Chapter 3 Atoms The Building Blocks of Matter

Chapter 3 Atoms: The Building Blocks of Matter

3. 3 Counting Atoms Objectives • Define atomic number and mass number, and describe how they apply to isotopes. • Given the identity of a nuclide, determine its number of protons, neutrons, and electrons. • Explain what isotopes are.

3. 3 Counting Atoms Atomic Number • Atoms of different elements have different numbers of protons. • Atoms of the same element all have the same number of protons. • The atomic number (Z) of an element is the number of protons of each atom of that element.

3. 3 Counting Atoms Atomic Number • The number of protons in each atom identifies it as an atom of a particular element • Each atom has a unique number of protons and, in effect, the number of protons cannot change for a particular atom • The Periodic Table is organized by increasing atomic number (left-to-right and top -to-bottom) • All atoms are neutral, so the # protons = the # of electrons because the number of positive charges equals the number of negative charges • Atomic # = # of protons = # electrons • For Helium, the atomic # is 2. So, we know that Helium also has 2 protons and 2 electrons

3. 3 Counting Atoms APE MAN • Protons; p+ • Electron; e • Neutron; n A P E Atomic # = # of p+ = # of e. M A N Atomic Mass – Atomic # = # n

3. 3 Counting Atoms Isotopes • All atoms of a particular element have the same number of protons and electrons • The number of neutrons may differ; the same element will have different a mass • In nature most elements are found as a mixture of isotopes • Isotopes are atoms of the same element that have different masses

3. 3 Counting Atoms Designating Isotopes • Hyphen Notation: The mass number is written with a hyphen after the name of the element. uranium-235 • Isotope Notation: The superscript indicates the mass number and the subscript indicates the atomic number. 235 U 92

3. 3 Counting Atoms Designating Isotopes, continued • The number of neutrons is found by subtracting the atomic number from the mass number. • mass number − atomic number = number of neutrons • 235 (protons + neutrons) − 92 protons = 143 neutrons • Nuclide is a general term for a specific isotope of an element.
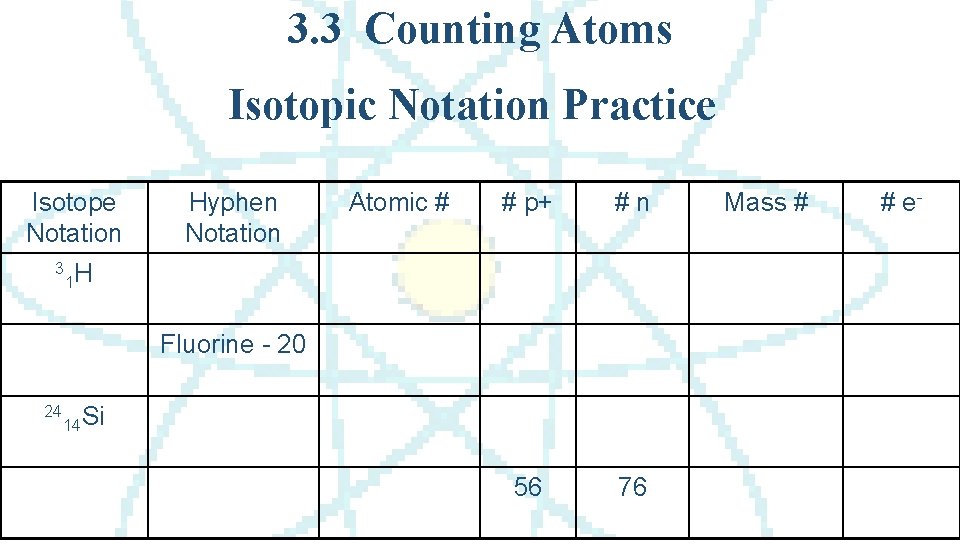
3. 3 Counting Atoms Isotopic Notation Practice Isotope Notation 3 Hyphen Notation Atomic # # p+ #n 56 76 1 H Fluorine - 20 24 14 Si Mass # # e-
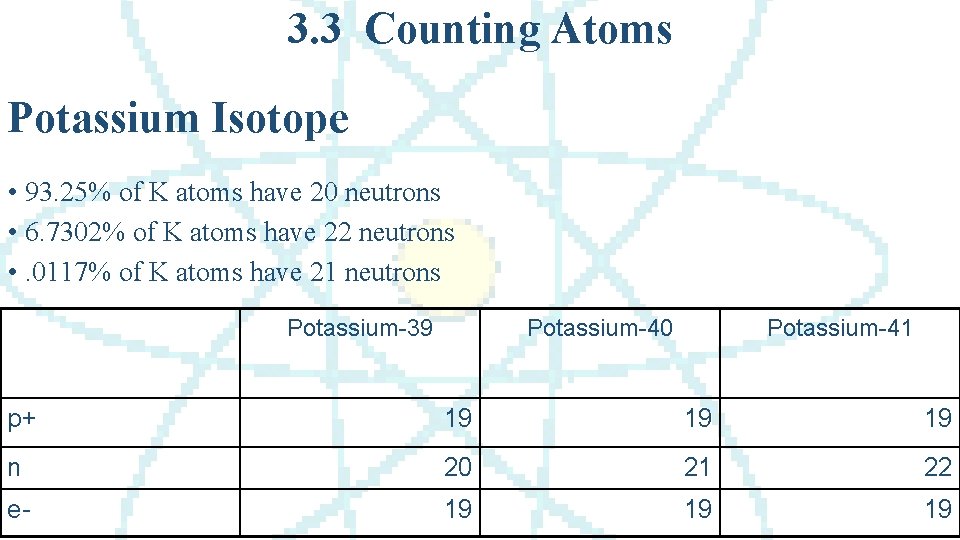
3. 3 Counting Atoms Potassium Isotope • 93. 25% of K atoms have 20 neutrons • 6. 7302% of K atoms have 22 neutrons • . 0117% of K atoms have 21 neutrons Potassium-39 Potassium-40 Potassium-41 p+ 19 19 19 n 20 21 22 e- 19 19 19

3. 3 Counting Atoms Mass of Individual Atoms • The atomic mass of an element is the weighted average mass of the all the known isotopes of that element • To determine the weighted average atomic mass of an element, calculate the mass contribution of each of the isotopes and add the values together

3. 3 Counting Atoms Average Atomic Mass 17 Cl 35 37 17 Cl Atomic mass: 34. 969 amu Atomic mass: 36. 966 amu % abundance: 75. 770% % abundance: 24. 230% Mass : (34. 969)(. 75770)= 26. 496 amu Mass: (36. 966)(. 24230)=8. 957 amu Average atomic mass of chlorine = (26. 496 amu + 8. 957 amu) = 35. 453 amu

3. 3 Counting Atoms Steps to Solving Weighted Averages of Isotopes 1. Turn the % into a decimal for the % abundance. 2. Multiply the decimal by the atomic mass of each particular isotope. 3. Add the masses of the isotopes together. 4. Your answer will be in amu (atomic mass units).

3. 3 Counting Atoms Practice 1 Calculate the atomic mass of Magnesium. The three Magnesium isotopes have atomic masses and relative abundances of 23. 985 amu (79. 99%), 24. 986 amu (10. 00%), and 25. 982 amu (11. 01%).

3. 3 Counting Atoms Practice 2 What is the atomic mass of this sample of Silver? 107 Ag 106. 905 amu 52 % 109 Ag 108. 905 amu 48 %

3. 3 Counting Atoms Practice 3 What is the average atomic mass of this sample of Hydrogen? 1 H 12 % 2 H 48 % 3 H 40 %
- Slides: 16