CHAPTER 3 MATTER AND ATOMIC STRUCTURE Questions of









































- Slides: 50

CHAPTER 3 MATTER AND ATOMIC STRUCTURE

Questions of the Day – 1/9/14 • 1. Which of the following is defined as something that cannot be broken down into simpler substances? • A. Mixture • C. Compound B. Element D. Panda • 2. What of the following is the smallest part of a substance which still has the characteristics of that substance? • A. Proton • C. Atom B. Element D. Quark • 3. How many protons does Helium have? • A. 1 B. 2 • C. 3 D. 4

Today’s Objectives • Define and describe element, atom, and the periodic table • Identify and describe the sub-atomic particles within atoms • Use the atomic number and mass number of elements to create diagrams of their atoms

What Makes Up All Matter? Element • Something that can’t be broken down into simpler pieces. • Exp: Gold Atom • The smallest part of the element that still has all of the same characteristics of the element. • Can be melted, divided into small pieces, hammered flat…. still gold • 92 Naturally Occurring • 20 Man-made • Atoms are made up of • 1). Protons • 2). Neutrons • 3). Electrons

Parts of an Atom Proton • Small particle found inside the nucleus of an atom with a positive charge • Usually represented by a plus sign when atoms are drawn out. • Atomic Number = Tells you the number of protons an element has. • Located above the element abbreviation on the periodic table of elements. • Periodic Table is organized by atomic number. • You cannot change the number of protons found within an atom without changing the element you are working with.

Parts of an Atom Neutrons • Small particle found inside the nucleus of an atom with a neutral charge/ no charge • Hold together all the positive charges in the nucleus. • Act as the glue to prevent all the positive charges from repelling each other. • Stabilizes the nucleus • Have to use a little subtraction to find the number of neutrons. Mass Number • The rounded whole number located beneath the abbreviation of the element. • Round the decimal point to the nearest whole number to obtain the mass number (the exact numbers of protons and neutrons in the nucleus of an particular atom). • Use the Mass Number of an element to find the number of neutrons by subtracting the number of protons (Atomic Number) from it.

Parts of an Atom Electrons • Smallest particle with a negative charge found organized into clouds outside the nucleus of an atom. • For all ‘normal’ atoms you can find the number of electrons because positive and negative charges must cancel out…. • So, an atom must have the same number of protons and electrons. • Atomic Number also tells you the number of electrons.

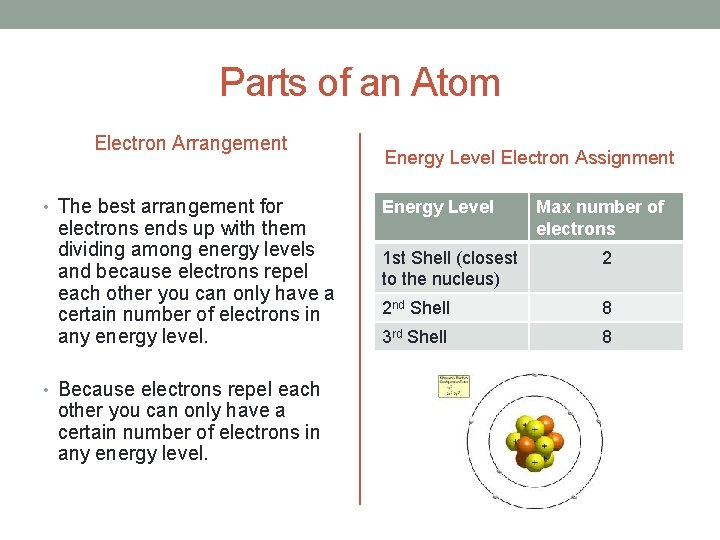
Parts of an Atom Electron Arrangement • These negative particles • Electrons are negatively are held in by their attraction to the positively charged nucleus. charged and want to repel each other. • Remember opposites attract are as far away from another electron as possible. • But, usually you can’t fit all of the electrons right around the nucleus. • Why? • They sort themselves so they

Question of the Day – 1/10/14 • 1. How many protons does the element Silver have? • Answer: 47 • 2. How many neutrons does the element Phosphorous have? • Answer: 16

Today’s Objectives • Practice using the Periodic Table to determine an atom’s proton, electron, and neutron counts. • Use the atomic number and atomic mass number of elements to create diagrams of their atoms

Parts of an Atom Electron Arrangement • The best arrangement for electrons ends up with them dividing among energy levels and because electrons repel each other you can only have a certain number of electrons in any energy level. • Because electrons repel each other you can only have a certain number of electrons in any energy level. Energy Level Electron Assignment Energy Level Max number of electrons 1 st Shell (closest to the nucleus) 2 2 nd Shell 8 3 rd Shell 8

Parts of an Atom Valence Electrons • The electrons that are in the energy level farthest away from the nucleus. • These electrons give us a lot of information about the element. • How/If it is reactive. • Chemical Properties

Question of the Day – 1/13/14 • 1. How many valence electrons does Magnesium have? • Answer: 2 • 2. How many valence electrons does Beryllium have? • Answer: 2 • 3. Without drawing or calculating it out, (look at the trend in your periodic table from questions 1 and 2), take an educated guess on how many valence electrons Strontium has. Answer: 2 The trend is the vertical grouping of these elements. All elements in a vertical group have the same number of valence electrons.

You do not need your clicker today! • Find a blank sheet of paper (Not notebook! No lines!) • From yourself • From the black files under the bookshelf • Study/prepare for quiz • Any questions?

Quiz - On your blank piece of paper: • For the element: Aluminum • Identify the total number of Protons -> P = _____ • Identify the total number of Neutrons -> N = _____ • Identify the total number of Electrons -> E = _____ • Draw the Bohr Diagram • You do not need to draw the protons or neutrons inside the nucleus • You may simply leave the inside of the nucleus blank • Draw the Lewis Diagram – also known as the Electron Dot Diagram • After your are done, put your paper in the wire basket. Be sure your name is on it! Grab 2 new papers. Please remain quiet while others finish their quizzes. You may read (check out my book/magazine selection) or do some other quiet activity.

Periodic Table Vocabulary • • • Periods = Rows that travel across the periodic table horizontally • • Groups = Columns of elements that are grouped vertically on the periodic table.

Periodic Table Trends • For Atoms, 8 is the magic number of valence electrons they want (or in hydrogen and helium’s case ~ 2).

Group 1: Alkali Metals • All of these elements have 1 electron in their outer shells. • Like to get rid of their electron. • Highly reactive • Metals • Low Density

Group 2: Alkaline Earth Metals • All of the elements have 2 electrons in their valence shell. • Like to get rid of their electrons. • Reactive • Silver Colored • Soft Metals

Groups 3 -12 Transition Elements • Have a vast array of properties • Characteristics vary greatly

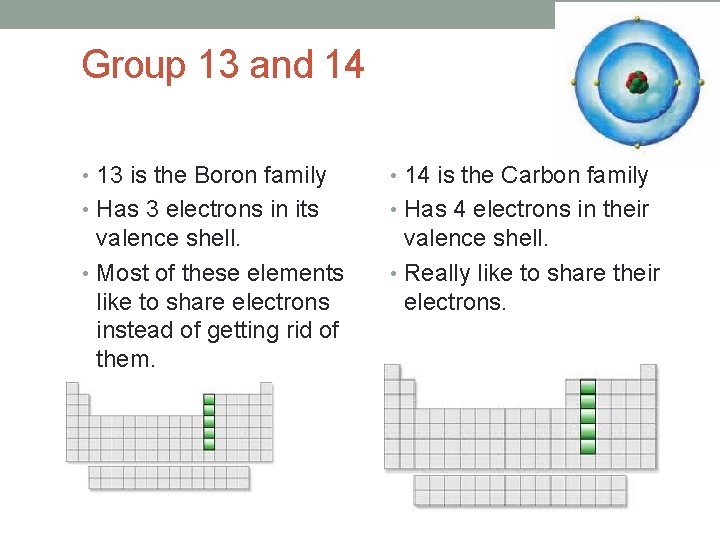
Group 13 and 14 • 13 is the Boron family • 14 is the Carbon family • Has 3 electrons in its • Has 4 electrons in their valence shell. • Most of these elements like to share electrons instead of getting rid of them. valence shell. • Really like to share their electrons.

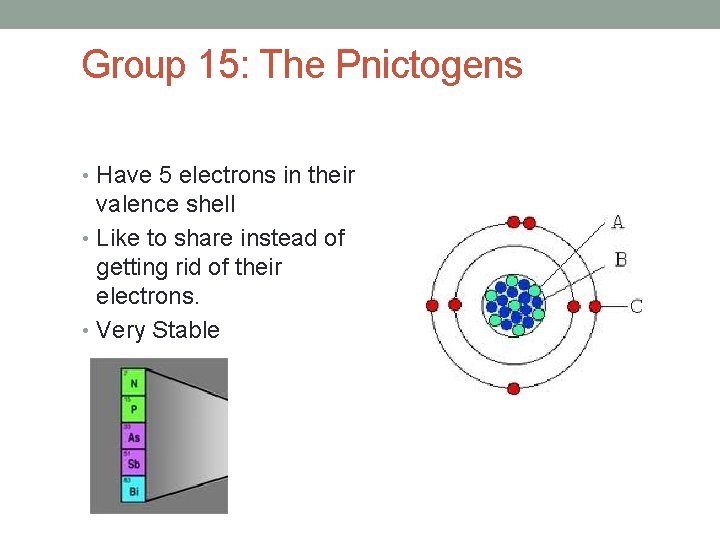
Group 15: The Pnictogens • Have 5 electrons in their valence shell • Like to share instead of getting rid of their electrons. • Very Stable

Group 16 and 17 • 16 = Chalcogens • Have 6 valence shell electrons • Most elements in this family like to steal electrons. • 17 = Halogens • Have 7 electrons in their valence shell. • Elements in this family really like to steal electrons. • Elements in all states of matter

Group 18: The Noble Gases • Why are these so • Odorless special/unique? • Content with the number of electrons they have. • Colorless • Full valence shell of 8. • Low Reactivity • Very Few Compounds Exist

Electron Arrangement Examples Hydrogen = atomic number 1 1 p Helium = atomic number 2 2 p Lithium = atomic number 3 3 p Beryillium = atomic number 4 4 p

Electron Arrangement Examples Carbon = Atomic number 6 Neon = Atomic number 10 6 p Oxygen = Atomic number 8 8 p 10 p Sodium = Atomic number 11 11 p

Ionic Bonds Electron Exchange • Some atoms bond by gaining or losing electrons. • Ion = an atom with more or less electrons. Opposites Attract • If an atom gains electrons then it will be negative • Anions • If an atom loses electrons then it will be positive. • Cations - cats are positive people • Once atoms become charged they attract • Positive and Negative charges attract.

Question of the Day – 1/15/14 • 1. In order to get to the magic number 8, what does Calcium “want” to do? • A. Give 2 - correct answer • C. Steal 2 B. Give 1 D. Steal 1 • 2. A cation is a… • A. Negatively charged ion B. Negatively charged isotope • C. Positively charged ion–correct D. Positively charged isotope • 3. What is the official name of Group 18? • A. Pnictogens B. Noble Gases–correct • C. Boron Group D. Alkaline Earth Metals

Ionic Poker • For each round, say a person’s name aloud. • Decide what the element on the card “wants” to do • “Give 2” or “Steal 2” etc • Say it aloud. Do that action with the person whose name you called at the beginning of the round. • If you are incorrect, you must give everyone in the group the correct amount (even if it is supposed to be steal) • Be the highest chip holder at the end of the class period.

Ionic Poker Rules Change • For each round, say a person’s name aloud. • Decide what the element on the card becomes • Becomes “Positive 2” or “Negative 2” • Say it aloud. Do that action with the person whose name you called at the beginning of the round. • If you are incorrect, you must give everyone in the group the correct amount (even if it is supposed to be steal). • If the other person does the wrong action, he/she has to give everyone in the group the correct amount. • Be the highest chip holder at the end of the class period.

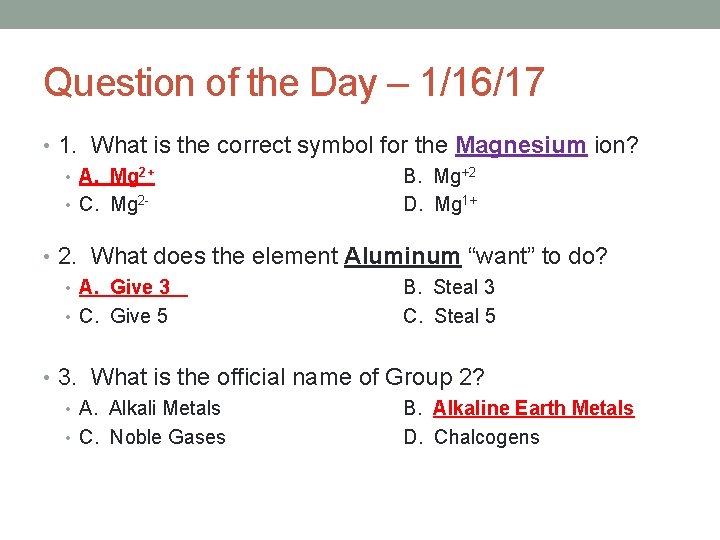
Question of the Day – 1/16/17 • 1. What is the correct symbol for the Magnesium ion? • A. Mg 2+ B. Mg+2 • C. Mg 2 D. Mg 1+ • 2. What does the element Aluminum “want” to do? • A. Give 3 B. Steal 3 • C. Give 5 C. Steal 5 • 3. What is the official name of Group 2? • A. Alkali Metals B. Alkaline Earth Metals • C. Noble Gases D. Chalcogens

Question of the Day – 1/17/14 • 1. What would the symbol for the Bromine ion look like? • A. Br 2+ B. Br 1 • C. Br 1+ D. Br 2 • 2. What is the official name for Group 17? • A. Halogens B. Boron Group • C. Chalcogens D. Pcnitogens

Making Compounds • A compound is a substance made of atoms from 2 or more different elements. • Chemical Bond = the forces that hold elements together in compounds. • Makes sure that the valence shells of elements are filled. • Most compounds are very different from the elements that make them up. • Exp Water and Salt • Occurs through giving and receiving electrons or by sharing electrons

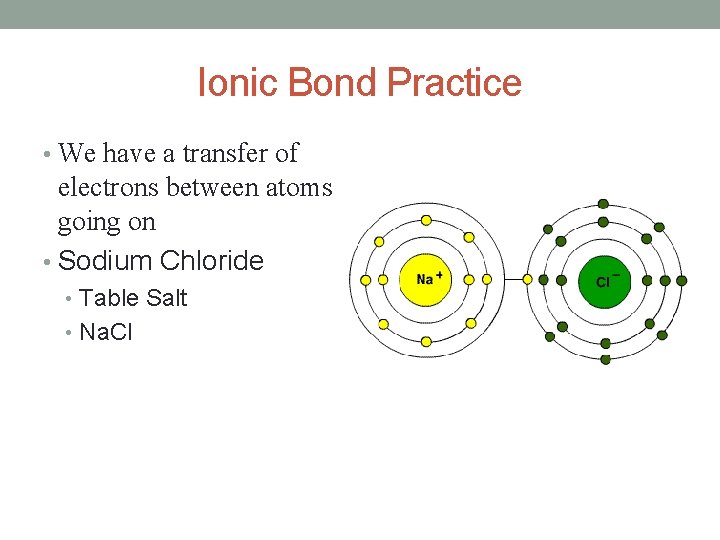
Ionic Bonds • Once atoms become ions positive and negative charges attract. • Each atom is charged; either positive or negative. • But, the compound is • The attraction between two atoms with opposite charges is called an Ionic Bond. • Strongest type of bond. neutral. • Positive and negative charges cancel out. • http: //www. youtube. com/w atch? v=x. Tx_DWbo. EVs

Ionic Bond Practice • We have a transfer of electrons between atoms going on • Sodium Chloride • Table Salt • Na. Cl

More Practice Ionic Bond • Build Li. F • Make Lithium (Li) and Fluorine (F) join in an ionic bond. Ionic Bon • Build Magnesium Chloride • Make Magnesium (Mg) and Chlorine (Cl) join in an ionic bond • Mg 2+ • Cl 1 • Mg. Cl 2

Question of the Day • 1. What is the charge on a Calcium (Ca) ion? • A. 2+ B. 2 • C. 1+ D. 1 • 2. What does Sodium (Na) “want” to do? • A. Steal one B. Give one • C. Steal two C. Give two • 3. How many electron shells (energy levels) does Phosphorous (P) have? • A. 1 • C. 3 B. 2 D. 4

Covalent Bonds • In a covalent bond, atoms fill up their valence shells by sharing electrons. • When two atoms are held together by a shared pair of electrons we call this process a covalent bond. • • Molecule = 2 or more elements held together by a covalent bond.

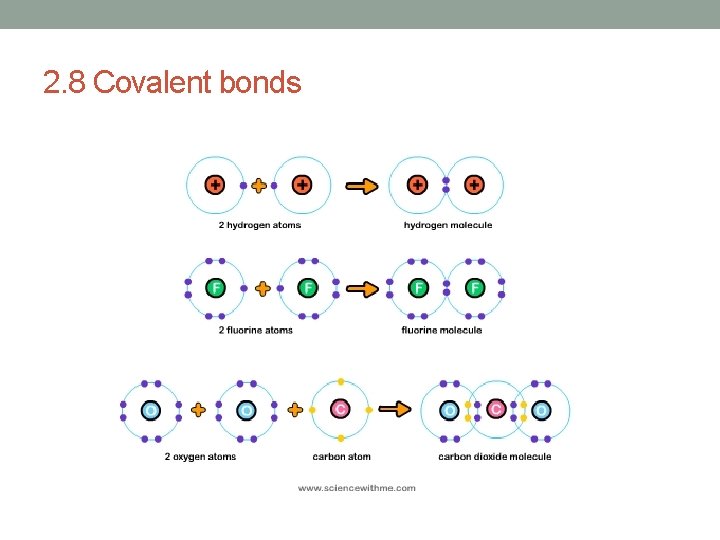
2. 8 Covalent bonds

Bond Strength • Ionic > Covalent!!

Question of the day • 1. What word should you pair with “covalent? ” • A. Sharing B. Stealing • C. Giving D. Penguins • 2. If you were going to bond Calcium (Ca) and Fluorine (F), how many of each would you need? • A. 2 Ca; 1 F • C. 1 Ca; 1 F B. 2 Ca; 2 F D. 1 Ca; 2 F

Different Types of Atoms Isotopes • What is an isotope? • When an element has more or less neutrons then it is supposed to. • Gains or loses neutrons due to high energy particle interactions What Stays the Same? • The number of protons does not change. • The element identity would change. • The number of electrons does not change. • Has the same properties • No charge is present • Atom weighs less or more

Mass Number – individual atom’s mass • This is what you used to find neutron count before • What is an individual atom’s mass number? • Protons Mass + Neutrons Mass + Electrons Mass • But electrons don’t matter! • 1, 000, 000 + 0. 00000001 = 2, 000, 000

Isotopes Mass Number This averaging is why we see decimals on the periodic table for the element’s mass number. • Existence • We know that isotopes exist because we have observed them in nature. • Scientists averaged out all of the mass numbers they discovered of an element and came up with atomic mass. • Atomic Mass = The average mass number for an element. • Abundance matters!!!! • http: //www. youtube. com/w atch? v=Jdtt 3 Lsod. AQ

Isotope Practice • Lithium: Make a normal atom of Lithium (Li)(Lithium-7) • Transform this normal atom into the isotope of Lithium-6 • Fluorine: Make a normal atom of Fluorine ( F) (Fluorine- 19) • Transform this normal atom into Fluorine-17 • Phosphorus: Make a normal atom of Phosphorus (P) (Phosphorus-31) • Transform this normal atom into Phosphorus-27 Protons =M side up Neutrons = Blank side Electrons = Pennies

Isotopes Existence • How do isotopes form? • Some Isotopes already exist • Others are formed by collision of particles. • In upper layers of the atmosphere. • Matter is exposed to intense light; UV rays, gamma rays, and x-rays. • These intense rays can break matter up and force particles in or knock particles out. • General Rule: The larger the element’s nucleus the harder it is for it to hold on to an extra neutron.

Radioactivity • All isotopes eventual want to return to their “normal state” • Some Isotopes take a very long time to get back to their normal number of neutrons. • We call these isotopes stable. • They can stay as isotopes for a long time • Exp: Carbon Isotopes 13, Oxygen 17 • Other isotopes return to their normal number of neutrons very quickly • These Isotopes are unstable • These want to get back to their normal amount of neutrons badly. • Exp: Radium, Palonium • Begin to throw away particles • This can release lots of energy. Unstable Isotopes • If Isotopes get rid of their neutrons slowly, then the emit energy (radiation) slowly over time. • If Isotopes throw away their neutrons quickly, then they emit huge amounts of energy quickly. • We call this radioactivity

Nuclear Bombs • Uranium 235 + 1 neutron => Uranium 236 • Very unstable • Decays really quickly but goes overboard (gives off too many neutrons) releasing a ton of energy in the process • Creates a chain reaction of neutrons hitting other uranium 235 atoms which creates more neutrons which hit other uranium 235 atoms, etc.


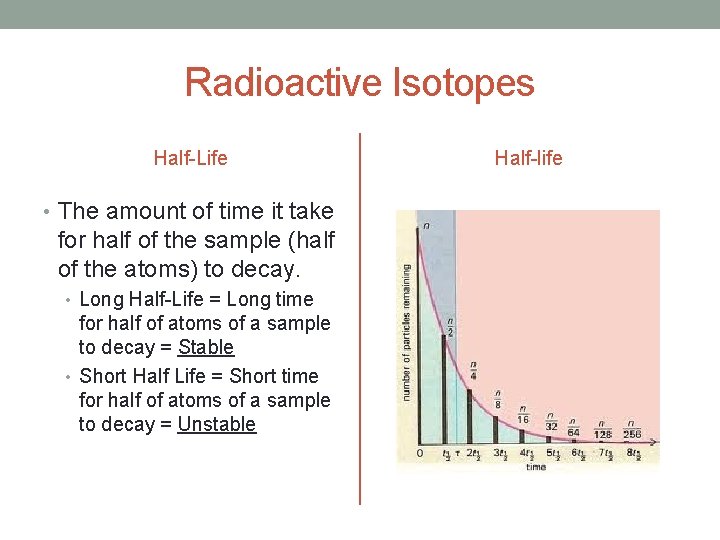
Radioactive Isotopes Half-Life • The amount of time it take for half of the sample (half of the atoms) to decay. • Long Half-Life = Long time for half of atoms of a sample to decay = Stable • Short Half Life = Short time for half of atoms of a sample to decay = Unstable Half-life