Chapter 3 Crystal Binding Bondingand Elastic Constants Overview

Chapter 3: Crystal Binding (Bonding)and Elastic Constants Overview & Survey of Bonding Types What kinds of forces hold the atoms together in a solid? That is, What is their physical origin? ?

Types of Bonding Mechanisms 1. Van der Waals (“Secondary”) 2. Ionic 3. Covalent 4. Metallic 5. Hydrogen (Special case of Van der Waals) Of course, as we’ve said, All bonding is a consequence of electrostatic interaction between nuclei & electrons.

Van der Waals Bonding Chemists often call it London Bonding Sometimes, it is called Van der Waals-London Bonding Johannes D. van der Waals (1837 -1923) Studied intermolecular forces in in liquids & gases Nobel Prize: 1910 in Physics Fritz London (1900 -1954) Studied intermolecular induced-dipole interactions.

Van der Waals Bondıng Very weak! Typical strength ~ 0. 2 e. V/atom. Most important in interactions between neutral atoms or molecules. • The cause of these weak attractive forces is the fact that the lattice vibrations result in slight charge separations between the electrons & the nuclei. Thus, (oscillating) Electric Dipoles are induced on each atom. These dipoles attract each other. • The “larger” an atom is, the easier it is to polarize (to form a dipole). So, Van der Waal's Forces are stronger between “large” atoms than between “small” atoms.

• Van der Waals (London) Bonding is very weak in comparison to all other bonding types. This effect is present in most bonds, but other effects usually are much, much larger than it. Exceptions are bonds between Noble or Rare Gas atoms (with completely filled valence electron shells). This type of bonding is important between any neutral molecules. Dipole-dipole interactions occur between the induced dipoles.
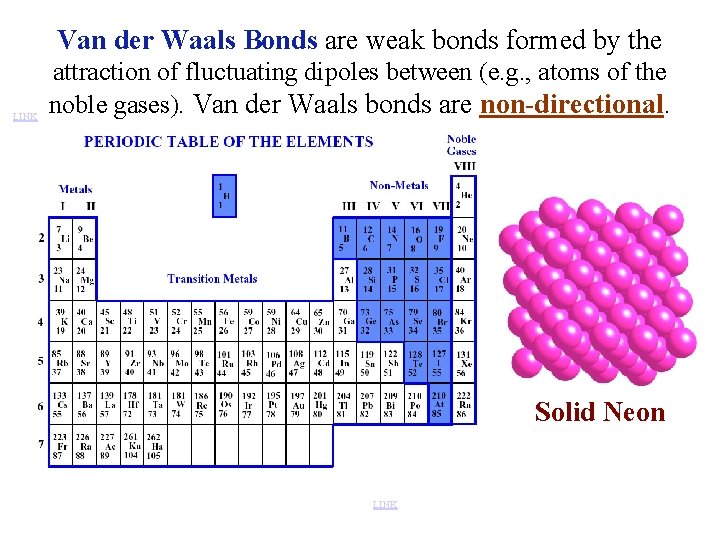
Van der Waals Bonds are weak bonds formed by the LINK attraction of fluctuating dipoles between (e. g. , atoms of the noble gases). Van der Waals bonds are non-directional. Solid Neon LINK
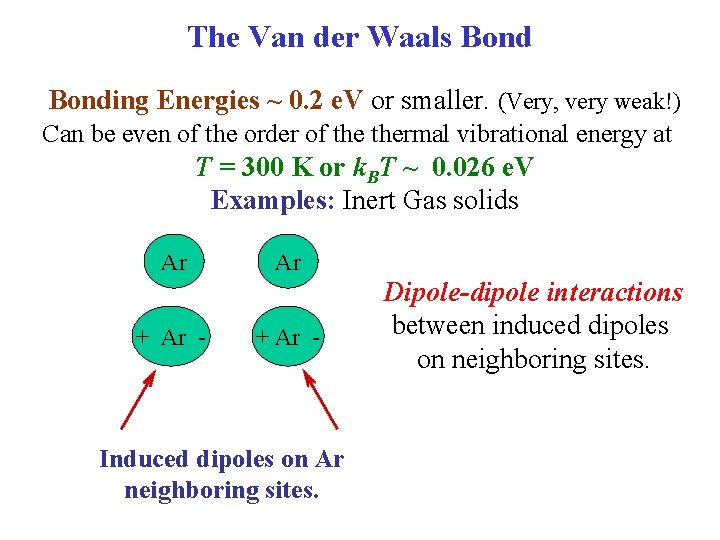
The Van der Waals Bonding Energies ~ 0. 2 e. V or smaller. (Very, very weak!) Can be even of the order of thermal vibrational energy at T = 300 K or k. BT ~ 0. 026 e. V Examples: Inert Gas solids Ar + Ar - Induced dipoles on Ar neighboring sites. Dipole-dipole interactions between induced dipoles on neighboring sites.

Van der Waals Forces (or London “Dispersion” Forces) • Electrons on one atom are attracted to the nucleus on a neighboring atom, creating an “instantaneous” dipole on the 1 st atom. That dipole induces an instantaneous dipole on the 2 nd atom. The 2 induced dipoles then attract each other. • These forces are proportional to the polarizability of the atom. This property is a measure of the ease with which the electron “cloud can be deformed. The polarizability is approximately proportional to the number of electrons in the molecule.
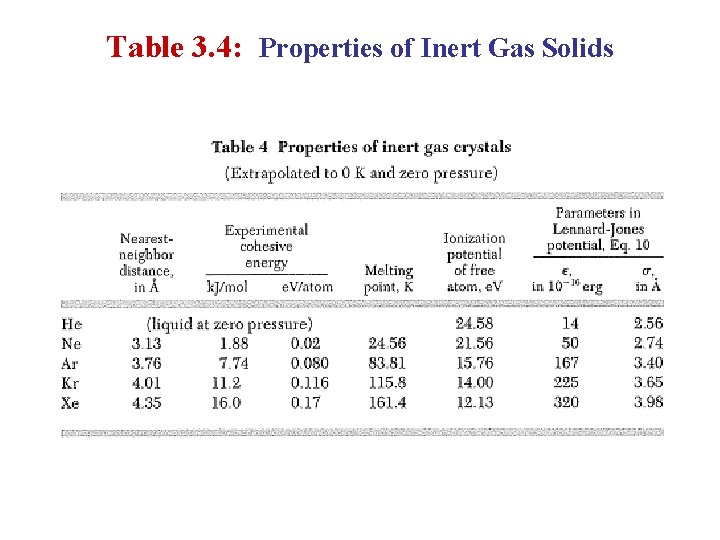
Table 3. 4: Properties of Inert Gas Solids

Ionıc Bondıng Ionic Bonding is caused by the Electrostatic Attraction between positively & negatively charged ions (usually non-metal atoms & metal atoms). • The ions are produced by a transfer of electrons between two atoms with a large difference in electro-negativities. • All ionic compounds are crystalline solids at room temperature. • Na. Cl & Cs. Cl are typical examples of ionically bonded solids.
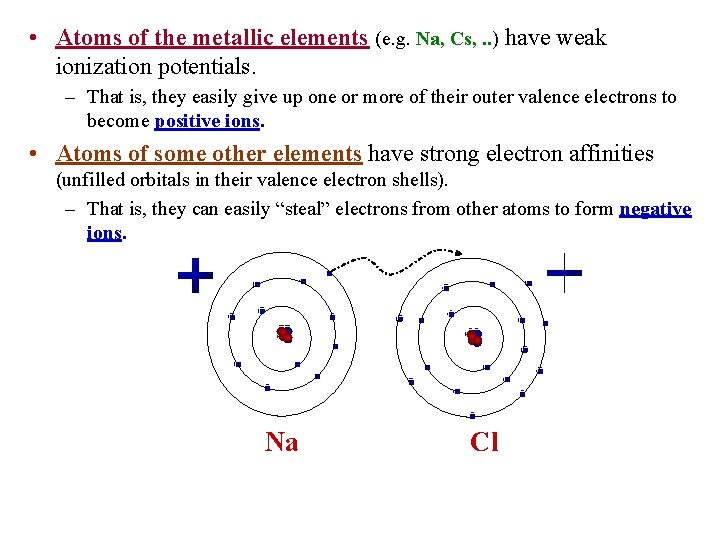
• Atoms of the metallic elements (e. g. Na, Cs, . . ) have weak ionization potentials. – That is, they easily give up one or more of their outer valence electrons to become positive ions. • Atoms of some other elements have strong electron affinities (unfilled orbitals in their valence electron shells). – That is, they can easily “steal” electrons from other atoms to form negative ions. Na Cl

Example: Na. Cl • Notice that when a sodium (Na) atom loses its one valence electron it gets “smaller” in size, while chlorine grows larger when it gains an additional valence electron. After the reaction takes place, the charged Na+ and Cl- ions are held together by electrostatic forces, thus forming an ionic bond.

• Of course, there also repulsive forces between ions. • When the Na+ & Cl- ions approach each other close enough that the electron orbitals in the ions begin to overlap, the electrons begin to repel each other because of the repulsive electrostatic force. Of course, the closer together the ions are, the grater the repulsive force. • The Pauli Exclusion Principle also plays an important role in the repulsive force. To prevent a violation of this principle, the potential energy of the system increases very rapidly.
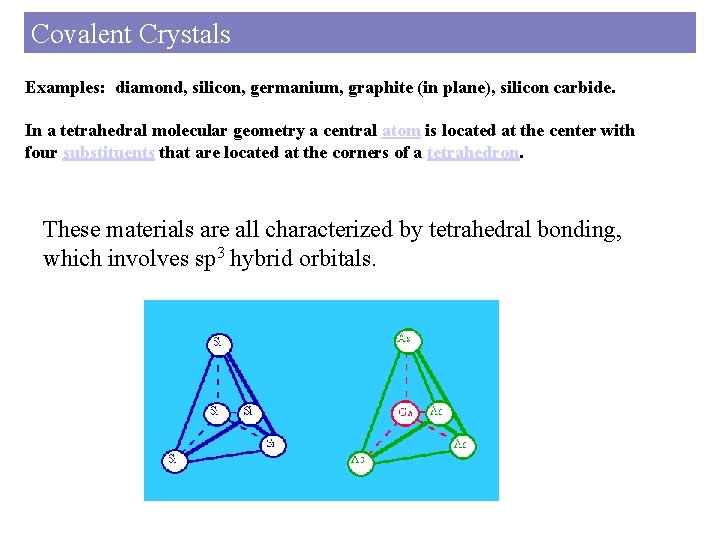
Covalent Crystals Examples: diamond, silicon, germanium, graphite (in plane), silicon carbide. In a tetrahedral molecular geometry a central atom is located at the center with four substituents that are located at the corners of a tetrahedron. These materials are all characterized by tetrahedral bonding, which involves sp 3 hybrid orbitals.

3 sp • hybridization The mixing of atomic orbitals to form special orbitals for bonding. • The atoms are responding as needed to give the minimum energy for the molecule.
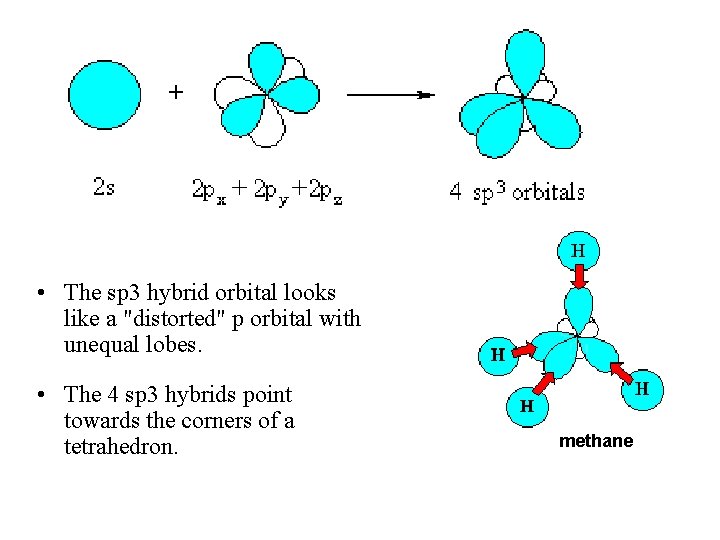
• The sp 3 hybrid orbital looks like a "distorted" p orbital with unequal lobes. • The 4 sp 3 hybrids point towards the corners of a tetrahedron. methane
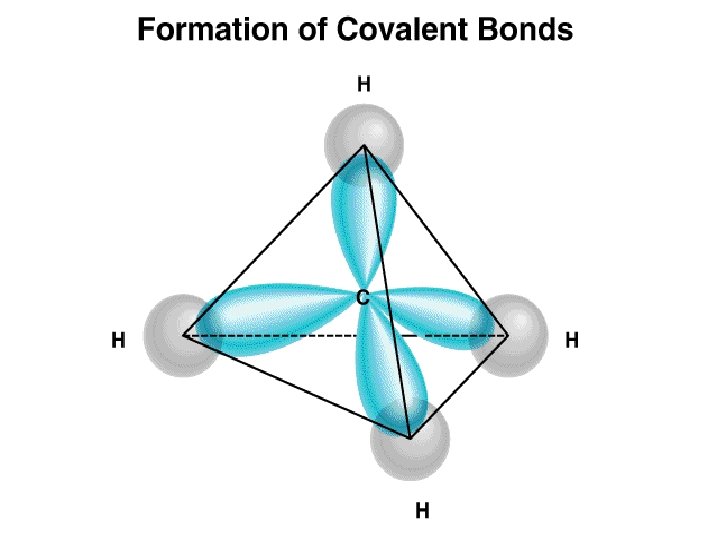
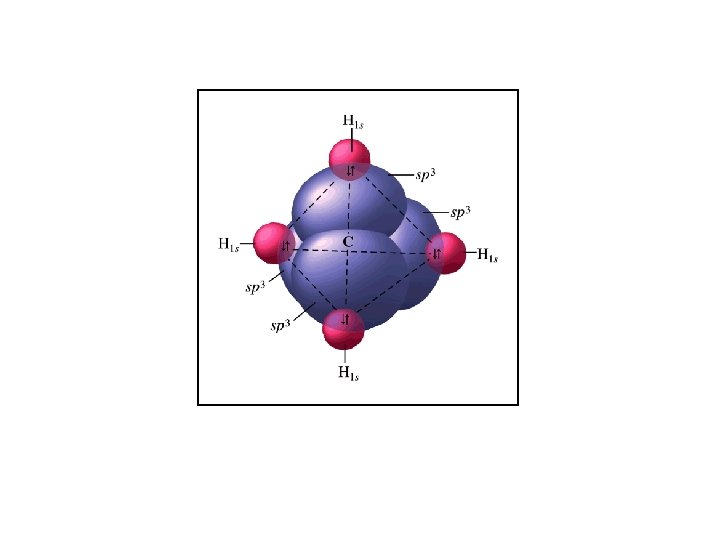
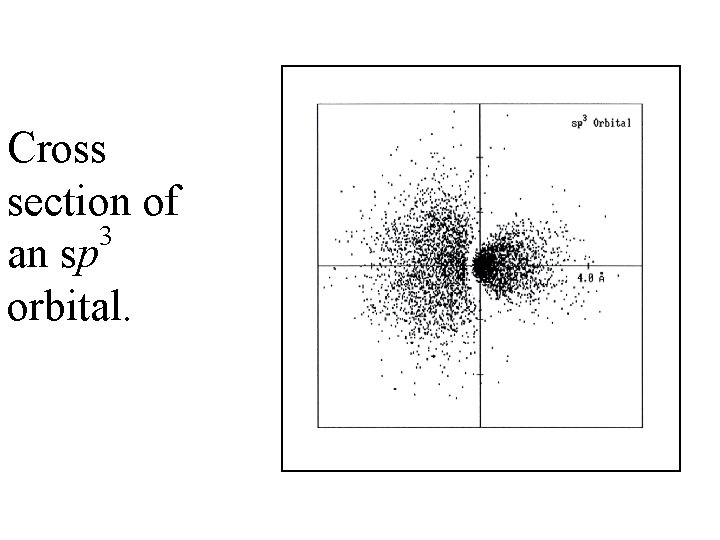
Cross section of 3 an sp orbital.
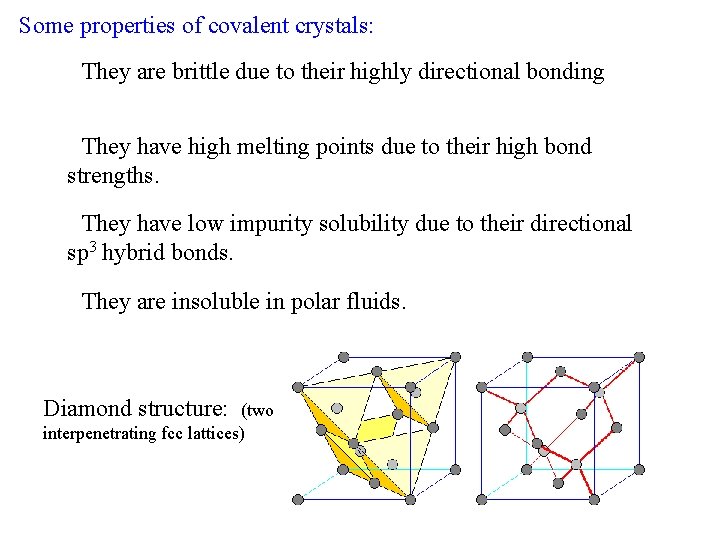
Some properties of covalent crystals: • They are brittle due to their highly directional bonding. • They have high melting points due to their high bond strengths. • They have low impurity solubility due to their directional sp 3 hybrid bonds. • They are insoluble in polar fluids. Diamond structure: (two interpenetrating fcc lattices)
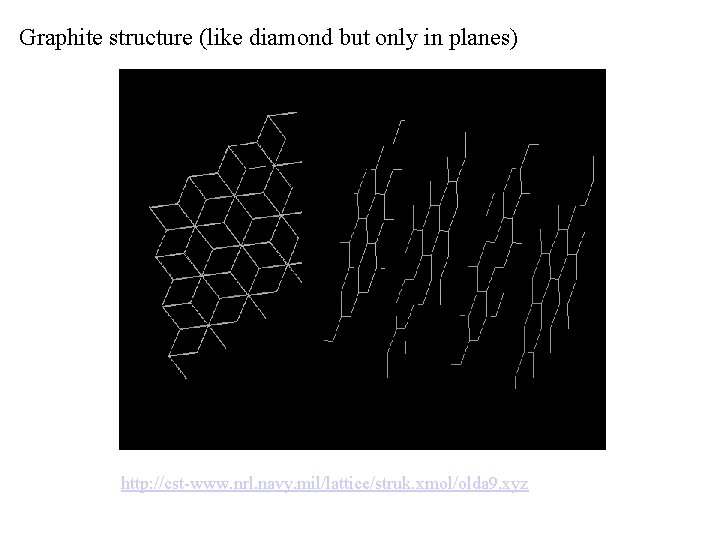
Graphite structure (like diamond but only in planes) http: //cst-www. nrl. navy. mil/lattice/struk. xmol/olda 9. xyz
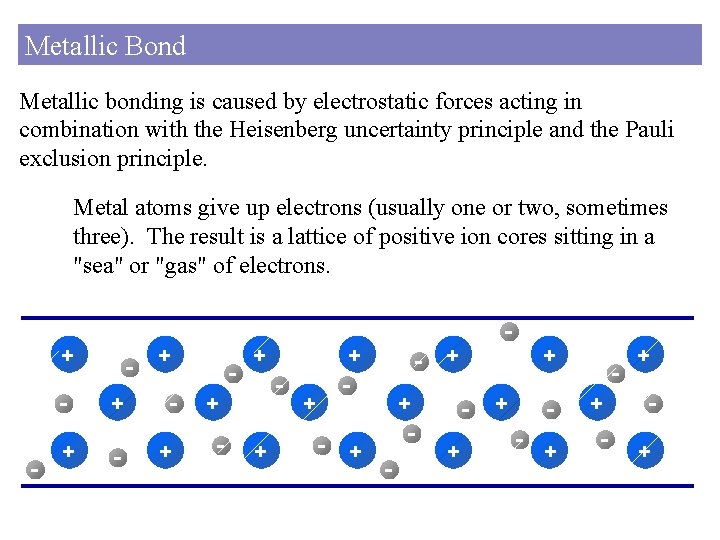
Metallic Bond Metallic bonding is caused by electrostatic forces acting in combination with the Heisenberg uncertainty principle and the Pauli exclusion principle. Metal atoms give up electrons (usually one or two, sometimes three). The result is a lattice of positive ion cores sitting in a "sea" or "gas" of electrons. + - + + + - + - - + + + - + +

Hydrogen bonding: “The attraction of the partially positive end of one highly polar molecule for the partially negative end of another highly polar molecule is called a hydrogen bond. ” http: //207. 10. 97. 102/chemzone/lessons/03 bonding/mleebonding/hydrogen_bonds. htm
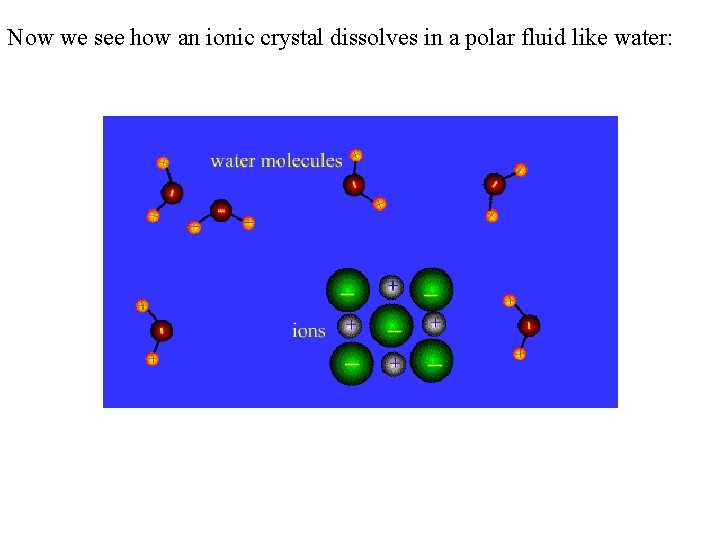
Now we see how an ionic crystal dissolves in a polar fluid like water: (I probably should have shown this back in the ionic crystals section. )
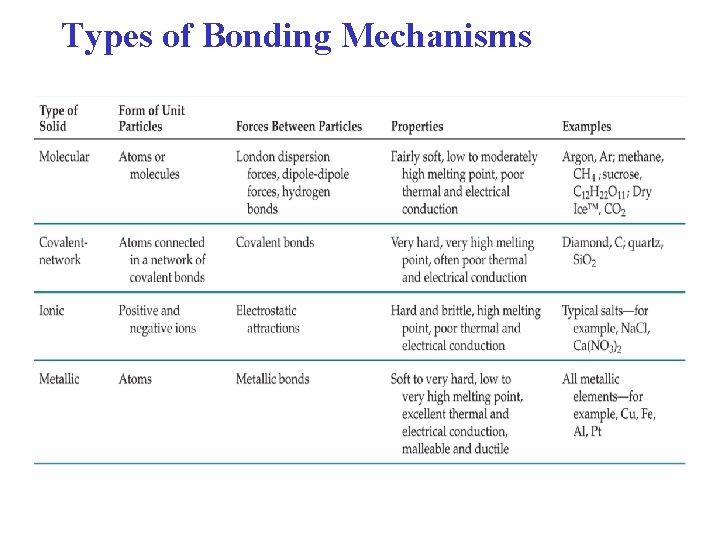
Types of Bonding Mechanisms
- Slides: 25