Chapter 3 Fatty Acids and Prostaglandins Biosynthesis of
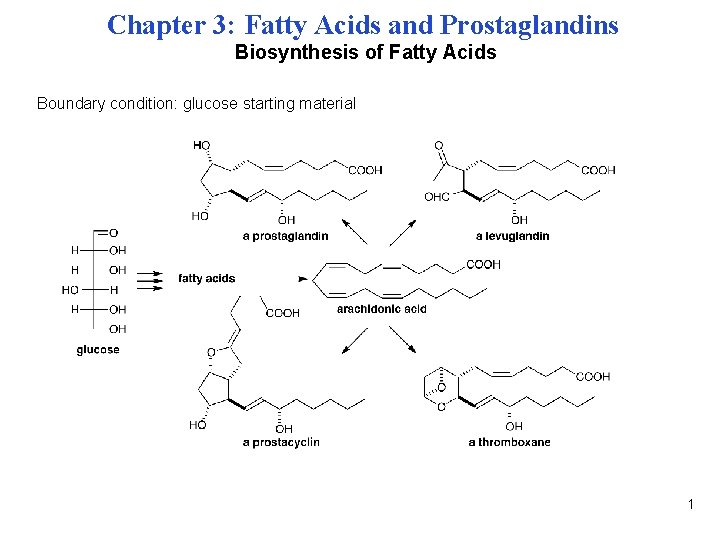
Chapter 3: Fatty Acids and Prostaglandins Biosynthesis of Fatty Acids Boundary condition: glucose starting material 1

Chapter 3: Fatty Acids and Prostaglandins Biosynthesis of Fatty Acids Unimplimented strategy for fatty acid synthesis from glucose 2
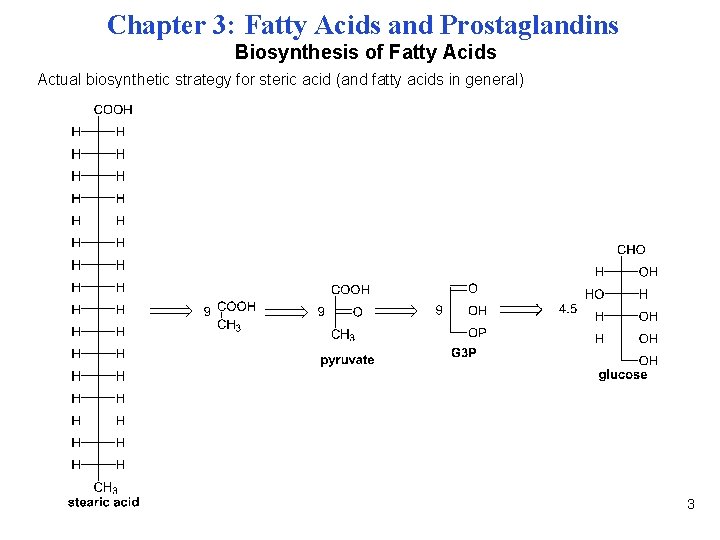
Chapter 3: Fatty Acids and Prostaglandins Biosynthesis of Fatty Acids Actual biosynthetic strategy for steric acid (and fatty acids in general) 3
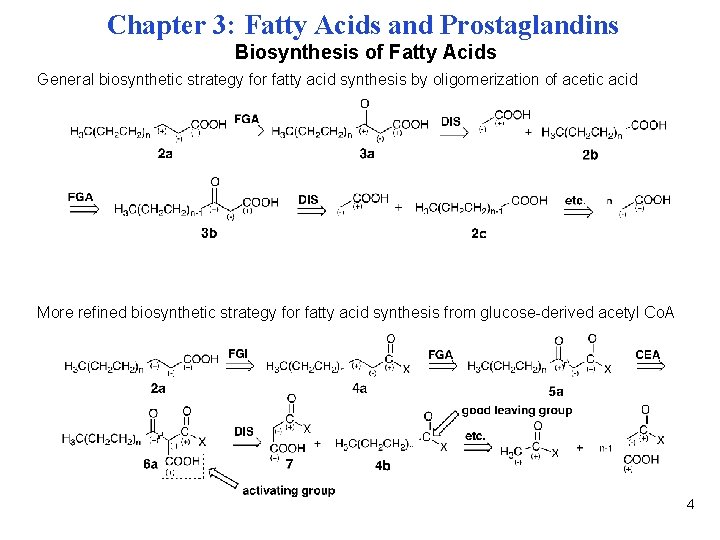
Chapter 3: Fatty Acids and Prostaglandins Biosynthesis of Fatty Acids General biosynthetic strategy for fatty acid synthesis by oligomerization of acetic acid More refined biosynthetic strategy for fatty acid synthesis from glucose-derived acetyl Co. A 4
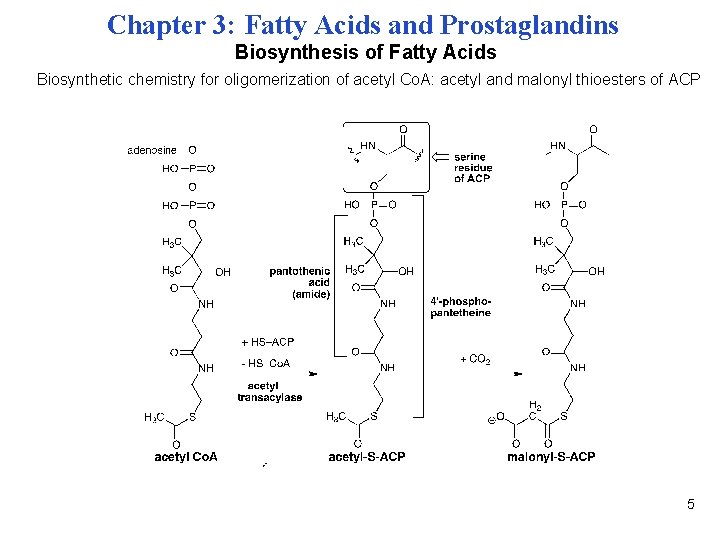
Chapter 3: Fatty Acids and Prostaglandins Biosynthesis of Fatty Acids Biosynthetic chemistry for oligomerization of acetyl Co. A: acetyl and malonyl thioesters of ACP 5
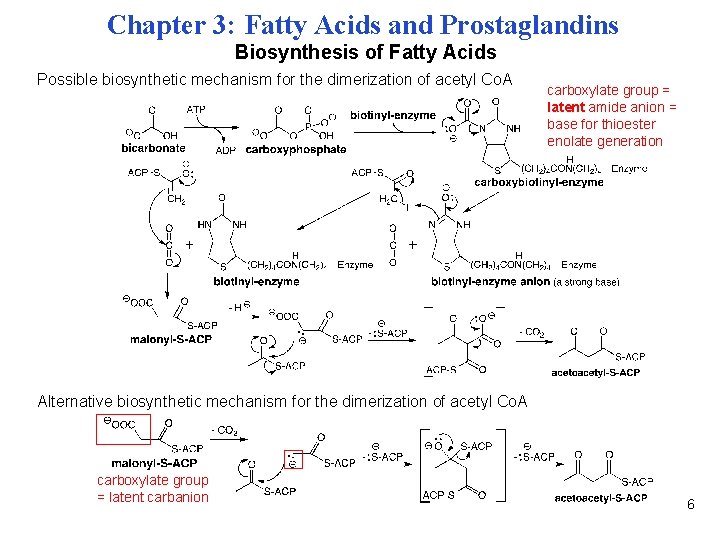
Chapter 3: Fatty Acids and Prostaglandins Biosynthesis of Fatty Acids Possible biosynthetic mechanism for the dimerization of acetyl Co. A carboxylate group = latent amide anion = base for thioester enolate generation Alternative biosynthetic mechanism for the dimerization of acetyl Co. A carboxylate group = latent carbanion 6
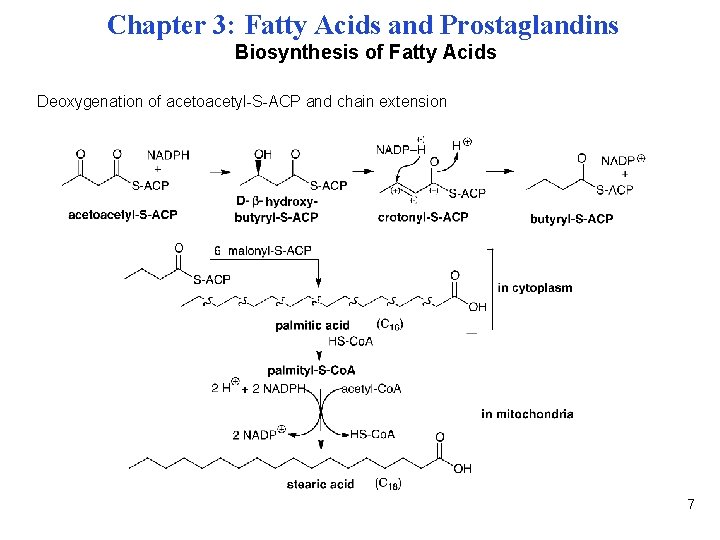
Chapter 3: Fatty Acids and Prostaglandins Biosynthesis of Fatty Acids Deoxygenation of acetoacetyl-S-ACP and chain extension 7
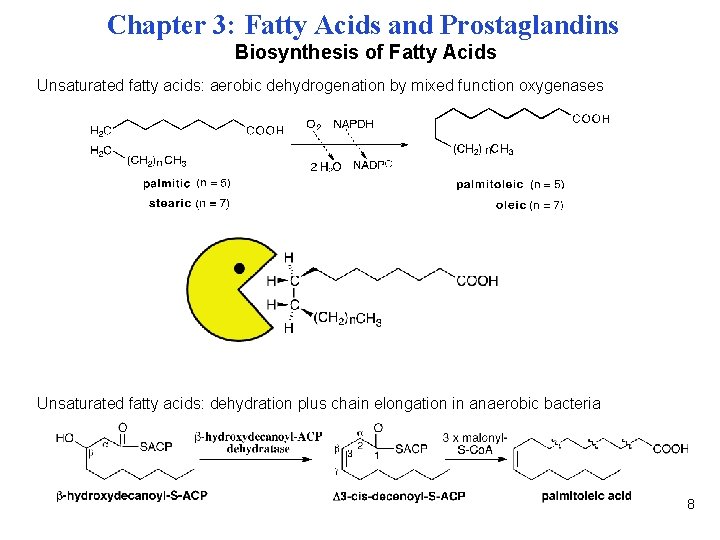
Chapter 3: Fatty Acids and Prostaglandins Biosynthesis of Fatty Acids Unsaturated fatty acids: aerobic dehydrogenation by mixed function oxygenases Unsaturated fatty acids: dehydration plus chain elongation in anaerobic bacteria 8

Chapter 3: Fatty Acids and Prostaglandins Biosynthesis of Fatty Acids Essential fatty acids: humans do not have some dehydrogenases 9
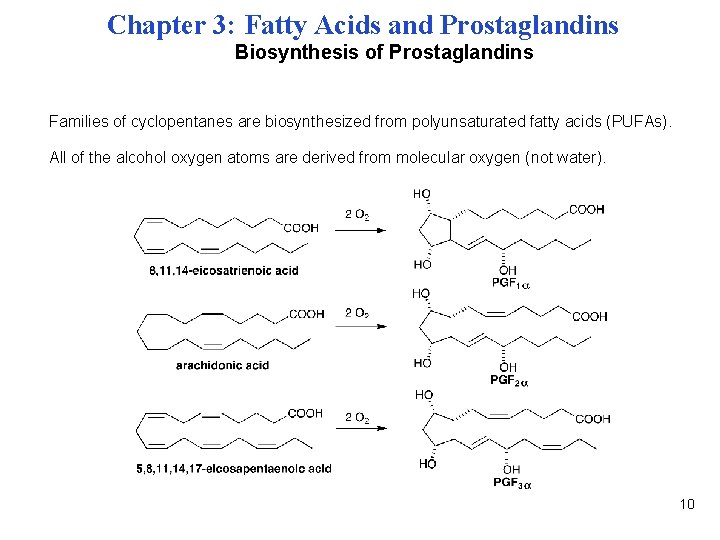
Chapter 3: Fatty Acids and Prostaglandins Biosynthesis of Prostaglandins Families of cyclopentanes are biosynthesized from polyunsaturated fatty acids (PUFAs). All of the alcohol oxygen atoms are derived from molecular oxygen (not water). 10
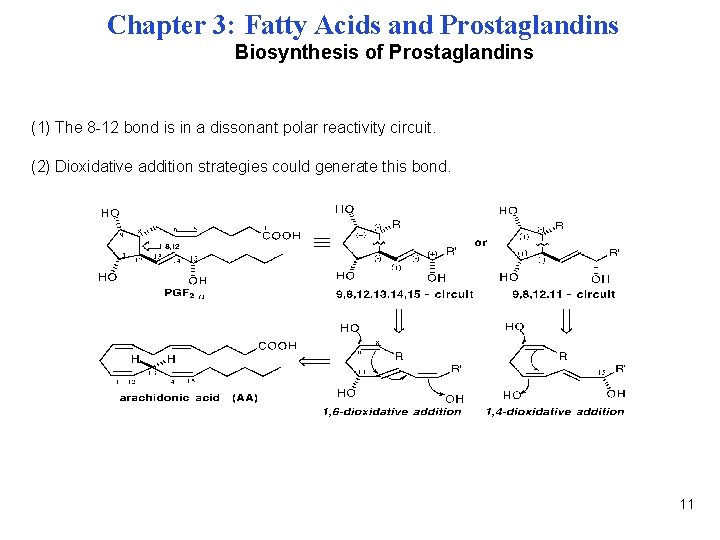
Chapter 3: Fatty Acids and Prostaglandins Biosynthesis of Prostaglandins (1) The 8 -12 bond is in a dissonant polar reactivity circuit. (2) Dioxidative addition strategies could generate this bond. 11
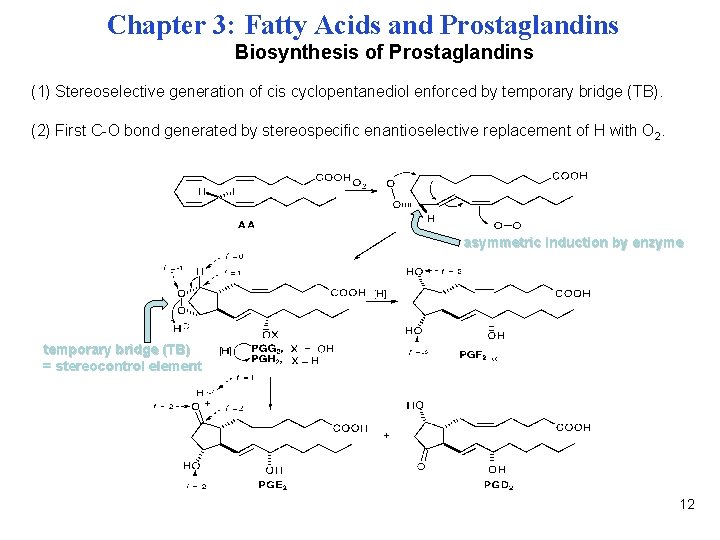
Chapter 3: Fatty Acids and Prostaglandins Biosynthesis of Prostaglandins (1) Stereoselective generation of cis cyclopentanediol enforced by temporary bridge (TB). (2) First C-O bond generated by stereospecific enantioselective replacement of H with O 2. asymmetric induction by enzyme temporary bridge (TB) = stereocontrol element 12
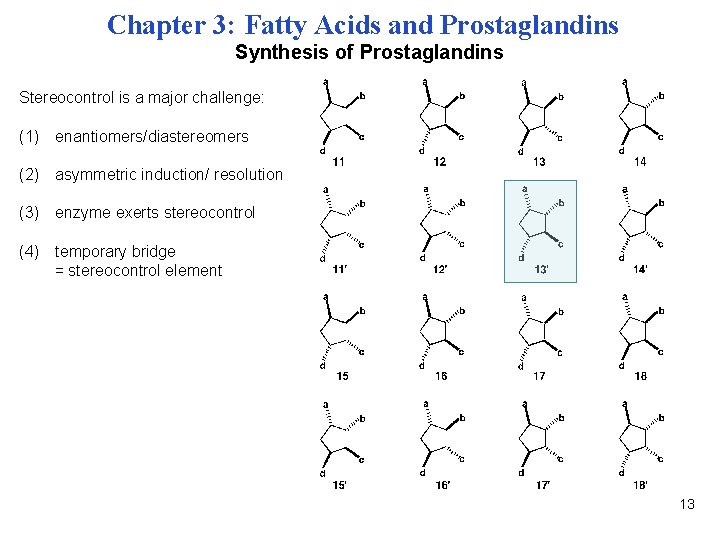
Chapter 3: Fatty Acids and Prostaglandins Synthesis of Prostaglandins Stereocontrol is a major challenge: (1) enantiomers/diastereomers (2) asymmetric induction/ resolution (3) enzyme exerts stereocontrol (4) temporary bridge = stereocontrol element 13
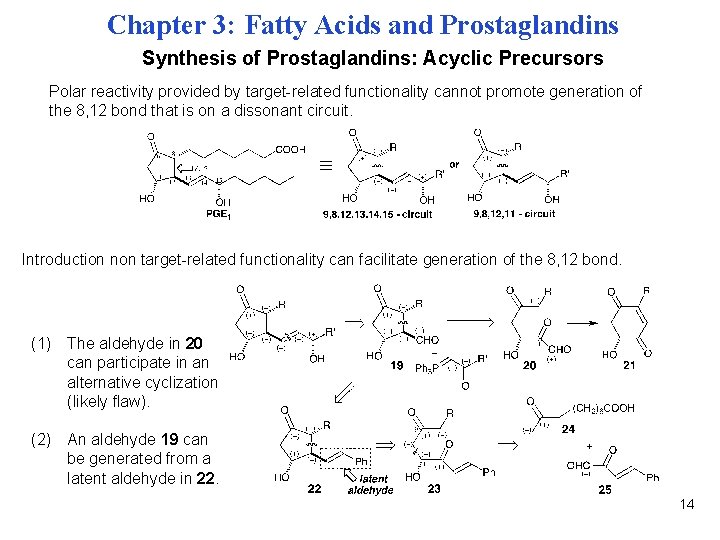
Chapter 3: Fatty Acids and Prostaglandins Synthesis of Prostaglandins: Acyclic Precursors Polar reactivity provided by target-related functionality cannot promote generation of the 8, 12 bond that is on a dissonant circuit. Introduction non target-related functionality can facilitate generation of the 8, 12 bond. (1) The aldehyde in 20 can participate in an alternative cyclization (likely flaw). (2) An aldehyde 19 can be generated from a latent aldehyde in 22. 14
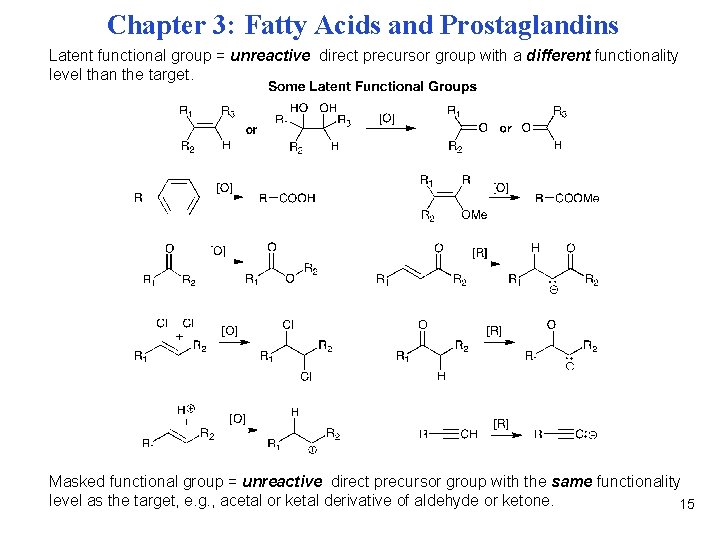
Chapter 3: Fatty Acids and Prostaglandins Latent functional group = unreactive direct precursor group with a different functionality level than the target. Masked functional group = unreactive direct precursor group with the same functionality level as the target, e. g. , acetal or ketal derivative of aldehyde or ketone. 15
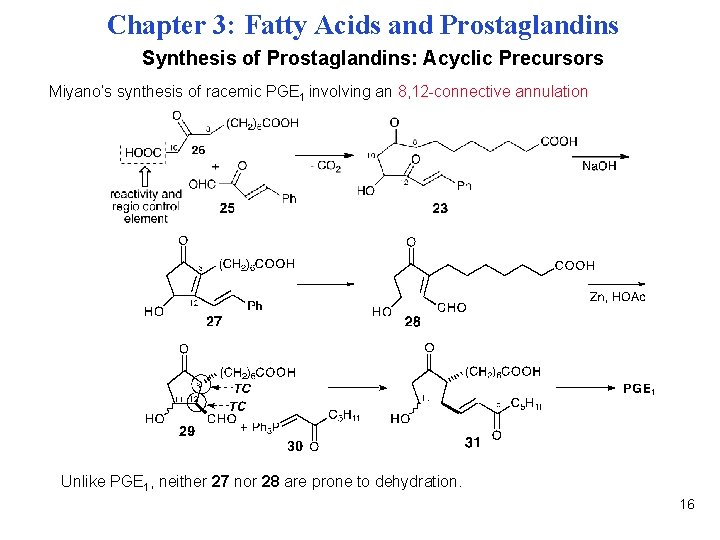
Chapter 3: Fatty Acids and Prostaglandins Synthesis of Prostaglandins: Acyclic Precursors Miyano’s synthesis of racemic PGE 1 involving an 8, 12 -connective annulation Unlike PGE 1, neither 27 nor 28 are prone to dehydration. 16
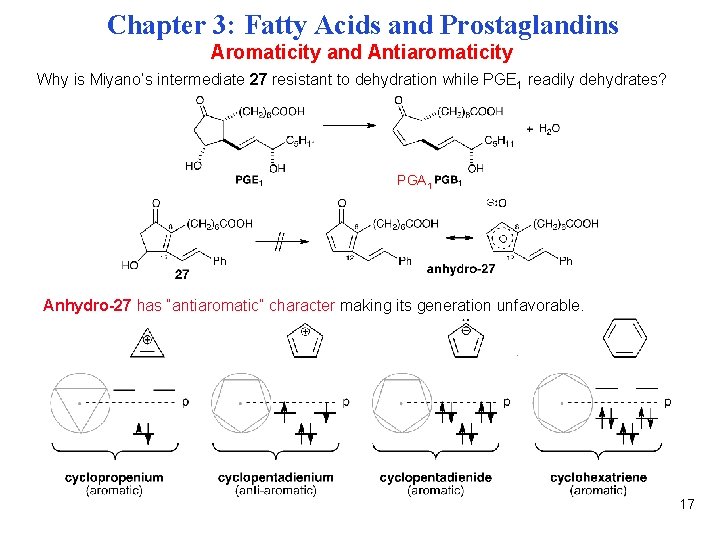
Chapter 3: Fatty Acids and Prostaglandins Aromaticity and Antiaromaticity Why is Miyano’s intermediate 27 resistant to dehydration while PGE 1 readily dehydrates? PGA 1 Anhydro-27 has “antiaromatic” character making its generation unfavorable. 17
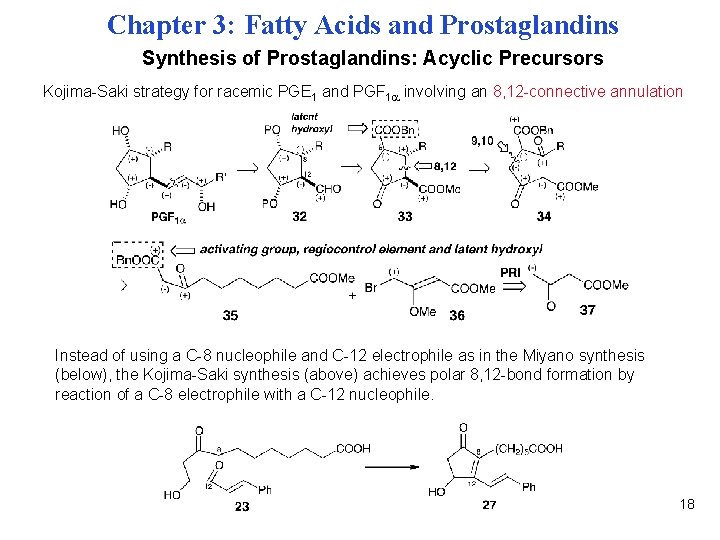
Chapter 3: Fatty Acids and Prostaglandins Synthesis of Prostaglandins: Acyclic Precursors Kojima-Saki strategy for racemic PGE 1 and PGF 1 involving an 8, 12 -connective annulation Instead of using a C-8 nucleophile and C-12 electrophile as in the Miyano synthesis (below), the Kojima-Saki synthesis (above) achieves polar 8, 12 -bond formation by reaction of a C-8 electrophile with a C-12 nucleophile. 18
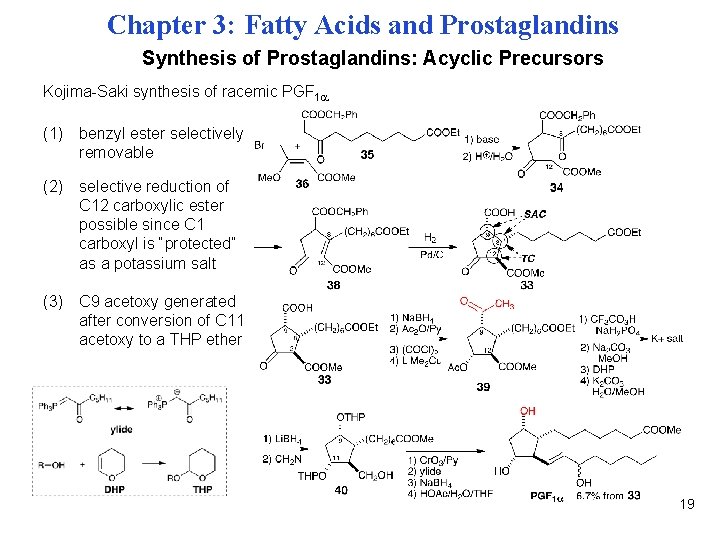
Chapter 3: Fatty Acids and Prostaglandins Synthesis of Prostaglandins: Acyclic Precursors Kojima-Saki synthesis of racemic PGF 1 (1) benzyl ester selectively removable (2) selective reduction of C 12 carboxylic ester possible since C 1 carboxyl is “protected” as a potassium salt (3) C 9 acetoxy generated after conversion of C 11 acetoxy to a THP ether 19
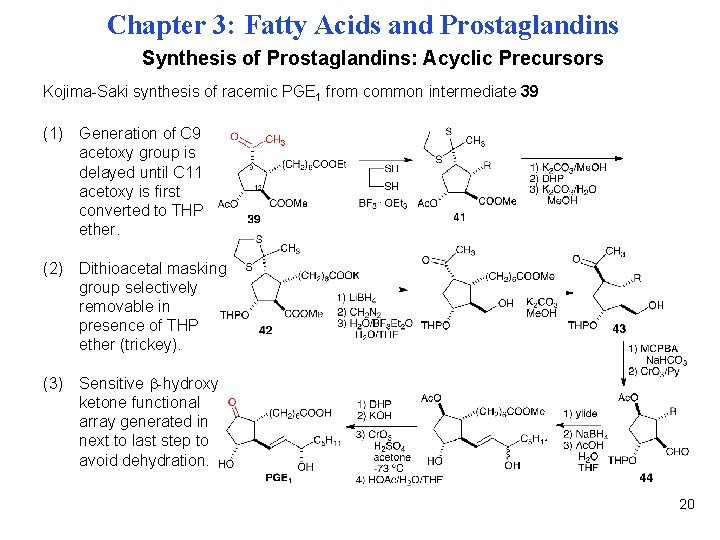
Chapter 3: Fatty Acids and Prostaglandins Synthesis of Prostaglandins: Acyclic Precursors Kojima-Saki synthesis of racemic PGE 1 from common intermediate 39 (1) Generation of C 9 acetoxy group is delayed until C 11 acetoxy is first converted to THP ether. (2) Dithioacetal masking group selectively removable in presence of THP ether (trickey). (3) Sensitive -hydroxy ketone functional array generated in next to last step to avoid dehydration. 20
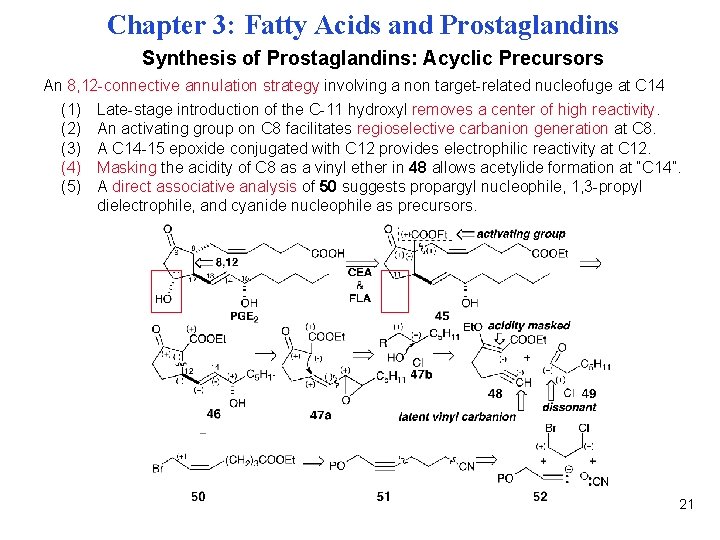
Chapter 3: Fatty Acids and Prostaglandins Synthesis of Prostaglandins: Acyclic Precursors An 8, 12 -connective annulation strategy involving a non target-related nucleofuge at C 14 (1) Late-stage introduction of the C-11 hydroxyl removes a center of high reactivity. (2) An activating group on C 8 facilitates regioselective carbanion generation at C 8. (3) A C 14 -15 epoxide conjugated with C 12 provides electrophilic reactivity at C 12. (4) Masking the acidity of C 8 as a vinyl ether in 48 allows acetylide formation at “C 14”. (5) A direct associative analysis of 50 suggests propargyl nucleophile, 1, 3 -propyl dielectrophile, and cyanide nucleophile as precursors. 21
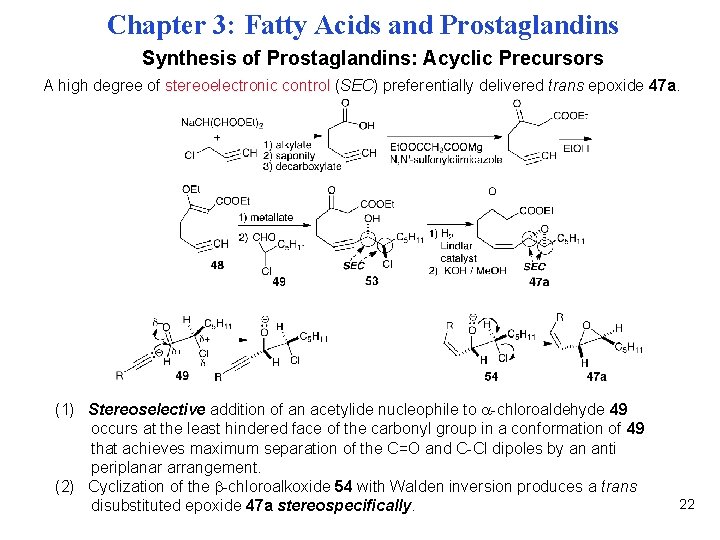
Chapter 3: Fatty Acids and Prostaglandins Synthesis of Prostaglandins: Acyclic Precursors A high degree of stereoelectronic control (SEC) preferentially delivered trans epoxide 47 a. (1) Stereoselective addition of an acetylide nucleophile to -chloroaldehyde 49 occurs at the least hindered face of the carbonyl group in a conformation of 49 that achieves maximum separation of the C=O and C-Cl dipoles by an anti periplanar arrangement. (2) Cyclization of the -chloroalkoxide 54 with Walden inversion produces a trans disubstituted epoxide 47 a stereospecifically. 22
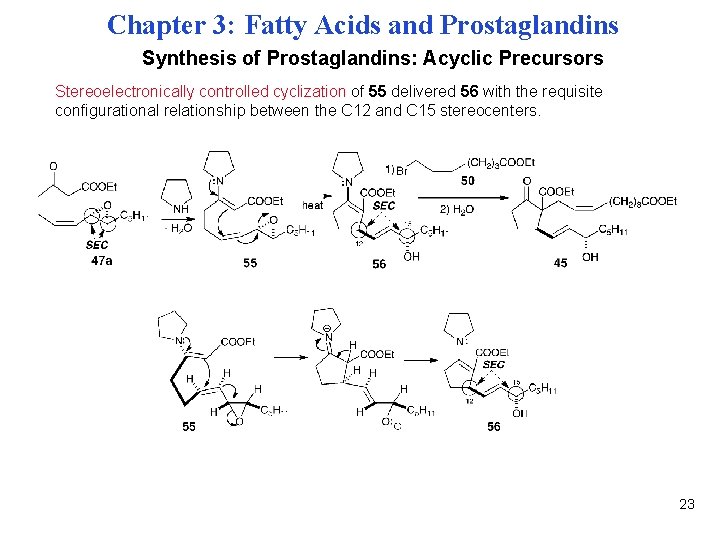
Chapter 3: Fatty Acids and Prostaglandins Synthesis of Prostaglandins: Acyclic Precursors Stereoelectronically controlled cyclization of 55 delivered 56 with the requisite configurational relationship between the C 12 and C 15 stereocenters. 23
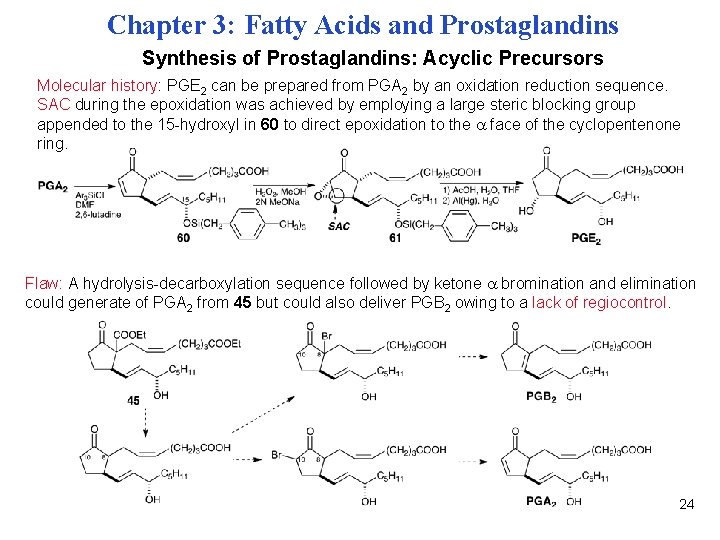
Chapter 3: Fatty Acids and Prostaglandins Synthesis of Prostaglandins: Acyclic Precursors Molecular history: PGE 2 can be prepared from PGA 2 by an oxidation reduction sequence. SAC during the epoxidation was achieved by employing a large steric blocking group appended to the 15 -hydroxyl in 60 to direct epoxidation to the face of the cyclopentenone ring. Flaw: A hydrolysis-decarboxylation sequence followed by ketone bromination and elimination could generate of PGA 2 from 45 but could also deliver PGB 2 owing to a lack of regiocontrol. 24
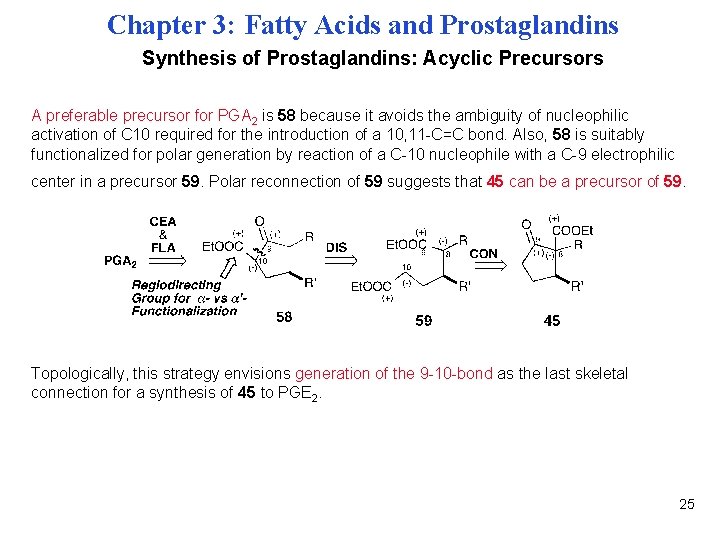
Chapter 3: Fatty Acids and Prostaglandins Synthesis of Prostaglandins: Acyclic Precursors A preferable precursor for PGA 2 is 58 because it avoids the ambiguity of nucleophilic activation of C 10 required for the introduction of a 10, 11 -C=C bond. Also, 58 is suitably functionalized for polar generation by reaction of a C-10 nucleophile with a C-9 electrophilic center in a precursor 59. Polar reconnection of 59 suggests that 45 can be a precursor of 59. Topologically, this strategy envisions generation of the 9 -10 -bond as the last skeletal connection for a synthesis of 45 to PGE 2. 25
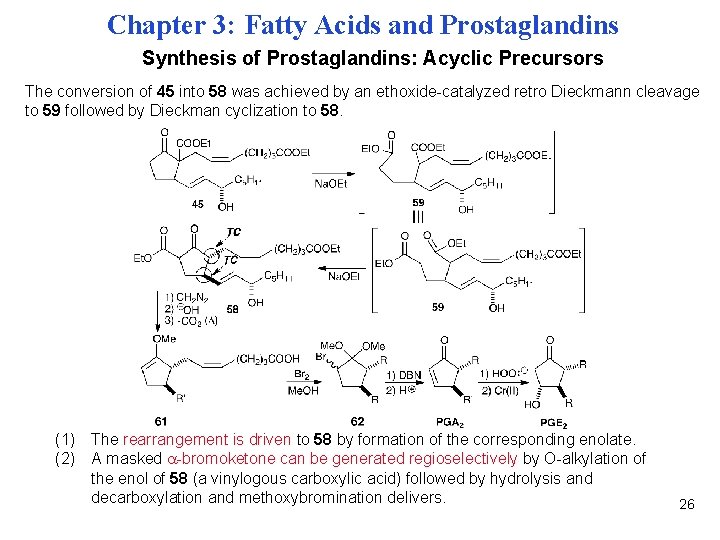
Chapter 3: Fatty Acids and Prostaglandins Synthesis of Prostaglandins: Acyclic Precursors The conversion of 45 into 58 was achieved by an ethoxide-catalyzed retro Dieckmann cleavage to 59 followed by Dieckman cyclization to 58. (1) The rearrangement is driven to 58 by formation of the corresponding enolate. (2) A masked -bromoketone can be generated regioselectively by O-alkylation of the enol of 58 (a vinylogous carboxylic acid) followed by hydrolysis and decarboxylation and methoxybromination delivers. 26
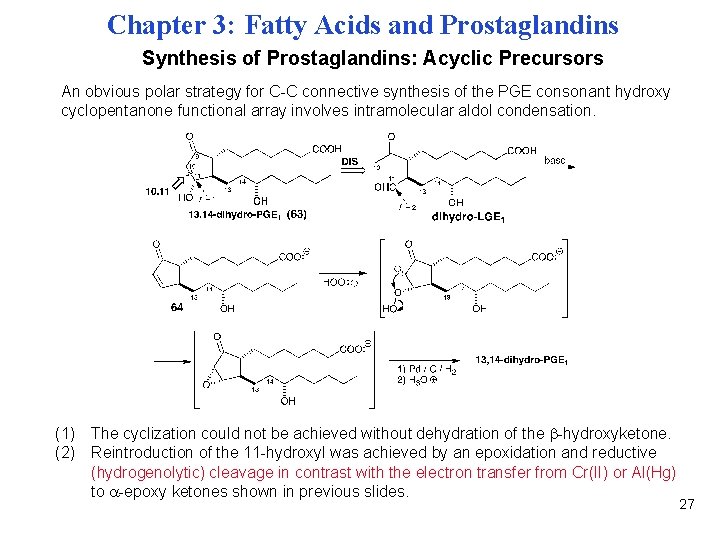
Chapter 3: Fatty Acids and Prostaglandins Synthesis of Prostaglandins: Acyclic Precursors An obvious polar strategy for C-C connective synthesis of the PGE consonant hydroxy cyclopentanone functional array involves intramolecular aldol condensation. (1) The cyclization could not be achieved without dehydration of the -hydroxyketone. (2) Reintroduction of the 11 -hydroxyl was achieved by an epoxidation and reductive (hydrogenolytic) cleavage in contrast with the electron transfer from Cr(II) or Al(Hg) to -epoxy ketones shown in previous slides. 27
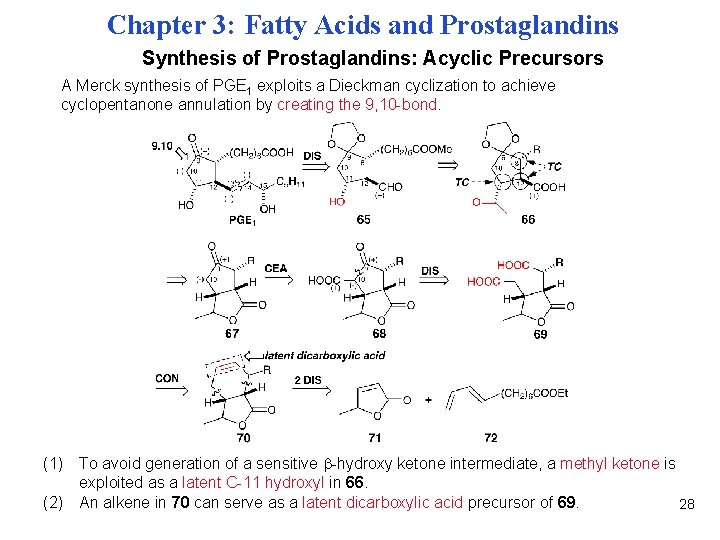
Chapter 3: Fatty Acids and Prostaglandins Synthesis of Prostaglandins: Acyclic Precursors A Merck synthesis of PGE 1 exploits a Dieckman cyclization to achieve cyclopentanone annulation by creating the 9, 10 -bond. (1) To avoid generation of a sensitive -hydroxy ketone intermediate, a methyl ketone is exploited as a latent C-11 hydroxyl in 66. (2) An alkene in 70 can serve as a latent dicarboxylic acid precursor of 69. 28
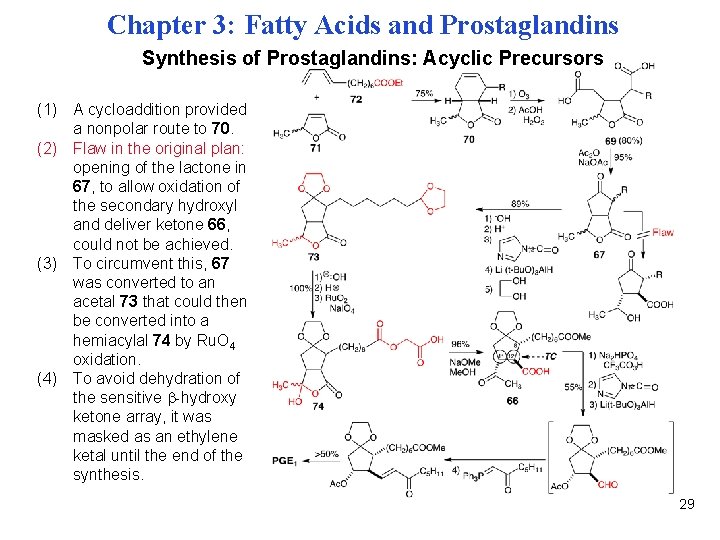
Chapter 3: Fatty Acids and Prostaglandins Synthesis of Prostaglandins: Acyclic Precursors (1) A cycloaddition provided a nonpolar route to 70. (2) Flaw in the original plan: opening of the lactone in 67, to allow oxidation of the secondary hydroxyl and deliver ketone 66, could not be achieved. (3) To circumvent this, 67 was converted to an acetal 73 that could then be converted into a hemiacylal 74 by Ru. O 4 oxidation. (4) To avoid dehydration of the sensitive -hydroxy ketone array, it was masked as an ethylene ketal until the end of the synthesis. 29
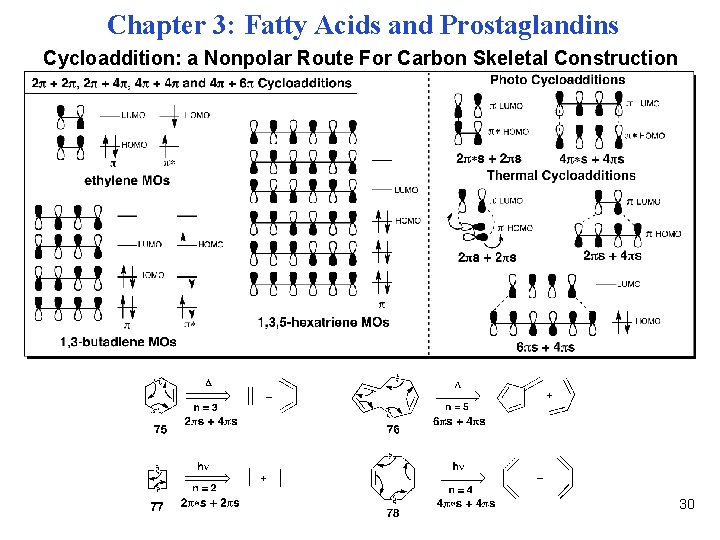
Chapter 3: Fatty Acids and Prostaglandins Cycloaddition: a Nonpolar Route For Carbon Skeletal Construction 30
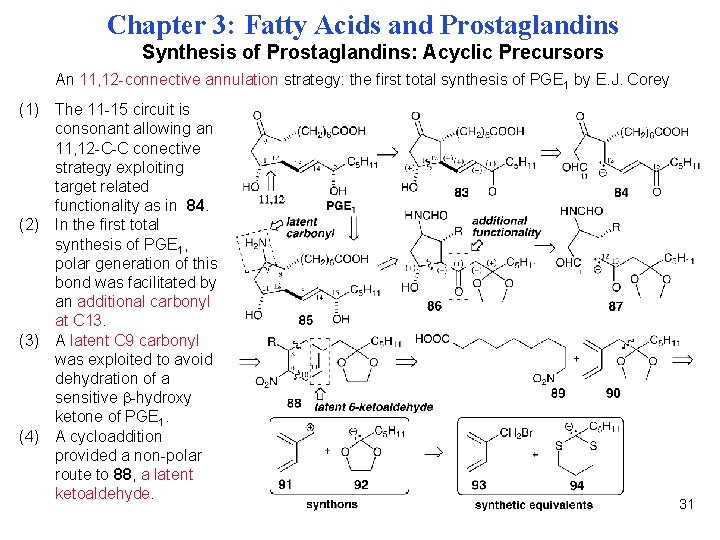
Chapter 3: Fatty Acids and Prostaglandins Synthesis of Prostaglandins: Acyclic Precursors An 11, 12 -connective annulation strategy: the first total synthesis of PGE 1 by E. J. Corey (1) The 11 -15 circuit is consonant allowing an 11, 12 -C-C conective strategy exploiting target related functionality as in 84. (2) In the first total synthesis of PGE 1, polar generation of this bond was facilitated by an additional carbonyl at C 13. (3) A latent C 9 carbonyl was exploited to avoid dehydration of a sensitive -hydroxy ketone of PGE 1. (4) A cycloaddition provided a non-polar route to 88, a latent ketoaldehyde. 31
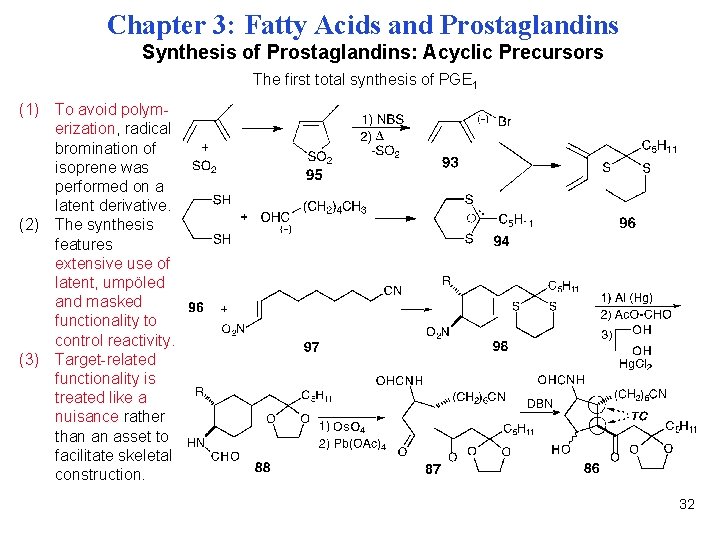
Chapter 3: Fatty Acids and Prostaglandins Synthesis of Prostaglandins: Acyclic Precursors The first total synthesis of PGE 1 (1) To avoid polymerization, radical bromination of isoprene was performed on a latent derivative. (2) The synthesis features extensive use of latent, umpöled and masked functionality to control reactivity. (3) Target-related functionality is treated like a nuisance rather than an asset to facilitate skeletal construction. 32
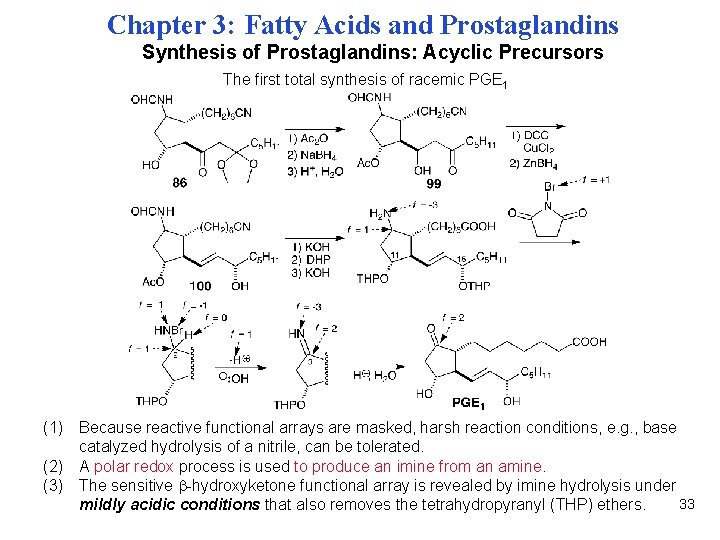
Chapter 3: Fatty Acids and Prostaglandins Synthesis of Prostaglandins: Acyclic Precursors The first total synthesis of racemic PGE 1 (1) Because reactive functional arrays are masked, harsh reaction conditions, e. g. , base catalyzed hydrolysis of a nitrile, can be tolerated. (2) A polar redox process is used to produce an imine from an amine. (3) The sensitive -hydroxyketone functional array is revealed by imine hydrolysis under 33 mildly acidic conditions that also removes the tetrahydropyranyl (THP) ethers.
- Slides: 33