Chapter 3 Spectral lines in stars Emission and



















- Slides: 32

Chapter 3: Spectral lines in stars

Emission and absorption of light Continuous spectrum (thermal, blackbody) Independent of composition Emission line spectrum Dependent on composition

Each element has its own unique spectrum

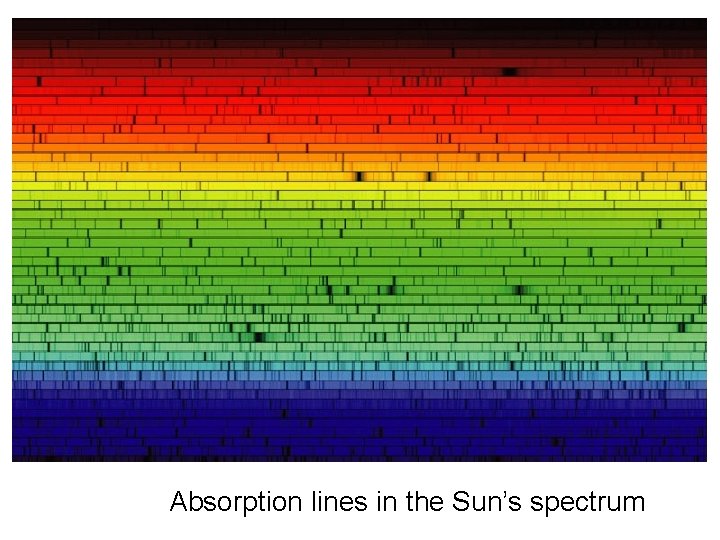
Absorption lines in the Sun’s spectrum

Absorption Line Spectrum Gustav Kirchhoff (1824 -1887)

absorption lines of hydrogen

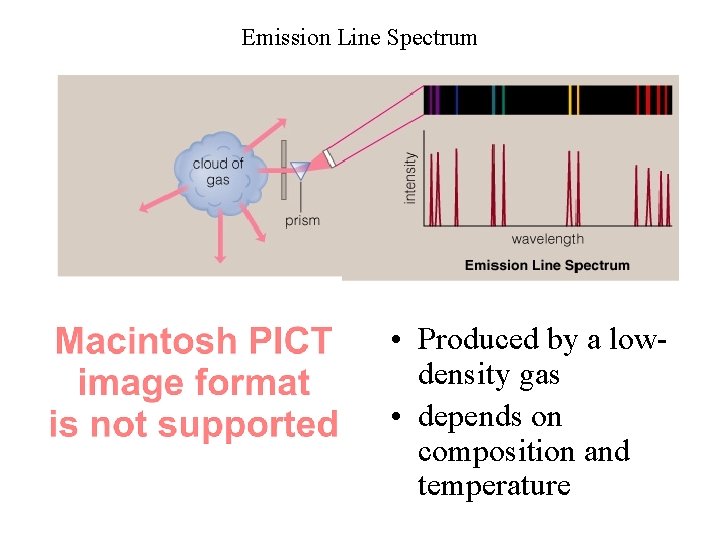
Emission Line Spectrum • Produced by a lowdensity gas • depends on composition and temperature

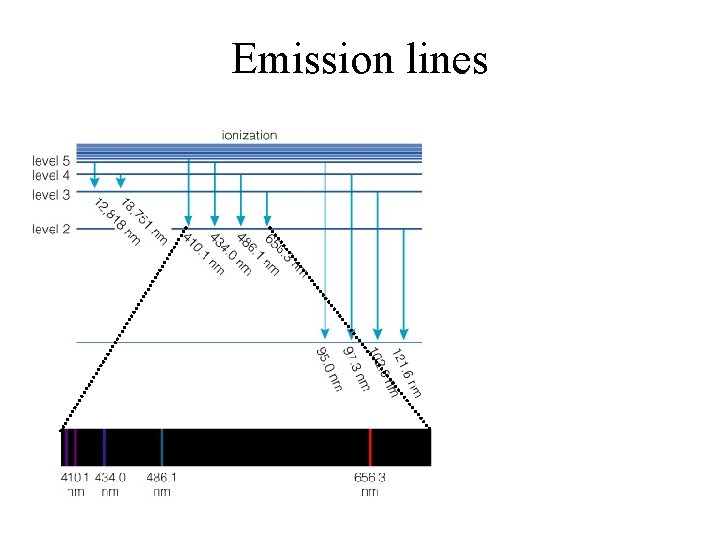
Emission lines

The Balmer series for hydrogen: Visible light electrons falling to n=2 Rydberg formula (Balmer for nf = 2) : 1 / = R (1/nf 2 - 1/ni 2) R = Rydberg constant = 1. 097 x 107 m-1

The hydrogen atom


Energy levels and transitions of the many-electron atom: Sodium Quantum states of the valence electron

The Bohr Model • • Classical physics predicts that the electron should spiral into the nucleus Cannot explain emission spectra

The Bohr model: • The e- stays in certain stable orbits, emits no radiation unless it jumps to a lower level • The angular momentum of the e- is quantized • the attaction between p and e- provides the centripetal acceleration n = principal quantum number

From Coulomb’s law, the force between the proton and electron is F = 1 4 0 q 1 q 2 r 2 This is the centripetal force, Where q 1 = q 2 = e for the hydrogen atom mv 2 / r

So when the electron is in any energy level n: Bohr radius a 0 = 0 h 2 / me 2 = 5. 29 x 10 -11 m KE of the electron in the nth level: Kn = 1/2 mv 2 PE of the electron in the nth level: Un = -1 4 0 Total energy En = Kn + Un = ? ? ? Compare to Rydberg formula! e 2 r

Reduced mass: the nucleus is not infinite in mass, Bohr model is off by 0. 1% m 1 m 2 mr = m 1 + m 2 isotopes

Ionized Helium is also a 1 -electron atom

Why is the emission spectrum of ionized helium similar to that of hydrogen? 1. Because hydrogen and helium are similar chemically 2. Because several of the energy levels of hydrogen and helium are the same 3. Because hydrogen and helium have similar atomic masses 4. It is a total coincidence

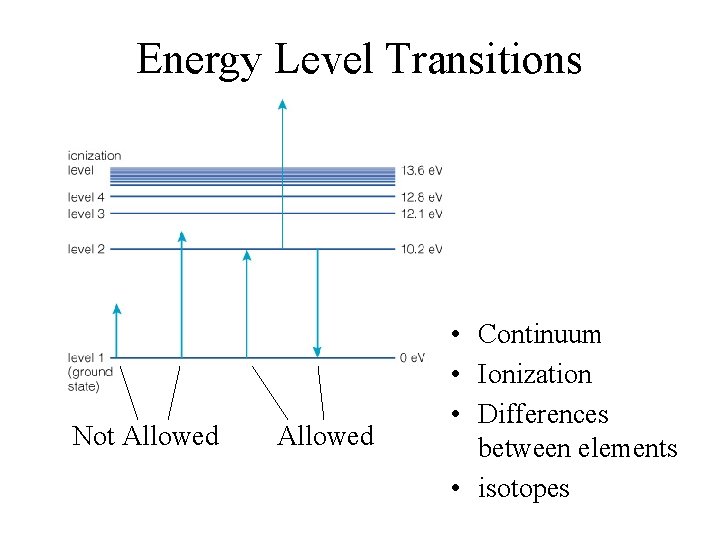
Energy Level Transitions Not Allowed • Continuum • Ionization • Differences between elements • isotopes

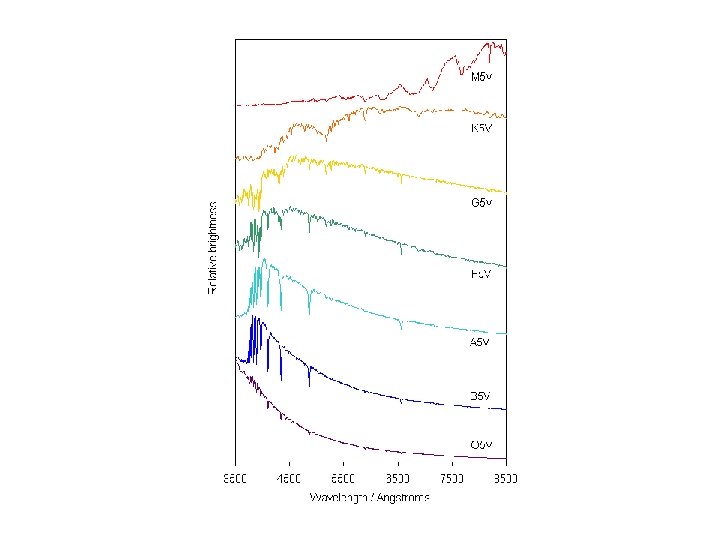
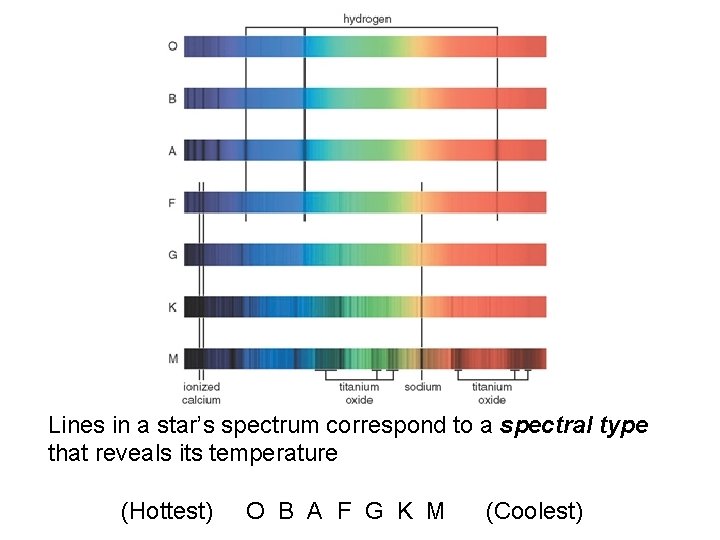
Stellar classification scheme


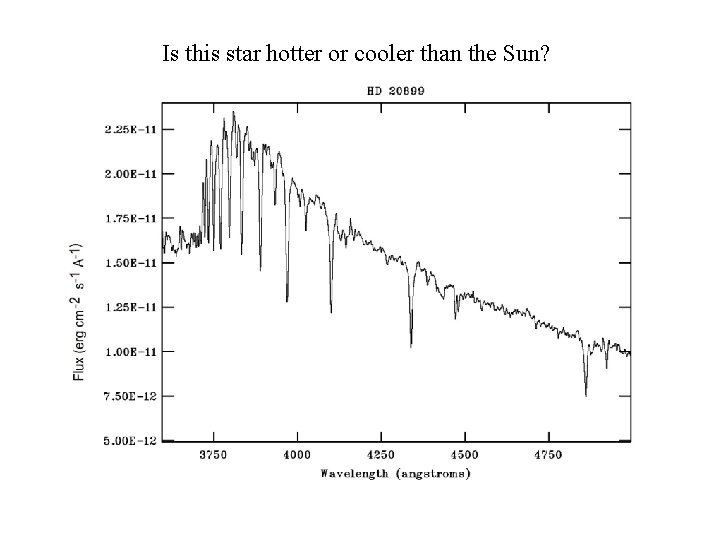
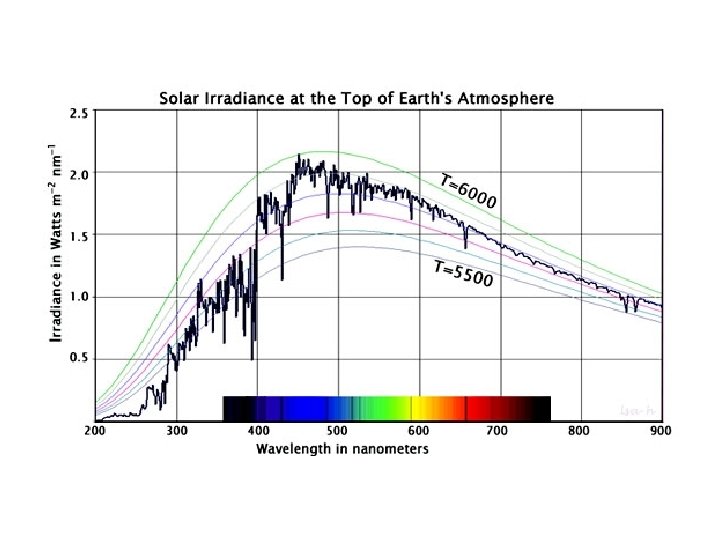
Is this star hotter or cooler than the Sun?


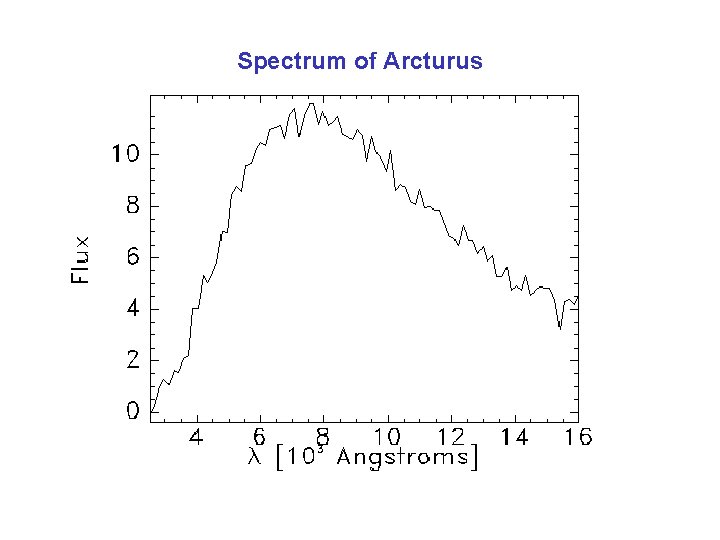
Spectrum of Arcturus


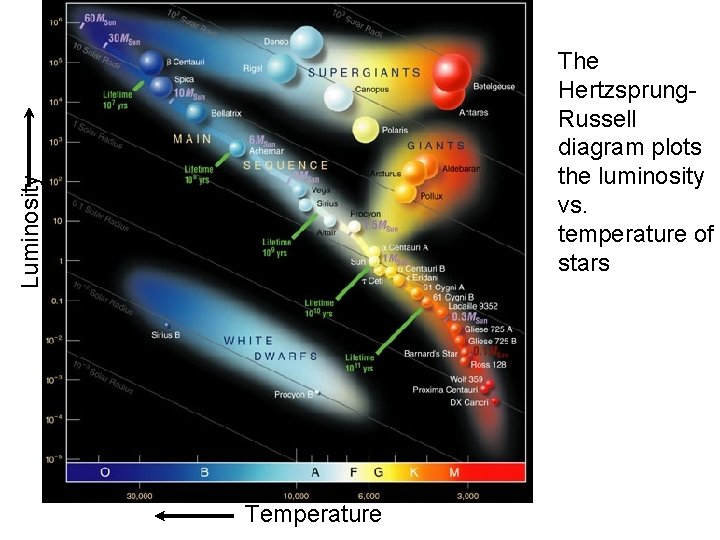
Luminosity The Hertzsprung. Russell diagram plots the luminosity vs. temperature of stars Temperature

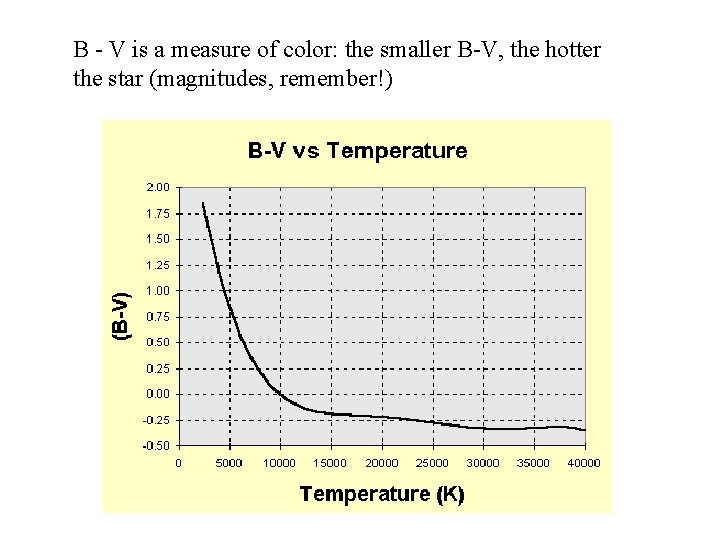
B - V is a measure of color: the smaller B-V, the hotter the star (magnitudes, remember!)




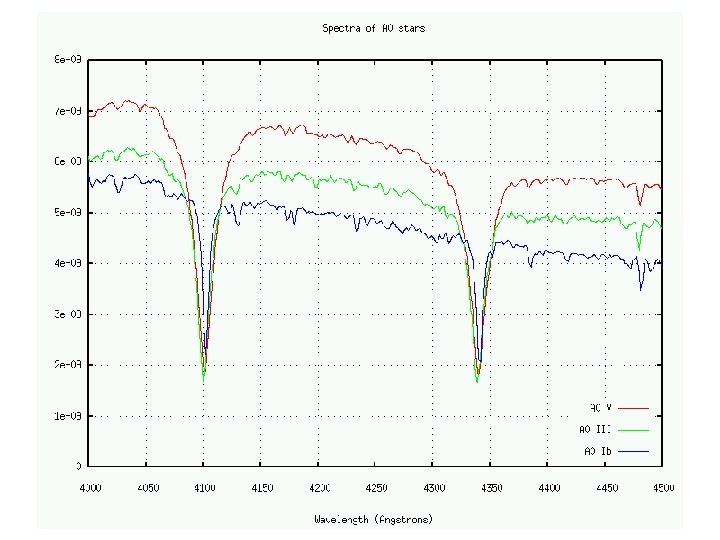
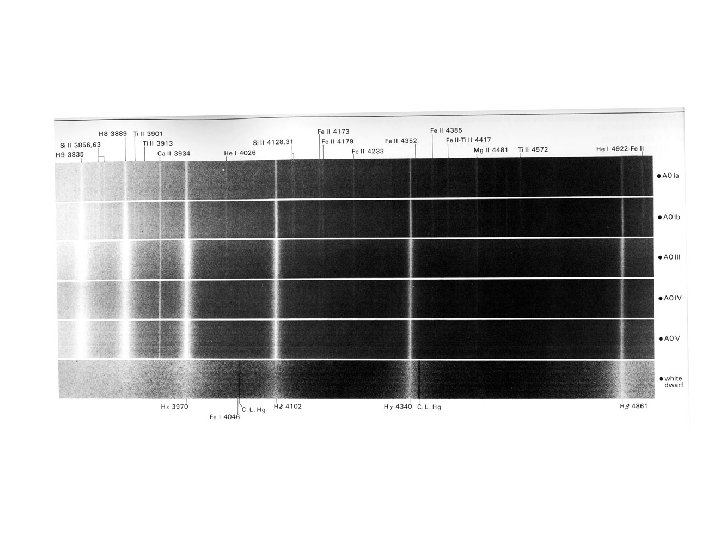
Lines in a star’s spectrum correspond to a spectral type that reveals its temperature (Hottest) O B A F G K M (Coolest)