Chapter 26 Fluid Electrolyte and Acid Base Balance

Chapter 26 Fluid, Electrolyte, and Acid. Base Balance © Annie Leibovitz/Contact Press Images © 2016 Pearson Education, Inc. Power. Point® Lecture Slides prepared by Karen Dunbar Kareiva Ivy Tech Community College

26. 1 Body Fluid Compartments Body Water Content • Infants are 73% or more water (low body fat, low bone mass) • Adult males: ~60% water • Adult females: ~50% water (higher fat content, less skeletal muscle mass) – Adipose tissue is least hydrated of all – Total body water in adults averages ~40 L • Water content declines to ~45% in old age © 2016 Pearson Education, Inc.
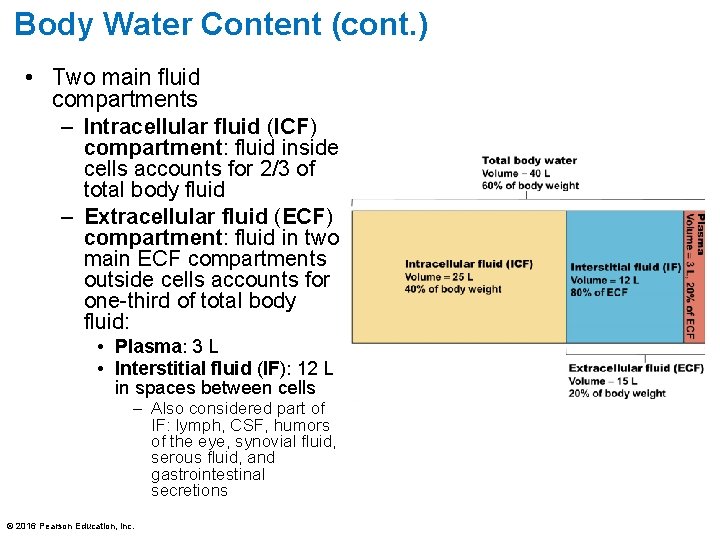
Body Water Content (cont. ) • Two main fluid compartments – Intracellular fluid (ICF) compartment: fluid inside cells accounts for 2/3 of total body fluid – Extracellular fluid (ECF) compartment: fluid in two main ECF compartments outside cells accounts for one-third of total body fluid: • Plasma: 3 L • Interstitial fluid (IF): 12 L in spaces between cells – Also considered part of IF: lymph, CSF, humors of the eye, synovial fluid, serous fluid, and gastrointestinal secretions © 2016 Pearson Education, Inc.

Composition of Body Fluids • Water is the universal solvent • Solutes are substances dissolved in water • Solutes are classified as nonelectrolytes and electrolytes • Electrolytes and nonelectrolytes – Nonelectrolytes: most are organic molecules • Do not dissociate in water • Examples: glucose, lipids, creatinine, and urea – No charged particles are created © 2016 Pearson Education, Inc.

Composition of Body Fluids (cont. ) – Electrolytes • Dissociate into ions in water – Examples: inorganic salts, all acids and bases, some proteins • Ions conduct electrical current • Greater osmotic power than nonelectrolytes – Greater ability to cause fluid shifts due to ability to dissociate into two or more ions – Na. Cl Na+ + Cl− (electrolyte; 2 particles) – Mg. Cl 2 Mg 2+ + 2 Cl− (electrolyte; 3 particles) – glucose (nonelectrolyte; 1 particle) © 2016 Pearson Education, Inc.
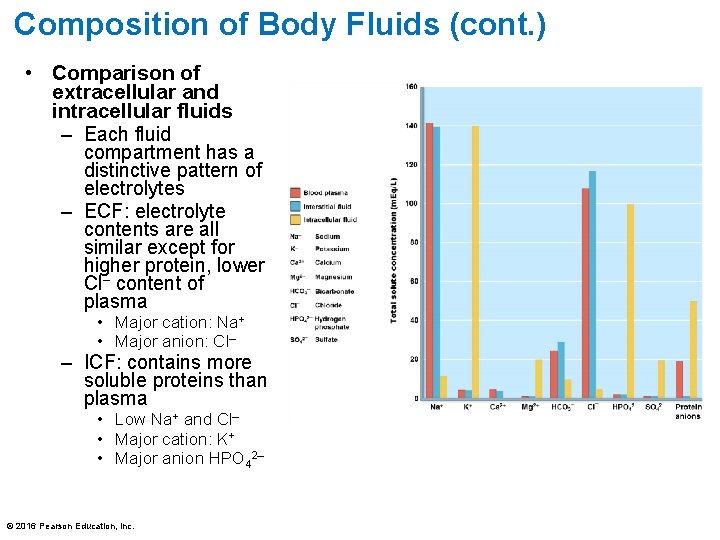
Composition of Body Fluids (cont. ) • Comparison of extracellular and intracellular fluids – Each fluid compartment has a distinctive pattern of electrolytes – ECF: electrolyte contents are all similar except for higher protein, lower Cl– content of plasma • Major cation: Na+ • Major anion: Cl– – ICF: contains more soluble proteins than plasma • Low Na+ and Cl– • Major cation: K+ • Major anion HPO 42– © 2016 Pearson Education, Inc.
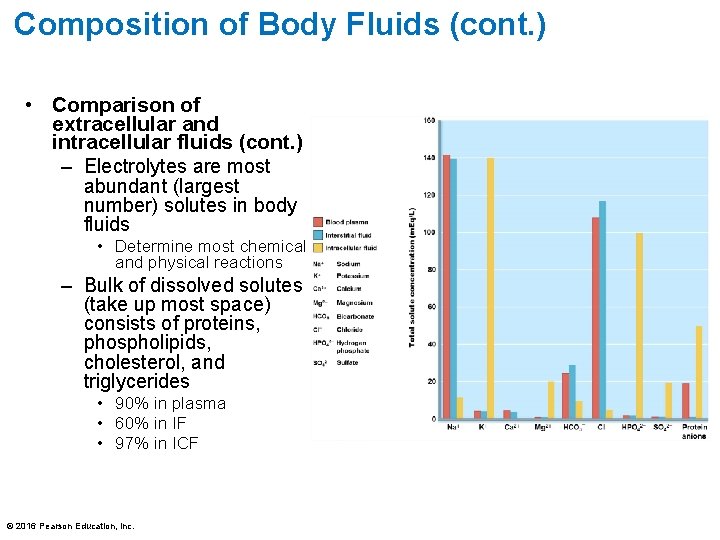
Composition of Body Fluids (cont. ) • Comparison of extracellular and intracellular fluids (cont. ) – Electrolytes are most abundant (largest number) solutes in body fluids • Determine most chemical and physical reactions – Bulk of dissolved solutes (take up most space) consists of proteins, phospholipids, cholesterol, and triglycerides • 90% in plasma • 60% in IF • 97% in ICF © 2016 Pearson Education, Inc.

Fluid Movement among Compartments • Osmotic and hydrostatic pressures regulate continuous exchange and mixing of fluids – Water moves freely along osmotic gradients – All body fluid osmolality is almost always equal – Change in solute concentration of any compartment leads to net water flow • ECF osmolality water leaves cell • ECF osmolality water enters cell © 2016 Pearson Education, Inc.

Fluid Movement among Compartments (cont. ) • Exchanges between plasma and IF occur across capillary walls – Fluid leaks from arteriolar end of capillary, reabsorbed at venule end – Lymphatics pick up remaining fluid and return it to blood • Exchanges between IF and ICF occur across cell membrane – Two-way osmotic flow of water – Ions move selectively into or out of cell – Nutrients, wastes, gases have unidirectional flow © 2016 Pearson Education, Inc.
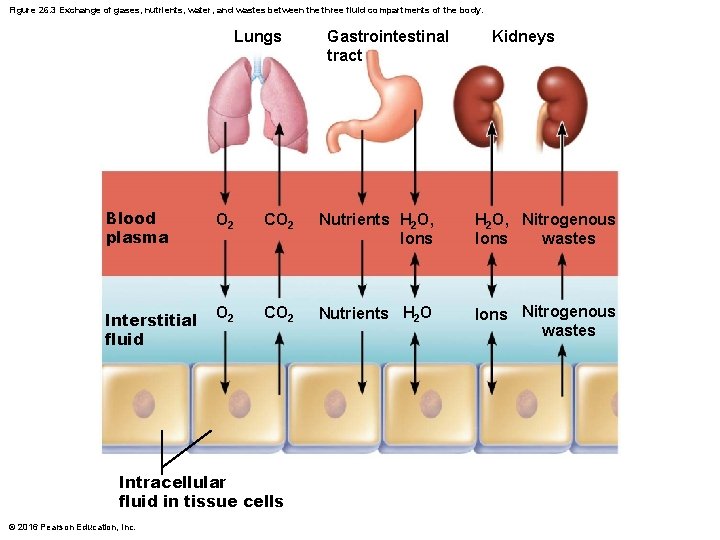
Figure 26. 3 Exchange of gases, nutrients, water, and wastes between the three fluid compartments of the body. Lungs Gastrointestinal tract Kidneys Blood plasma O 2 CO 2 Nutrients H 2 O, Ions H 2 O, Nitrogenous wastes Ions Interstitial fluid O 2 CO 2 Nutrients H 2 O Ions Nitrogenous wastes Intracellular fluid in tissue cells © 2016 Pearson Education, Inc.

26. 2 Water Balance and ECF Osmolality • Water intake must equal water output: ~2500 ml/day • Water intake: most water is taken in via ingested foods and beverages, but small amount from metabolism – Metabolic water (water of oxidation): water produced by cellular metabolism • Water output: urine (60%), insensible water loss (lost through skin and lungs), perspiration, and feces © 2016 Pearson Education, Inc.

26. 2 Water Balance and ECF Osmolality • Osmolality is maintained around 280– 300 m. Osm • Rise in osmolality – Stimulates thirst – Causes ADH release • Decrease in osmolality – Causes inhibition – Causes ADH inhibition © 2016 Pearson Education, Inc.
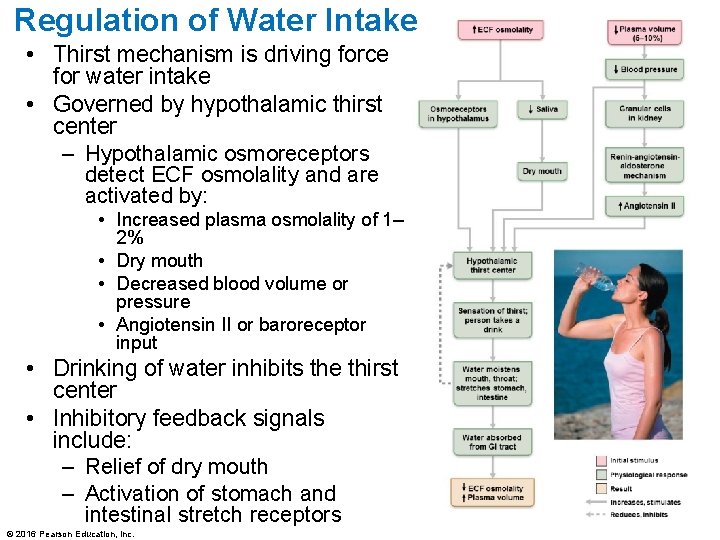
Regulation of Water Intake • Thirst mechanism is driving force for water intake • Governed by hypothalamic thirst center – Hypothalamic osmoreceptors detect ECF osmolality and are activated by: • Increased plasma osmolality of 1– 2% • Dry mouth • Decreased blood volume or pressure • Angiotensin II or baroreceptor input • Drinking of water inhibits the thirst center • Inhibitory feedback signals include: – Relief of dry mouth – Activation of stomach and intestinal stretch receptors © 2016 Pearson Education, Inc.

Regulation of Water Output • Obligatory water losses: explain why we cannot live without water very long – Include: • Insensible water loss from lungs, skin, and feces • Sensible water loss from urine to excrete wastes – Minimum daily loss of 500 ml • Volume of urine excreted and solute concentration also depend on fluid intake, diet, and water loss via other avenues © 2016 Pearson Education, Inc.
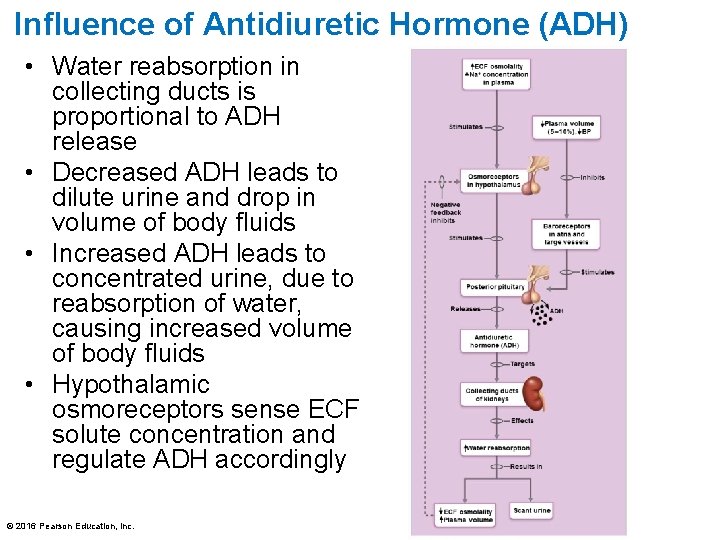
Influence of Antidiuretic Hormone (ADH) • Water reabsorption in collecting ducts is proportional to ADH release • Decreased ADH leads to dilute urine and drop in volume of body fluids • Increased ADH leads to concentrated urine, due to reabsorption of water, causing increased volume of body fluids • Hypothalamic osmoreceptors sense ECF solute concentration and regulate ADH accordingly © 2016 Pearson Education, Inc.
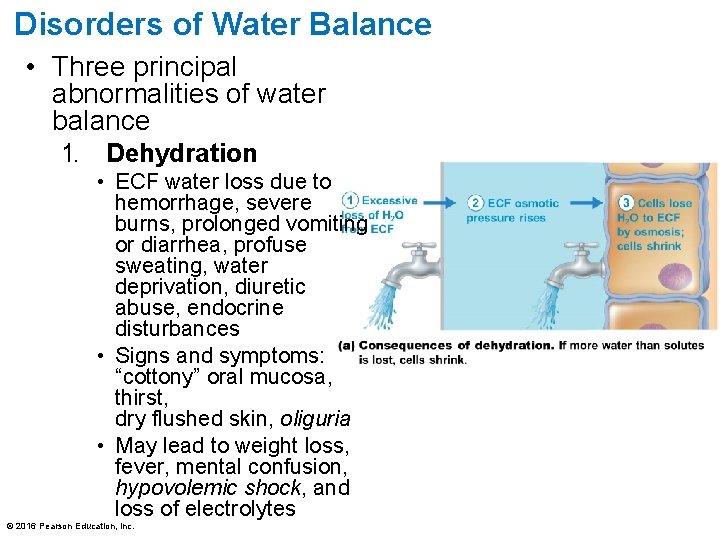
Disorders of Water Balance • Three principal abnormalities of water balance 1. Dehydration • ECF water loss due to hemorrhage, severe burns, prolonged vomiting or diarrhea, profuse sweating, water deprivation, diuretic abuse, endocrine disturbances • Signs and symptoms: “cottony” oral mucosa, thirst, dry flushed skin, oliguria • May lead to weight loss, fever, mental confusion, hypovolemic shock, and loss of electrolytes © 2016 Pearson Education, Inc.
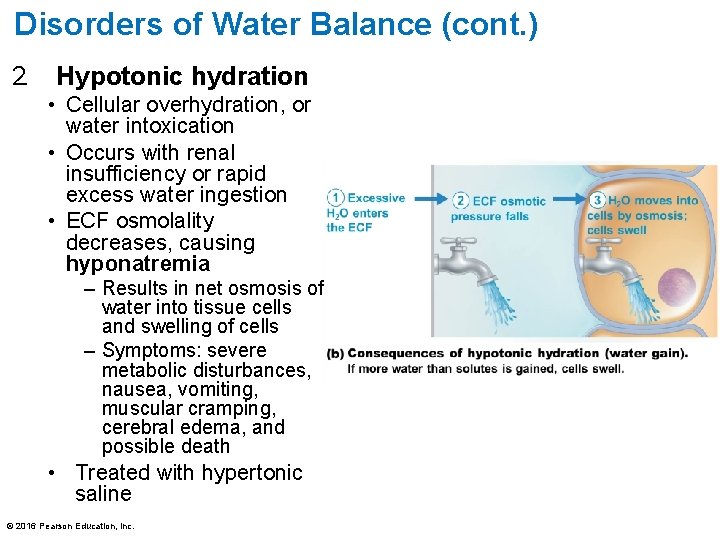
Disorders of Water Balance (cont. ) 2. Hypotonic hydration • Cellular overhydration, or water intoxication • Occurs with renal insufficiency or rapid excess water ingestion • ECF osmolality decreases, causing hyponatremia – Results in net osmosis of water into tissue cells and swelling of cells – Symptoms: severe metabolic disturbances, nausea, vomiting, muscular cramping, cerebral edema, and possible death • Treated with hypertonic saline © 2016 Pearson Education, Inc.
Disorders of Water Balance (cont. ) 3. Edema • Atypical accumulation of IF, resulting in tissue swelling (not cell swelling) – Only volume of IF is increased, not of other compartments • Can impair tissue function by increasing distance for diffusion of oxygen and nutrients from blood into cells • Could be caused by increased fluid flow out of blood or decreased return of fluid to blood © 2016 Pearson Education, Inc.

26. 3 Electrolyte Balance • Electrolyte balance usually refers only to salt balance even though electrolytes also include acids, bases, and some proteins • Salts control fluid movements, provide minerals for excitability, secretory activity, and membrane permeability • Salts enter body by ingestion and metabolism and are lost via perspiration, feces, urine, vomit © 2016 Pearson Education, Inc.
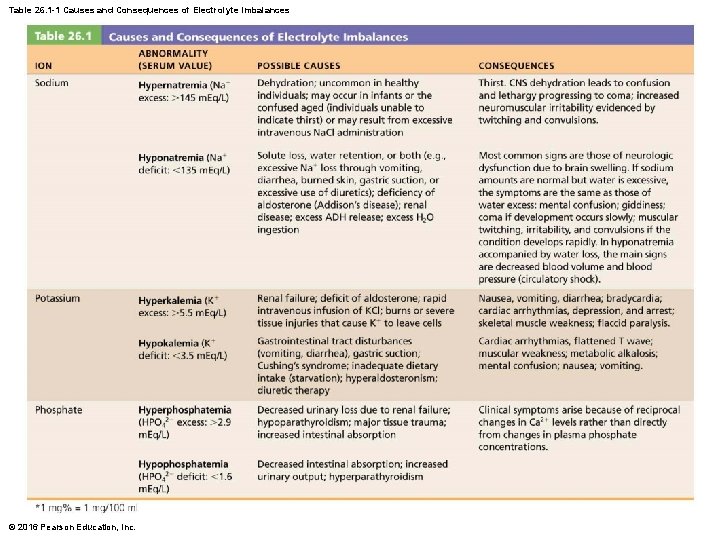
Table 26. 1 -1 Causes and Consequences of Electrolyte Imbalances © 2016 Pearson Education, Inc.
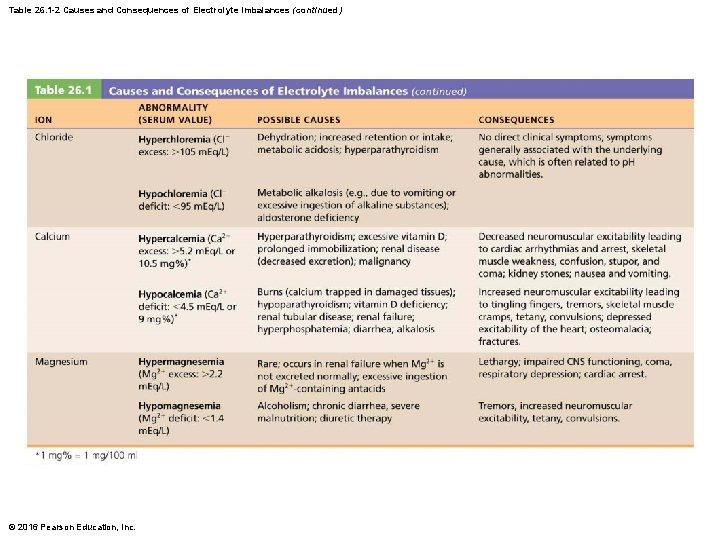
Table 26. 1 -2 Causes and Consequences of Electrolyte Imbalances (continued) © 2016 Pearson Education, Inc.

Central Role of Sodium in Fluid and Electrolyte Balance • Sodium is most abundant cation in ECF – Sodium salts in ECF contribute 280 m. Osm of total 300 m. Osm ECF solute concentration • Only cation exerting significant osmotic pressure – Controls ECF volume and water distribution because water follows salt – Changes in Na+ levels affects plasma volume, blood pressure, and ECF and IF volumes © 2016 Pearson Education, Inc.

Central Role of Sodium in Fluid and Electrolyte Balance (cont. ) • Na+ that leaks into cells is pumped out against its electrochemical gradient • Na+ moves back and forth between ECF and body secretions (example: digestive secretions) • Renal acid-base control mechanisms are coupled to sodium ion transport © 2016 Pearson Education, Inc.

Central Role of Sodium in Fluid and Electrolyte Balance (cont. ) • Sodium concentration versus sodium content – Concentration of Na+ • Determines osmolality of ECF and influences excitability of neurons and muscles • Remains stable because of water shifts out of or into ICF – Content of Na+ • Total body content determines ECF volume and therefore blood pressure © 2016 Pearson Education, Inc.

Regulation of Sodium Balance • There are no known receptors that monitor Na+ levels in body fluids • Na+-water balance is linked to blood pressure and blood volume control mechanisms • Changes in blood pressure or volume trigger neural and hormonal controls to regulate Na+ content © 2016 Pearson Education, Inc.
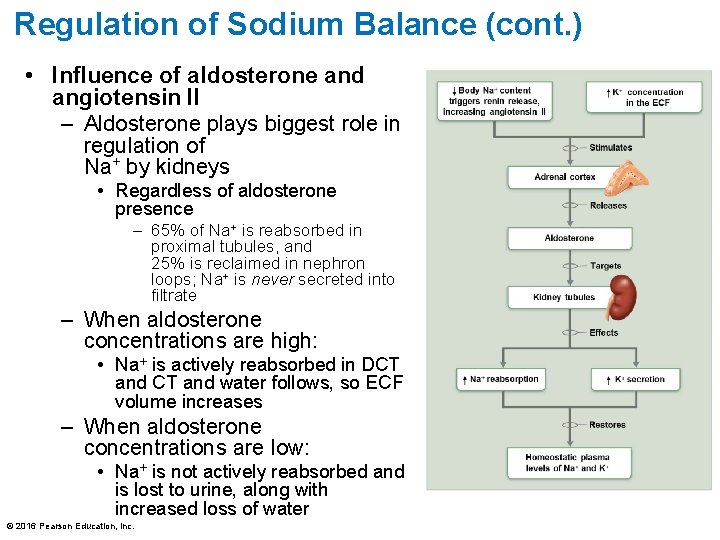
Regulation of Sodium Balance (cont. ) • Influence of aldosterone and angiotensin II – Aldosterone plays biggest role in regulation of Na+ by kidneys • Regardless of aldosterone presence – 65% of Na+ is reabsorbed in proximal tubules, and 25% is reclaimed in nephron loops; Na+ is never secreted into filtrate – When aldosterone concentrations are high: • Na+ is actively reabsorbed in DCT and water follows, so ECF volume increases – When aldosterone concentrations are low: • Na+ is not actively reabsorbed and is lost to urine, along with increased loss of water © 2016 Pearson Education, Inc.
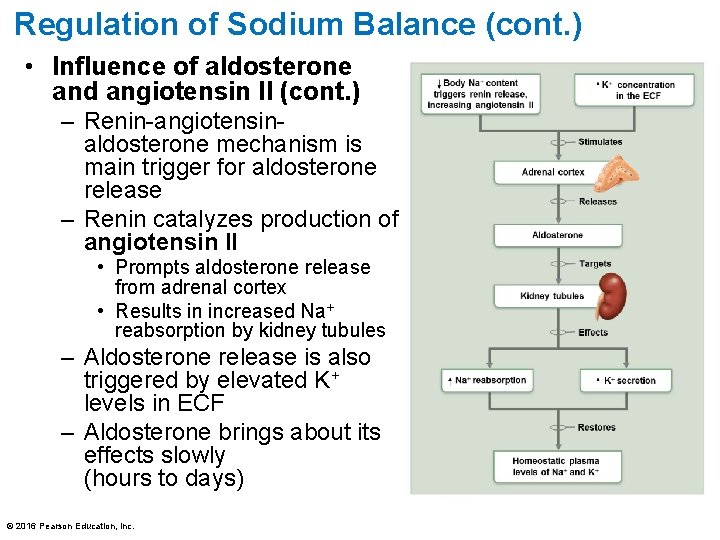
Regulation of Sodium Balance (cont. ) • Influence of aldosterone and angiotensin II (cont. ) – Renin-angiotensinaldosterone mechanism is main trigger for aldosterone release – Renin catalyzes production of angiotensin II • Prompts aldosterone release from adrenal cortex • Results in increased Na+ reabsorption by kidney tubules – Aldosterone release is also triggered by elevated K+ levels in ECF – Aldosterone brings about its effects slowly (hours to days) © 2016 Pearson Education, Inc.
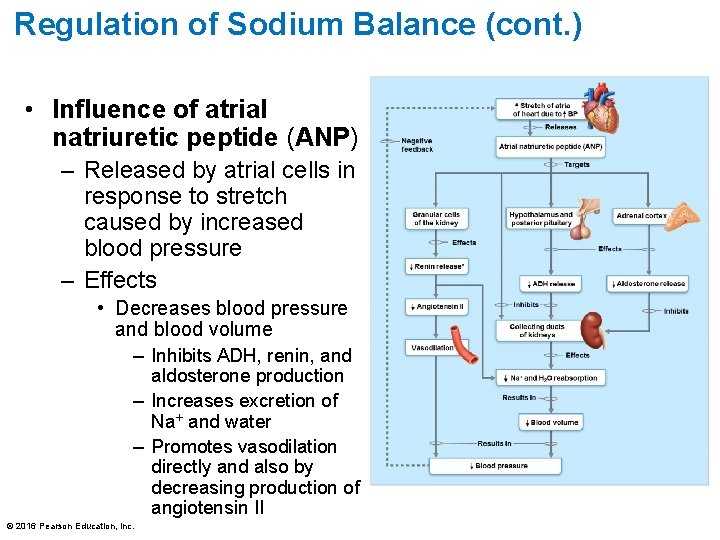
Regulation of Sodium Balance (cont. ) • Influence of atrial natriuretic peptide (ANP) – Released by atrial cells in response to stretch caused by increased blood pressure – Effects • Decreases blood pressure and blood volume – Inhibits ADH, renin, and aldosterone production – Increases excretion of Na+ and water – Promotes vasodilation directly and also by decreasing production of angiotensin II © 2016 Pearson Education, Inc.
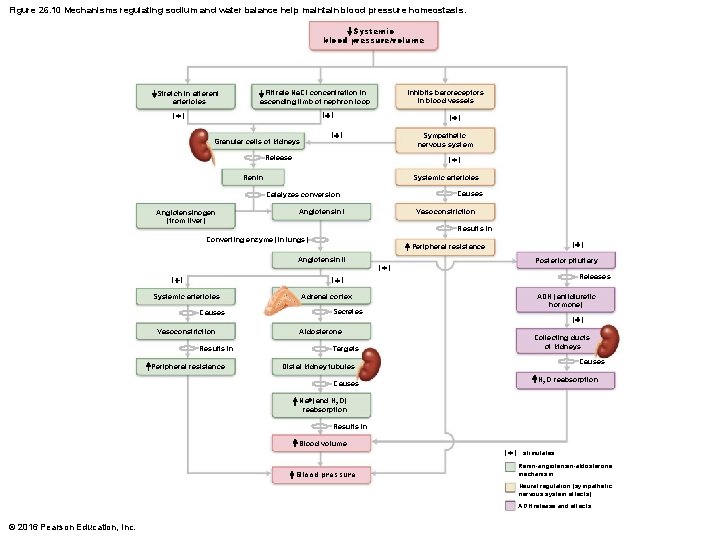
Figure 26. 10 Mechanisms regulating sodium and water balance help maintain blood pressure homeostasis. Systemic blood pressure/volume Stretch in afferent arterioles Inhibits baroreceptors in blood vessels Filtrate Na. Cl concentration in ascending limb of nephron loop (+) (+) Granular cells of kidneys Sympathetic nervous system Release (+) Renin Systemic arterioles Causes Catalyzes conversion Angiotensinogen (from liver) Vasoconstriction Angiotensin I Results in Converting enzyme (in lungs) Angiotensin II (+) Peripheral resistance (+) Posterior pituitary Releases (+) Systemic arterioles Causes Vasoconstriction Results in Peripheral resistance Adrenal cortex Secretes Aldosterone Targets ADH (antidiuretic hormone) (+) Collecting ducts of kidneys Causes Distal kidney tubules Causes H 2 O reabsorption Na+ (and H 2 O) reabsorption Results in Blood volume ( + ) stimulates Blood pressure Renin-angiotensin-aldosterone mechanism Neural regulation (sympathetic nervous system effects) ADH release and effects © 2016 Pearson Education, Inc.

Regulation of Potassium Balance • Importance of potassium – Affects resting membrane potential (RMP) in neurons and muscle cells (especially cardiac muscle) • Increases in ECF [K+] (hyperkalemia) cause decreased RMP, causing depolarization, followed by reduced excitability • Decreases in ECF [K+] (hypokalemia) cause hyperpolarization and nonresponsiveness – Disruption in [K+] (hyper- or hypokalemia) in heart can interfere with electrical conduction, leading to sudden death © 2016 Pearson Education, Inc.

Regulation of Potassium Balance (cont. ) • K+ is also part of body’s buffer system • H+ shifts in and out of cells in opposite direction of K+ to maintain cation balance, so: – ECF K+ levels rise with acidosis – ECF K+ levels fall with alkalosis • Interferes with activity of excitable cells © 2016 Pearson Education, Inc.

Regulation of Potassium Balance (cont. ) • Influence of plasma potassium concentration – Most important factor affecting K+ secretion is its concentration in ECF – High K+ diet leads to increased K+ content of ECF • K+ entry into principal cells leads to increased K+ secretion – Low K+ diet or accelerated K+ loss reduces its secretion and promotes its limited reabsorption © 2016 Pearson Education, Inc.

Regulation of Potassium Balance (cont. ) • Influence of aldosterone – Aldosterone stimulates K+ secretion (and Na+ reabsorption) by principal cells – Adrenal cortical cells are directly sensitive to K+ content of ECF • Increased K+ in adrenal cortex causes release of aldosterone, which increases K+ secretion – Means that K+ controls its own ECF concentrations via feedback regulation of aldosterone release © 2016 Pearson Education, Inc.
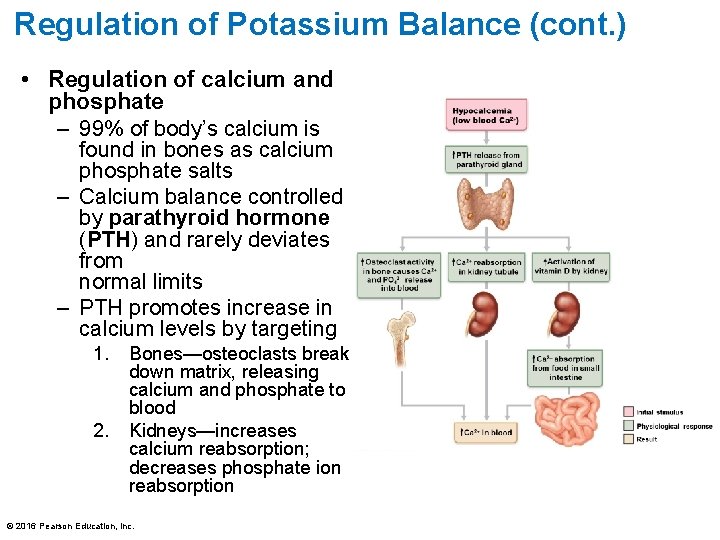
Regulation of Potassium Balance (cont. ) • Regulation of calcium and phosphate – 99% of body’s calcium is found in bones as calcium phosphate salts – Calcium balance controlled by parathyroid hormone (PTH) and rarely deviates from normal limits – PTH promotes increase in calcium levels by targeting 1. 2. Bones—osteoclasts break down matrix, releasing calcium and phosphate to blood Kidneys—increases calcium reabsorption; decreases phosphate ion reabsorption © 2016 Pearson Education, Inc.
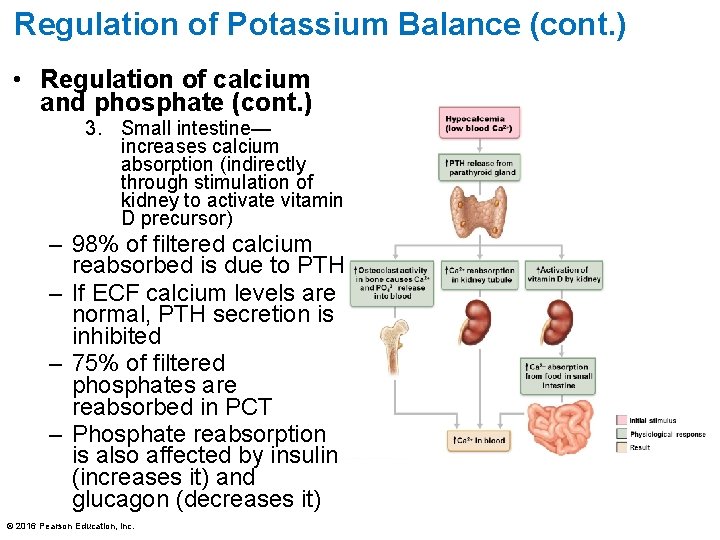
Regulation of Potassium Balance (cont. ) • Regulation of calcium and phosphate (cont. ) 3. Small intestine— increases calcium absorption (indirectly through stimulation of kidney to activate vitamin D precursor) – 98% of filtered calcium reabsorbed is due to PTH – If ECF calcium levels are normal, PTH secretion is inhibited – 75% of filtered phosphates are reabsorbed in PCT – Phosphate reabsorption is also affected by insulin (increases it) and glucagon (decreases it) © 2016 Pearson Education, Inc.

Clinical – Homeostatic Imbalance 26. 4 • Ca 2+ in ECF is important for: – Blood clotting – Cell membrane permeability – Secretory activities – Neuromuscular excitability: most important • Hypocalcemia: increases neuromuscular excitability and can lead to muscle tetany • Hypercalcemia: inhibits neurons and muscle cells and may cause heart arrhythmias © 2016 Pearson Education, Inc.

26. 4 Acid-Base Balance • p. H affects all functional proteins and biochemical reactions, so it is closely regulated by the body • Normal p. H of body fluids – Arterial blood: p. H 7. 4 – Venous blood and interstitial fluid: p. H 7. 35 – ICF: p. H 7. 0 • Alkalosis or alkalemia: arterial p. H >7. 45 • Acidosis or acidemia: arterial p. H <7. 35 © 2016 Pearson Education, Inc.

26. 4 Acid-Base Balance • Small amounts of acidic substances enter body in food but most H+ is produced as a by-product of metabolism: – Phosphorus-containing protein breakdown releases phosphoric acid into ECF – Lactic acid from anaerobic respiration of glucose – Fatty acids and ketone bodies from fat metabolism – H+ is liberated when CO 2 is converted to HCO 3– in blood © 2016 Pearson Education, Inc.

26. 4 Acid-Base Balance • Concentration of hydrogen ions is regulated sequentially by three mechanisms 1. Chemical buffer systems • Rapid, first line of defense 2. Brain stem respiratory centers • Acts within 1– 3 minutes 3. Renal mechanisms • Most potent, but require hours to days to effect p. H changes © 2016 Pearson Education, Inc.
- Slides: 39