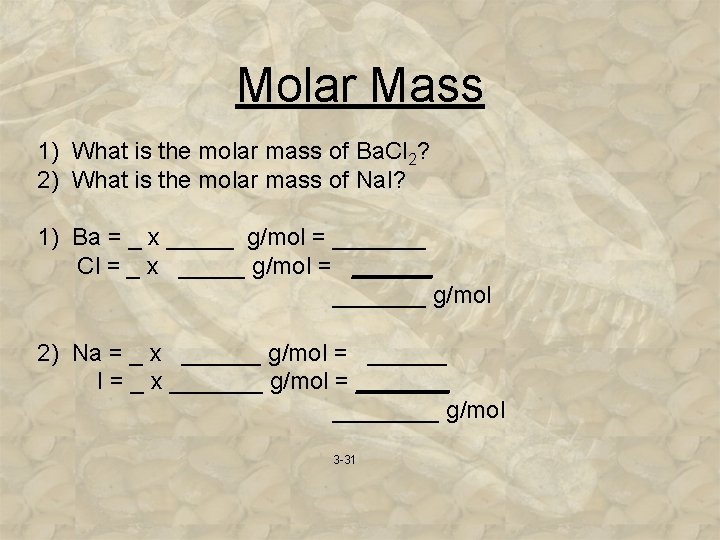
Molar Mass 1) What is the molar mass of Ba. Cl 2? 2) What is the molar mass of Na. I? 1) Ba = _ x _____ g/mol = _______ Cl = _ x _____ g/mol = _______ g/mol 2) Na = _ x ______ g/mol = ______ I = _ x _______ g/mol = ________ g/mol 3 -31

Molar Mass This photograph shows one mole of _______ (Na. Cl 58. 44 g/mol), ____ (H 2 O 18. 02 g/mol), and ____(N 2 28. 02 g/mol). 3 -33

Mass/Mole Conversions When given the number of _______, you can find the _______by: Moles x _g__ = grams mole Ex. 5. 0 moles of H 2 O = X g 5. 0 moles x 18. 02 g = 90. g H 2 O in 5. 0 moles 3 -34

Mass/Mole Conversions Moles x _g_ = moles mol Now try these problems: 3) 8. 32 moles of barium chloride equals how many grams? 4) 20. 1 moles of sulfur dioxide equals how many grams? 3 -35

Mass/Mole Conversions 3) 8. 32 moles of barium chloride equals how many grams? 8. 32 moles Ba. Cl 2 x _____ g/mol = _____ grams Ba. Cl 2 4) 20. 1 moles of sulfur dioxide equals how many grams? 20. 1 moles SO 2 x ______ g/mol = _____ g SO 2 3 -36

Mass/Mole Conversions
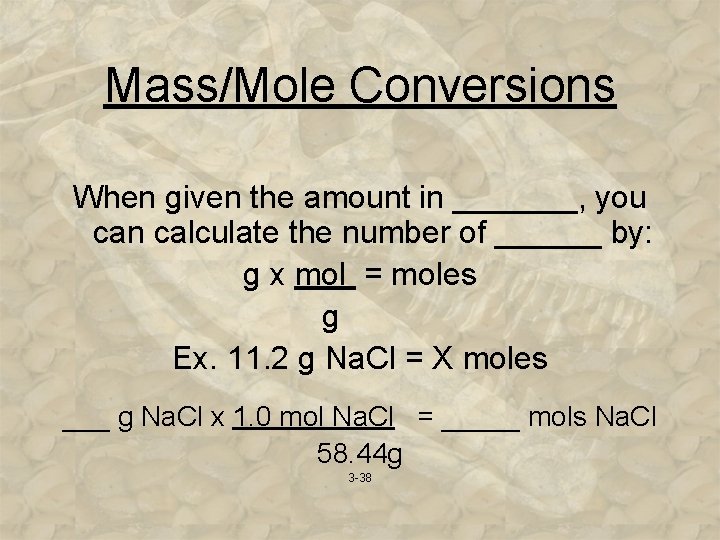
Mass/Mole Conversions When given the amount in _______, you can calculate the number of ______ by: g x mol = moles g Ex. 11. 2 g Na. Cl = X moles ___ g Na. Cl x 1. 0 mol Na. Cl = _____ mols Na. Cl 58. 44 g 3 -38

Mass/Mole Conversions g x mol = moles g Now try these problems: 5) 50. 56 g of sodium chloride equals how many moles? 6) 329. 8 g of ammonia equals how many moles? 3 -39

Mass/Mole Conversions 5) 50. 56 g of sodium chloride equals how many moles? _______ g Na. Cl x mole = _____ moles Na. Cl _____g 6) 329. 8 g of ammonia equals how many moles? _____ g NH 3 x mole = _____ moles NH 3 ______g 3 -40

Particle/Mole Conversions You can also calculate between _______ and number of ____: (1. 0 moles = 6. 02 x 1023 particles) To enter this number into your calculator, punch in 6. 02 _____ button (____) 23. 3 -41

Particle/Mole Conversions Ex. 2. 59 moles of marble (Ca. CO 3) contains how many molecules? _____ mol Ca. CO 3 x 6. 02 x 1023 molecules = ______ 1. 0 mol Ca. CO 3 molecules *Particles can be molecules, atoms or formula units 7) How many molecules are in 5. 0 moles of carbon dioxide? 5. 0 mol CO 2 x ____ molecules = 1. 0 mole CO 2 _____ molecules CO 2 3 -42
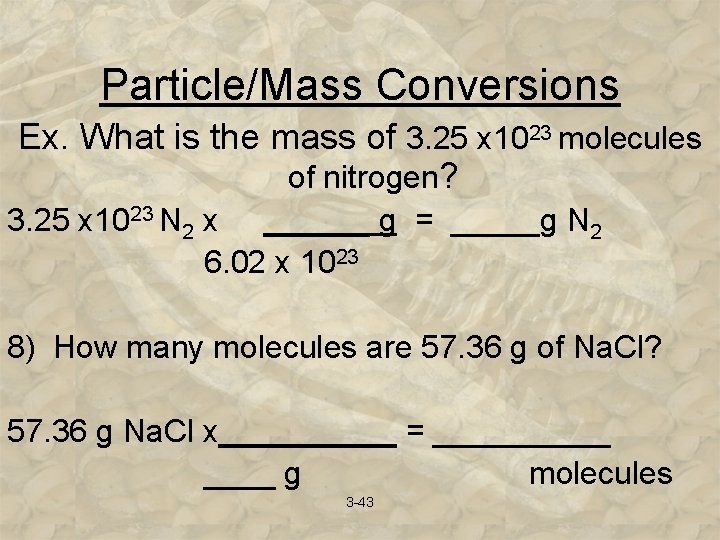
Particle/Mass Conversions Ex. What is the mass of 3. 25 x 1023 molecules of nitrogen? 3. 25 x 1023 N 2 x ______ g = _____g N 2 6. 02 x 1023 8) How many molecules are 57. 36 g of Na. Cl? 57. 36 g Na. Cl x_____ = _____ g molecules 3 -43


The End!
- Slides: 45