Chapter 3 Molecular Shape and Structure i ii













- Slides: 23

Chapter 3: Molecular Shape and Structure • • • i. ii. Molecular Shape and Structure ALL MOLECULAR FUNCTION can be related to MOLECULAR SHAPE In this chapter, we are going to cover 2 models that relate to chemical bonding VSEPR: Valence Shell Electron Pair Repulsion Model Valence Bond Theory


VSEPR Theory • In the Valence Shell Electron Pair Repulsion model (VSEPR), molecular shapes are determined by empiricial observations • Before we jump into the details of theory, we need to consider some of the geometry that we will be dealing with this chapter

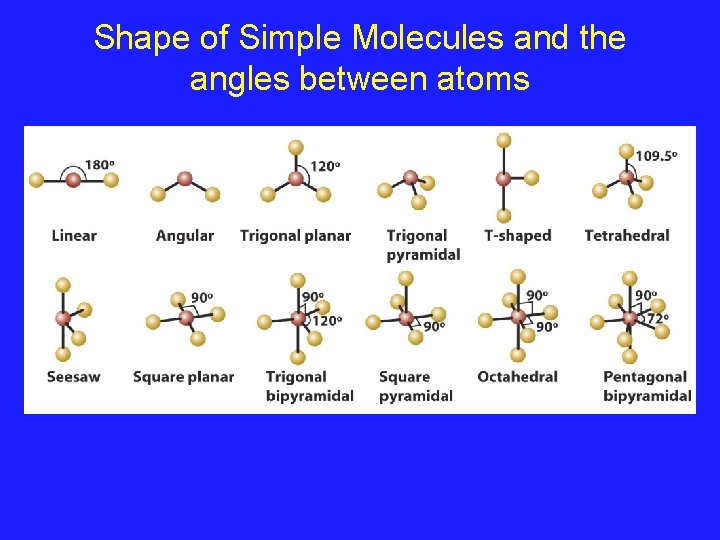
Shape of Simple Molecules and the angles between atoms






Now that we know the possible geometries… • VSEPR model builds on the Lewis structures we learned how to draw last chapter – If you didn’t learn how to draw them, SEE ME!! – The the VSEPR model, we apply rules to the final Lewis structures that determine bond angles • Rule #1: Regions of high electron concentration (Bonds and Lone pairs on the central atom) repel one another and, to minimize these repulsions, these regions move as far apart as possible from each other without moving away from the central atom.

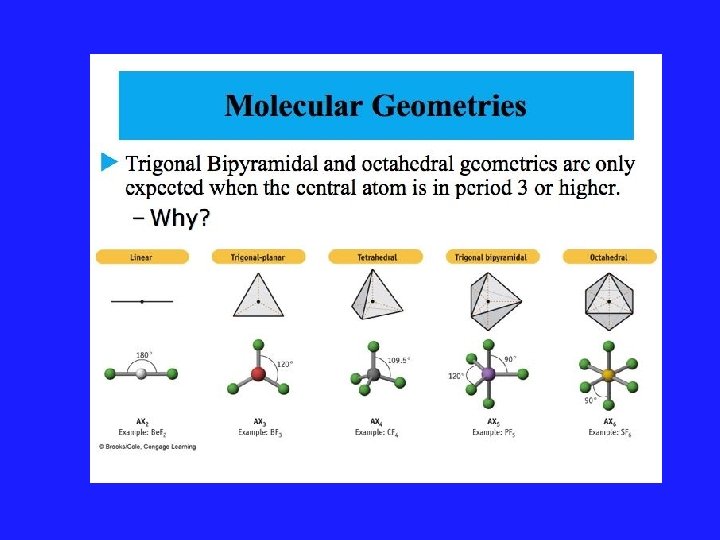
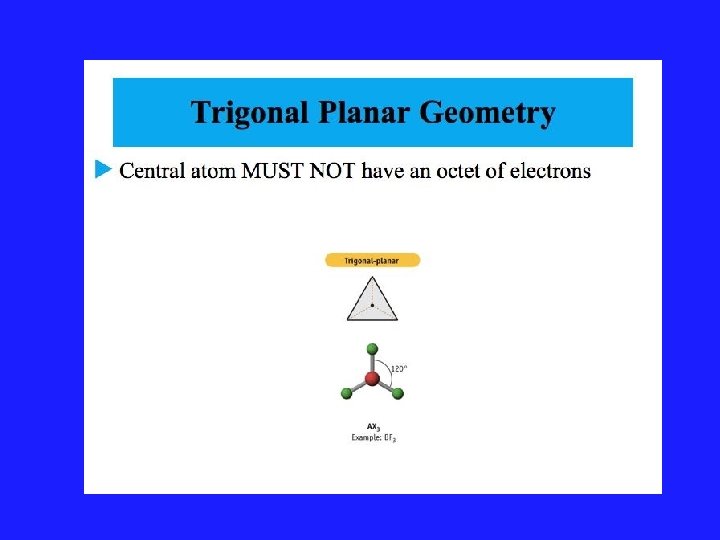
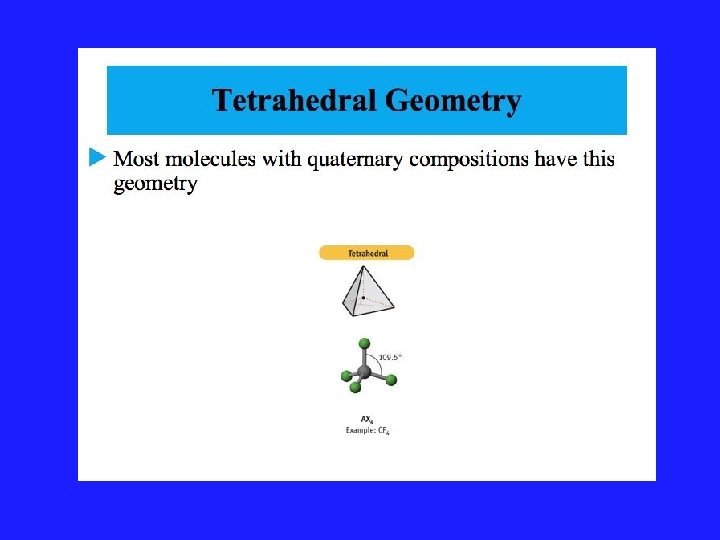
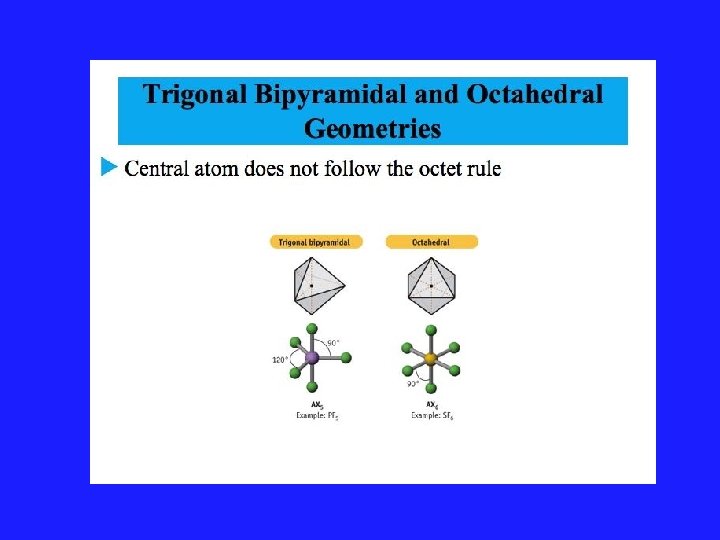
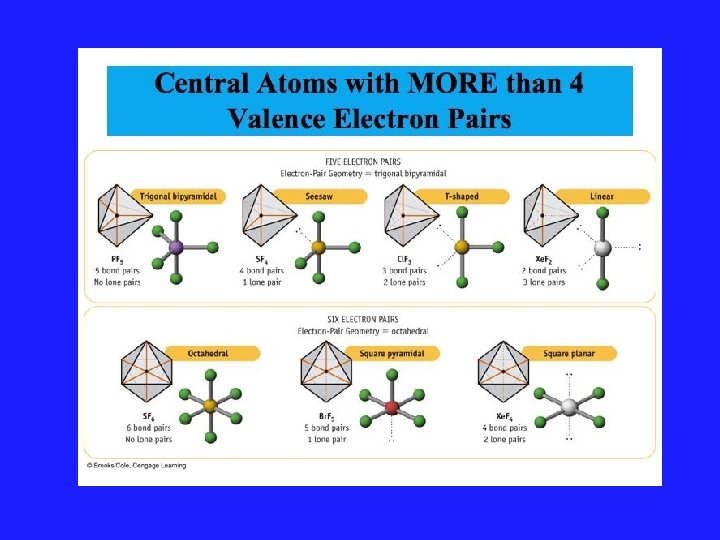
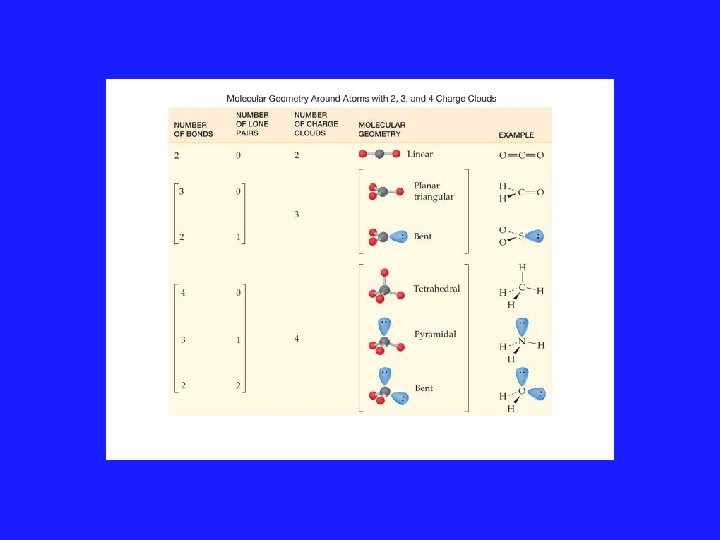
Using VSEPR for a central atom without Lone Pairs 1. Once we have drawn the Lewis structure and know the basic arrangement of the bonds, we further arrange them to maximize the distance 2. We name them in accordance with the names given in Figure 3. 1 of your text

VSEPR Model and Multiple Bonds Rule #2: There is no distinction between multiple and single bonds for geometry purposes • The pairs are groups together in a multiple bond, so we consider them to act as a single unit


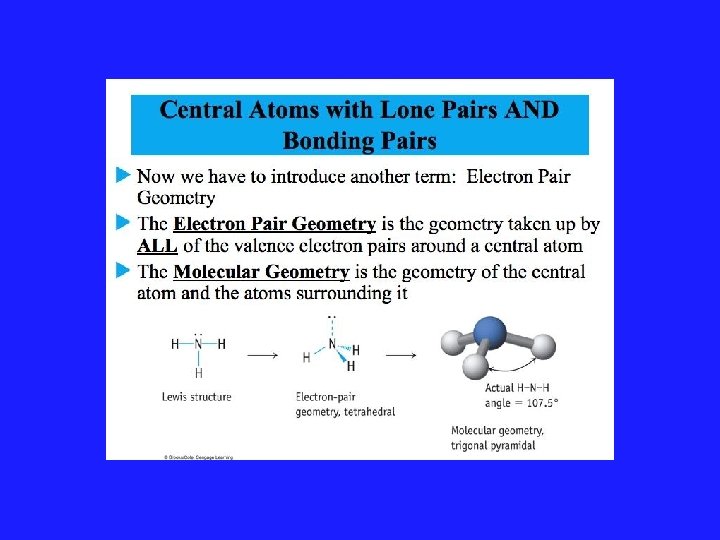
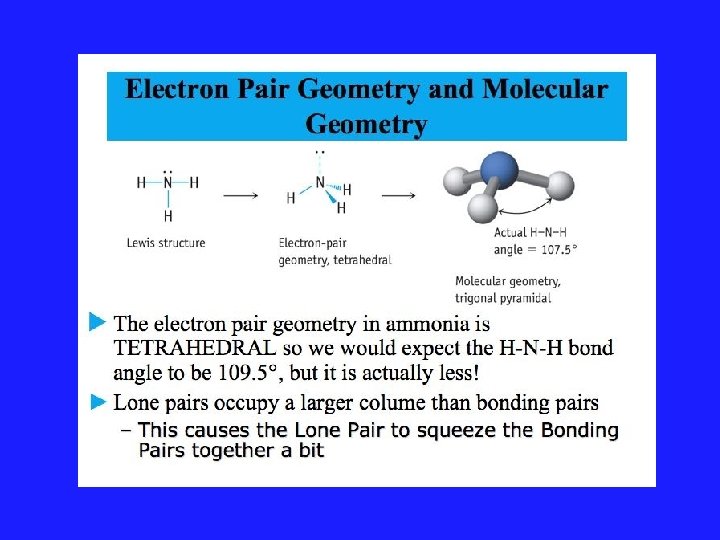
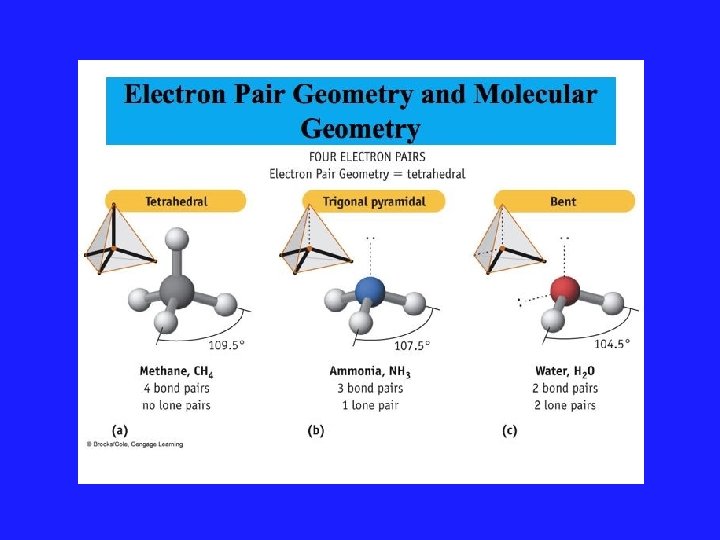
VSEPR Model and Lone Pairs • Rule #3: All regions of high electron density, lone pairs and bonds, are included in a description of the electronic arrangement. But only the position of the atoms are considered when reporting the shape of the molecule.






VSEPR Model Rule for Repulsion • Rule #4: Electron repulsions are ranked in decreasing order: Lone pair - lone pair Lone pair - atom Atom - atom • Lone pairs must be as far apart as possible to minimize energy

VSEPR and Polarity • Last chapter, we saw that some coavelnt bonds exist in which electrons are not shared equally between 2 atoms – We call these Polar Bonds • For polyatomic molecules, several polar bonds may exist – The sum total of all the polar bonds in a molecule will determine its polarity • So what? – Molecular polarity determines many of the chemical properties for a molecule

VSEPR and Polarity • We use molecular geometries to determine the polarity of a molecule

VSEPR and Polarity: Summary A diatomic molecule is polar if its bond is polar. (We can tell this from the electronegativity of the bonded atoms) A polyatomic molecule is polar if it has polar bonds that are arranged in space in such a way that the dipole moments associated with the bonds do not cancel each other out.