Chapter 3 Atoms The Building Blocks of Matter

Chapter 3 - Atoms: The Building Blocks of Matter

• There were two schools of thought of the composition of the cosmos… – is everything in the universe continuous and infinitely divisible – Or, is there a limit to how small you can get? • Particle theory was not the most popular early opinion, but was supported as early as Democritus in ancient Greece. From Philosophy to Science

• Democritus proposed that all the matter is composed of tiny particles called “Atomos” – These “particles” were thought to be indivisible • Aristotle did not accept Democritus’ atom, he was of the “matter is continuous” philosophy – Because of Aristotle’s popularity his theory was adopted as the standard From Philosophy to Science

• By the 1700’s nearly all chemists had accepted the modern definition of an element as a particle that is indivisible • It was also understood at that time that elements combine to form compounds that are different in their properties than the elements that composed them – However, these understandings were based on observations not empirical evidence From Philosophy to Science

• There was controversy as to whether elements always combine in the same proportion when forming a particular compound. – In the 1790’s, chemistry was revolutionized by a new emphasis on quantitative analysis because of new and improved balances • This new technology led to the discovery of some new scientific understandings From Philosophy to Science

• The Law of Conservation of Mass: – States that mass is neither created nor destroyed during ordinary chemical reactions or physical changes. – Which means the total mass of the reactants must equal the total mass of the products. From Philosophy to Science

+ Carbon, C Mass x Oxygen, O Mass y Carbon Monoxide, CO Mass x + Mass y + Carbon Monoxide, CO Mass x + Mass y Carbon, C Mass x Oxygen, O Mass y

• The Law of Definite Proportions: – The fact that a chemical compound contains the same elements in exactly the same proportions by mass regardless of the size of the sample or the source of the compound • Na. Cl is Na. Cl no matter if it is table salt (small crystals) or rock salt (large crystals) From Philosophy to Science

• The Law of Multiple Proportions: – If 2 or more different compounds are composed of the same 2 elements, then the ratio of the masses of the 2 nd element combined with a certain mass of the 1 st element is always a ratio of small whole numbers From Philosophy to Science

+ Carbon 1 = Oxygen 1 + Carbon Monoxide, 1: 1 = Carbon Oxygen 1 2 Carbon Dioxide, 1: 2

• In 1808, John Dalton proposed an explanation for each of the proposed laws – He reasoned that elements were composed of atoms & that only whole #’s of atoms can combine to form compounds – His ideas are now called the Atomic Theory of Matter Atomic Theory

1. All matter is composed of extremely small particles called atoms. 2. Atoms of a given element are identical in size, mass, and other properties; atoms of different elements differ in size, mass, & other properties. Atomic Theory ELEMENT 1 ELEMENT 2 ELEMENT 3 ELEMENT 4
3. Atoms cannot be subdivided, created, or destroyed 4. atoms of different elements combine in simple whole # ratios to form chem compds 5. in chemical rxns, atoms are combined, separated, or rearranged Atomic Theory + +

• Through these statements, evidence could be gathered to confirm or discount its claims – Not all of Dalton’s claims held up to the scrutiny of experimentation – Atoms CAN be divided into even smaller particles – Not every atom of an element has an identical mass Atomic Theory

• Dalton’s Atomic Theory of Matter has been modified. • What remains unchanged is… 1. All matter is composed of atoms 2. Atoms of any one element differ in properties from atoms of another element • One of the disputed statements of Dalton was that atoms are indivisible Atomic Theory

– In the 1800’s it was determined that atoms are actually composed of several basic types of smaller particles – it’s the number and arrangement of these particles that determine the atom’s chemical properties. • The def. of an atom that emerged was, the smallest particle of an element that retains the chemical properties of that original element. Atomic Theory

• All atoms consist of 2 regions that contain the subatomic particles – The nucleus – The electron cloud around the nucleus • The nucleus is a very small region located near the center of the atom – In every atom the nucleus contains at least 1 proton, which is positively charged particle and usually contains 1 or more neutral particles called neutrons Atomic Structure

• The electron cloud is the region that surrounds the nucleus – This region contains 1 or more electrons, which are negatively charged subatomic particles – The volume of the electron cloud is much larger than the nucleus Atomic Structure

• The discovery of the first subatomic particle took place in the late 1800’s. – A power source was attached to two metal ends of an evacuated glass tube, called a cathode ray tube. – A beam of “light” appears between the two electrodes called a cathode ray. Discovery of the Electron

• Investigators began to study the ray and they observed that… 1. An object placed in the path of the ray cast a shadow on the glass 2. A paddle wheel placed in the path of the cathode ray began to spin 3. Cathode rays were deflected by a magnetic field 4. The rays were deflected away from a negatively charged object Discovery of the Electron

• The first 2 observations support the idea that the ray is composed of tiny individual particles traveling through the vacuum tube • The second set of observations support the evidence that the ray is composed of a substance that is negatively charged. Discovery of the Electron

• J. J. Thomson studied the rays and proved that they were tiny negative particles being emitted from the metal atoms. – Dubbed these tiny particles “electrons” – And it was later determined that the electrons were not part of the mass of the atom. Discovery of the Electron

• What can their work help us conclude about the atom? – atoms are composed of smaller particles, and one of these components is negatively charged – atoms are neutral, so there must be an opposing (+) charge – because electrons are essentially mass-less, an opposing substance makes up the mass of the atom First Atomic Model

• In 1886, E. Goldstein observed in the cathode-ray tube a new set of rays traveling in the opposite direction than the cathode rays – The new rays were called canal rays and they proved to be positively charged – And the particles mass were about 2000 X’s that of the electron Discovery of the Proton

• In 1932, the English physicist James Chadwick discovered yet another subatomic particle. – the neutron is electrically neutral – It’s mass is nearly equal to the proton • Therefore the subatomic particles are the electron, proton, and neutron. Discovery of the Neutron
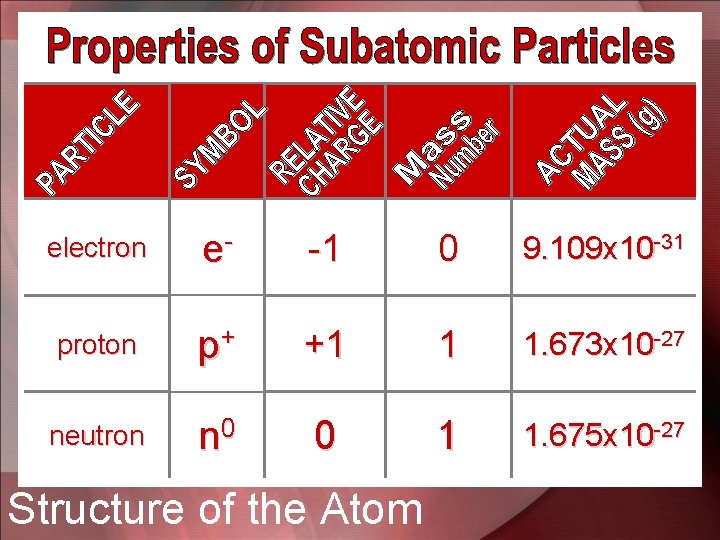
electron e- -1 0 9. 109 x 10 -31 proton p+ +1 1 1. 673 x 10 -27 neutron n 0 0 1 1. 675 x 10 -27 Structure of the Atom

• With the exception of Hydrogen, every nucleus contains 2 kinds of particles protons and neutrons – they make up the mass of the atom (Mass Number = Protons + Neutrons) • Proton has a charge equal to but opposite of the charge of an electron. – Atoms are neutral because they contain equal #’s of protons & electrons Atomic Structure

• The atoms of different elements differ in the # of protons in their nuclei and therefore in their positive charge – The # of protons the atom contains determines the atom’s identity, also known as atomic number. • Only Oxygen contains 8 protons • Only Fluorine contains 9 protons • Only Neon contains 10 protons Structure of the Atom

• The nucleus is composed of a densely packed cluster of protons, which are all electrically positive – Don’t like charges repel? – Why don’t they fly apart? • When 2 protons are in very close proximity, there is a strong force of attraction between them. – similar attraction exists when neutrons are close Structure of the Atom

• These short-range p+-n 0, p+-p+, & n 0 -n 0 forces hold the nuclear particles together, A. K. A strong nuclear forces. – When these nuclear forces are strong enough the atom is stable – If the forces are not strong enough the atom (heavier atoms) the atom is unstable and becomes radioactive. Structure of the Atom
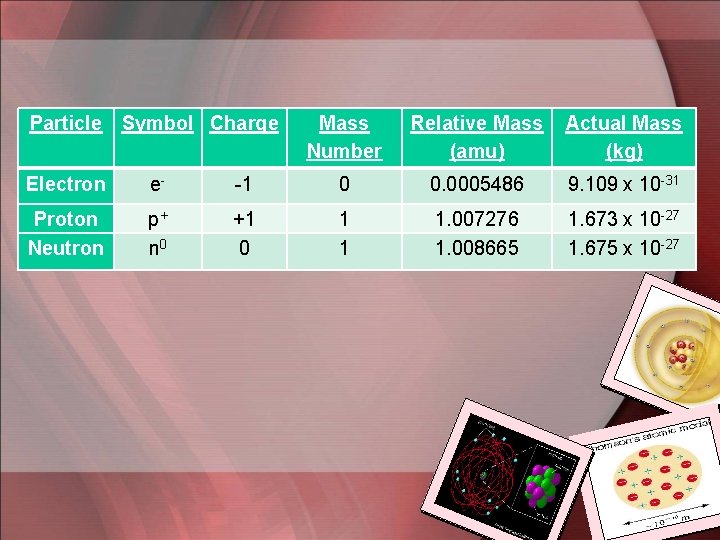
Particle Symbol Charge Mass Number Relative Mass (amu) Actual Mass (kg) Electron e- -1 0 0. 0005486 9. 109 x 10 -31 Proton Neutron p+ n 0 +1 0 1 1 1. 007276 1. 008665 1. 673 x 10 -27 1. 675 x 10 -27

Ch 3. 3: Atomic Number

• Elements are identified by the number of PROTONS they contain. • The “atomic number” of an element is the number of protons in the nucleus – PROTONS IDENTIFIES AN ELEMENT!!! • # protons in an atom = # electrons – Why? Because atoms are neutral!
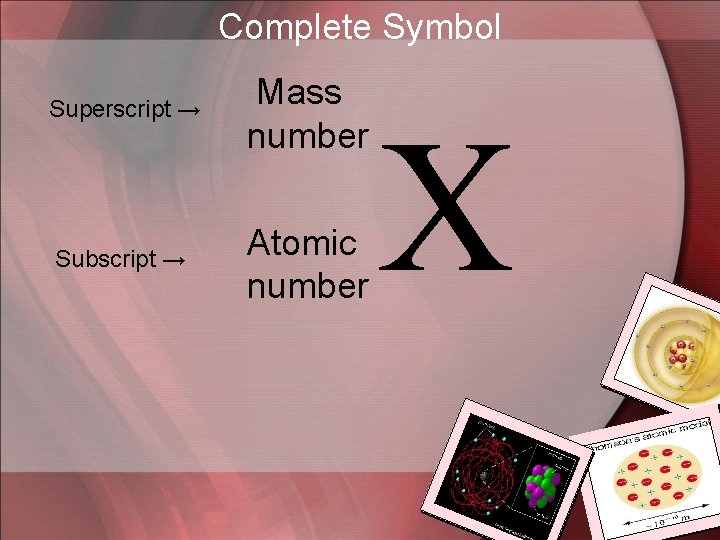
Complete Symbol Superscript → Mass number Subscript → Atomic number X

# OF PROTONS + # OF NEUTRONS MASS NUMBER 35 ATOMIC NUMBER 17 Cl NUMBER OF PROTONS
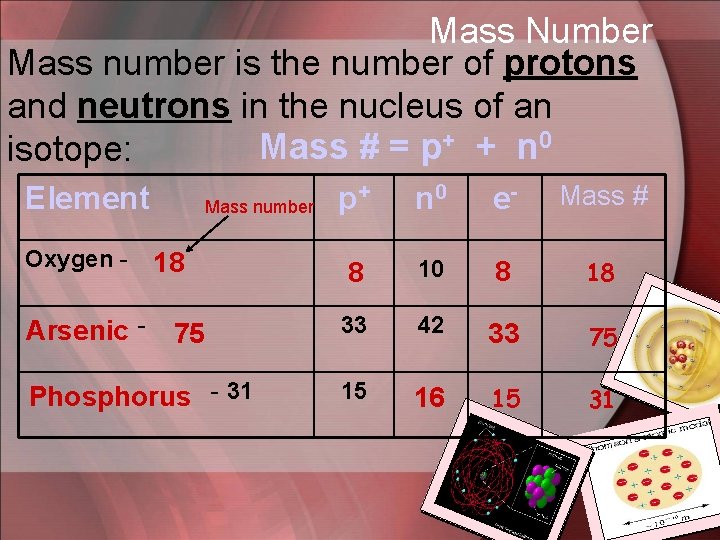
Mass Number Mass number is the number of protons and neutrons in the nucleus of an + + n 0 Mass # = p isotope: p+ n 0 e- Mass # 8 10 8 18 Arsenic - 75 33 42 33 75 Phosphorus - 31 15 16 15 31 Element Oxygen - Mass number 18

Practice Problems (1) Find the # of e-, p+ and n 0 for sodium. (mass # = 23) Atomic # = 11 = # e- = # p+ # neutrons = 23 -11 = 12 2) Find the # of e-, p+ and n 0 for uranium. (mass # = 238) Atomic # = 92 = # e- = # p+ # neutrons = 238 -92 = 146

Check for understanding: n If an element has 91 protons and 140 neutrons find the: a) Atomic number 91 b) Mass number 231 c) number of electrons 91 d) element name protactinium

Isotopes • An isotope refers to atoms that have the same # of protons, but a different number of neutrons. • Because of this, they have different mass #’s. Ex---> (1) Carbon-12 & Carbon-13 (2) Chlorine-35 & Chlorine-37 (Isotopes: The # after the name is the mass #. )
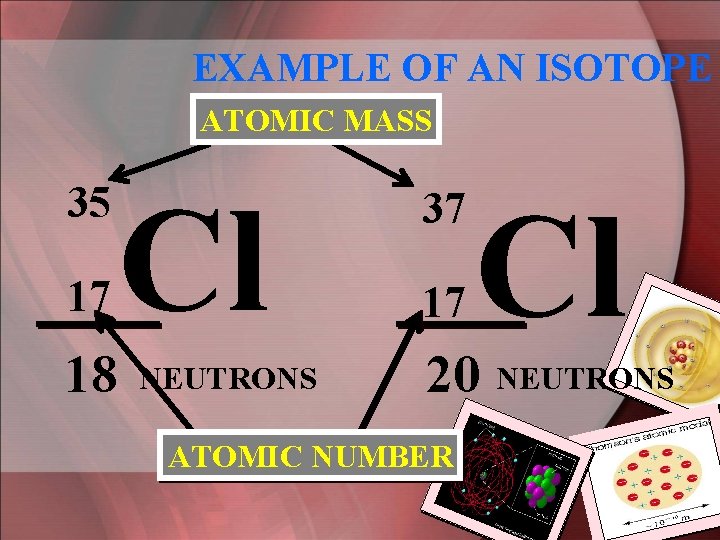
EXAMPLE OF AN ISOTOPE ATOMIC MASS 35 17 18 Cl NEUTRONS 37 17 Cl 20 NEUTRONS ATOMIC NUMBER

Question #1 n Find each of these: a) Atomic number b) Mass Number c) number of protons d) number of neutrons e) number of electrons 80 35 Br

Question #2 n If an element has an atomic number of 34 and a mass number of 78, what is the: a) number of protons b) number of neutrons c) number of electrons d) complete symbol

Atomic Mass 12 • Units = atomic mass unit (amu) • The atomic masses listed in the Periodic Table are a “weighted average” of all the isotopes of the element.

Weighted Average Practice Problems: (1) In chemistry, chlorine has 2 isotopes: Cl-35 (75. 8% abundance) Cl-37 (24. 23 % abundance) What is the weighted average atomic mass of chlorine? 35 x 0. 758 = 26. 53 amu 37 x 0. 2423 = + 8. 9651 amu 35. 4951 amu Add them up!!!

Relating Mass Numbers to Atoms • The Mole: the amount of a substance that contains as many particles as there atoms in exactly 12 grams of carbon-12. • Avogadro’s Number: the number of particles in exactly one mole of a pure substance = 6. 022 x 1023. • Molar Mass: the mass of one mole of a pure substance. Units = g/mol

• This is when we get to use dimensional analysis! • The conversion factors we need are: and of course…molar mass
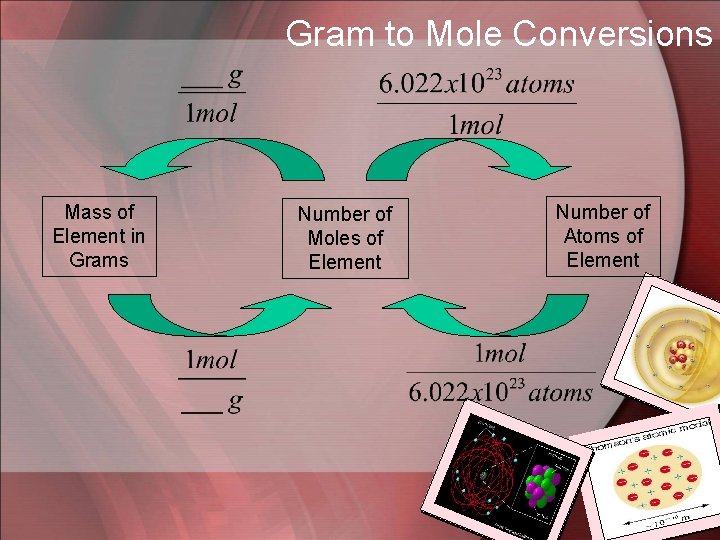
Gram to Mole Conversions Mass of Element in Grams Number of Moles of Element Number of Atoms of Element

Practice Problem • ALWAYS USE PARANTHESES AROUND YOUR CONVERSION FACTORS!! • You have 3. 50 mol of Copper. What is it mass in grams?
- Slides: 48