Chapter 3 Orbitals and Bonding Atomic Orbitals AOs
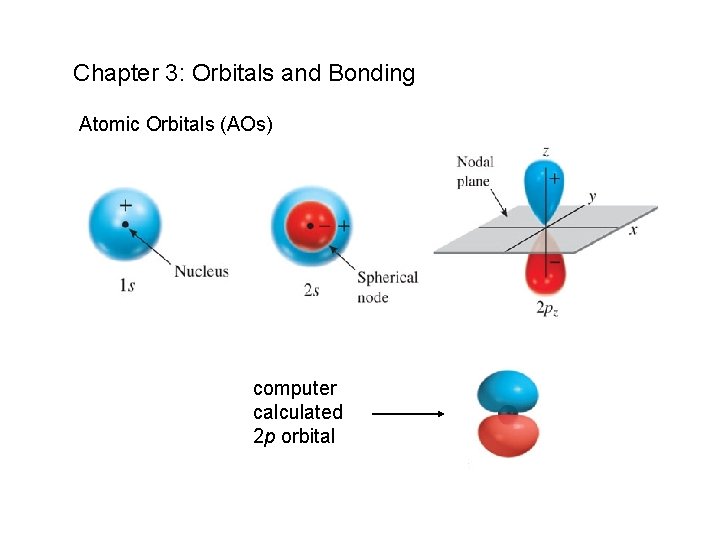
Chapter 3: Orbitals and Bonding Atomic Orbitals (AOs) computer calculated 2 p orbital

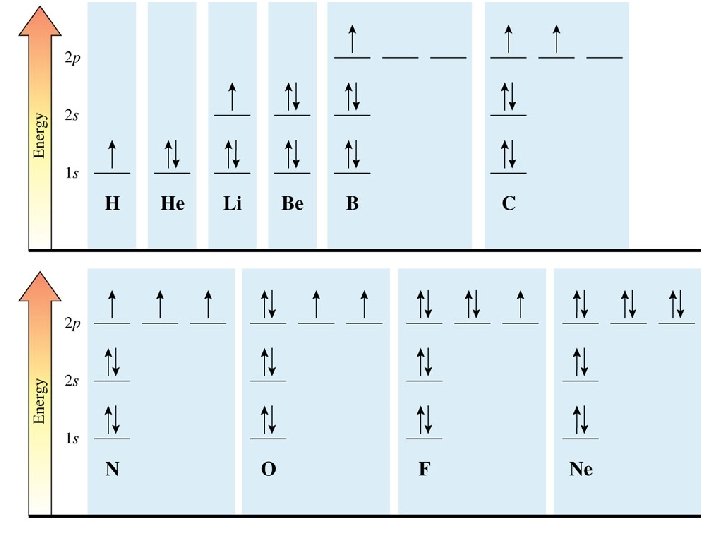
electron configuration for atoms 1. Each electron is placed in the lowest-energy orbital available. 2. Each orbital can contain a maximum of two electrons with opposite spins (Pauli exclusion principle). 3. When degenerate orbitals (orbitals with the same energy) are available, the electrons first occupy them singly with the same (parallel) spins. After all of the degenerate orbitals contain one electron, additional electrons with opposite spins are added to each (Hund’s rule).

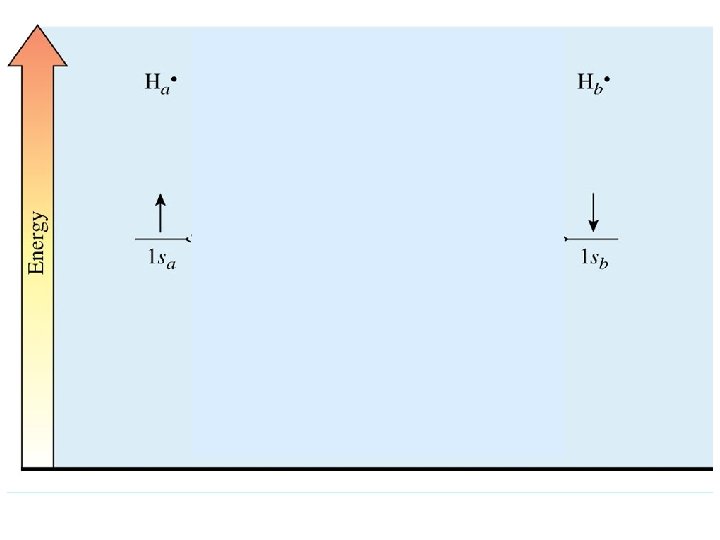
Molecular Orbitals (MOs): orbitals that extend around more than one atom H: H nuclei approach to bonding distance 1 s atomic orbitals begin to overlap electron waves reinforce to give lower energy MO (bonding) or electron waves cancel to give higher energy MO (antibonding)

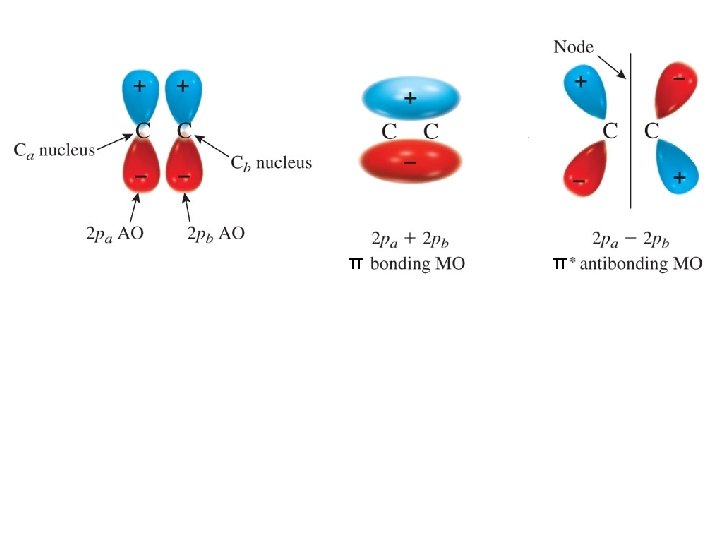
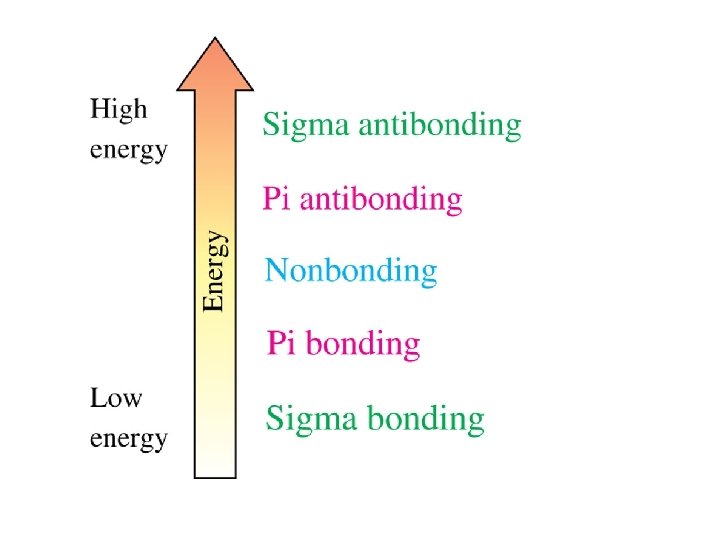
These are sigma (s) MOs: symmetrical about the internuclear axis

Valence Bond Theory overlap of AOs on two atoms at a time forms localized MOs: delocalized MOs: extend around only two atoms extend around more than two atoms

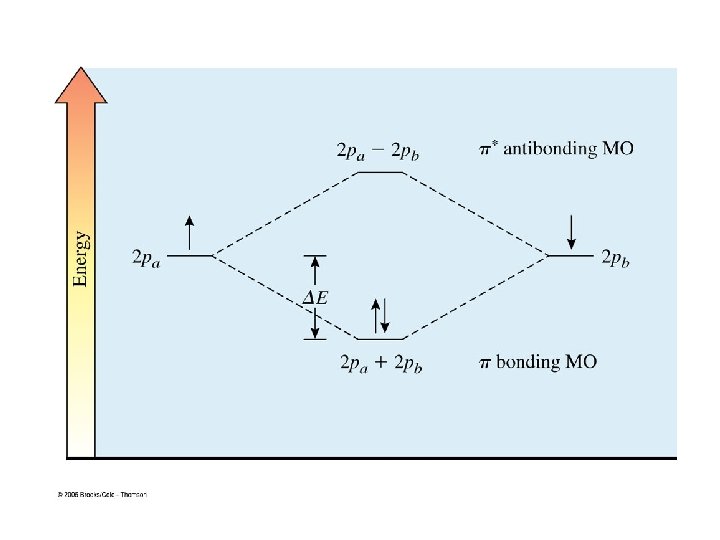
Generalizations for Simple MO Theory 1. two AOs overlap to form two MOs number of MOs = number of AOs 2. lower energy MO (bonding) higher energy MO with a node (antibonding) 3. use the same rules to assign electrons to MOs as used for AOs fill lowest energy MO first two electrons per MO (Pauli exclusion principle) Hund’s rule 4. bond energy is approximately equal to the number of electrons times DE 5. magnitude of DE increases with increasing overlap of the AOs
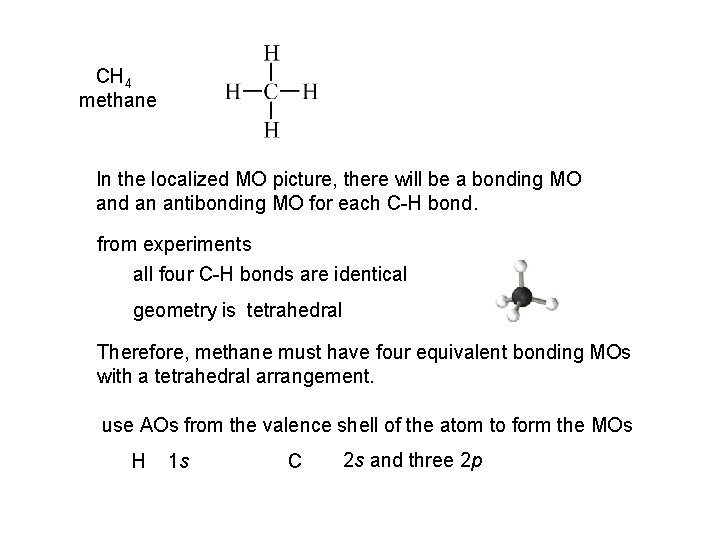
CH 4 methane In the localized MO picture, there will be a bonding MO and an antibonding MO for each C-H bond. from experiments all four C-H bonds are identical geometry is tetrahedral Therefore, methane must have four equivalent bonding MOs with a tetrahedral arrangement. use AOs from the valence shell of the atom to form the MOs H 1 s C 2 s and three 2 p
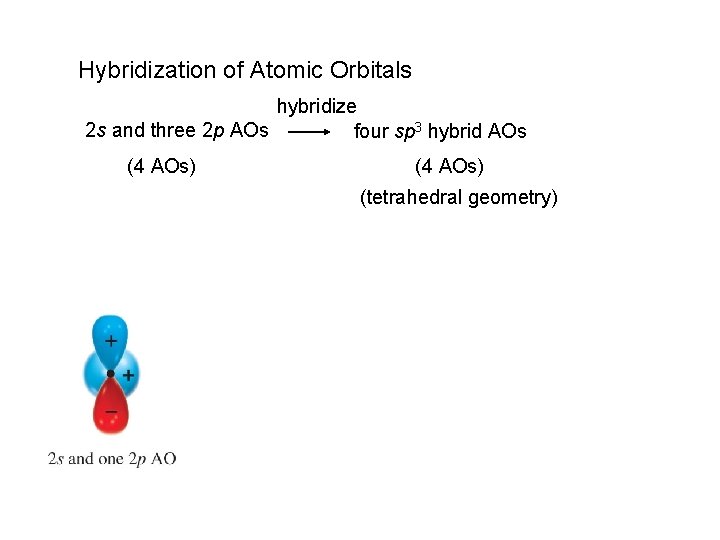
Hybridization of Atomic Orbitals hybridize 2 s and three 2 p AOs four sp 3 hybrid AOs (4 AOs) (tetrahedral geometry)
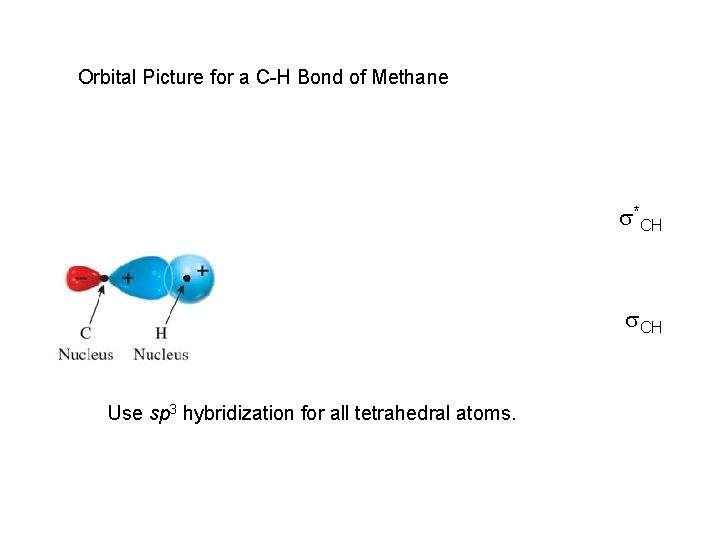
Orbital Picture for a C-H Bond of Methane s*CH s. CH Use sp 3 hybridization for all tetrahedral atoms.
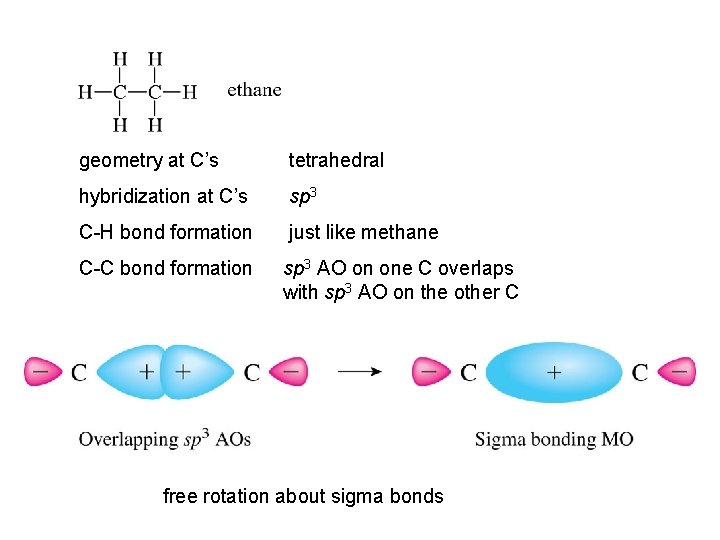
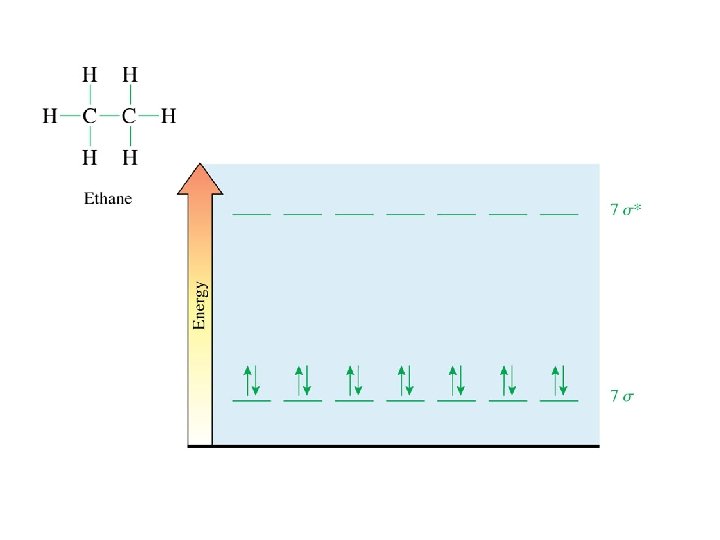
geometry at C’s tetrahedral hybridization at C’s sp 3 C-H bond formation just like methane C-C bond formation sp 3 AO on one C overlaps with sp 3 AO on the other C free rotation about sigma bonds
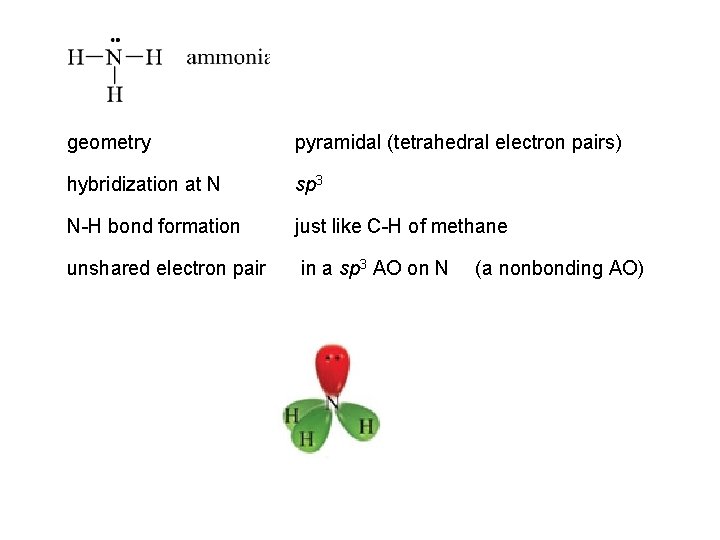
geometry pyramidal (tetrahedral electron pairs) hybridization at N sp 3 N-H bond formation just like C-H of methane unshared electron pair in a sp 3 AO on N (a nonbonding AO)
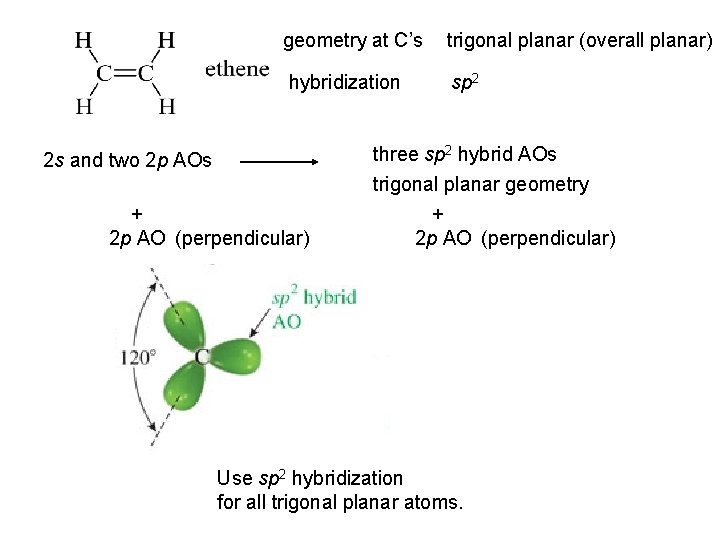
geometry at C’s trigonal planar (overall planar) hybridization sp 2 2 s and two 2 p AOs + 2 p AO (perpendicular) three sp 2 hybrid AOs trigonal planar geometry + 2 p AO (perpendicular) Use sp 2 hybridization for all trigonal planar atoms.
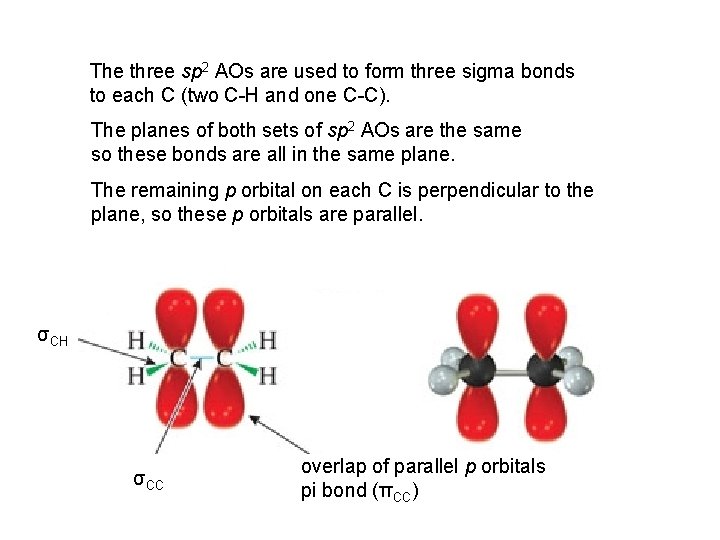
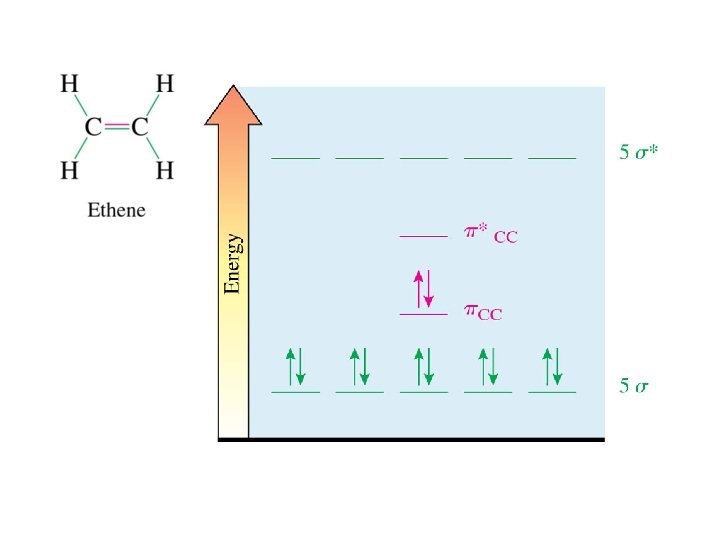
The three sp 2 AOs are used to form three sigma bonds to each C (two C-H and one C-C). The planes of both sets of sp 2 AOs are the same so these bonds are all in the same plane. The remaining p orbital on each C is perpendicular to the plane, so these p orbitals are parallel. σCH σCC overlap of parallel p orbitals pi bond (πCC)
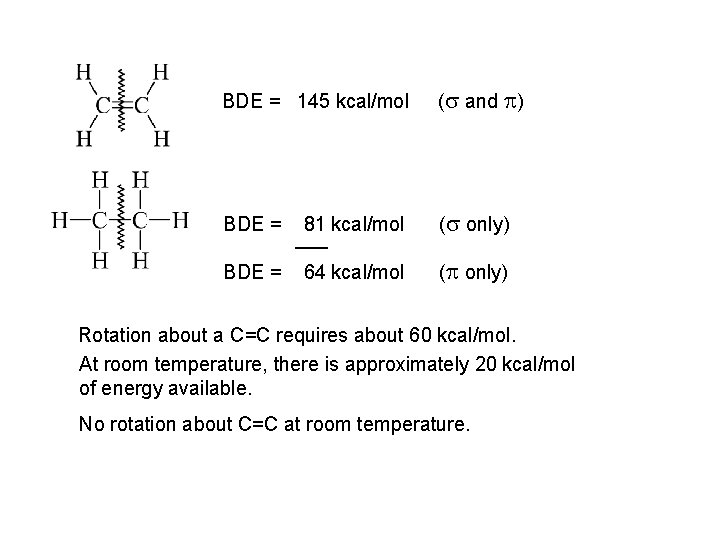
BDE = 145 kcal/mol (s and p) BDE = 81 kcal/mol ___ (s only) BDE = 64 kcal/mol (p only) Rotation about a C=C requires about 60 kcal/mol. At room temperature, there is approximately 20 kcal/mol of energy available. No rotation about C=C at room temperature.
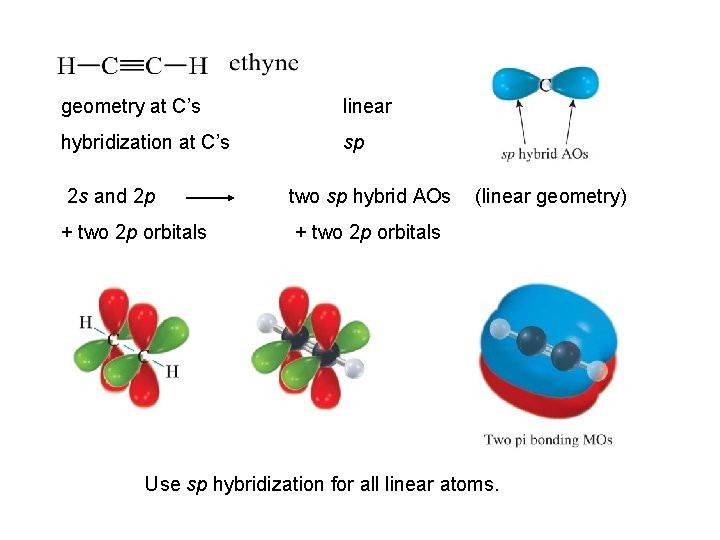
geometry at C’s linear hybridization at C’s sp 2 s and 2 p two sp hybrid AOs + two 2 p orbitals (linear geometry) Use sp hybridization for all linear atoms.
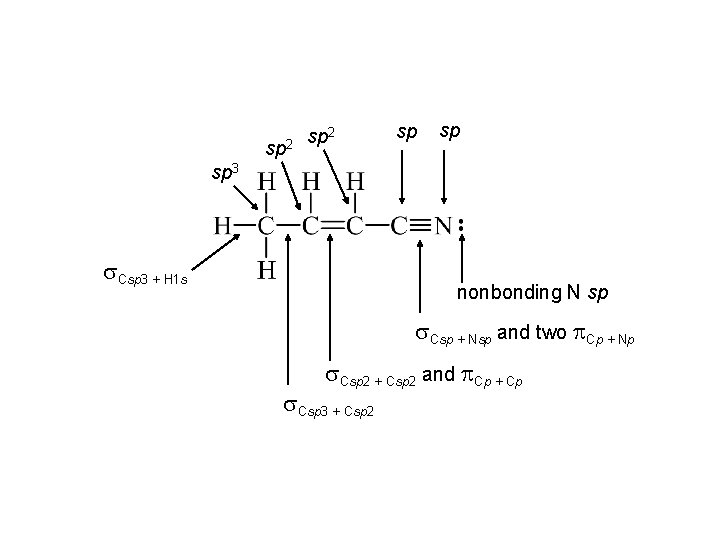
sp 2 sp sp sp 3 s. Csp 3 + H 1 s nonbonding N sp s. Csp + Nsp and two p. Cp + Np s. Csp 2 + Csp 2 and p. Cp + Cp s. Csp 3 + Csp 2
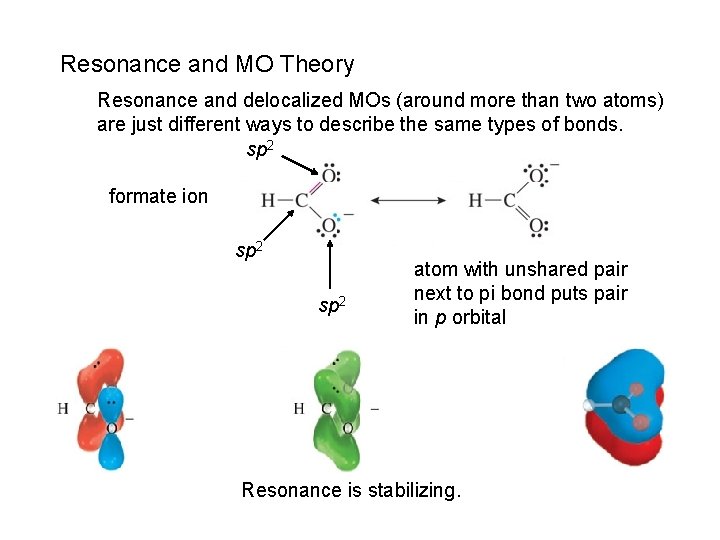
Resonance and MO Theory Resonance and delocalized MOs (around more than two atoms) are just different ways to describe the same types of bonds. sp 2 formate ion sp 2 atom with unshared pair next to pi bond puts pair in p orbital Resonance is stabilizing.
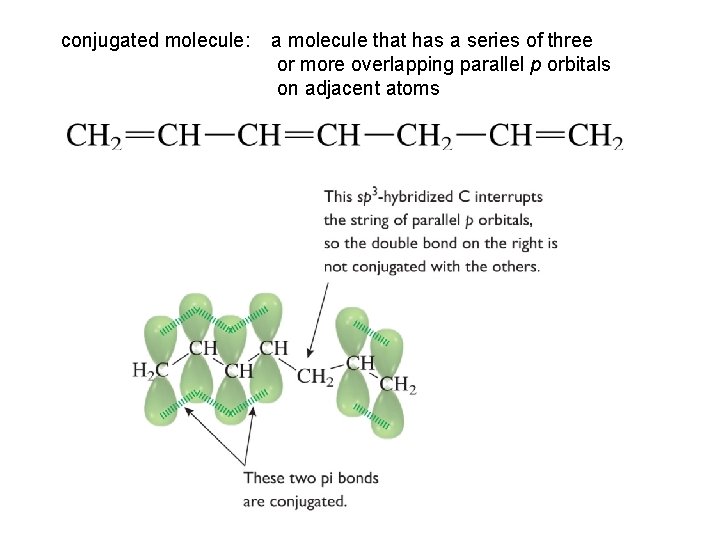
conjugated molecule: a molecule that has a series of three or more overlapping parallel p orbitals on adjacent atoms

Rules for Resonance Structures Rule 1: In drawing resonance structures, the nuclei of atoms may not be moved; only pi and nonbonding electrons in conjugated p orbitals may be moved. H has moved Rule 2: Each structure must have the same number of electrons and the same total charge.

Rule 3: The relative stability of resonance structures is judged by the same rules used to judge the stability of Lewis structures: octet rule number and location of formal charges interaction between charges more important - charge on more electronegative N less important unimportant (octet rule) Arrows always point from where the electrons are in a structure to where they are going in the next structure.

Rule 4: The actual structure most resembles the most stable resonance structure. Rule 5: The resonance stabilization energy increases as the number of important resonance structures increases.
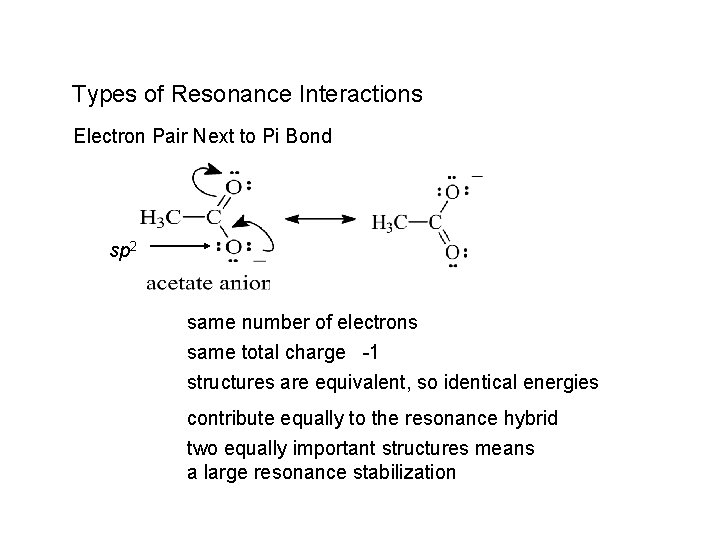
Types of Resonance Interactions Electron Pair Next to Pi Bond sp 2 same number of electrons same total charge -1 structures are equivalent, so identical energies contribute equally to the resonance hybrid two equally important structures means a large resonance stabilization
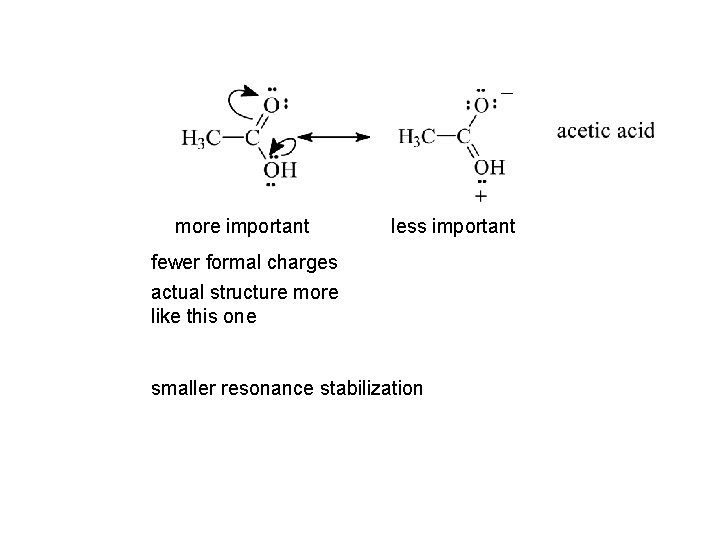
more important less important fewer formal charges actual structure more like this one smaller resonance stabilization
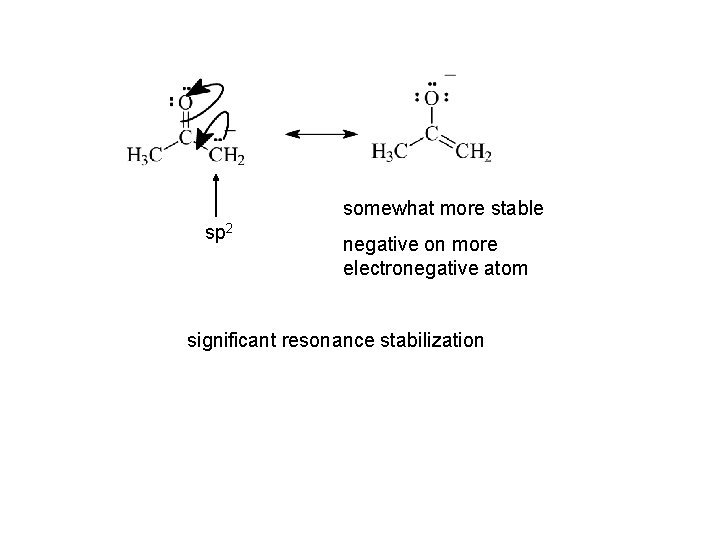
somewhat more stable sp 2 negative on more electronegative atom significant resonance stabilization
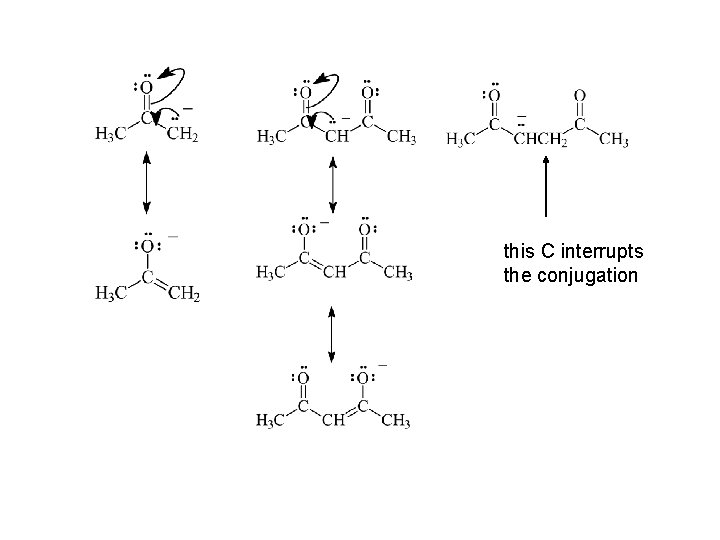
this C interrupts the conjugation
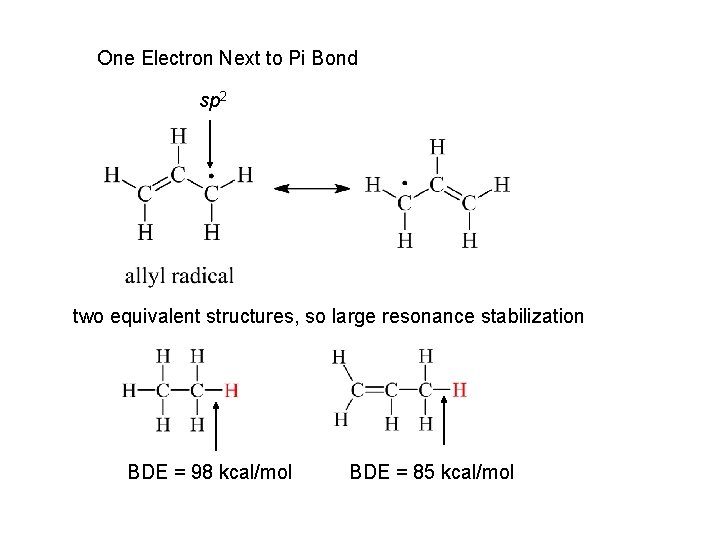
One Electron Next to Pi Bond sp 2 two equivalent structures, so large resonance stabilization BDE = 98 kcal/mol BDE = 85 kcal/mol
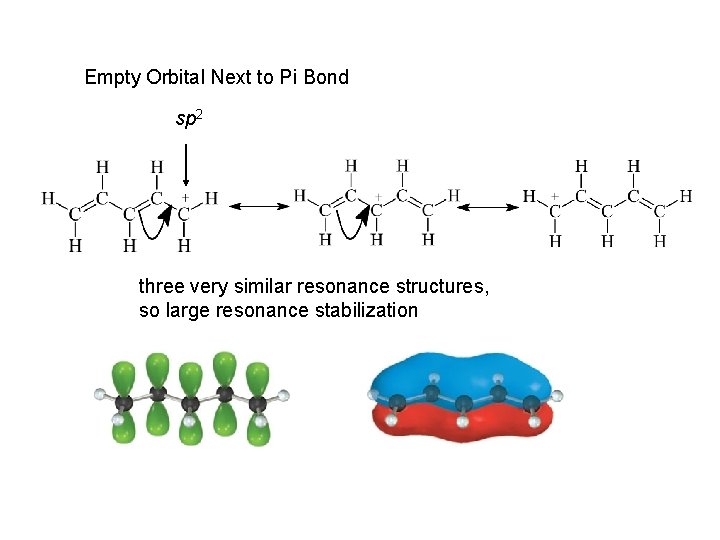
Empty Orbital Next to Pi Bond sp 2 three very similar resonance structures, so large resonance stabilization
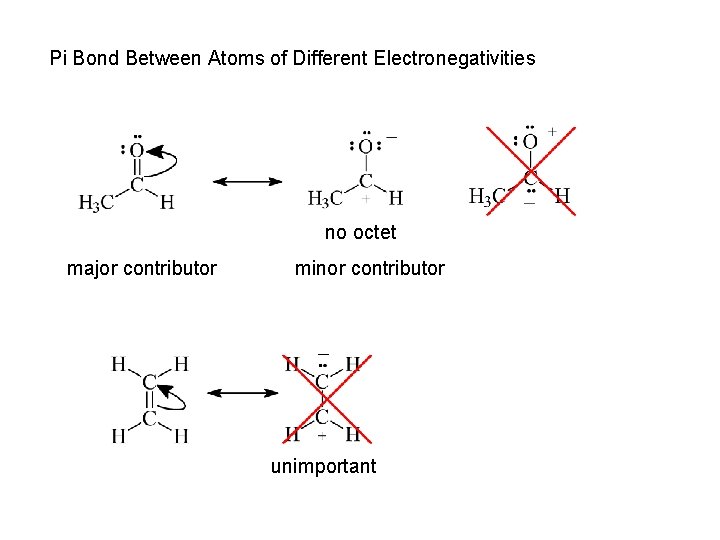
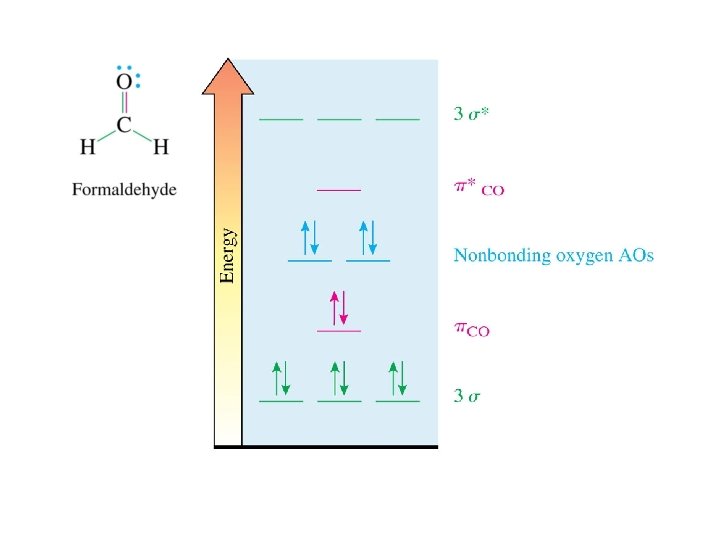
Pi Bond Between Atoms of Different Electronegativities no octet major contributor minor contributor unimportant
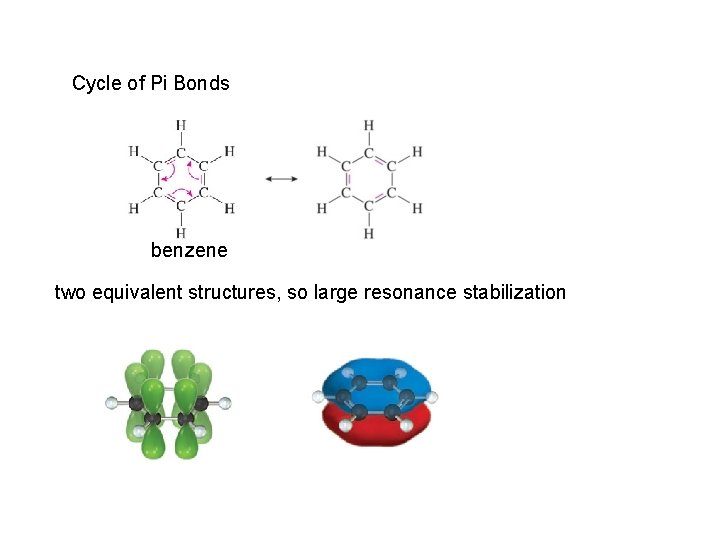
Cycle of Pi Bonds benzene two equivalent structures, so large resonance stabilization
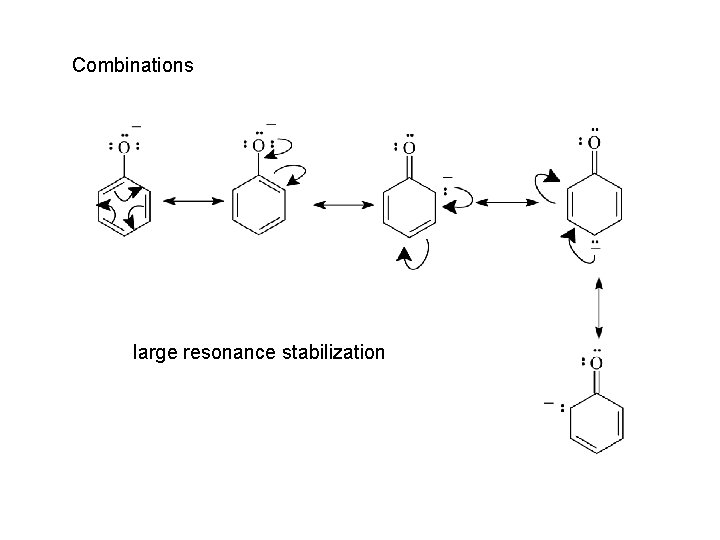
Combinations large resonance stabilization
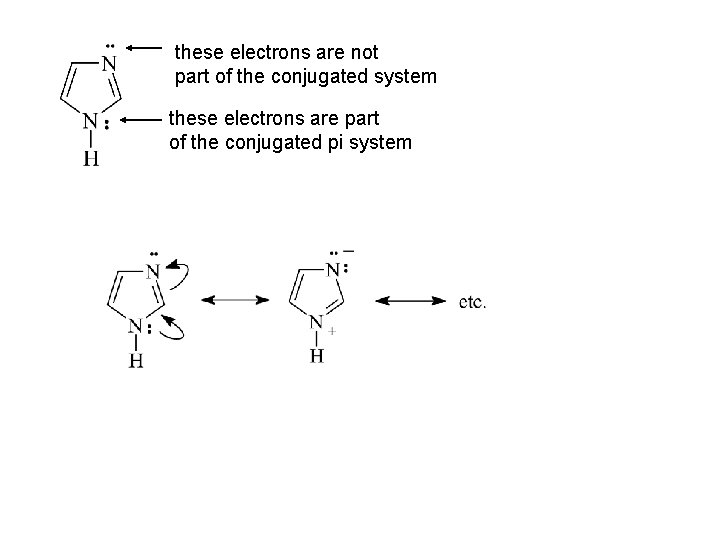
these electrons are not part of the conjugated system these electrons are part of the conjugated pi system
- Slides: 40