Chapter 3 Atoms The Building Blocks of Matter

Chapter 3 Atoms: The Building Blocks of Matter

The Atomic Theory § Law of conservation of mass § Mass is neither destroyed nor created § Law of definite proportions § Compound is always composed of same proportion of elements § Law of multiple proportions § If two different compounds are composed of the same elements, the two elements form a ratio of small number

Law of Conservation of Mass
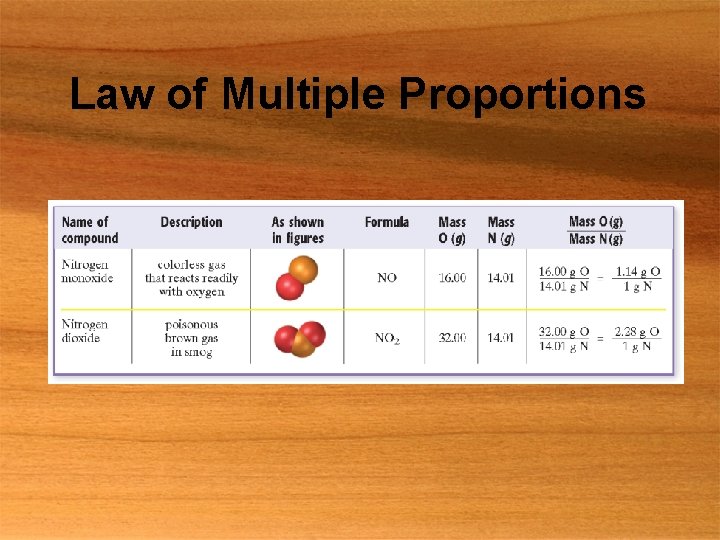
Law of Multiple Proportions

Dalton § All matter is composed of atoms § Atoms of an element are identical in size, mass and other properties § Atoms cannot be subdivided, created, or destroyed § Atoms combine in simple whole-number ratios § Atoms are combined, separated, or rearranged in chemical reactions

Modern Atomic Theory § All matter is composed of atoms § Atoms of one element differ in properties from atoms of another element § Atom - smallest particle of an element that retains the chemical properties of that element

Thompson § Current passed through cathode-ray tube § Current produced a stream of particles (cathode ray) that traveled towards the anode § Rays deflected from negatively charged object § Ratio of charge to mass of particles

Milikan § Oil-drop experiment § Charge of electron determined § Calculated mass of electron using ratio of charge to mass

The Electron § Mass of 9. 109 x 10 -31 kg § Atoms are neutral, so must contain same number of electrons and protons § Electrons mass has negligible effect on total mass of atom

The Nucleus § Rutherford’s gold foil experiment § § Thin, gold foil bombarded with α particles Some α particles deflected Caused by positively charged nucleus Volume of nucleus small
Rutherford
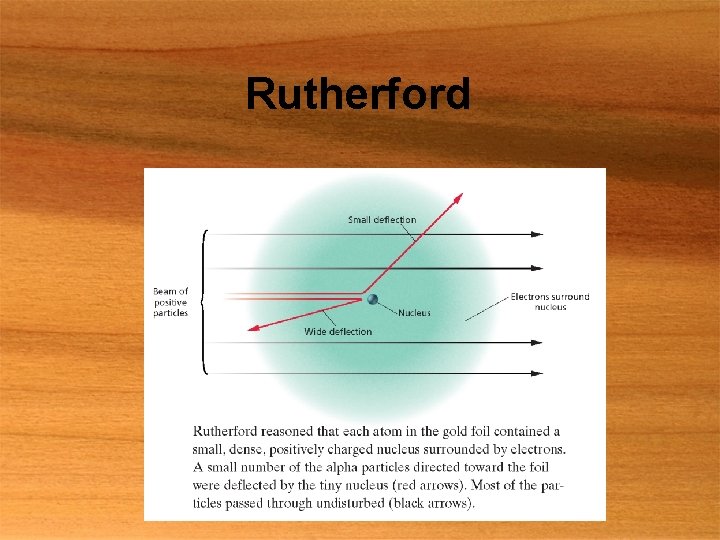
Rutherford

The Nucleus § Composed of two particles § Protons § Neutrons § Protons mass = 1. 673 x 10 -27 kg § 1826 times greater than e- § Nuclear strong force § Holds positive particles together

Atomic Number § Atoms of different elements have different number of protons § Atomic number (Z) § number of protons in the nucleus § Elements arranged in order of their atomic number on periodic table

Mass Number § Total number of protons and neutrons § Ex. Hydrogen Isotopes

Isotopes § Element may contain different number of neutrons § Isotopes have differing mass § Naming § Helium-4 (hyphen notation) § 4 Helium (nuclear symbol) 2

Problems § How many protons, electrons, and neutrons are in an atom of carbon-13? § Write the nuclear symbol for oxygen-16. § How many protons, electrons, and neutrons are in an atom of bromine-80? § Write the nuclear symbol for carbon-13. § Write the hyphen notation for the element that contains 15 electrons and 15 neutrons.

Relative Atomic Masses § Atomic mass unit (amu) § 1/12 the mass of carbon-12 § 1. 660 540 x 10 -27 kg § Hydrogen-1 = 1 amu § Average atomic mass § Weighted average of atomic masses of isotopes

Average Atomic Mass § Ex. § 25% weigh 2. 00 g § 75% weigh 3. 00 g § (2. 00 g x 0. 25) + (3. 00 g x 0. 75) = 2. 75 g

Problem § What is the average atomic mass of copper which consists of 69. 17% copper-63 (62. 929 598 amu), and copper-65 (64. 927 793 amu)?

Problems § Three isotopes of argon occur in nature - Ar-36, Ar-38, Ar-40. Calculate the average atomic mass of argon to two decimal places, given the following relative atomic masses and abundances of each of the isotopes: argon-36 (35. 97 amu; 0. 337%), argon-38 (37. 96 amu; 0. 063%), and argon-40 (39. 96 amu; 99. 600%).

The Mole § Amount of substance that contains as many particles as there atoms in exactly 12 g of carbon-12 § Counting unit like dozen § Dozen = 12 § Mole = 6. 022 x 1023 § Avogadro’s number § 6. 022 x 1023

Molar Mass § Mass of one mole of pure substance § Units →g/mol § Molar mass = atomic mass in amu
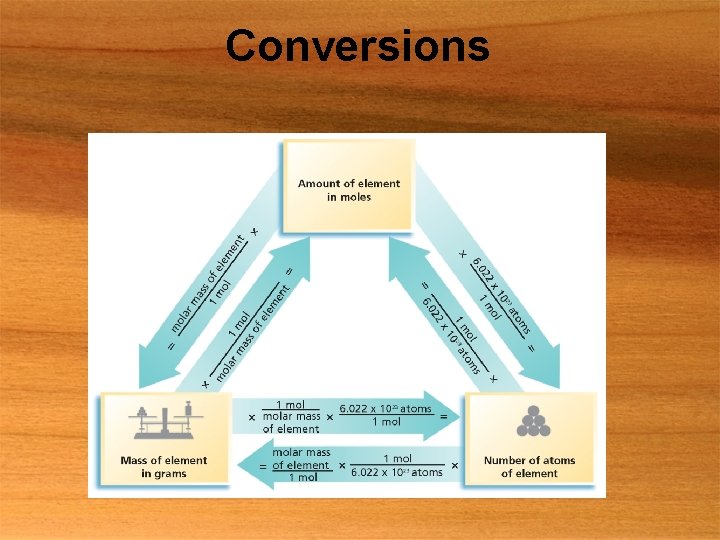
Conversions

Conversions § 1 mol/(6. 022 x 1023) atoms § Molar mass/mol

Problems § What is the mass in grams of 3. 6 mol of the element carbon, C? § What is the mass in grams of 0. 733 mol of the element chlorine, Cl? § How many moles of copper, Cu, are in 3. 22 g of copper? § How many moles of lithium, Li, are in 2. 72 x 10 -4 g of lithium?

Problems § How many moles of carbon, C, are in 2. 25 x 1022 atoms of carbon? § How many moles of oxygen, O are in 2 000 atoms of oxygen? § How many atoms of sodium, Na, are in 3. 80 mol of sodium? § What is the mass in grams of 5. 0 x 109 atoms of neon, Ne?

Problems § How many atoms of carbon, C, are in 0. 020 g of carbon? § What mass of silver, Ag, contains the same number of atoms as 10. 0 g of boron, B? § How many moles of CO 2 are in 66. 0 g of dry ice, which is solid CO 2

Chapter Review § Pg. 89 § 1, 8, 11, 17, 18 acd, 20, 21, 22 ade, 23 abcd
- Slides: 29