Chapter 25 The Body Fluid Compartments Extracellular and

Chapter 25: The Body Fluid Compartments: Extracellular and Intracellular Fluids; Interstitial Fluid and Edema Slides by John E. Hall, Ph. D.
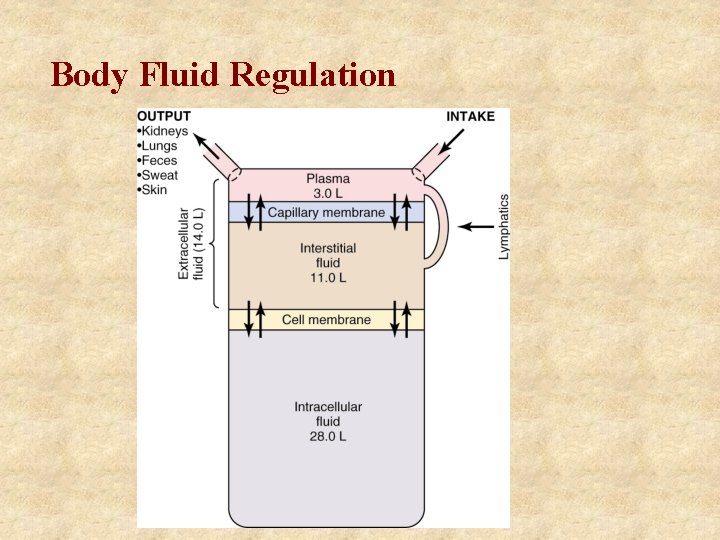
Body Fluid Regulation

Fluid Balance (ml/day) – 70 kg Adult Intake Fluids ingested From metabolism Output Insensible - skin Insensible - lungs Sweat Feces Urine Normal High Salt Diet 2100 2300 4100 200 4300 350 100 1400 2300 350 100 3400 4300

Balance Concept Balance is necessary, in the long-term, to maintain life. Fluid Loss = Fluid Intake Electrolyte Loss = Electrolyte Intake Fluid Intake: regulated by thirst mechanism, habits Electrolyte intake: governed by dietary habits Fluid Output: regulated mainly by kidneys Electrolyte output: regulated mainly by kidneys
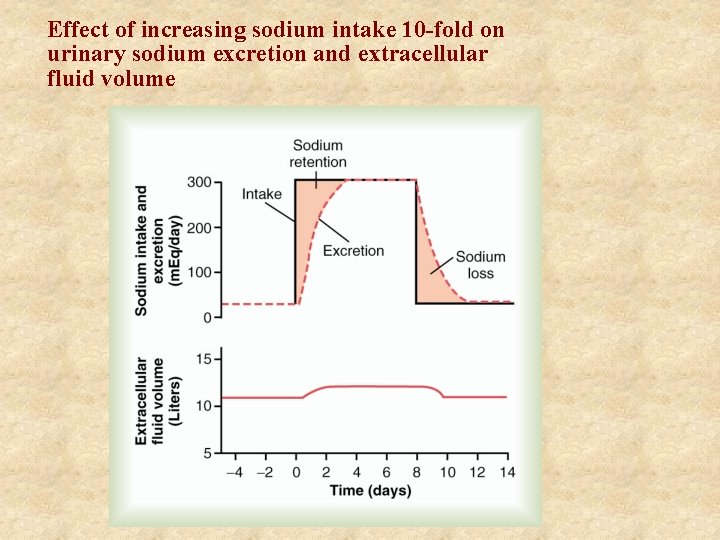
Effect of increasing sodium intake 10 -fold on urinary sodium excretion and extracellular fluid volume

Fluid Balance (ml/day) – 70 kg Adult Normal Intake Fluids ingested From metabolism Output Insensible - skin Insensible - lungs Sweat Feces Urine 2100 2300 350 100 1400 2300 Heavy Exercise ? 200 ? 350 650 5000 100 500 6500
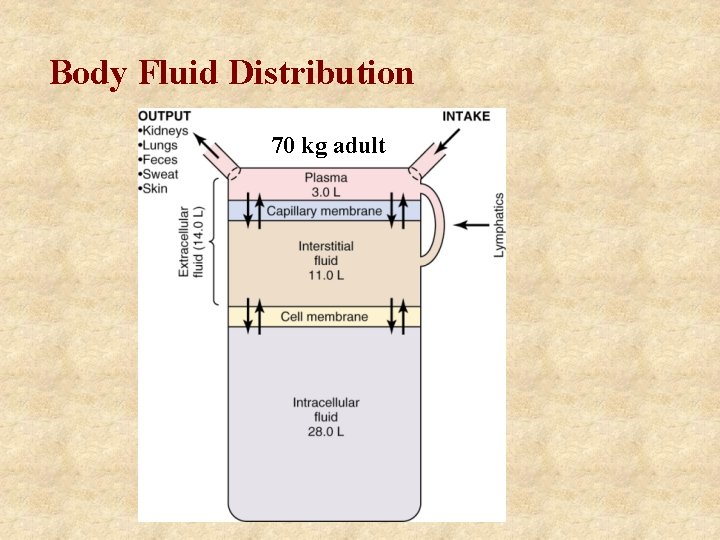
Body Fluid Distribution 70 kg adult

Total Body Water in Relation to Body Weight: Effects of Gender, Body Build and Age Body Build TBW (%) Adult Male TBW (%) Adult Female TBW (%) Infant Normal 60 50 70 Lean 70 60 80 Obese 50 42 60 Increasing obesity decreases TBW % Increasing age decreases TBW %

Measuring Body Fluid Volumes: Indicator Dilution Principle Indicator Mass A = Volume A x Concentration A A B

Measuring Body Fluid Volumes: Indicator Dilution Principle Indicator Mass A = Volume A x Concentration A A Indicator Mass A = Indicator Mass B Volume B = Indicator Mass B / Concentration B B Indicator Mass B = Volume B x Concentration B

Indicator Dilution Principle Volume (B) = Indicator Mass (B) = Q Concentration (B) C Valid if: • Indicator disperses only in compartment measured • Indicator disperses evenly in compartment • Indicator not metabolized or excreted
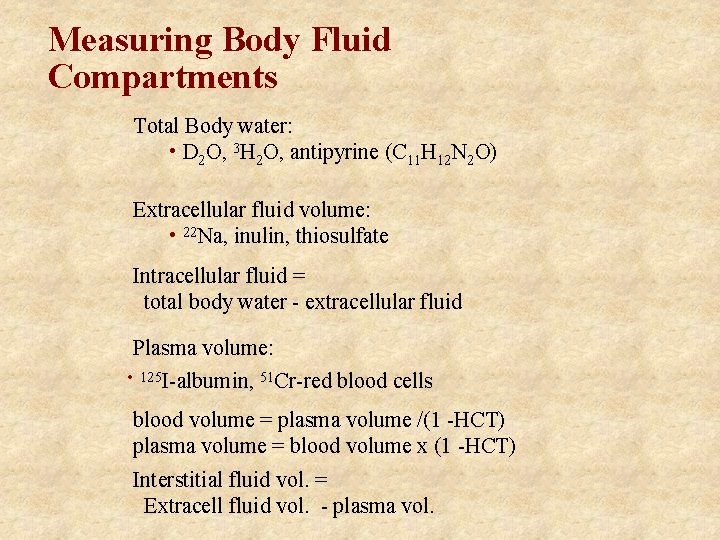
Measuring Body Fluid Compartments Total Body water: • D 2 O, 3 H 2 O, antipyrine (C 11 H 12 N 2 O) Extracellular fluid volume: • 22 Na, inulin, thiosulfate Intracellular fluid = total body water - extracellular fluid Plasma volume: • 125 I-albumin, 51 Cr-red blood cells blood volume = plasma volume /(1 -HCT) plasma volume = blood volume x (1 -HCT) Interstitial fluid vol. = Extracell fluid vol. - plasma vol.
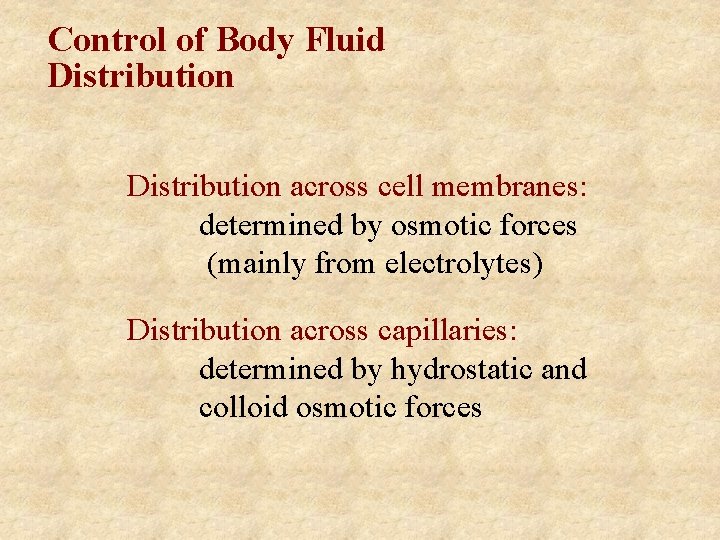
Control of Body Fluid Distribution across cell membranes: determined by osmotic forces (mainly from electrolytes) Distribution across capillaries: determined by hydrostatic and colloid osmotic forces

Osmotic Pressure ( ) = CRT C = concentration (osm/L) R = gas constant (62. 3 if = mm. Hg) T = Kelvino = 273 o + Co = 310 o (normal body temp. ) = 19, 300 mm. Hg / Osm If C = m. Osm/L, = C x 19. 3 mm. Hg

Principles of Osmotic Equilibria 300 Osmolarity m. Osm/ L ICF 7980 m. Osm 200 100 28 L 0 0 10 Normal State ECF 3920 m. Osm 14 L 20 30 40 VOLUME (L) • H 2 O moves across cell membranes easily i. e. ECFosm = ICFosm • Many solutes (e. g. Na. Cl) are relatively impermeant to cell membranes • Hydrostatic pressure differences have only a small effect on fluid movement across cell membrane
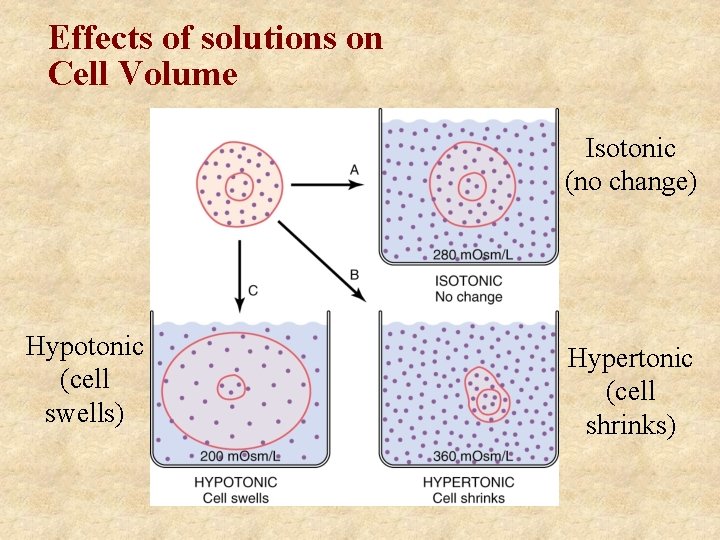
Effects of solutions on Cell Volume Isotonic (no change) Hypotonic (cell swells) Hypertonic (cell shrinks)

Isosmotic - has same osmolarity as body fluids Hyperosmotic - higher osmolarity than body fluids Hyposmotic- lower osmolarity than body fluids

Question • What is the osmolarity of a 5 % glucose solution? • Is the solution hyperosmotic, hypo-osmotic, or isosmotic?

Osmolarity of a 5 % Glucose Solution MW glucose = 180 gm/mol 5 % = 5 gm/100 ml = 50 gm/L 50 gm x 1 mol =. 278 mol = 278 m. Osm L L 180 gm L Isosmotic

Question • What is the osmolarity of a 3. 0% Na. Cl solution? • Is the solution hypertonic, hypotonic, or isotonic?

Osmolarity of a 3% Na. Cl Solution MW Na. Cl = 58. 5 3 % = 3 gm/100 ml = 30 gm/L 30 gm x 1 mol =. 513 mol = 513 mmol 58. 5 gm L L For Na. Cl, 1 mmol = 2 m. Osm 513 mmol x 2 m. Osm = 1026 m. Osm L Hypertonic mmol L L
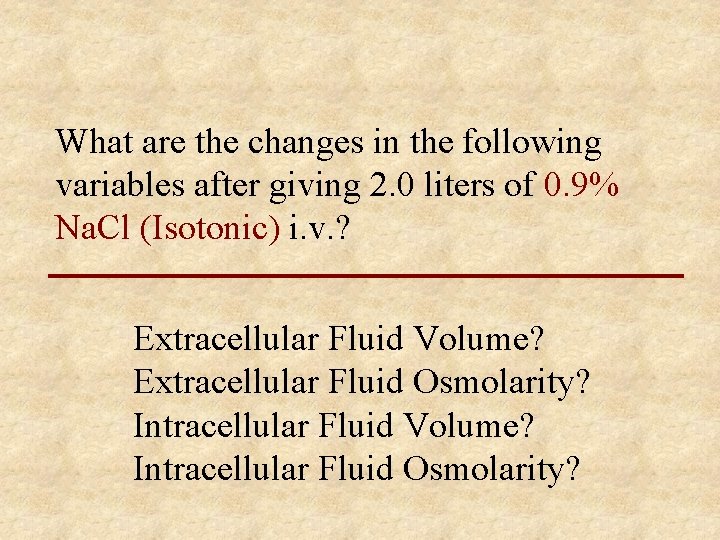
What are the changes in the following variables after giving 2. 0 liters of 0. 9% Na. Cl (Isotonic) i. v. ? Extracellular Fluid Volume? Extracellular Fluid Osmolarity? Intracellular Fluid Volume? Intracellular Fluid Osmolarity?
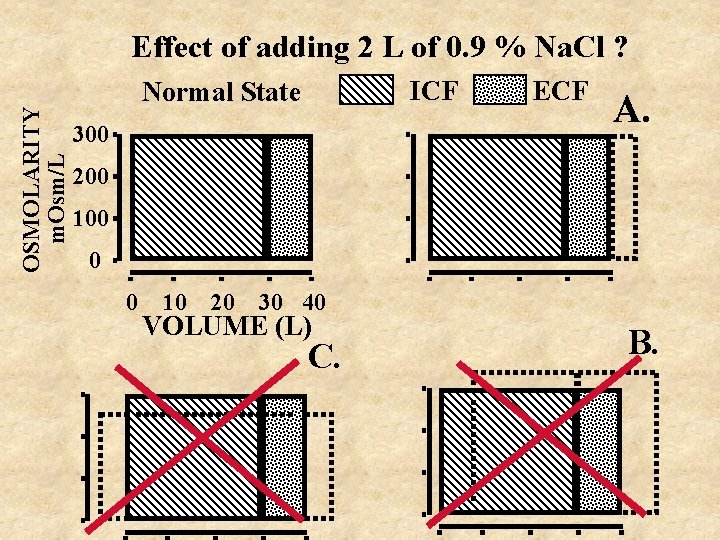
OSMOLARITY m. Osm/L Effect of adding 2 L of 0. 9 % Na. Cl ? ICF Normal State 300 ECF A. 200 100 0 0 10 20 30 40 VOLUME (L) C. B.
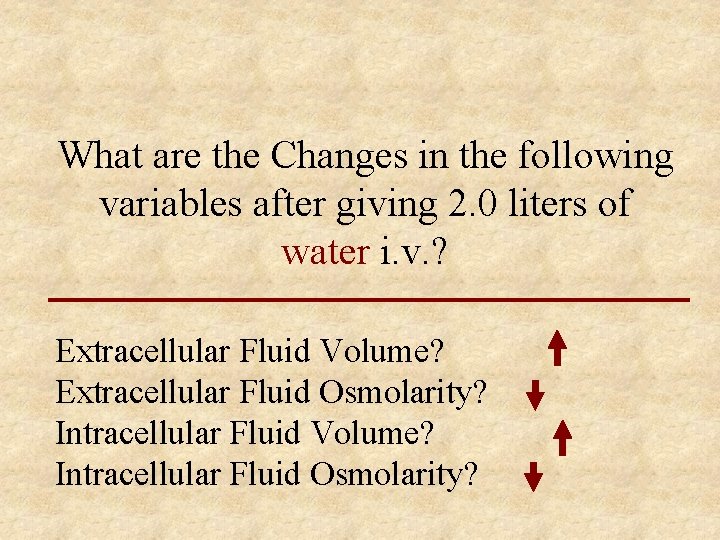
What are the Changes in the following variables after giving 2. 0 liters of water i. v. ? Extracellular Fluid Volume? Extracellular Fluid Osmolarity? Intracellular Fluid Volume? Intracellular Fluid Osmolarity?
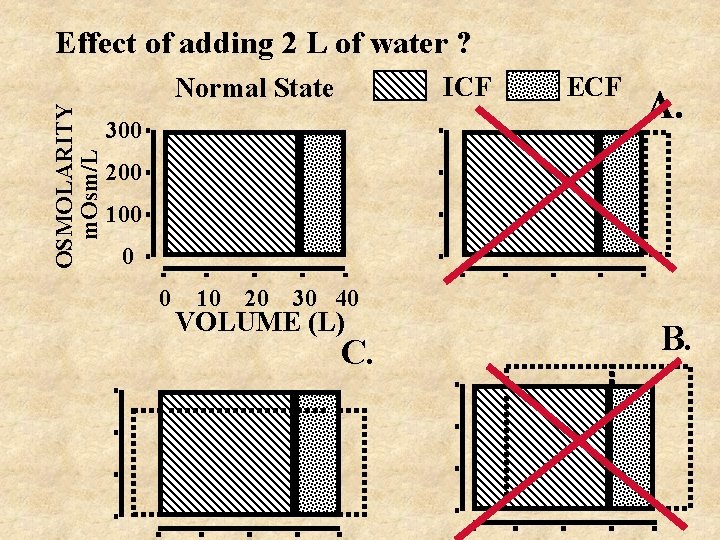
OSMOLARITY m. Osm/L Effect of adding 2 L of water ? ICF Normal State 300 ECF A. 200 100 0 0 10 20 30 40 VOLUME (L) C. B.
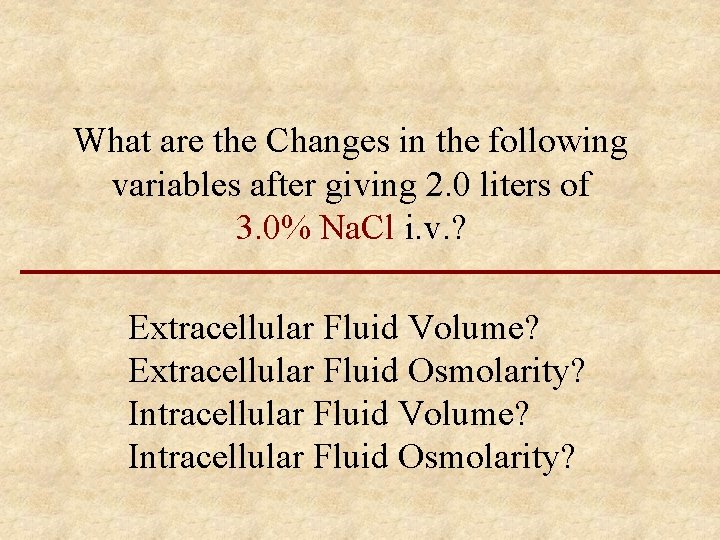
What are the Changes in the following variables after giving 2. 0 liters of 3. 0% Na. Cl i. v. ? Extracellular Fluid Volume? Extracellular Fluid Osmolarity? Intracellular Fluid Volume? Intracellular Fluid Osmolarity?
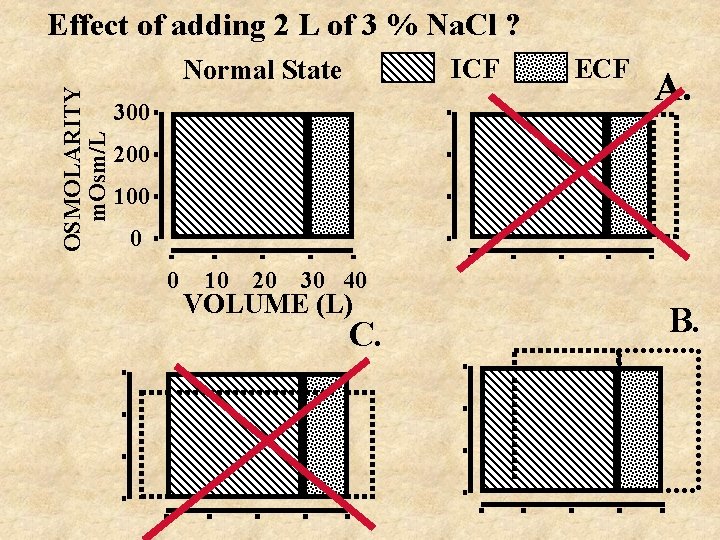
Effect of adding 2 L of 3 % Na. Cl ? ICF OSMOLARITY m. Osm/L Normal State 300 ECF A. 200 100 0 0 10 20 30 40 VOLUME (L) C. B.
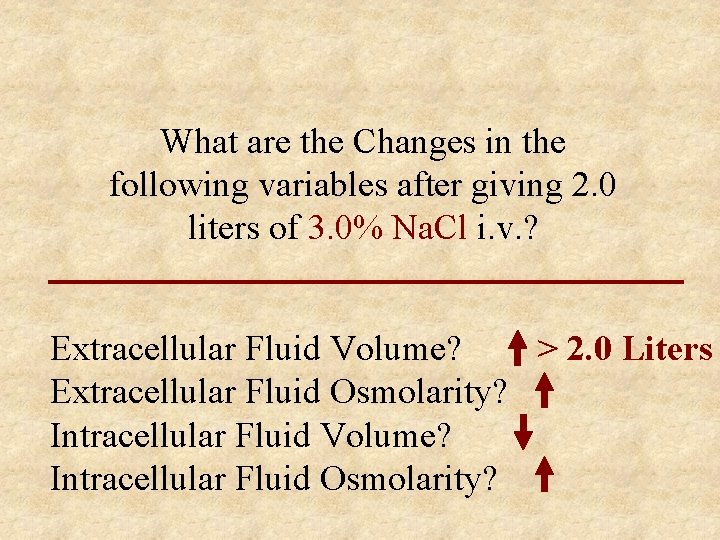
What are the Changes in the following variables after giving 2. 0 liters of 3. 0% Na. Cl i. v. ? Extracellular Fluid Volume? > 2. 0 Liters Extracellular Fluid Osmolarity? Intracellular Fluid Volume? Intracellular Fluid Osmolarity?
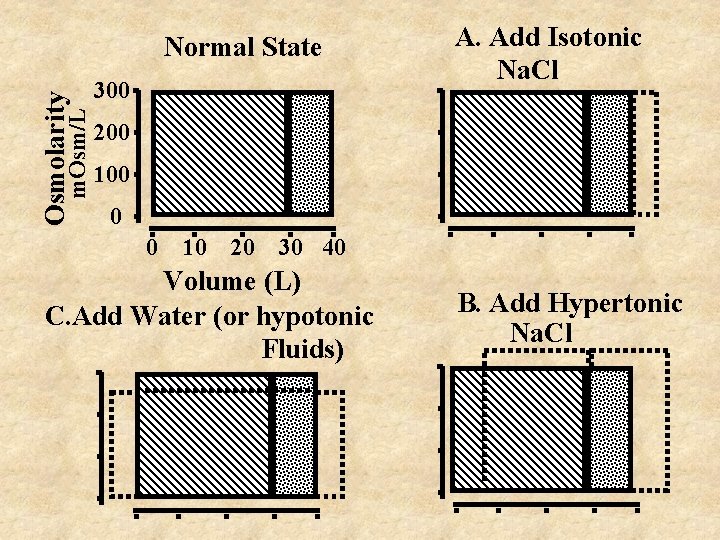
m. Osm/L Osmolarity Normal State 300 A. Add Isotonic Na. Cl 200 100 0 0 10 20 30 40 Volume (L) C. Add Water (or hypotonic Fluids) B. Add Hypertonic Na. Cl
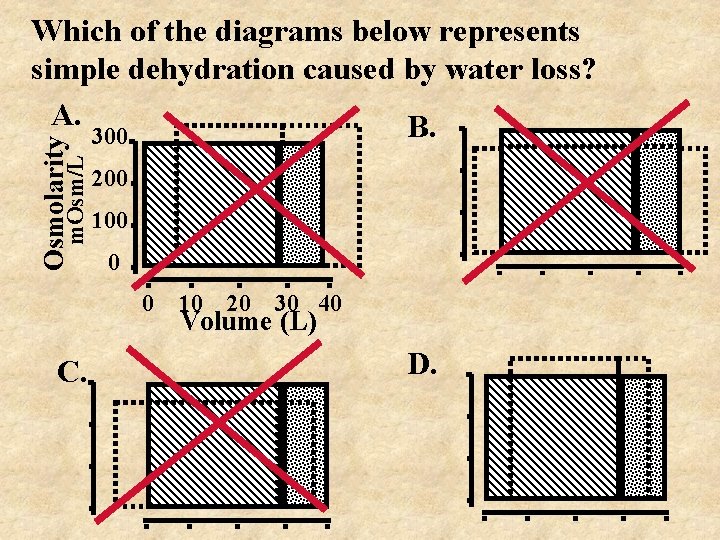
m. Osm/L Osmolarity Which of the diagrams below represents simple dehydration caused by water loss? A. B. 300 200 100 0 0 C. 10 20 30 40 Volume (L) D.

Osmotic Equilibrium Problems • Calculation of intracellular and extracellular fluid volumes and osmolarities after fluid administration.

Effect of Infusing 2. 0 liters of 3% Na. Cl to a 70 kg Person Assume: 1) no excretion of water or solutes, 2) osmotic equilibrium, 3) ECF =20% body wt, ICF =40 % body wt. • What is the extracellular osmolarity? • What are the intra- and extracellular fluid volumes?

Effect of Infusing 2. 0 liters of 3% Na. Cl to a 70 kg Person Assume: extracell vol. = 20 % BW; intracell. vol. = 40 % BW Step 1. Initial conditions Volume (L) Conc. m. Osm/L Total m. Osm Extracell. Fluid (. 2 x 70) = 14 280 3920 Intracell. Fluid (. 4 x 70) = 28 280 7840 = 42 280 11, 760 Total Fluid

Osmolarity of a 3% Na. Cl Solution MW Na. Cl = 58. 5 3% = 3 gm/100 ml = 30 gm/L 30 gm x 1 mol =. 513 mol = 513 mmol L 58. 5 gm L L For Na. Cl, 1 mmol = 2 m. Osm 513 mmol x 2 m. Osm = 1026 m. Osm mmol L L 2. 0 L x 1026 m. Osm/L = 2052 m. Osm

Effect of Infusing 2. 0 liters of 3% Na. Cl to a 70 kg Person Step 2. Instantaneous Effect Volume (L) Conc. m. Osm/L Total m. Osm Extracell. fluid 14+2= 16 ? 3920+2052= 5972 Intracell. fluid 28 +0= 28 280 7840 + 0 = 7840 Total Fluid 42+2= 44 ? 11, 760+2052= 13, 812 no equilibrium

Effect of Infusing 2. 0 liters of 3% Na. Cl to a 70 kg Person Step 2. After osmotic equilibrium Volume (L) Conc. m. Osm/L Total m. Osm Extracell. fluid 19 314 3920+2052= 5972 Intracell. fluid 25 314 7840 + 0 = Total Fluid Net effect: 42+2= 44 7840 314 11, 760+2052= 13, 812 Extracell. Volume by 5. 0 L Intracell volume by 3. 0 L

Abnormalities of Extracellular + Fluid Na Concentration (Osmolarity)

Abnormalities of Extracellular Fluid Na+ Concentration Hypernatremia : • water loss • Na+ excess Hyponatremia : Na+ conc. • water excess • Na+ loss Normal plasma [Na+] = 140 -145 mmol / L
Consequences of Hyponatremia and Hypernatremia • Water moves in and out of cells swell or shrink • This has profound effects on the brain. - Neurologic function is altered - Rapid shrinking can tear vessels and cause hemorrhage - Rapid swelling can cause herniation Because the skull is rigid, the brain cannot increase its volume by more than 10% without being forced down the neck (herniation).
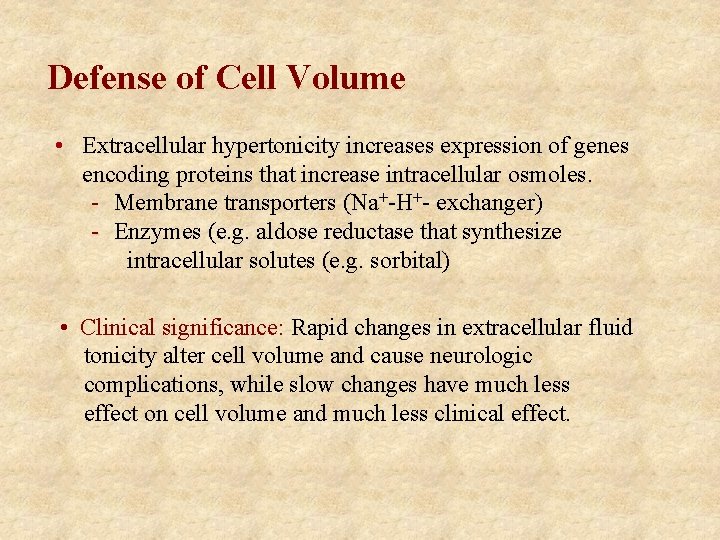
Defense of Cell Volume • Extracellular hypertonicity increases expression of genes encoding proteins that increase intracellular osmoles. - Membrane transporters (Na+-H+- exchanger) - Enzymes (e. g. aldose reductase that synthesize intracellular solutes (e. g. sorbital) • Clinical significance: Rapid changes in extracellular fluid tonicity alter cell volume and cause neurologic complications, while slow changes have much less effect on cell volume and much less clinical effect.

Hypernatremia With Decreased Extracellular Fluid Volume (water loss: hyperosmotic dehydration) With Increased Extracellular Fluid Volume ( Na. Cl excess: hyperosmotic overhydration) What lab measurements would help in diagnosis? • Hematocrit, plasma proteins • Urine volume

Hypernatremia With Decreased Extracellular Fluid Volume (water loss: hyperosmotic dehydration) • Increased hematocrit, plasma proteins • Small volume of concentrated urine - dehydration, excess sweating • Large volume of dilute urine - diabetes insipidus - lack of ADH (“central” diabetes insipidus) - “nephrogenic” diabetes insipidus

Hypernatremia With Increased Extracellular Fluid Volume ( Na. Cl excess: hyperosmotic overhydration) • Decreased hematocrit, plasma proteins • Primary aldosteronism • Cushing’s syndrome Usually the Hypernatremia is mild in these examples, but the person may have very low plasma K + (hypokalemia).

Hypernatremia – Clinical Case A 52 year old male comes to you office complaining of frequent headaches and large volumes of urine. His medical history indicates that he is being treated with lithium for manic-depressive disorder. your laboratory tests give the following results: Body weight Plasma Na+ Plasma K+ Blood pressure Heart rate Hematocrit Plasma protein = 70 kg = 162 mmol/L = 4. 7 mmol/L = 102/65 mm. Hg = 92 beats/min = 0. 50 = 8. 40 • What are some potential causes of his hypernatremia? • What additional tests would help in making a differential diagnosis?

Hypernatremia A. With Increased Extracellular Fluid Volume ( Na. Cl excess: hyperosmotic overhydration) • Primary aldosteronism • Cushing’s syndrome B. With Decreased Extracellular Fluid Volume (water loss: hyperosmotic dehydration) • Small volume of concentrated urine - dehydration, excess sweating • Large volume of dilute urine - diabetes insipidus - lack of ADH (“central” diabetes insipidus) - “nephrogenic” diabetes insipidus

Hyponatremia With Decreased Extracellular Fluid Volume ( Na. Cl Loss: hypo-osmotic dehydration) • diarrhea, vomiting • Addison’s disease (decreased aldosterone) • overuse of diuretics • sodium wasting renal diseases Increased hematocrit, plasma proteins

Hyponatremia With Increased Extracellular Fluid Volume (water excess: hypo-osmotic overhydration) • Inappropriate ADH syndrome • Bronchogenic tumors • Fibrosing tuberculosis lung lesions • Circulatory failure (e. g. , congestive heart failure) Decreased hematocrit, plasma proteins

Intracellular Edema • Hyponatremia • Depression of tissue metabolic systems (e. g. hypothyroidism) • Inadequate tissue nutrition (e. g. ischemia) • Inflammation of tissues (increased cell membrane permeability)

Extracellular Edema Causes of Extracellular Edema (increased interstitial fluid volume) • Increased Capillary Filtration • Failure of lymphatics to Return Interstitial Fluid to Circulation
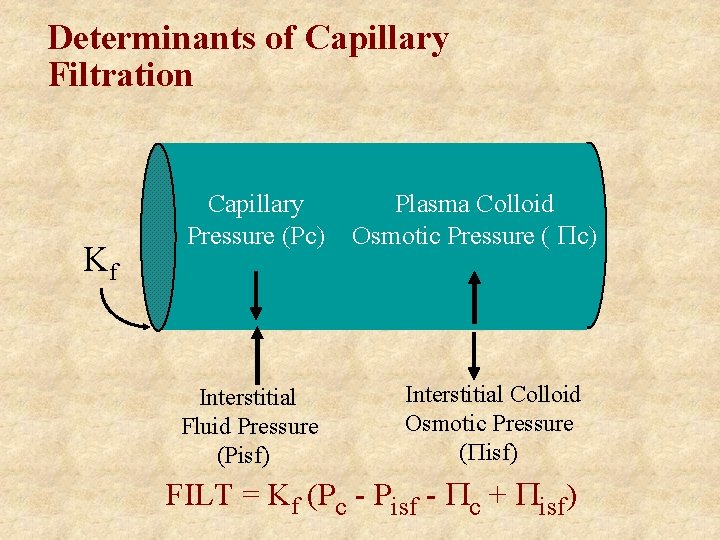
Determinants of Capillary Filtration Kf Capillary Pressure (Pc) Interstitial Fluid Pressure (Pisf) Plasma Colloid Osmotic Pressure ( c) Interstitial Colloid Osmotic Pressure ( isf) FILT = Kf (Pc - Pisf - c + isf)

Causes of Increased Capillary Filtration FILT = Kf (Pc - Pisf - c + isf) Increased Kf: toxins, ischemic damage, infections, etc. Increased Pc: • increased arterial pressure, excess fluid retention, • decreased arteriolar resistance • increased venous resistance, increased venous pressure Decreased c: • nephrotic syndrome • cirrhosis • malnutrition

Normal Filtration FILT = Kf x Net Filt Pressure = 6. 6 x ( 17. 3 - (-3) - 28 +8) = 6. 6 x ( + 0. 3) = 1. 89 ml/min FILT = 2722 ml/day Where does all of this fluid (and protein in it) go?
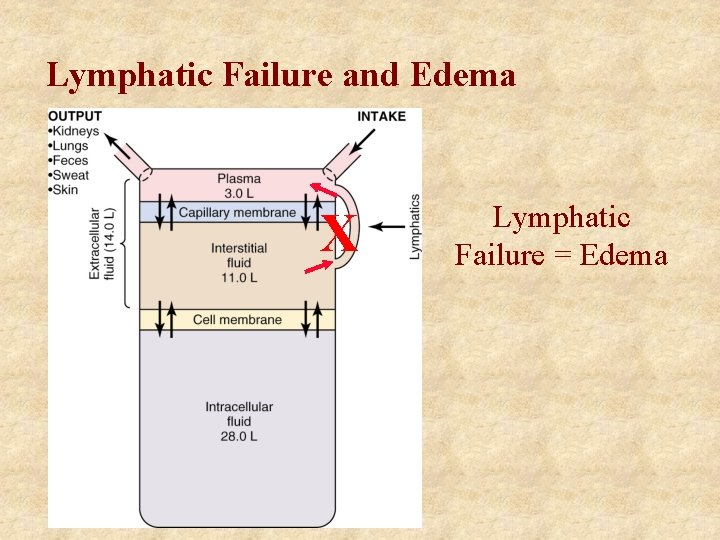
Lymphatic Failure and Edema X Lymphatic Failure = Edema

Safety Factors Against Edema • Low compliance of interstitium when interstitial fluid pressure is negative = 3 mm. Hg • Increased lymph flow = 7 mm. Hg • “ Washdown” of interstitial protein at high lymph flow rates = 7 mm. Hg Total Safety factor = 17 mm. Hg
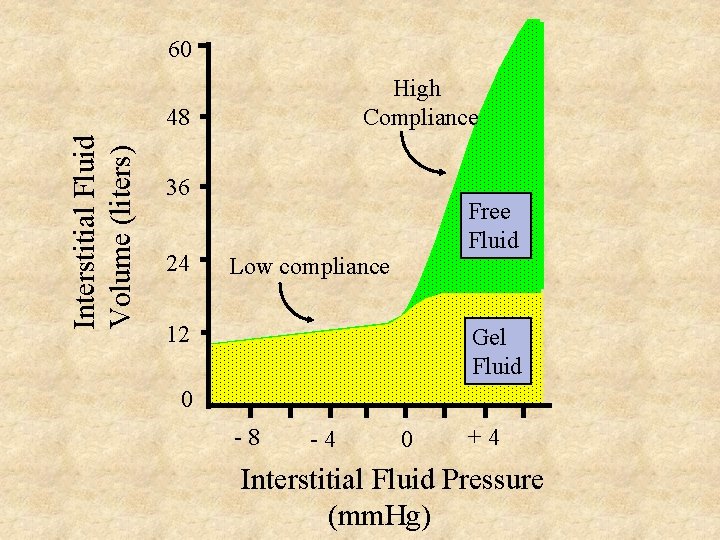
60 High Compliance Interstitial Fluid Volume (liters) 48 36 24 Free Fluid Low compliance 12 Gel Fluid 0 -8 -4 0 +4 Interstitial Fluid Pressure (mm. Hg)
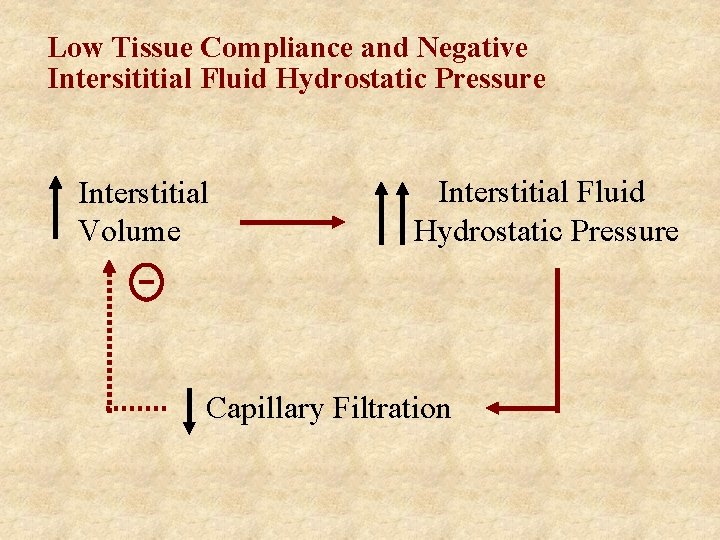
Low Tissue Compliance and Negative Intersititial Fluid Hydrostatic Pressure Interstitial Volume Interstitial Fluid Hydrostatic Pressure Capillary Filtration
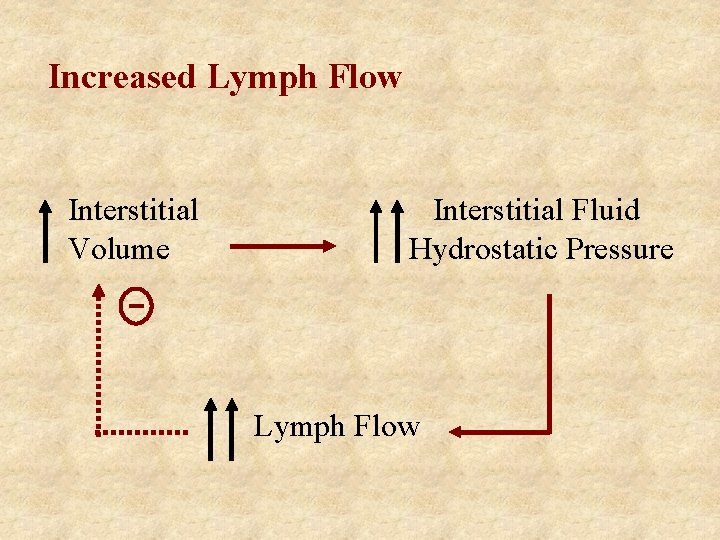
Increased Lymph Flow Interstitial Volume Interstitial Fluid Hydrostatic Pressure Lymph Flow
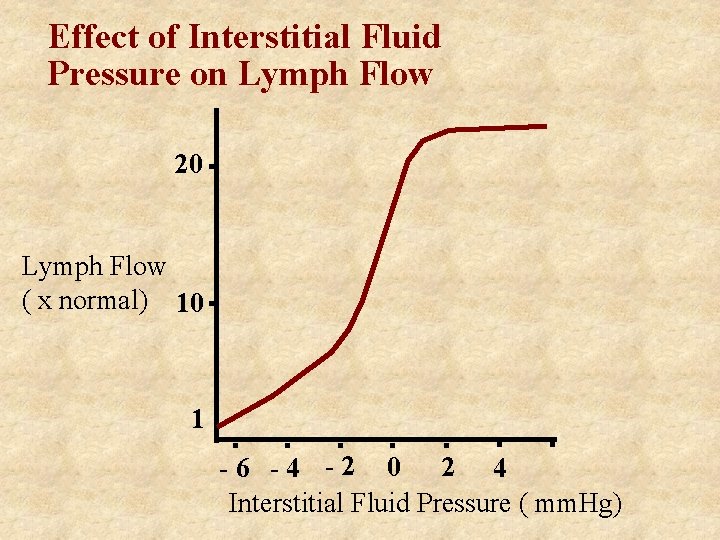
Effect of Interstitial Fluid Pressure on Lymph Flow 20 Lymph Flow ( x normal) 10 1 -6 -4 -2 0 2 4 Interstitial Fluid Pressure ( mm. Hg)
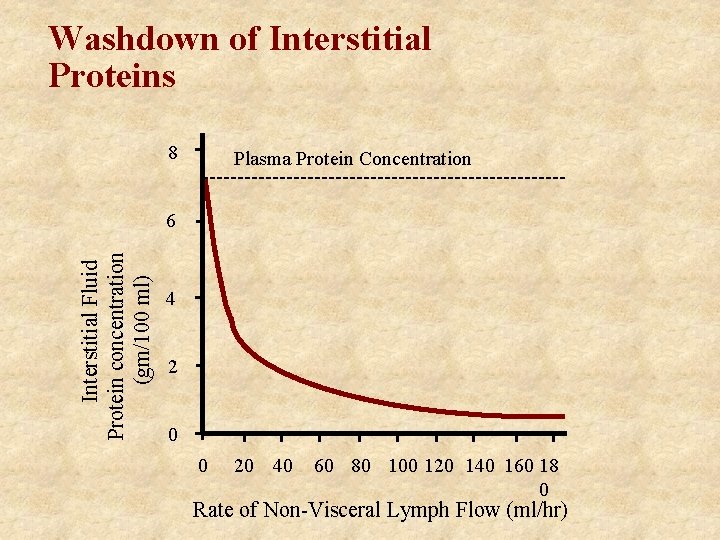
Washdown of Interstitial Proteins 8 Plasma Protein Concentration Interstitial Fluid Protein concentration (gm/100 ml) 6 4 2 0 0 20 40 60 80 100 120 140 160 18 0 Rate of Non-Visceral Lymph Flow (ml/hr)
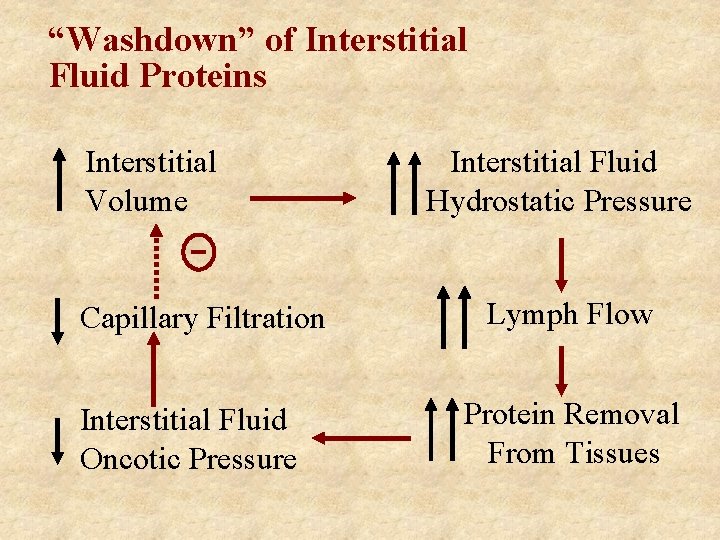
“Washdown” of Interstitial Fluid Proteins Interstitial Volume Capillary Filtration Interstitial Fluid Oncotic Pressure Interstitial Fluid Hydrostatic Pressure Lymph Flow Protein Removal From Tissues

Saftey Factors Against Edema • Low compliance of interstitium when interstitial fluid pressure is negative = 3 mm. Hg • Increased lymph flow = 7 mm. Hg • “ Washdown of interstitial protein at high lymph flow rates = 7 mm. Hg Total Safety factor = 17 mm. Hg

Conditions Causing Edema 1. High capillary hydrostatic pressure • Excess fluid retention by kidneys - acute or chronic kidney failure - glomerulonephritis - mineralocorticoid excess

Conditions Causing Edema 1. High capillary hydrostatic pressure • Decreased arteriolar resistance - vasodilator drugs - autonomic insufficiency • Increased venous pressure - congestive heart failure - high output heart failure (e. g. anemia) - venous obstruction - venous valve failure - cirrhosis

Conditions Causing Edema 2. Decreased plasma proteins (low oncotic pressure) • Loss of proteins - burns, wounds - nephrosis - gastroenteropathy • Failure to produce proteins - malnutrition (“kwashiorkor”) - cirrhosis - analbuminemia

Conditions Causing Edema 3. Increased capillary permeability • Immune reactions (histamine) • Toxins • Burns • Prolonged ischemia • Vitamin deficiency (e. g. vitamin C) • Pre-eclampsia and eclampsia in pregnancy

Conditions Causing Edema 4. Blockage of lymphatics • Cancer • Surgery • Infections (e. g. filaria nematodes)
- Slides: 66