Chapter 27 Fluid Electrolyte and Acidbase Balance BIO

Chapter 27: Fluid, Electrolyte and Acid-base Balance BIO 211 Lecture Instructor: Dr. Gollwitzer 1

• Today in class we will discuss: – The importance of water and its significance to fluid balance in the body – Definitions and the importance of: • Fluid Balance • Electrolyte balance • Acid-base balance – Extracellular fluid (ECF) and intracellular fluid (ICF) and compare their composition – Fluid and electrolyte balance • Hormones that regulate them • Importance of key electrolytes 2

Introduction • Water critical to survival – 50 -60% total body weight – 99% of extracellular fluid (ECF) – Essential component of cytosol (intracellular fluid, ICF) • All cellular operations rely on water – Diffusion medium for gases, nutrients, waste products 3

Body Fluid Compartments • Body must maintain normal volume and composition of: – ICF – ECF = all other body fluids • Major - IF, plasma • Minor - lymph, CSF, serous and synovial fluids • ICF > total body water than ECF – Acts as water reserve 4
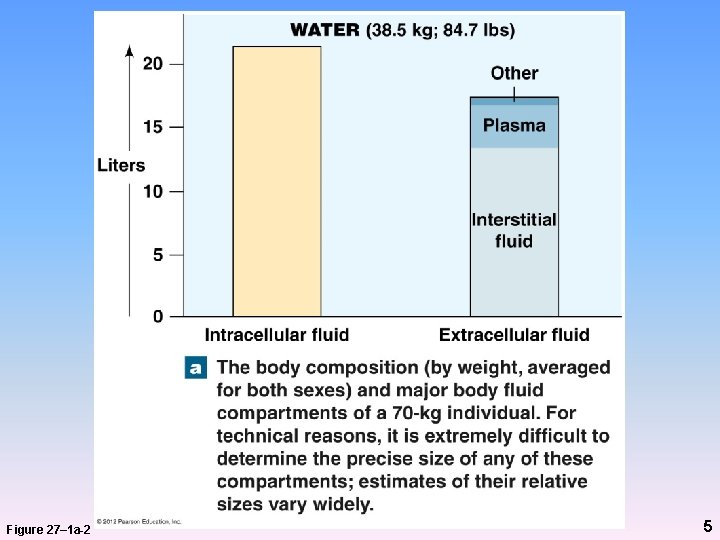
Body Fluid Compartments Figure 27– 1 a-2 5

Body Fluid Balance • Must maintain body fluid: – Volume (fluid balance) – Ionic concentration (electrolyte balance) – p. H (acid-base balance) • Gains (input) must equal loss (output) • Balancing efforts involve/affect almost all body systems 6

Fluid (Water) Balance • = amount of H 20 gained each day equal to amount of H 2 O lost • Regulates content and exchange of body water between ECF and ICF • Gains – GI (from food, liquid)* – Catabolism • Losses – Urine* – Evaporation (from skin, lungs) – Feces * Primary route 7
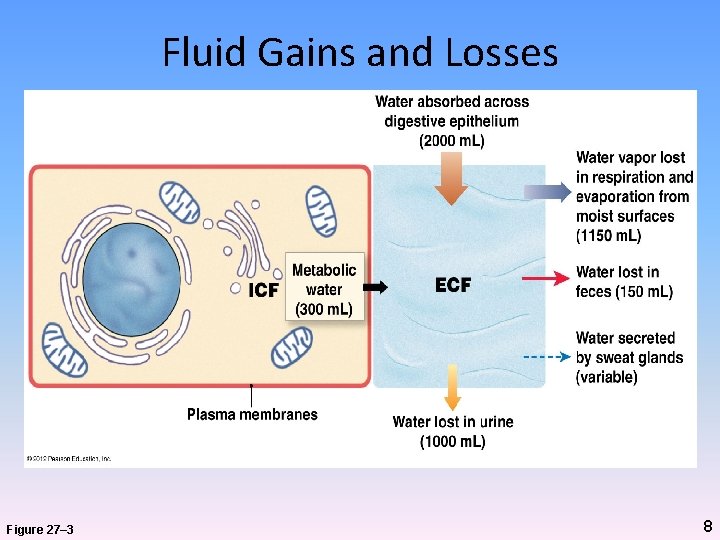
Fluid Gains and Losses Figure 27– 3 8

Electrolyte (Ion) Balance • Balances gains and losses of all electrolytes (ions that can conduct electrical current in solution) • Gains – GI (from food, liquid) • Losses – Urine – Sweat – Feces 9

Acid-base (p. H) Balance • Balances production and loss of H+ • Gains – GI (from food and liquid) – Metabolism • Losses – Kidneys (secrete H+) – Lungs (eliminate CO 2) 10

Fluid Components • ECF components (plasma and IF) very similar • Major differences between ECF and ICF • ICF very different because of cell membrane – Selectively permeable – Specific channels for ions – Active transport into/out of cell • Water exchange between ECF and ICF occurs across cell membranes by: – Diffusion – Osmosis – Carrier-mediated transport 11
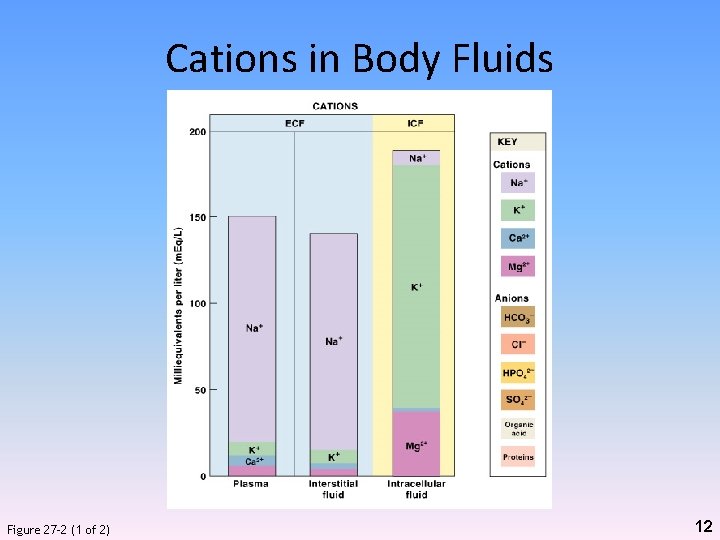
Cations in Body Fluids Figure 27– 2 (1 of 2) 12

Anions in Body Fluids Figure 27– 2 (2 of 2) 13

Cations and Anions in Body Fluids • In ECF – Na+ – Cl– HCO 3 - • In ICF – K+ (98% of body content) – Mg 2+ – HPO 42– Negatively charge proteins 14

Principles of Fluid and Electrolyte Regulation • All homeostatic mechanisms that monitor and adjust body fluid composition respond to changes in ECF, not ICF – Because: • A change in ECF spreads throughout body and affects many or all cells • A change in ICF in one cell does not affect distant cells 15

Principles of Fluid and Electrolyte Regulation • No receptors directly monitor fluid or electrolyte balance – Electrolyte balance = electrolytes gained equals the electrolytes lost • Monitor secondary indicators – Baroreceptors – for plasma volume/pressure – Osmoreceptors – for osmotic (solute) concentration • Solutes = ions, nutrients, hormones, all other materials dissolved in body fluids 16

Principles of Fluid and Electrolyte Regulation • Cells cannot move water by active transport – Passive in response to osmotic gradients • Fluid balance and electrolyte balance are interrelated • Body’s content of water and electrolytes: – Increases if gains exceed losses – Decreases if losses exceed gains 17

Primary Hormones for Fluid and Electrolyte Balance • ADH • Aldosterone • Natriuretic peptides (e. g. , ANP) 18

ADH • Produced by osmoreceptor neurons in supraoptic nuclei in hypothalamus (and released by posterior pituitary) – Osmoreceptors monitor osmotic concentrations in ECF – Osmotic concentration increases/decreases when: • Na+ increases/decreases or • H 2 O decreases/increases • Increased osmotic concentration increased ADH 19

ADH • Water conservation – Increases water absorption decreased osmotic concentration (by diluting Na+) – Stimulates thirst center in hypothalamus increased fluid intake 20
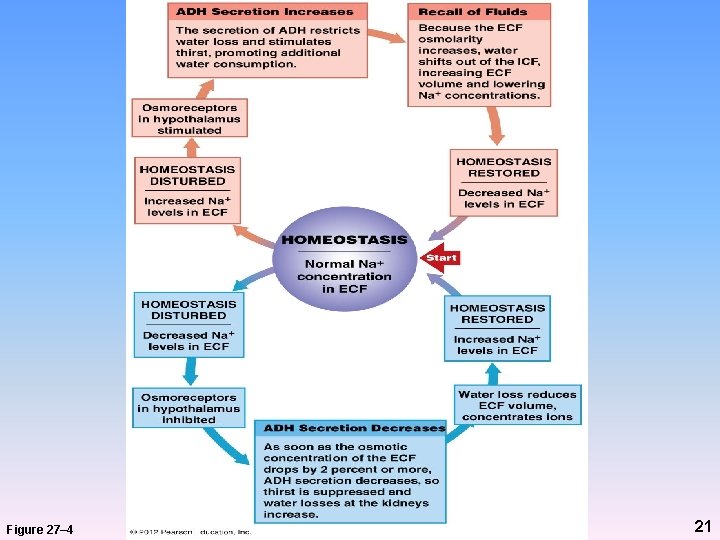
Figure 27– 4 21

Aldosterone • Mineralocorticoid secreted by adrenal cortex • Produced in response to: – Decreased Na+ or increased K+ – In blood arriving at: • Adrenal cortex • Kidney (renin-angiotensin system 22

Renin-Angiotensin System • Renin released in response to: – Decreased Na+ or increased K+ in renal circulation – Decreased plasma volume or BP at JGA – Decreased osmotic concentration at DCT • Renin angiotensin II activation in lung capillaries • Angiotensin II – Adrenal cortex increased aldosterone – Posterior pituitary ADH – Increased BP (hence it’s name) 23

Aldosterone • In DCT and collecting system of kidneys – Increased Na+ absorption (and associated Cl- and H 2 O absorption) – Increased K+ loss • Increased sensitivity of salt receptors on tongue crave salty foods 24

Natriuretic Peptides • Released by cardiac muscle cells stretched by: – Increased BP or blood volume • Oppose angiotensin II and cause diuresis – Decreased ADH increased H 2 O loss at kidneys – Decreased aldosterone increased Na+ and H 2 O loss at kidneys – Decreases thirst decreased H 2 O intake • Net result = decreased stretching of cells 25
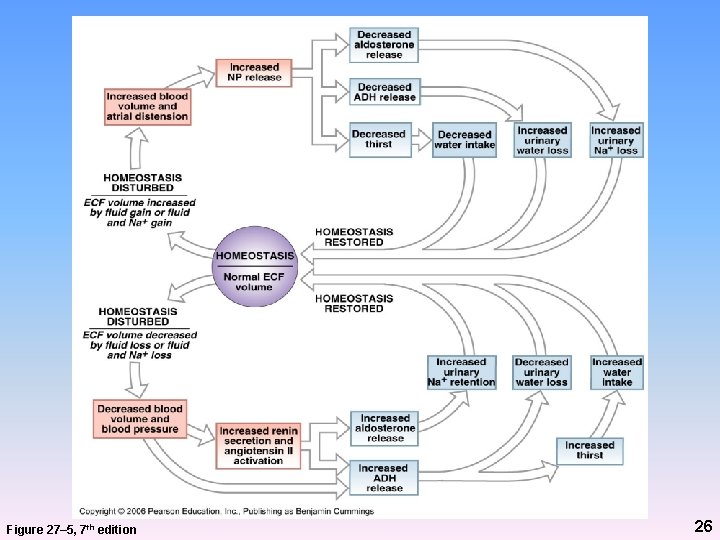
Figure 27– 5, 7 th edition 26

Fluid and Electrolyte Balance • When body loses water: – Plasma volume decreases – Electrolyte concentrations increase • When body loses electrolytes: – Electrolyte concentrations decrease – Water also lost 27

Disorders of Fluid and Electrolyte Balance • Dehydration = water depletion – Due to: • Inadequate water intake • Fluid loss, e. g. , vomiting, diarrhea • Inadequate ADH (hypothalamic/pituitary malfunction) – Leads to: • • Too high Na+ = hypernatremia Thirst, wrinkled skin Decreased blood volume and BP Fatal circulatory shock 28

Disorders of Fluid and Electrolyte Balance • Overhydration = water excess – Due to: • Excess water intake (>6 -8 L/24 hours) – Seen in hazing rituals (water torture) – Marathon runners/paddlers – Ravers on ecstasy who overcompensate for thirst • Chronic renal failure • Excess ADH – Leads to • Too low Na+ = hyponatremia • Increased blood volume and BP • CNS symptoms (water intoxication); can proceed to convulsions, coma, death 29

Disorders of Fluid and Electrolyte Balance • Hypokalemia – Too low K+ – Caused by diuretics, diet, chronic alkalosis (plasma p. H >7. 45) – Results in muscle weakness and paralysis • Hyperkalemia – Too high K+ – Caused by diuretics (that block Na+ reabsorption) – Renal failure, chronic acidosis (plasma p. H<7. 35) – Results in severe cardiac arrhythmias 30

Summary: Disorders of Electrolyte Balance • Most common problems with electrolyte balance – Caused by imbalance between gains/losses of Na+ • Uptake across digestive epithelium • Excretion in urine and perspiration • Problems with potassium balance – Less common, but more dangerous 31

• Today in class we will discuss: – Acid-base balance and • Three major buffer systems that balance p. H of ECF and ICF • Compensatory mechanisms involved in maintaining acidbase balance – Respiratory compensation – Renal compensation • Causes, effects, and the body’s response to acid-base disturbances that occur when p. H varies – Respiratory acid-base disorders » Respiratory acidosis » Respiratory alkalosis – Metabolic acid-base disorders » Metabolic acidosis » Metabolic alkalosis 32

Acid-Base Balance • Control of p. H – Acid-base balance = = production of H+ is precisely offset by H+ loss • Body generates acids (H+) during metabolic processes – Decrease p. H • Normal p. H of ECF = 7. 35 – 7. 45 – <7. 35 = acidosis (more common than alkalosis) – >7. 45 = alkalosis • <6. 8 or >7. 7 = lethal 33

Acid-Base Balance • Deviations outside normal range extremely dangerous – Disrupt cell membranes – Alter protein structure (remember hemoglobin? ) – Change activities of enzymes • Affects all body systems – Especially CNS and CVS 34

Acid-Base Balance • CNS and CVS especially sensitive to p. H fluctuations – Acidosis more lethal than alkalosis – CNS deteriorates coma death – Cardiac contractions grow weak and irregular heart failure – Peripheral vasodilation decreased BP and circulatory collapse 35

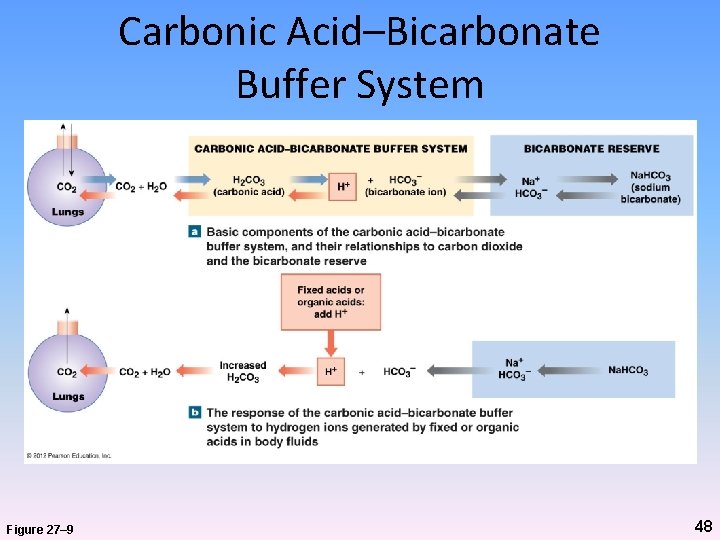
Acid-Base Balance • Carbonic acid (H 2 CO 3) – Most important factor affecting p. H of ECF • CO 2 + H 2 O H 2 CO 3 H+ + HCO 3 - 36
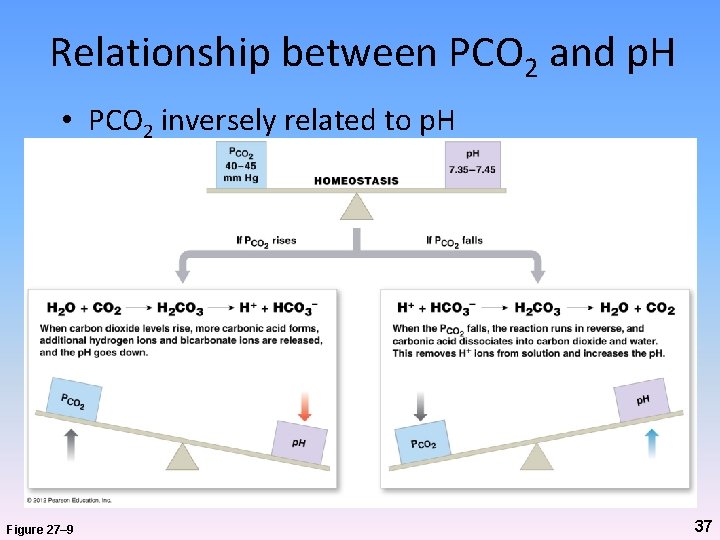
Relationship between PCO 2 and p. H • PCO 2 inversely related to p. H Figure 27– 9 37

Acid-Base Balance • H+ – Gained • At digestive tract • Through cellular metabolic activities – Eliminated • At kidneys by secretion of H+ into urine • At lungs by forming H 2 O and CO 2 from H+ and HCO 3 - – Sites of elimination far from sites of production – As H+ travels through body, must be neutralized to avoid tissue damage – Accomplished through buffer systems 38

Buffers • Compounds dissolved in body fluids • Stabilize p. H • Can provide or remove H+ 39

Buffer Systems in Body Fluids • Phosphate buffer system (H 2 PO 4 -) – In ICF and urine • Protein buffer systems – In ICF and ECF – Includes: • Hb buffer system (RBCs only) • Amino acid buffers (in proteins) • Plasma protein buffers (albumins, globulins…) • Carbonic acid-bicarbonate buffer system – Most important in ECF 40
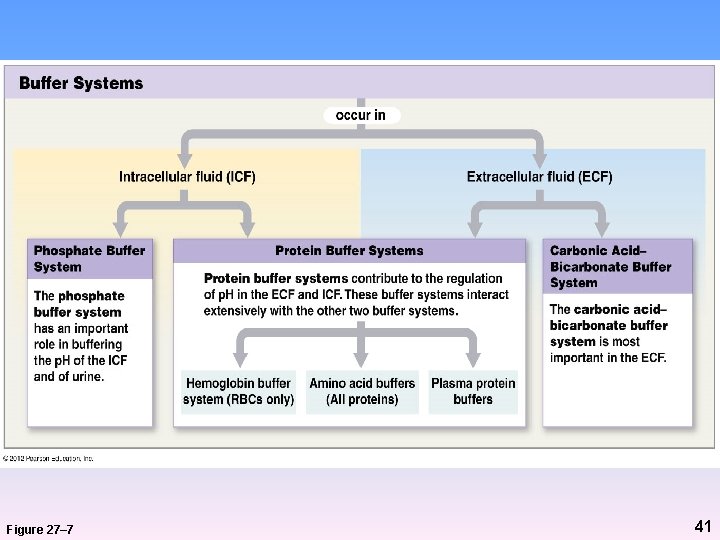
Figure 27– 7 41
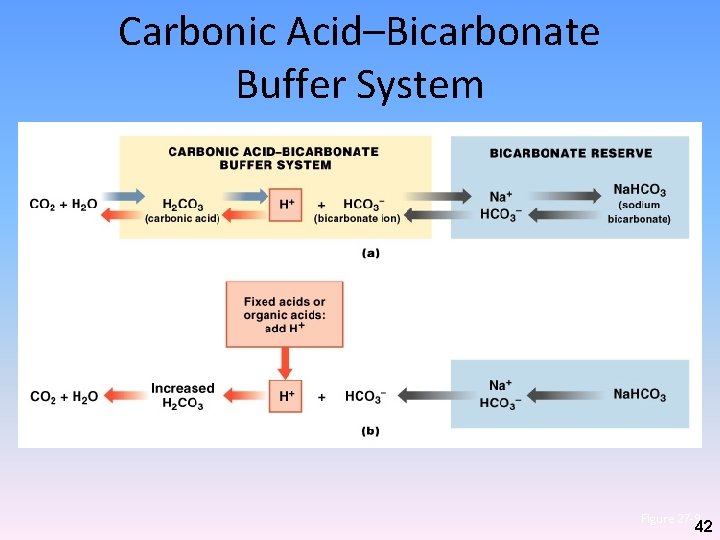
Carbonic Acid–Bicarbonate Buffer System Figure 27– 9 42

Maintenance of Acid-Base Balance • For homeostasis to be preserved: – H+ gains and losses must be balanced • Excess H+ must be: – Tied up by buffers • Temporary; H+ not eliminated, just not harmful – Permanently tied up in H 2 O molecules • Associated with CO 2 removal at lungs – Removed from body fluids • Through secretion at kidneys • Accomplished by: – Respiratory mechanisms – Renal mechanisms 43

Conditions Affecting Acid-Base Balance • Disorders affecting buffers, respiratory or renal function – Emphysema, renal failure • Cardiovascular conditions – Heart failure or hypotension – Can affect p. H, change glomerular filtration rates, respiratory efficiency • Conditions affecting CNS – Neural damage/disease that affects respiratory and cardiovascular reflexes that regulate p. H 44

Disturbances of Acid-Base Balance • Serious abnormalities have an: – Acute (initial) phase • p. H moves rapidly out of normal range – Compensated phase • If condition persists • Physiological adjustments move p. H back into normal range • Cannot be completed unless underlying problem corrected • Types of compensation – Respiratory – Renal 45

Respiratory Compensation • Changes respiratory rate – Increasing/decreasing respiratory rate changes p. H by lowering/raising PCO 2 – Helps stabilize p. H of ECF • Occurs whenever p. H moves outside normal limits • Has a direct effect on carbonic acidbicarbonate buffer system 46
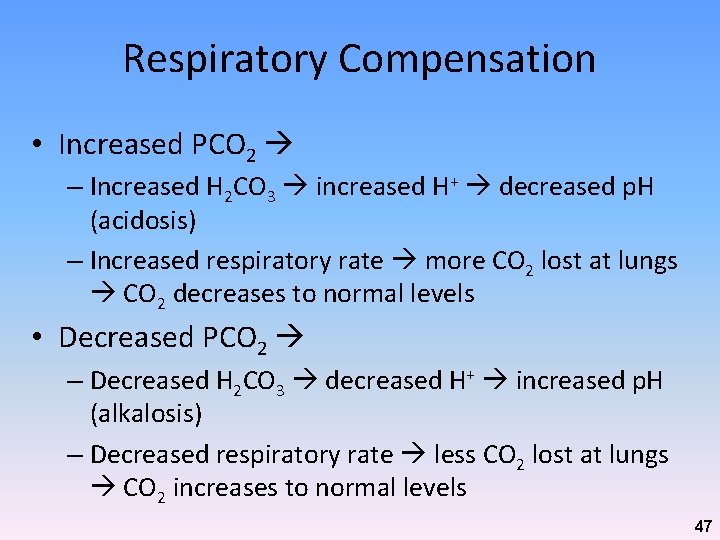
Respiratory Compensation • Increased PCO 2 – Increased H 2 CO 3 increased H+ decreased p. H (acidosis) – Increased respiratory rate more CO 2 lost at lungs CO 2 decreases to normal levels • Decreased PCO 2 – Decreased H 2 CO 3 decreased H+ increased p. H (alkalosis) – Decreased respiratory rate less CO 2 lost at lungs CO 2 increases to normal levels 47
Carbonic Acid–Bicarbonate Buffer System Figure 27– 9 48

Renal Compensation • Changes renal rates of H+ and HCO 3– Secretion – Reabsorption • In response to changes in plasma p. H – Increased H+ or decreased HCO 3 - • Decreased p. H (acidosis) more H+ secreted and/or less HCO 3 - reabsorbed – Decreased H+ or increased HCO 3 - • Increased p. H (alkalosis) less H+ secreted and/or more HCO 3 - reabsorbed 49
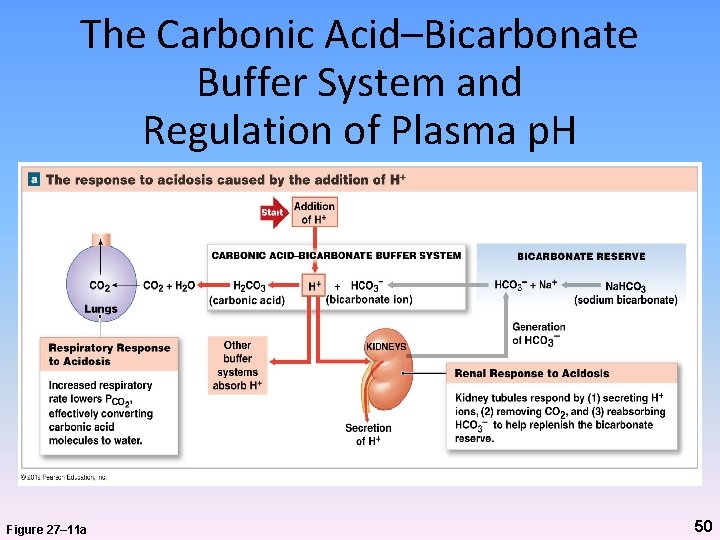
The Carbonic Acid–Bicarbonate Buffer System and Regulation of Plasma p. H Figure 27– 11 a 50
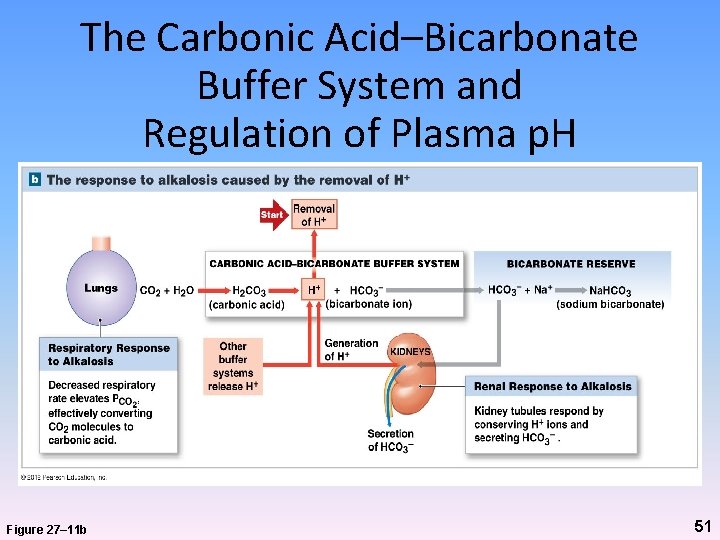
The Carbonic Acid–Bicarbonate Buffer System and Regulation of Plasma p. H Figure 27– 11 b 51

Disturbances of Acid-Base Balance • Conditions named for: – Uncompensated or • Compensated – Primary source of problem • Respiratory or metabolic • Mixed (both) – Primary effect • Acidosis or alkalosis • e. g. , – Compensated or uncompensated • Respiratory acidosis or alkalosis • Metabolis acidosis or alkalosis 52

Respiratory Acid-Base Disorders • Result from imbalance between: – CO 2 generated in peripheral tissues (ECF) – CO 2 excreted at lungs • Cause abnormal CO 2 levels in ECF • Respiratory – Acidosis – Alkalosis 53

Respiratory Acidosis • Most common challenge to acid-base equilibrium • Primary sign is hypercapnia (increased PCO 2) • Develops when respiratory system cannot eliminate all CO 2 generated by peripheral tissues • Usual cause is hypoventilation • Acute situation may be immediate, life-threatening condition – Requires bronchodilation or mechanical breathing assistance (ventilator) • p. H can get as low as 7. 0 54
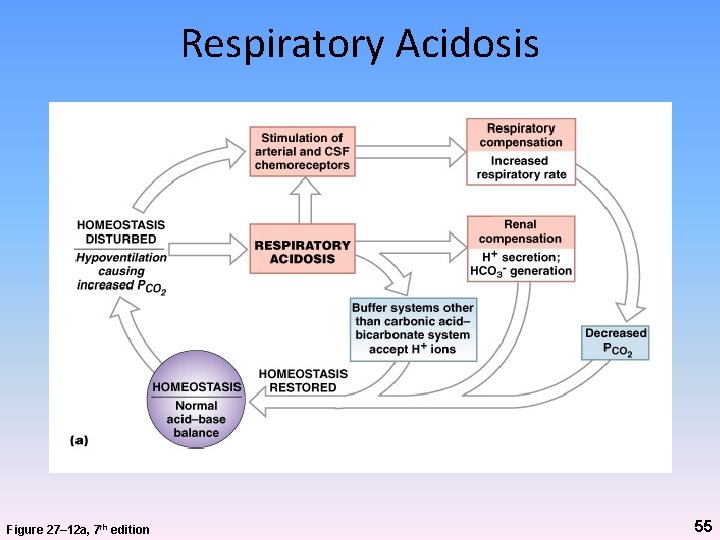
Respiratory Acidosis Figure 27– 12 a, 7 th edition 55

Respiratory Alkalosis • Relatively uncommon • Primary sign is high p. H • Develops when increased respiratory activity (hyperventilation) lowers plasma PCO 2 to below normal levels (hypocapnia) • Seldom of clinical significance • p. H can get as high as 7. 8 – 8. 0 56
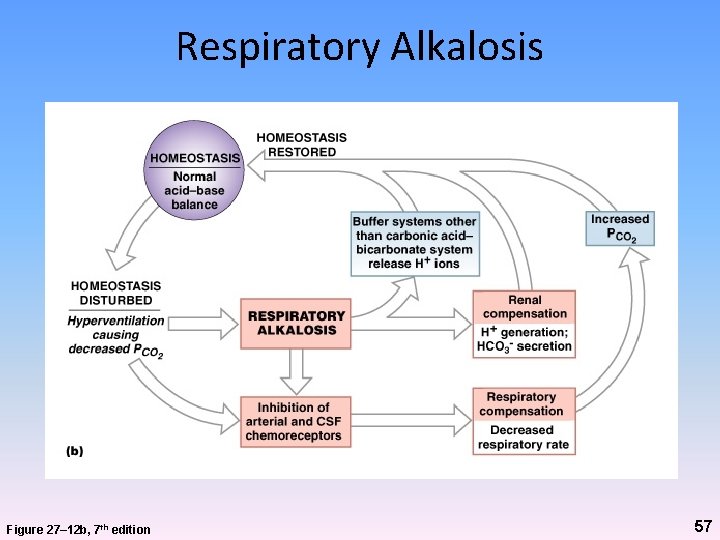
Respiratory Alkalosis Figure 27– 12 b, 7 th edition 57

Metabolic Acid-Base Disorders • Result from: – Production of acids during metabolic processes – Conditions that affect concentration of HCO 3 - in ECF • Metabolic – Acidosis – Alkalosis 58

Metabolic Acidosis • Results from: – Production of large numbers of acids • H+ overloads buffer systems – Inability to excrete H+ at kidneys – Severe HCO 3 - loss 59

Metabolic Acidosis • Production of large number of acids – Lactic acidosis from anaerobic respiration • After strenuous exercise • From prolonged tissue hypoxia (O 2 starvation) – Ketoacidosis from generation of ketone bodies during metabolism • When peripheral tissues cannot obtain adequate glucose from bloodstream and begin metabolizing lipids and ketone bodies), e. g. , – Starvation – Complication of poorly controlled diabetes mellitus 60

Metabolic Acidosis • Inability to excrete H+ at kidneys – With severe kidney damage (glomerulonephritis) – Caused by diuretics that interfere with H+ secretion into urine • Severe HCO 3 - loss – From chronic diarrhea – Loss interferes with buffer system ability to remove H+ 61
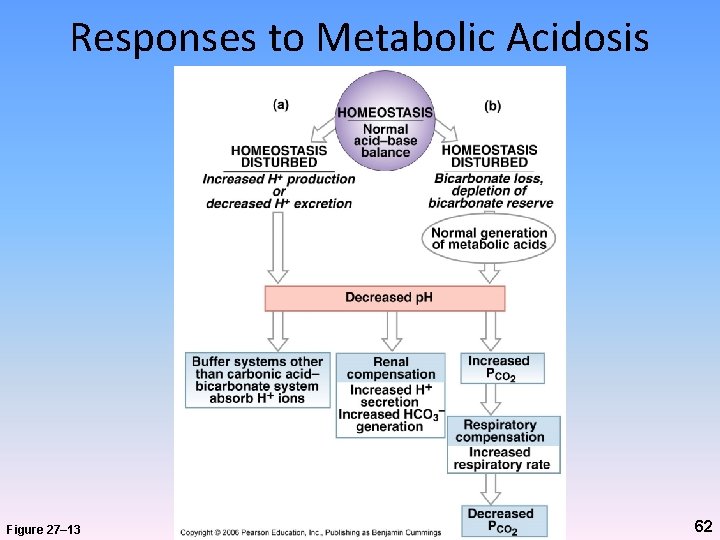
Responses to Metabolic Acidosis Figure 27– 13 62

Metabolic Alkalosis • Relatively rare • Occurs after repeated vomiting – Stomach continues to generate HCl to replace lost acids – Is associated with increased HCO 3 - in ECF – HC 03 - + H+ H 2 CO 3 • Reduces H+ alkalosis 63
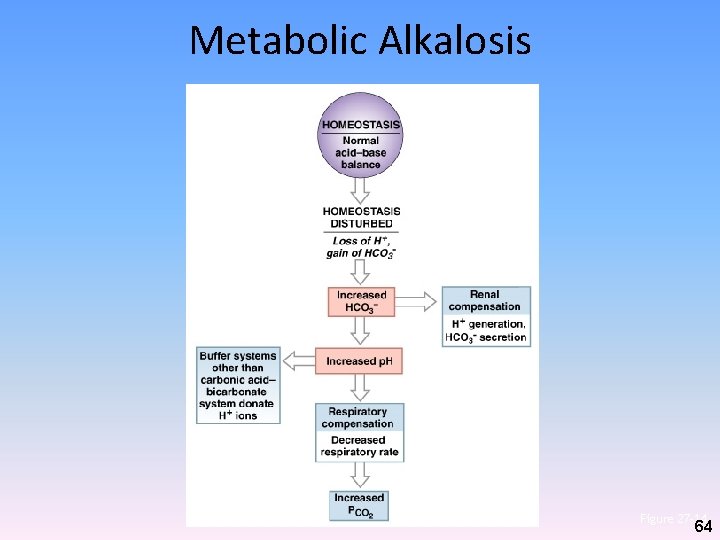
Metabolic Alkalosis Figure 27– 14 64

Detection of Acidosis and Alkalosis • Includes blood tests for: – p. H – PCO 2 – HCO 3 - • Recognition of acidosis or alkalosis • Classification as respiratory or metabolic 65
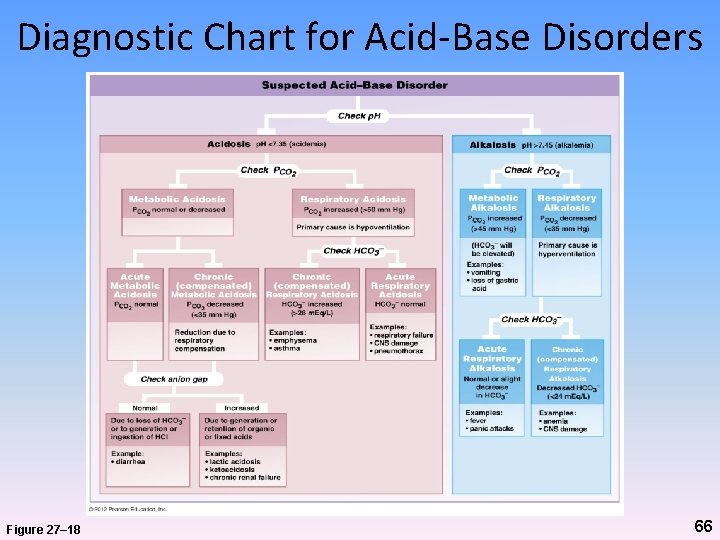
Diagnostic Chart for Acid-Base Disorders Figure 27– 18 66
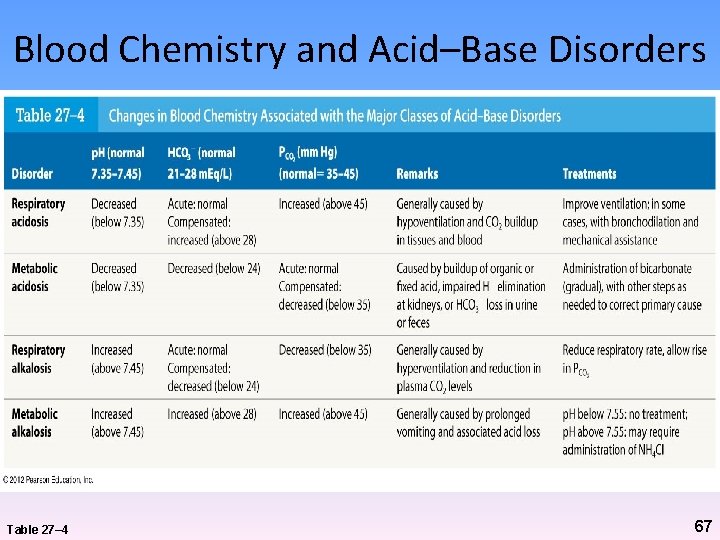
Blood Chemistry and Acid–Base Disorders Table 27– 4 67
- Slides: 67