Chapter 3 3 1 Properties of Matter Substances

Chapter 3 3. 1 – Properties of Matter

Substances Matter – has mass & takes up space Substance – matter with uniform and definite composition Ex: Salt, pure water, air, etc.

States of Matter All matter can be classified as one of 3 physical forms: 1. Solid – definite shape and volume -Particles tightly packed & do not conform to container.

States of Matter 2. Liquid – Matter that flows, constant volume, & takes shape of container -Particles are NOT rigidly held in place and are less closely packed than those of a solid.

States of Matter 3. Gas – flows to conform to container & fills entire volume -Large amounts of space between particles & a gas will compress easily.
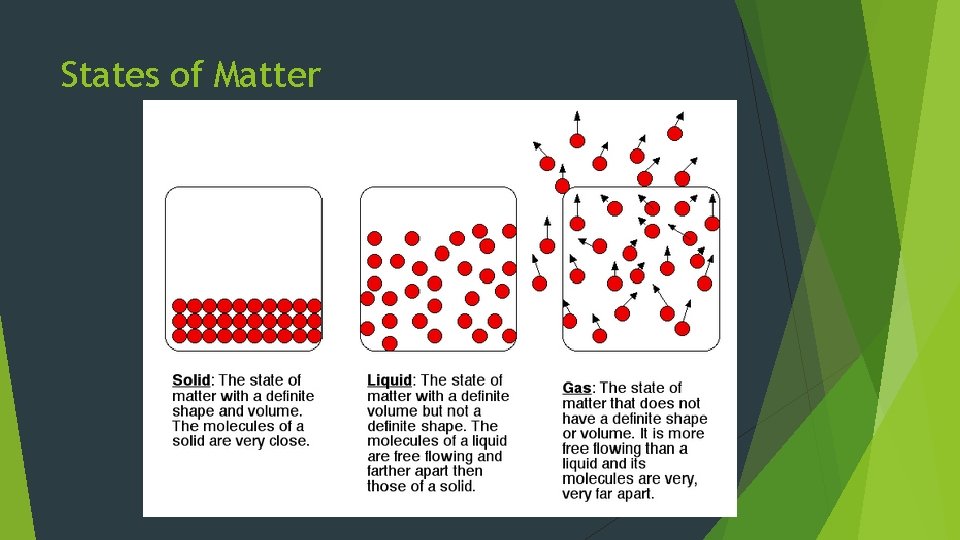
States of Matter

States of Matter **NOTE** Vapor is NOT the same as gas! ~ Vapor is a solid or liquid at room temp (a gas is still a gas at room temp!) ex: Steam is a vapor

Physical Properties of Matter Physical Property – characteristic that is observable or measured w/o changing composition. Ex: Denisty, color, melting point, boiling point, smell, etc.

Physical Properties of Matter – cont. Physical properties can be broken down further into two types: 1. Extensive Properties – dependent on amount of substance present (mass, length, etc. ) 2. Intensive Properties – amount of substance is not important (density, color, etc. )

Chemical Properties Chemical Property – ability of substance to combine w/ or change into one or more other substances Ex: Rust

Observing Properties of Matter Do surrounding conditions affect properties? How or why not? When making observations, be sure to state specific conditions such as temperature, pressure, etc. for more repeatable results! Ex: Density of Water: STP (1. 0 atm & 273. 15 K) = 1. 00 g/cm 3 >100°C <0 = 0. 0006 g/cm 3 °C = 0. 92 g/cm 3

Chapter 3 3. 2 – Changes in Matter

Physical Changes Physical Change – change that alters substance w/o changing composition ex: wadding up paper Phase Change – transition of matter from one state to another physical change ex: water cycle – (freeze, boil, condense, vaporize, etc. )

Chemical Change – one or more substances changing into new substance The new substance will have different composition and properties! Reactants Products – starting substance – new substance formed **Decompose, rust, explode, oxidize, corrode, tarnish, burn, rot, etc. = chemical rxn.

Conservation of Mass

Example 1

Chapter 3 3. 3 – Mixtures of Matter

Mixtures Pure Substance – uniform and unchanging composition Mixture Each – Combo of 2 or more pure substances pure substance retains individual chem properties **Most everyday matter occurs as mixtures since pure substances tend to mix naturally (difficult to keep pure!)

Types of Mixtures Heterogeneous Mixtures – does not blend smoothly throughout (composition is NOT uniform) Ex: Italian Salad Dressing Homogeneous throughout Ex: Mixture – Constant composition Tea AKA – solutions (can occur as solid, liquid and/or gas)

Tyndall Effect – Quick test to differentiate solution from heterogeneous mixture Shine thin beam (laser) of light through beaker (glass container) If light does not shine through => solution If light shines though – heterogeneous https: //youtu. be/So. Umz 8 L 87 Z 4

Separating Mixtures Since most substances are physically combined, most can be physically separated! **Take advantage of the physical properties to separate if possible!

Techniques for Separating Mixtures Filtration Use easy on heterogeneous solid/liquid mixtures porous barrier to separate solids from liquids (coffee filters) https: //youtu. be/Q 0 s 71 cj. CNWs Distillation – most homogeneous mixtures can be separated using distillation Based on ∆ in BP Mixture heated until substance w/ lowest BP boils to a vapor that can be condensed & collected as a liquid https: //youtu. be/V 5 ep 0 -oj. PGw

Techniques for Separating Mixtures – Cont. Crystallization – results in the formation of pure solid particles of a substance from soln containing dissolved substance (collects as crystals) Ex: rock candy https: //youtu. be/mrm 5 Dg. Qa 8 mo Sublimation – process of changing solid to vapor without melting/going through liquid phase Can be used to separate 2 solids in mixture when one solid will sublimate and not other https: //youtu. be/6 YYrc. HLck. Mw

Techniques for Separating Mixtures – Cont. Chromatography – separates compounds of mixture based on ability of each component (gas or liquid) to travel/be drawn across surface of another material (chromatography paper) Components w/ strongest attraction to surface travel slower https: //youtu. be/Pv. Hvx 7 k 7 UPU

Chapter 3 3. 4 – Elements and Compounds

Elements All matter can be broken down into small, basic building blocks Element – pure substance that can’t be separated into simpler substance by phys/chem means 92 elements occur naturally Each has unique chem name & symbol 1 st letter ALWAYS capitalized and other letters are lowercase. Ex: Gold - Au, Silver – Ag, Oxygen – O, etc.

1 st Periodic Table 1869 Russian Chemist (Mendeleev) created 1 st version of periodic table Classification based upon similarities and masses of elements. Periodic Table – organizes elements into a grid of horizontal rows (periods) & vertical columns (groups/families)

Compounds Compound – made up of 2 or more different elements that are combined chemically ~10 million known compounds Increases Chemical at a rate of ~100, 000/year symbols make it easy to write formulas for compounds (ex: Na. Cl, H 2 O)

Compounds – cont. Properties: Prop. of compound different than prop of individual elements.
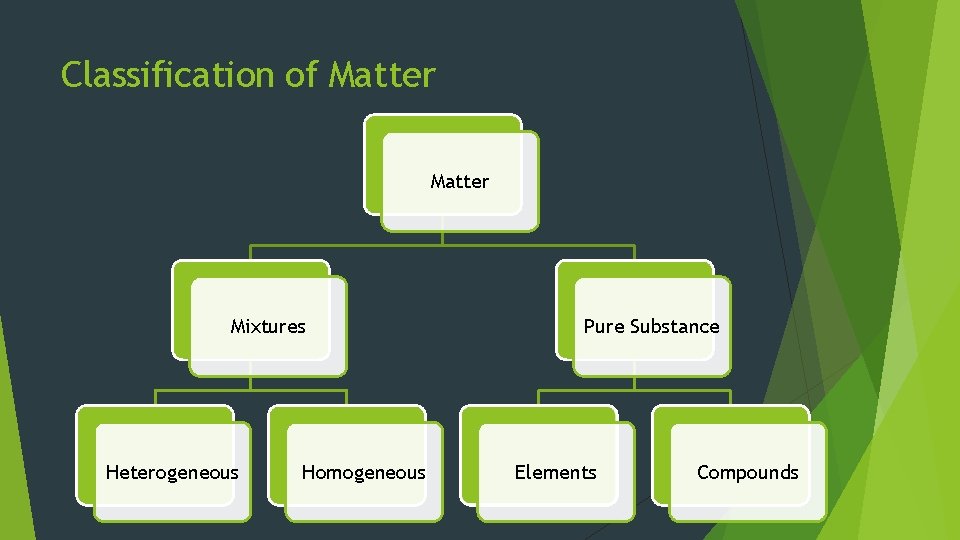
Classification of Matter Mixtures Heterogeneous Homogeneous Pure Substance Elements Compounds

Law of Definite Proportions Elements that make up compounds always combine in definite proportions. Law of Definite Proportions – compound is always composed of same elements in same proportion by mass, regardless of sample size

Law of Definite Proportions – cont.

Example 2

- Slides: 34