Chapter 3 Molecules compounds and chemical equations Combining

Chapter 3: Molecules, compounds, and chemical equations

• Combining two or more atoms forms a molecule • Combining two or more elements forms a compound • The composition and structure determine the physical and chemical properties of an element or compound Molecules

Chemical formula – tells you the elements and relative atoms in a compound • 2 types, molecular and empirical formula Chemical formulas Molecular formula give the actual number of atoms of each element in a compound • H 2 O 2 = two H atoms and two O atoms Empirical formula – gives the relative atom to atom ratio with whole numbers • H 2 O 2 , greatest common factor is 2 so in empirical form H 2 O 2 = HO • C 6 H 12 = CH 2
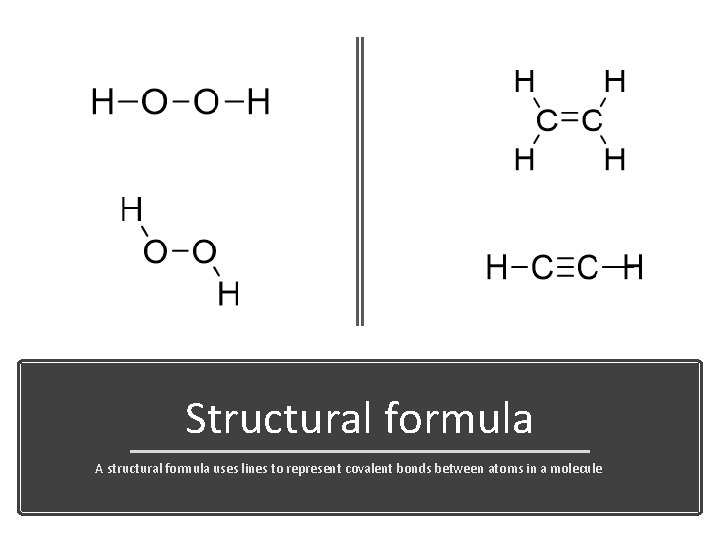
Structural formula A structural formula uses lines to represent covalent bonds between atoms in a molecule
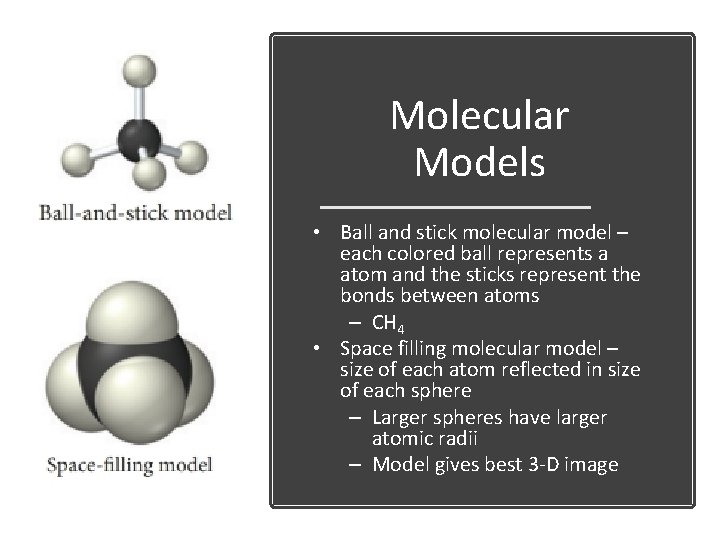
Molecular Models • Ball and stick molecular model – each colored ball represents a atom and the sticks represent the bonds between atoms – CH 4 • Space filling molecular model – size of each atom reflected in size of each sphere – Larger spheres have larger atomic radii – Model gives best 3 -D image
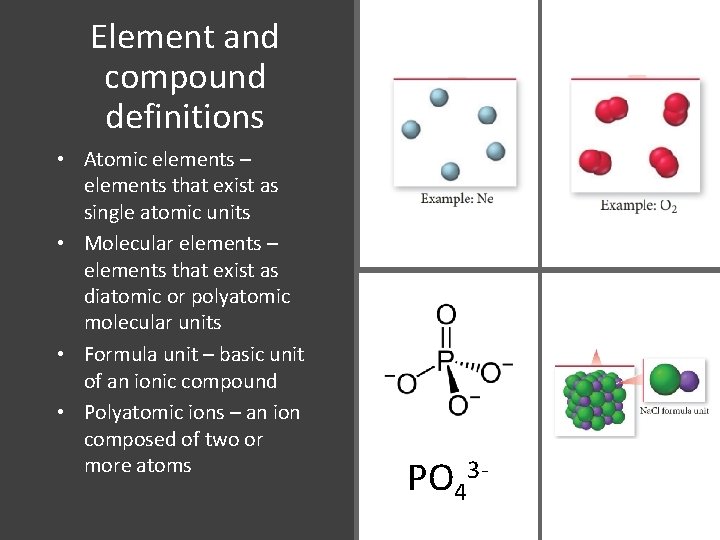
Element and compound definitions • Atomic elements – elements that exist as single atomic units • Molecular elements – elements that exist as diatomic or polyatomic molecular units • Formula unit – basic unit of an ionic compound • Polyatomic ions – an ion composed of two or more atoms PO 43 -

Ionic compounds vs Covalent compounds IONIC COVALENT
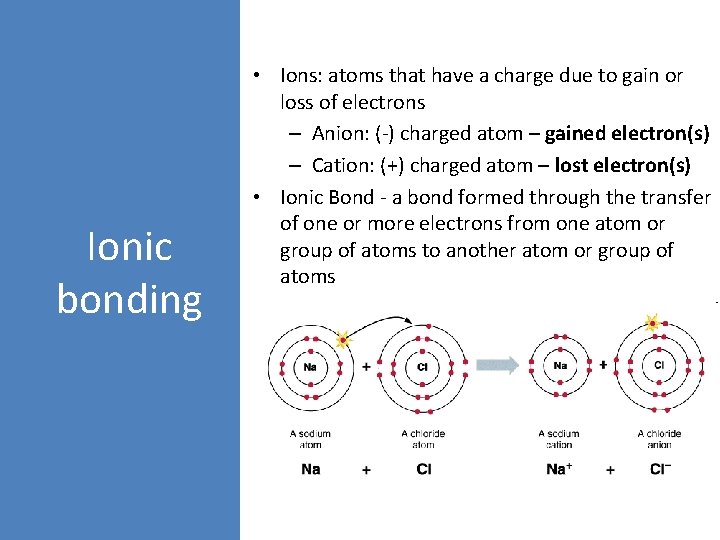
Ionic bonding • Ions: atoms that have a charge due to gain or loss of electrons – Anion: (-) charged atom – gained electron(s) – Cation: (+) charged atom – lost electron(s) • Ionic Bond - a bond formed through the transfer of one or more electrons from one atom or group of atoms to another atom or group of atoms
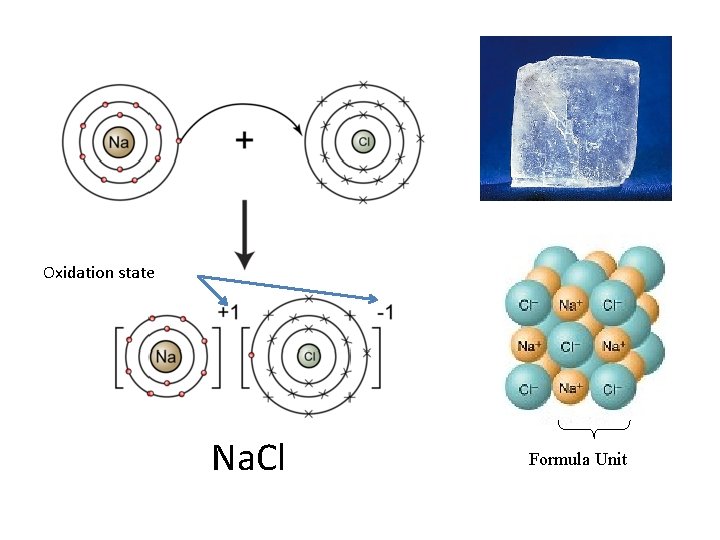
Oxidation state Na. Cl Formula Unit
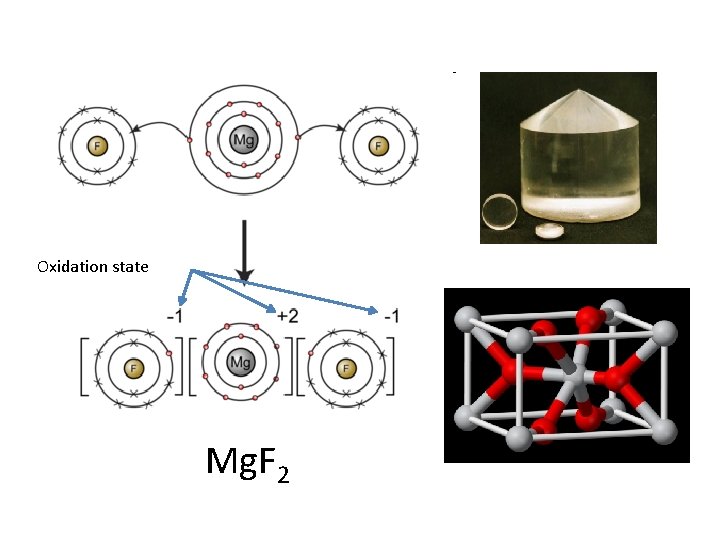
Oxidation state Mg. F 2

Ways to make an ionic compound • • • Metal + Non-metal – Na. Cl Metal + Polyatomic Ion – Na. NO 3 Polyatomic Ion + Non-metal – NH 4 Cl Polyatomic Ion + Polyatomic Ion – NH 4 NO 3 Net charge on compound equal to zero
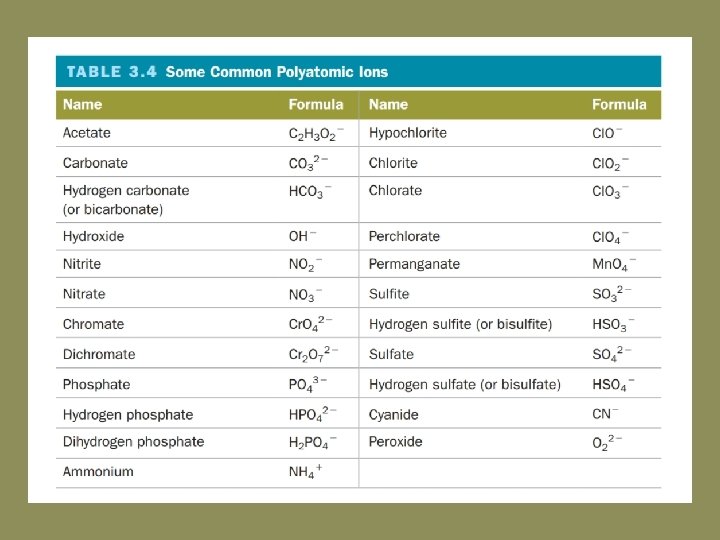
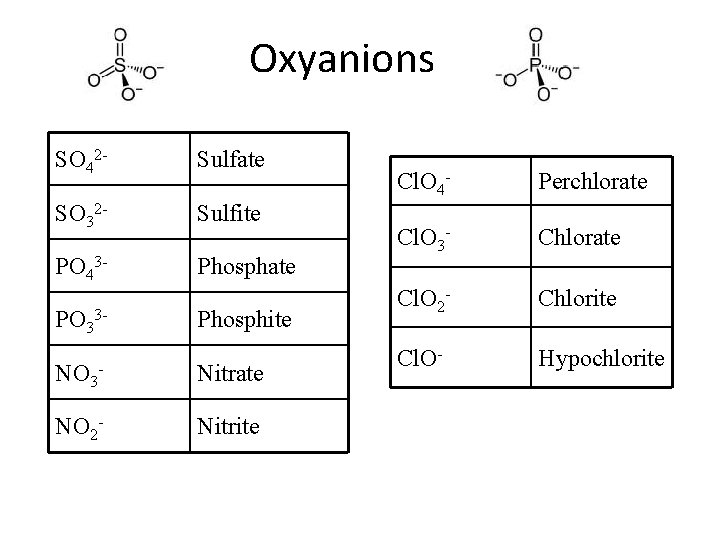
Oxyanions SO 42 - Sulfate SO 32 - Sulfite PO 43 - Phosphate PO 33 - Phosphite NO 3 - Nitrate NO 2 - Nitrite Cl. O 4 - Perchlorate Cl. O 3 - Chlorate Cl. O 2 - Chlorite Cl. O- Hypochlorite
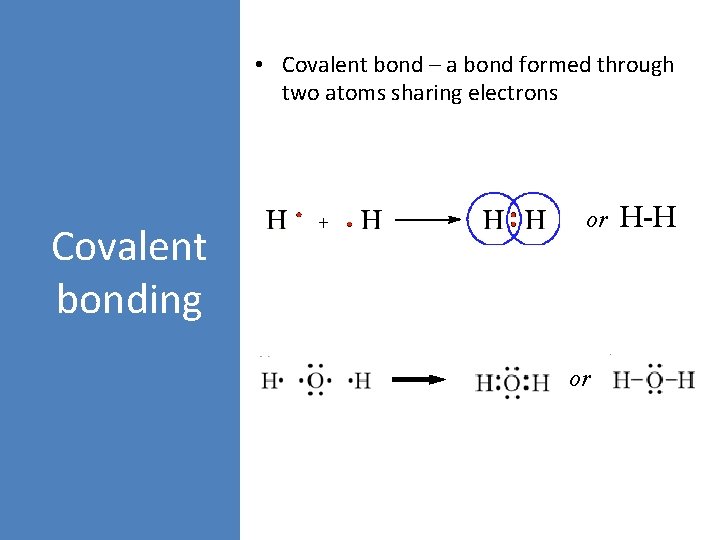
• Covalent bond – a bond formed through two atoms sharing electrons Covalent bonding or or H-H

Ways to make a covalent compound • Covalent compound - two or more nonmetals sharing electrons to make bonds create a covalent compound or molecular compound • nonmetal + nonmetal
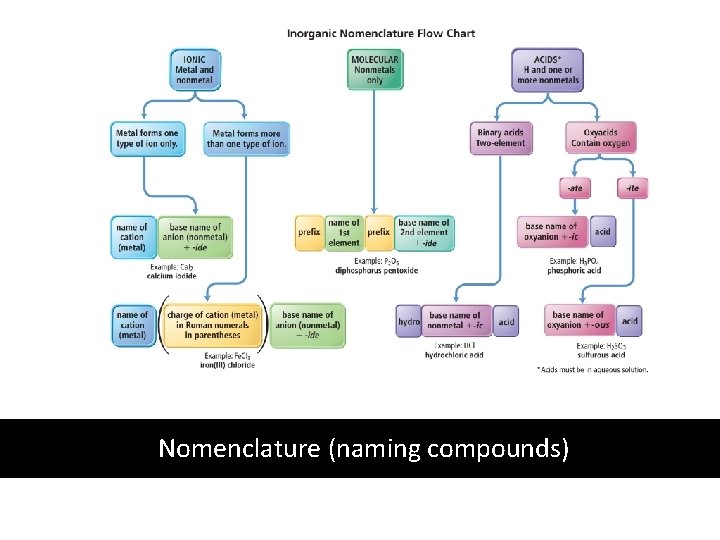
Nomenclature (naming compounds)

1) 2) Naming ionic compounds 3) 4) Name the cation by its elemental or polyatomic name If the metal is a transition metal with a variable charge, use a roman numeral in parentheses for its charge Next, name the anion and change its ending to “-ide” If the anion is polyatomic, do not change the ending to “-ide” Na. Br cation is Na+ = sodium anion is Br- = bromine bromide sodium bromide Fe. CO 3 Transition metal cation is Fe 2+ = iron(II) Polyatomic anion is CO 32 - = carbonate Iron(II) carbonate

Iron (III) Chloride Fe. Cl 3 Iron (II) Chloride Fe. Cl 2

Hydrated ionic compounds • Hydrates – contain a specific number of water molecules associated with each formula unit • The waters can typically be removed with heating • Ionic compound naming is the same, just add in hydrate and the prefix for the amount of waters

1) 2) 3) 4) Naming covalent compounds 5) Name the first non-metal by its elemental name Add a prefix to indicate how many If only one atom, don’t put mono Name the 2 nd non-metal and change its ending to “-ide” Add a prefix to indicate how many 1 mono 6 hexa 2 di 7 hepta 3 tri 8 octa 4 tetra 9 nona 5 penta 10 deca CO 2 First nonmetal C = carbon Second nonmetal O = oxygen oxide Carbon dioxide

Acids contain hydrogen atoms that can be donated to other molecules, indicated by being at the front of a chemical formula • Naming acids (acids w/o oxygen) 1) 2) 3) HBr Begin the name with “hydro” Name the anion, but change the ending to “ic” Add “acid” on the end The anion is Br = bromine bromic Hydrobromic acid

Naming acids (acids with oxygen) 1) Begin the name with the anion 2) If the anion has the ending “-ate, ” change this to “-ic acid” 3) If the anion has the ending “-ite, ” change this to “-ous acid” HNO 3 The anion is NO 3 - = nitrate nitric acid

Naming gas phase acids • • EXCEPTION, if in the gas phase, treat like a regular covalent compound for naming with no prefixes HF(g) = hydrogen fluoride

The Mole
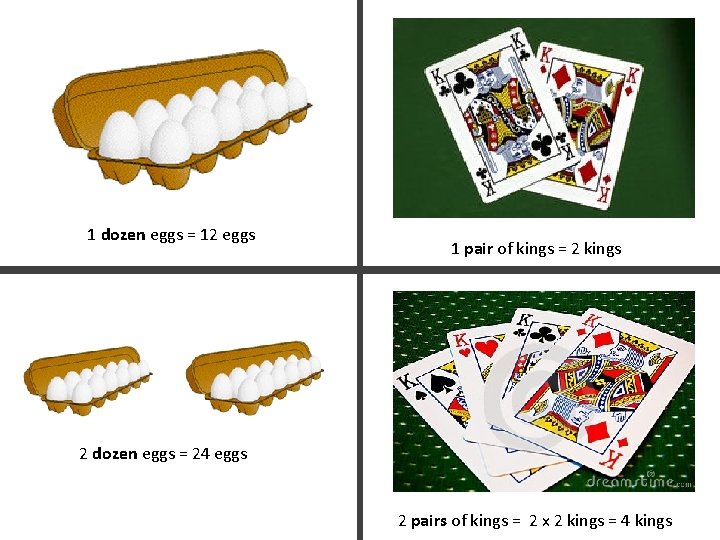
1 dozen eggs = 12 eggs 1 pair of kings = 2 kings 2 dozen eggs = 24 eggs 2 pairs of kings = 2 x 2 kings = 4 kings

The Mole • 1 mole = 6. 022 x 1023 things – Avogadro’s number • Abbreviation: “mol” • 1 mol = The number of carbon atoms in 12 g of C-12

• 1 mole C = 6. 022 x 1023 C atoms Moles to atoms or molecules • 1 mol H 2 O = 6. 022 x 1023 H 2 O molecules • If you have moles of something, you can convert to atoms or molecules of that something

Molar mass moles to grams • The mass, in grams, of one mole of any element or compound • Unit = grams/mole = g/mol • Gives us a way to go from grams to moles

Molar Masses for elements and molecules • Use periodic table to figure out Molar mass for each element. • Carbon – 1 atom of carbon is 12. 01 amu – 1 mole of carbon is 12. 01 grams – Molar Mass = 12. 01 g/mol • H 2 O – Made of 2 hydrogen atoms and 1 oxygen atom – Molar mass of each hydrogen is 1. 008 g/mol – Molar mass of each oxygen is 16. 00 g/mol Molar mass of H 2 O

Problems 1) You have 145. 9 g Hg. a. How many moles are in the sample? b. How many atoms of Hg are in the sample? 2) What is the molar mass for C 3 H 8 (propane)? 3) What is the amount of grams in 3. 57 moles of iron(III) chloride (Fe. Cl 3)?

Composition of compounds • Mass percent – the percentage of mass of an element in a compound (keep units consistent since it is a ratio) • Mass percent of H in H 2 O • 1. 008 amu per H atom x 2 H atoms = 2. 016 amu • 18. 016 amu for H 2 O

• Practice If you have 55. 4 grams of H 2 O, how many grams of that sample is hydrogen?

Chemical formula from empirical data • Decomposing a compound can give you the grams of each of the constituent elements of that compound • The grams can be converted into moles of each element • The whole numbered mole ratio between the elements gives the empirical formula

Grams to chemical formula • 25. 0 g gas decomposes into 18. 75 g of carbon and 6. 25 g of hydrogen. What is the chemical formula of the original gas? = =

Mass percent to chemical formula • Given the mass percentages of elements in a compound you can determine in a 100 g sample, how much grams of each element would be present • C 60. 00%, H 4. 48 %, O 35. 52 % • In 100 g sample – C 60. 00 g, H 4. 48 g, O 35. 52 g • Once you have the grams, continue the rest of the steps from previous slide

Mole ratios of a molecule • The atoms of an element in a molecule can be represented as a mole ratio • 2 atoms of H in 1 molecule of H 2 O • 2 mol of H in 1 mol of H 2 O – 2: 1 H: O ratio • 3 mol of Cl in 1 mol Fe. Cl 3 – 3: 1 Cl: Fe ratio • Mole ratio gets you from moles of one element to moles of another element
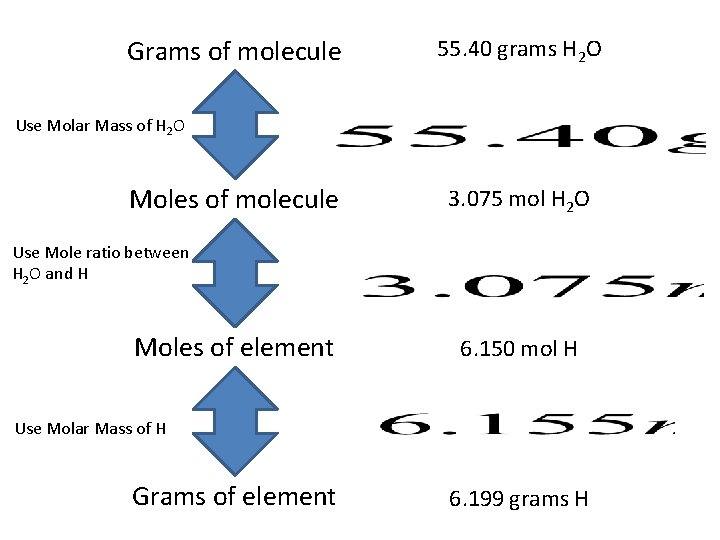
Grams of molecule 55. 40 grams H 2 O Use Molar Mass of H 2 O Moles of molecule 3. 075 mol H 2 O Use Mole ratio between H 2 O and H Moles of element 6. 150 mol H Use Molar Mass of H Grams of element 6. 199 grams H
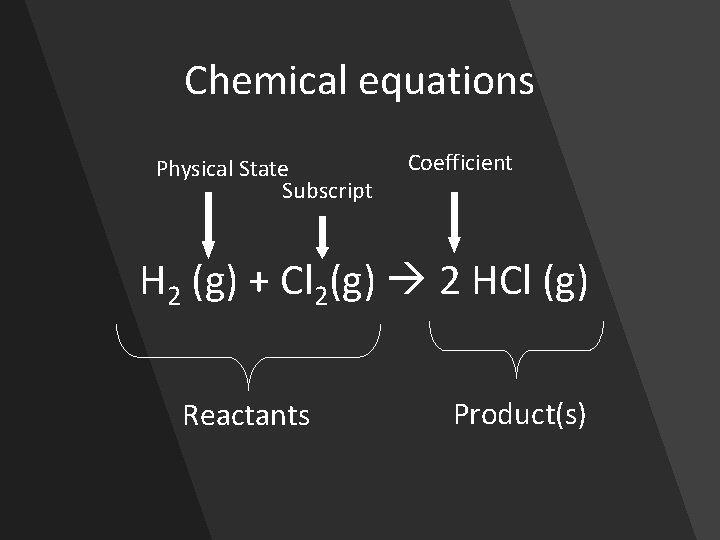
Chemical equations Physical State Subscript Coefficient H 2 (g) + Cl 2(g) 2 HCl (g) Reactants Product(s)

Law of conservation of Mass • Matter is neither created, nor destroyed, but is merely rearranged • The mass of the reactants must equal the mass of the products • Atoms in the reactants must equal atoms in the products • H 2 (g) + Cl 2(g) 2 HCl (g)

Balancing Chemical Equations • Write the unbalanced equation • Balance the atoms of one element (saving single elements for last) • Choose another element and balance it • Continue until all elements have the same number of atoms on both sides of the equation • Check yourself • __Zn(s) + __HCl(aq) __H 2(g) + __Zn. Cl 2(aq)

Problems 1) __ N 2(g) + __ H 2(g) __ NH 3(g) 2) __ Fe(s) + __ Cl 2(g) __ Fe. Cl 3(s) 3) __NH 3(g) + __O 2(g) __NO(g) + __H 2 O(g) 4) __C 5 H 12(l) + __O 2(g) __CO 2(g) + __H 2 O(g)
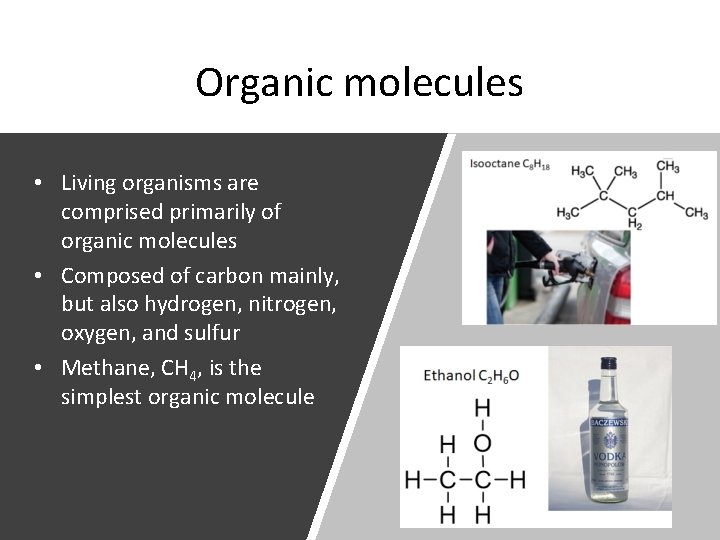
Organic molecules • Living organisms are comprised primarily of organic molecules • Composed of carbon mainly, but also hydrogen, nitrogen, oxygen, and sulfur • Methane, CH 4, is the simplest organic molecule
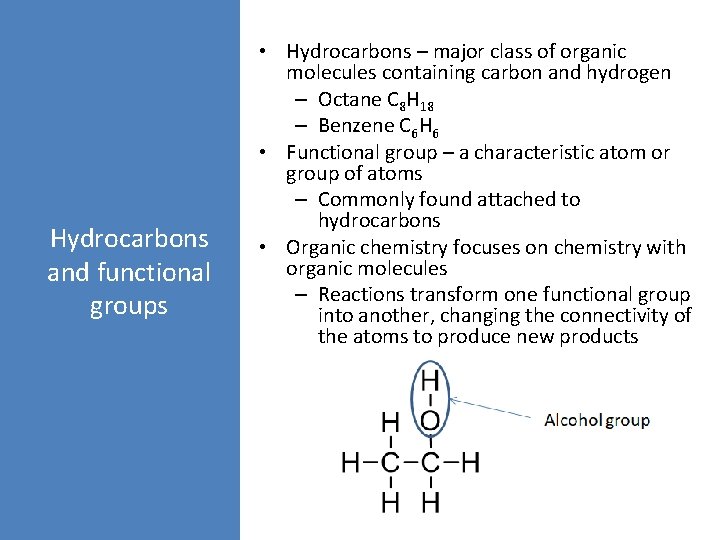
Hydrocarbons and functional groups • Hydrocarbons – major class of organic molecules containing carbon and hydrogen – Octane C 8 H 18 – Benzene C 6 H 6 • Functional group – a characteristic atom or group of atoms – Commonly found attached to hydrocarbons • Organic chemistry focuses on chemistry with organic molecules – Reactions transform one functional group into another, changing the connectivity of the atoms to produce new products
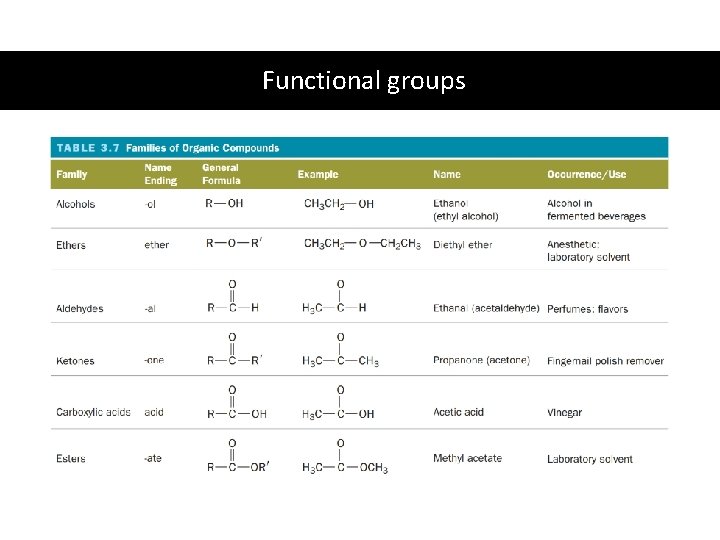
Functional groups

Chapter 3…all done
- Slides: 46