Chapter 3 Simple Bonding Theory Lewis Dot Structures

Chapter 3 Simple Bonding Theory • Lewis Dot Structures – Resonance – Formal Charge • VSEPR: the subtle effects

Lewis Dot Structures 1. Count valence electrons 2. Arrange atoms 3. Add bonds 4. Add lone pairs 5. Convert lone pairs to bonding pairs (octet rule and exceptions)

Lewis Dot Structures • Examples: CO 2 SO 3 N 2 O Xe. F 4 Cl. F 3 – PCl 6

Why does the octet rule work?

More complex NO 2 NO

Formal Charge = Group # - #unshared electrons on atom - # bonds to atom Example: O 3

Resonance Example: SO 3

Resonance and Formal Charge Example: SCN-

Resonance and Formal Charge Example: SCN-
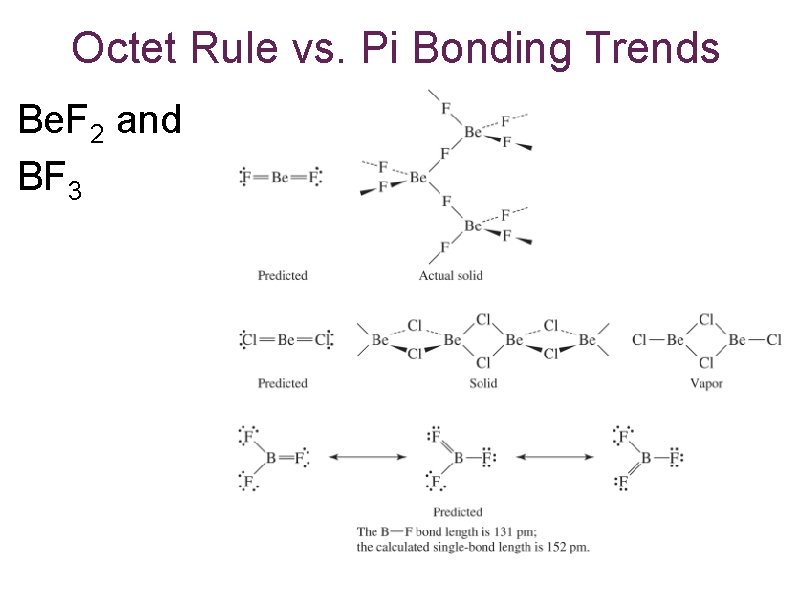
Octet Rule vs. Pi Bonding Trends Be. F 2 and BF 3

Octet Rule vs. Formal Charge • Always follow octet rule Exceptions? SO 42 -

VSEPR • Maximize “personal space” • CO 2, SO 3, SO 42–, PCl 5, SF 6 • Lone pairs vs. bonding pairs? • Single bonds vs. multiple bonds? • Electronegativity effects
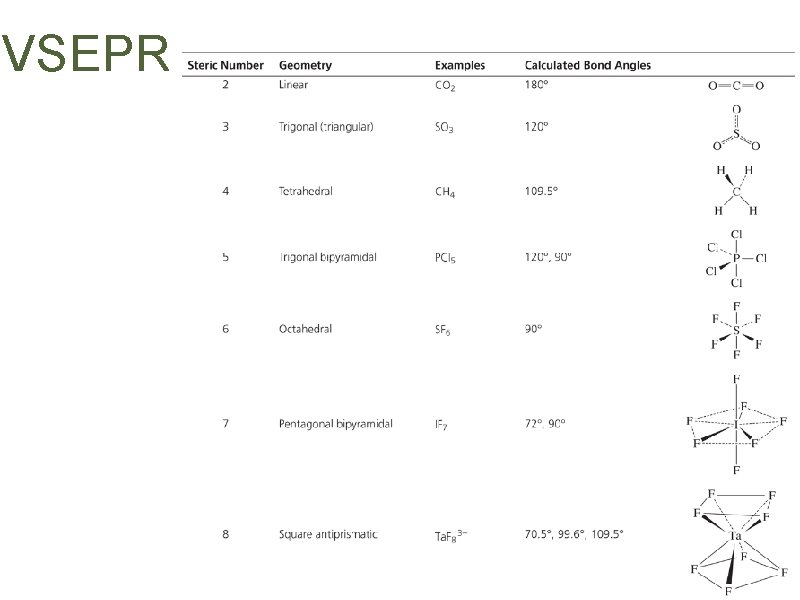
VSEPR
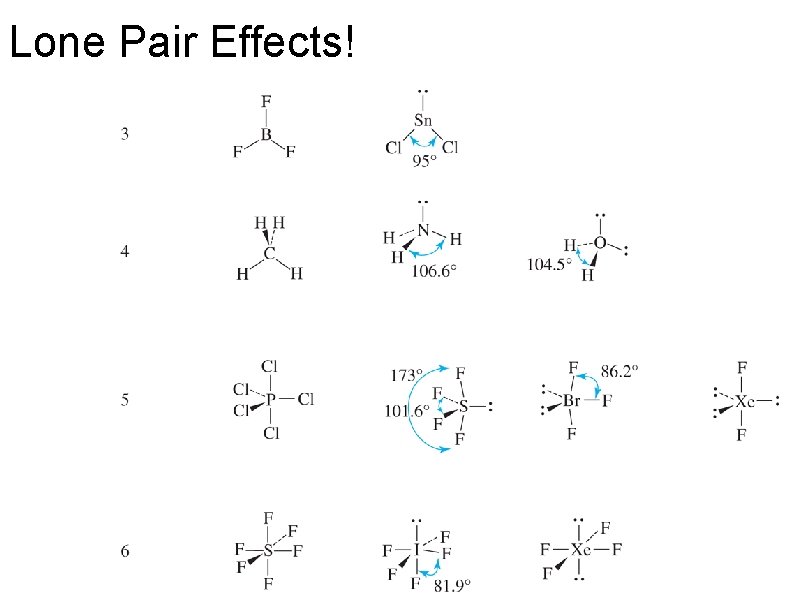
Lone Pair Effects!
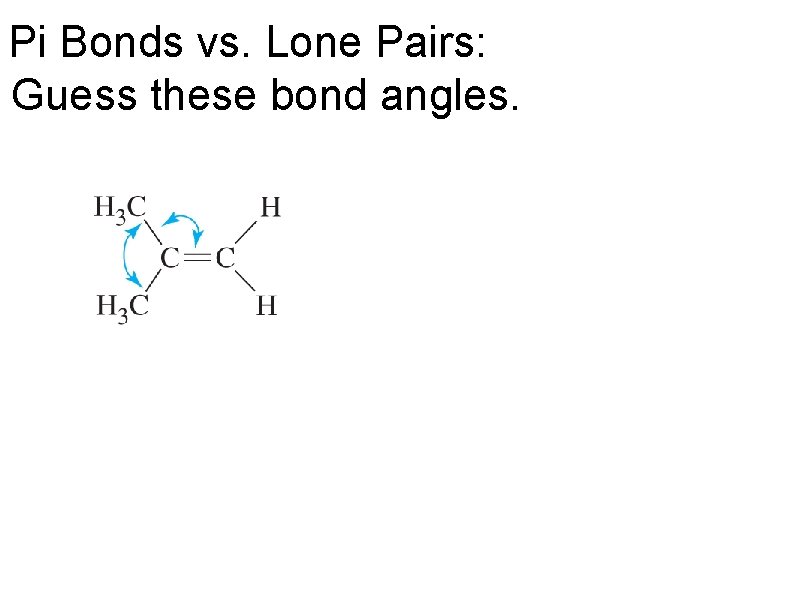
Pi Bonds vs. Lone Pairs: Guess these bond angles.
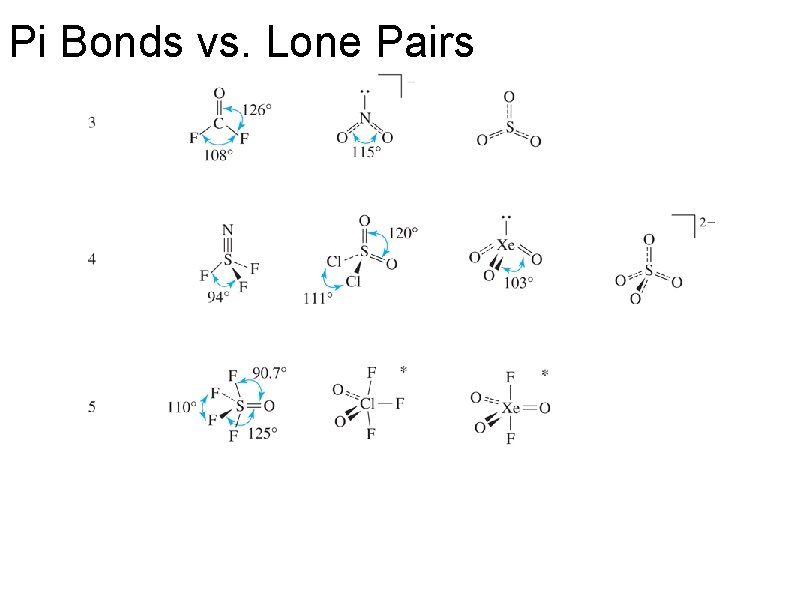
Pi Bonds vs. Lone Pairs
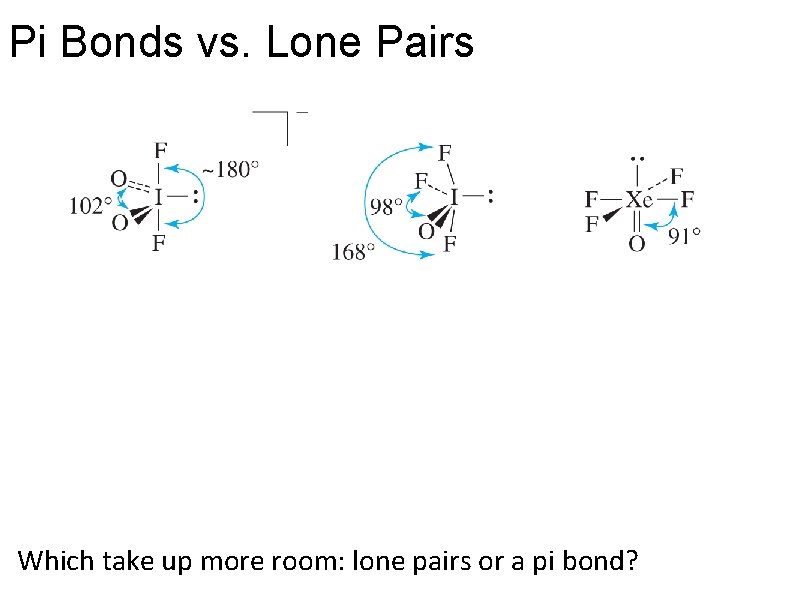
Pi Bonds vs. Lone Pairs Which take up more room: lone pairs or a pi bond?

Electronegativity Effects Molecule X-P-X Angle o PF 3 97. 8 PCl 3 100. 3 PBr 3 101 Explain this trend.

Electronegativity Effects Molecule H 2 O H 2 S H 2 Se Molecule As. F 3 As. Cl 3 As. Br 3 H-X-H Angle o 104. 4 92. 1 90. 6 Explain this trend. X-As-X Angle o Predict this trend.
- Slides: 19