Chapter 24 Carbohydrates Carbohydrates Sugars and their derivatives

Chapter 24 Carbohydrates

Carbohydrates • Sugars and their derivatives are classified as carbohydrates – Examples: Glucose, Sucrose, Starch, Glycogen • Molecular formulas fit a hydrate of carbon pattern: Cn(H 2 O)m • Sucrose: C 6 H 12 O 6 = C 6(H 2 O)6 24. 1 Properties and Classification of Carbohydrates 2

3
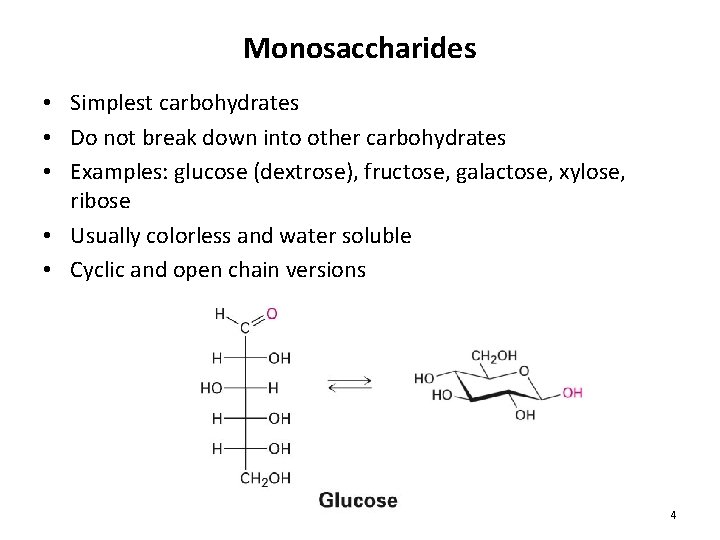
Monosaccharides • Simplest carbohydrates • Do not break down into other carbohydrates • Examples: glucose (dextrose), fructose, galactose, xylose, ribose • Usually colorless and water soluble • Cyclic and open chain versions 4
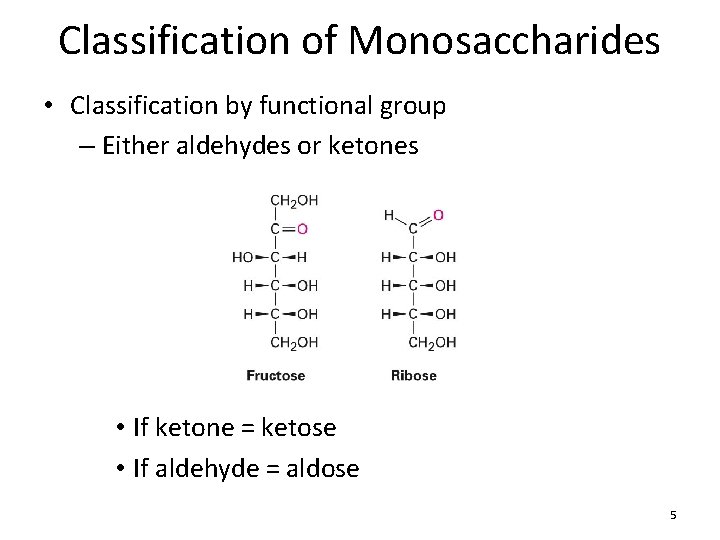
Classification of Monosaccharides • Classification by functional group – Either aldehydes or ketones • If ketone = ketose • If aldehyde = aldose 5
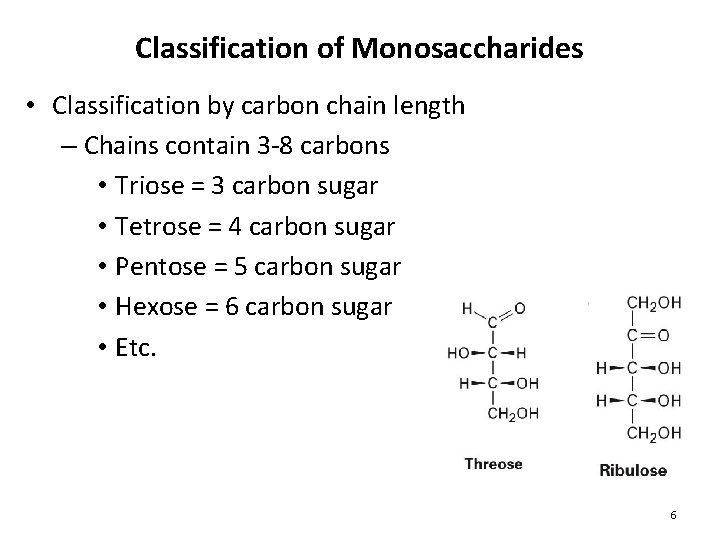
Classification of Monosaccharides • Classification by carbon chain length – Chains contain 3 -8 carbons • Triose = 3 carbon sugar • Tetrose = 4 carbon sugar • Pentose = 5 carbon sugar • Hexose = 6 carbon sugar • Etc. 6
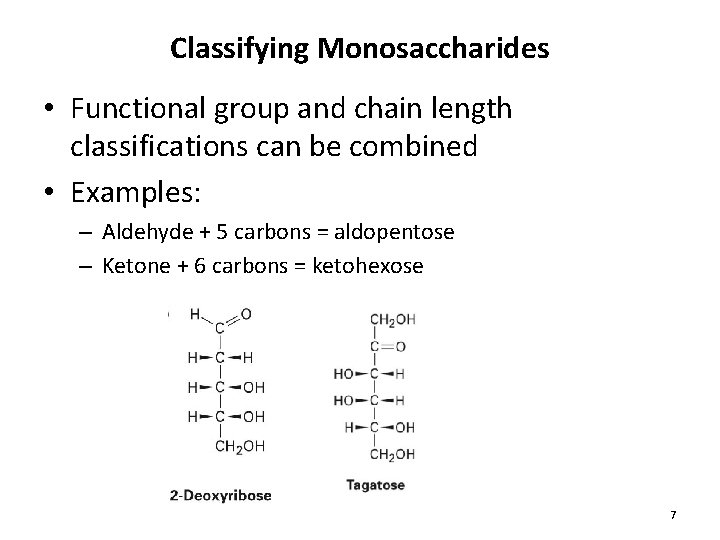
Classifying Monosaccharides • Functional group and chain length classifications can be combined • Examples: – Aldehyde + 5 carbons = aldopentose – Ketone + 6 carbons = ketohexose 7
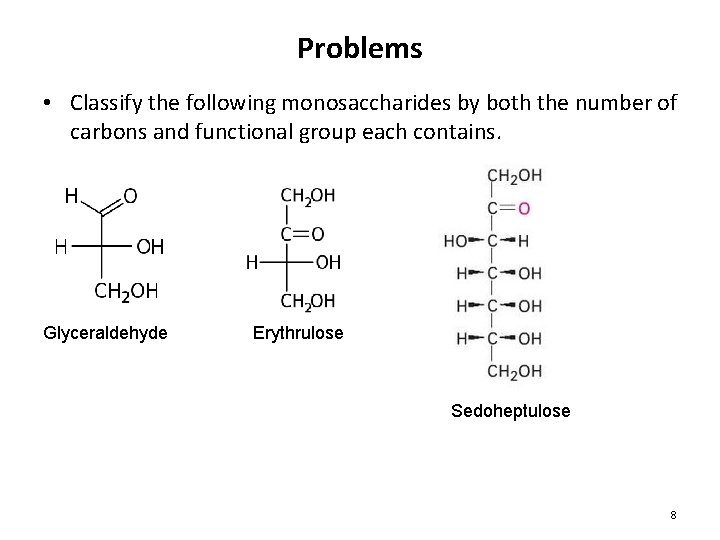
Problems • Classify the following monosaccharides by both the number of carbons and functional group each contains. Glyceraldehyde Erythrulose Sedoheptulose 8
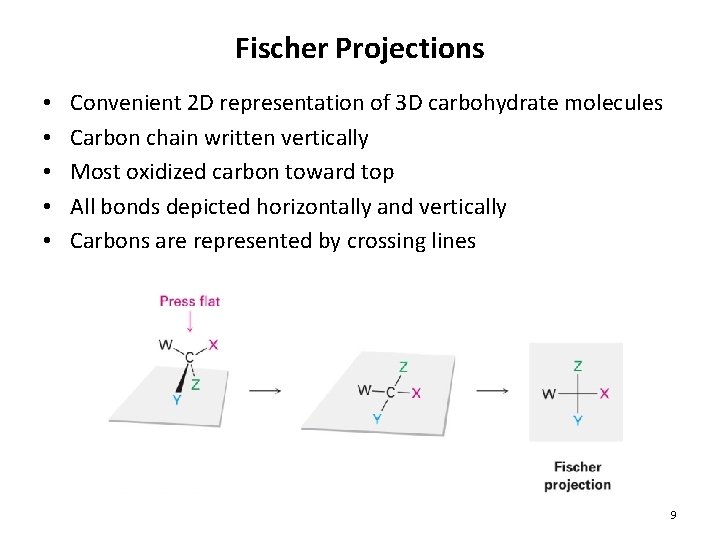
Fischer Projections • • • Convenient 2 D representation of 3 D carbohydrate molecules Carbon chain written vertically Most oxidized carbon toward top All bonds depicted horizontally and vertically Carbons are represented by crossing lines 9
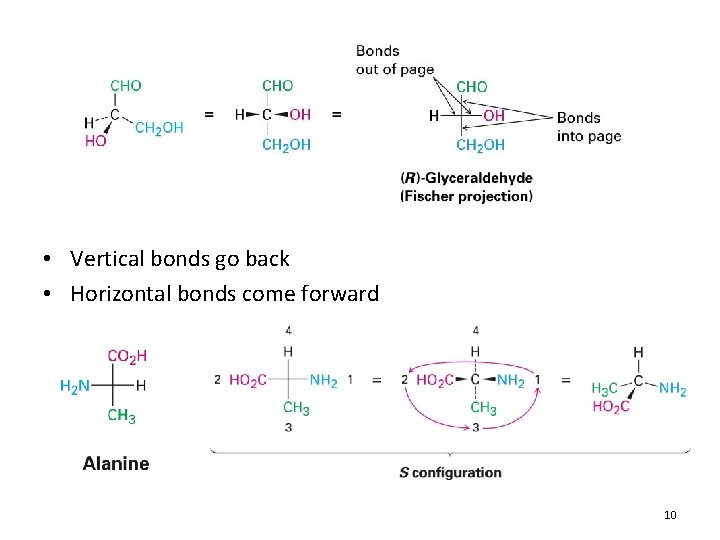
• Vertical bonds go back • Horizontal bonds come forward 10
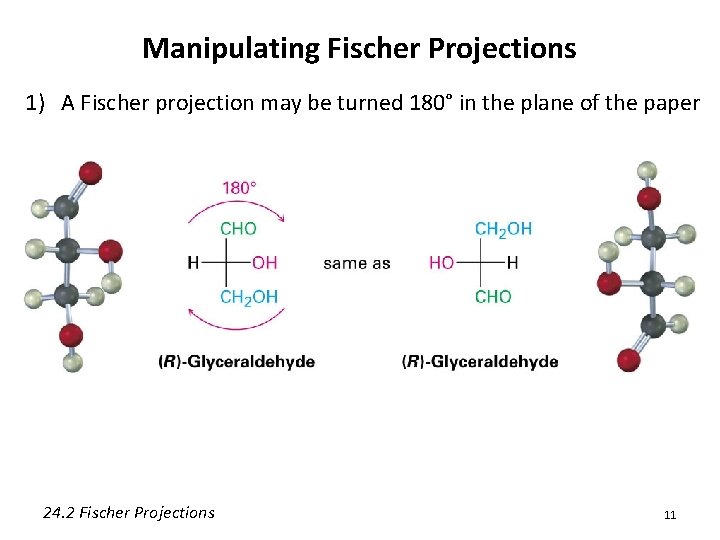
Manipulating Fischer Projections 1) A Fischer projection may be turned 180° in the plane of the paper 24. 2 Fischer Projections 11
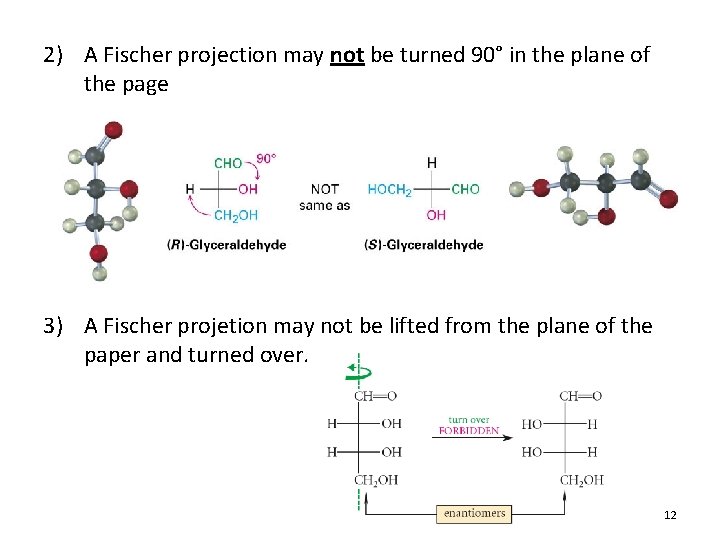
2) A Fischer projection may not be turned 90° in the plane of the page 3) A Fischer projetion may not be lifted from the plane of the paper and turned over. 12
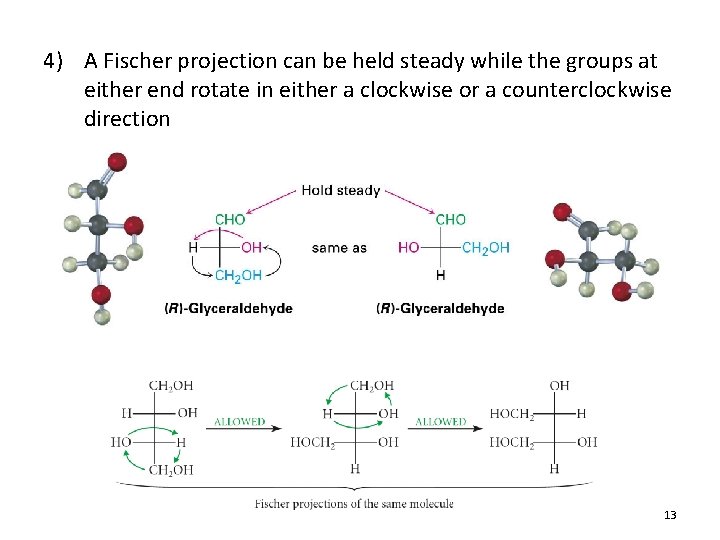
4) A Fischer projection can be held steady while the groups at either end rotate in either a clockwise or a counterclockwise direction 13
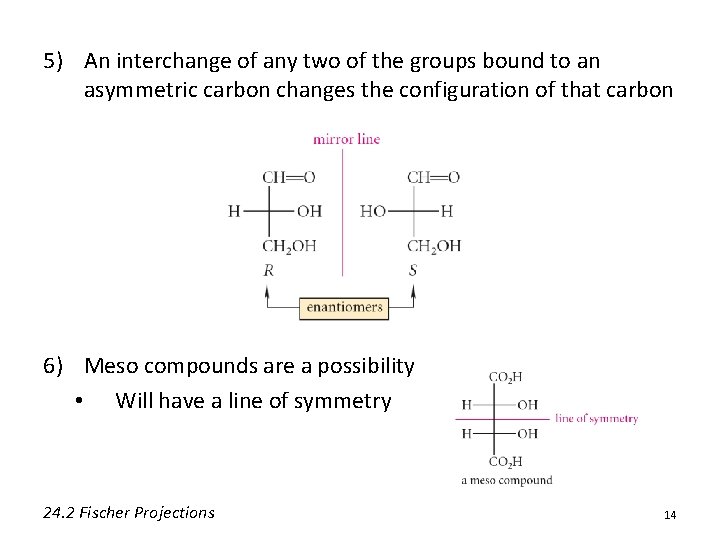
5) An interchange of any two of the groups bound to an asymmetric carbon changes the configuration of that carbon 6) Meso compounds are a possibility • Will have a line of symmetry 24. 2 Fischer Projections 14
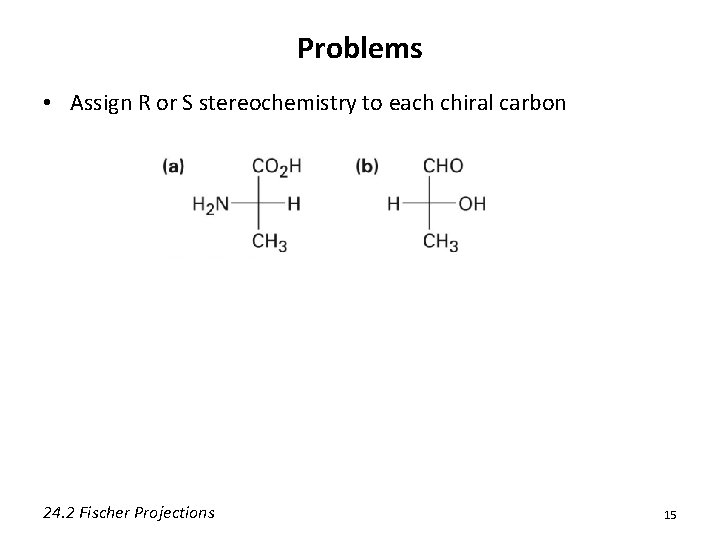
Problems • Assign R or S stereochemistry to each chiral carbon 24. 2 Fischer Projections 15
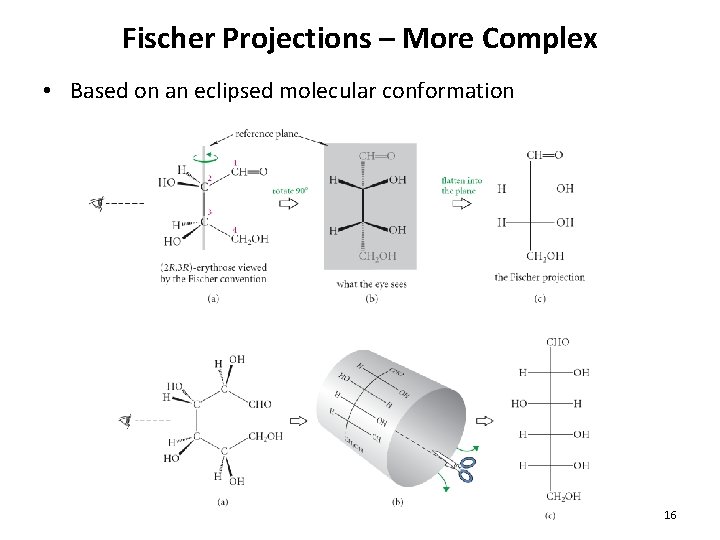
Fischer Projections – More Complex • Based on an eclipsed molecular conformation 16
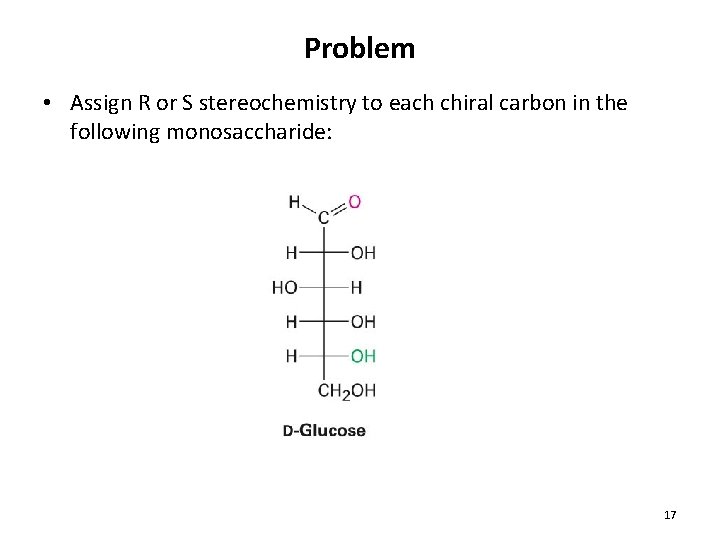
Problem • Assign R or S stereochemistry to each chiral carbon in the following monosaccharide: 17
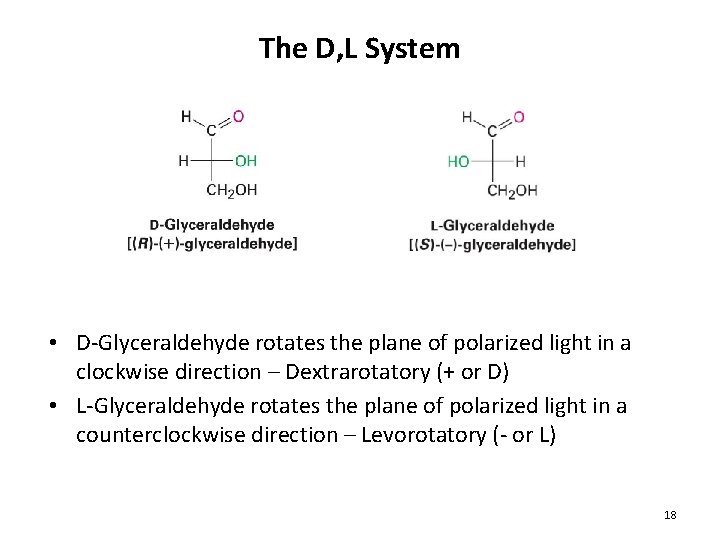
The D, L System • D-Glyceraldehyde rotates the plane of polarized light in a clockwise direction – Dextrarotatory (+ or D) • L-Glyceraldehyde rotates the plane of polarized light in a counterclockwise direction – Levorotatory (- or L) 18
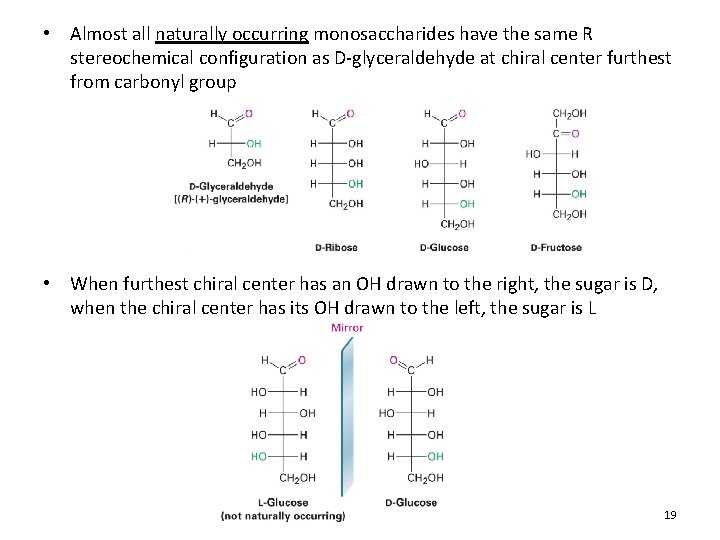
• Almost all naturally occurring monosaccharides have the same R stereochemical configuration as D-glyceraldehyde at chiral center furthest from carbonyl group • When furthest chiral center has an OH drawn to the right, the sugar is D, when the chiral center has its OH drawn to the left, the sugar is L 19
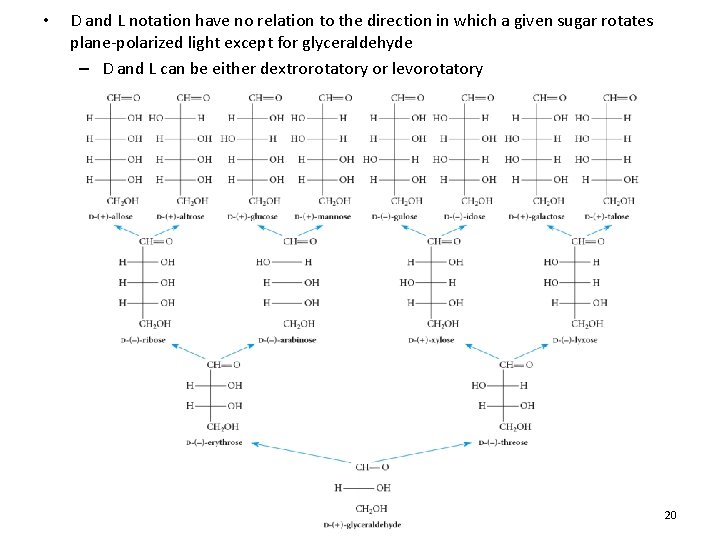
• D and L notation have no relation to the direction in which a given sugar rotates plane-polarized light except for glyceraldehyde – D and L can be either dextrorotatory or levorotatory 20
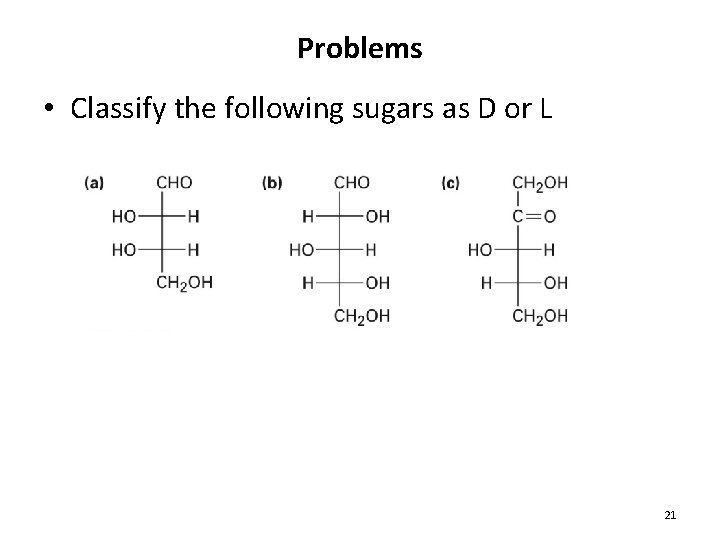
Problems • Classify the following sugars as D or L 21
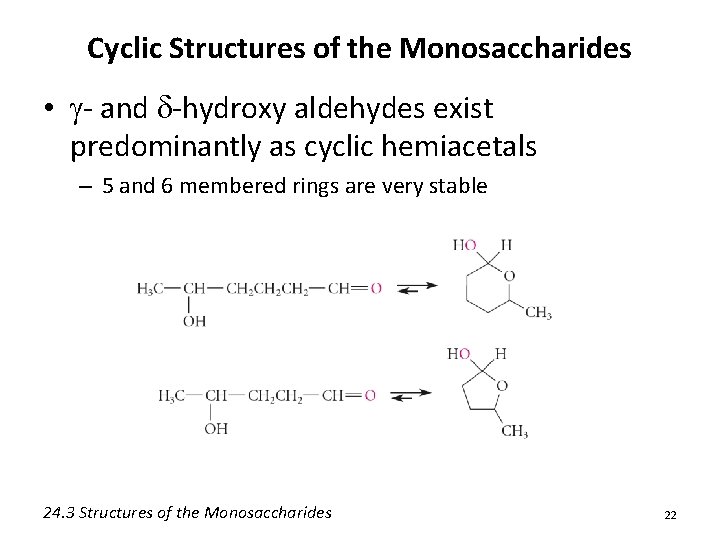
Cyclic Structures of the Monosaccharides • g- and d-hydroxy aldehydes exist predominantly as cyclic hemiacetals – 5 and 6 membered rings are very stable 24. 3 Structures of the Monosaccharides 22
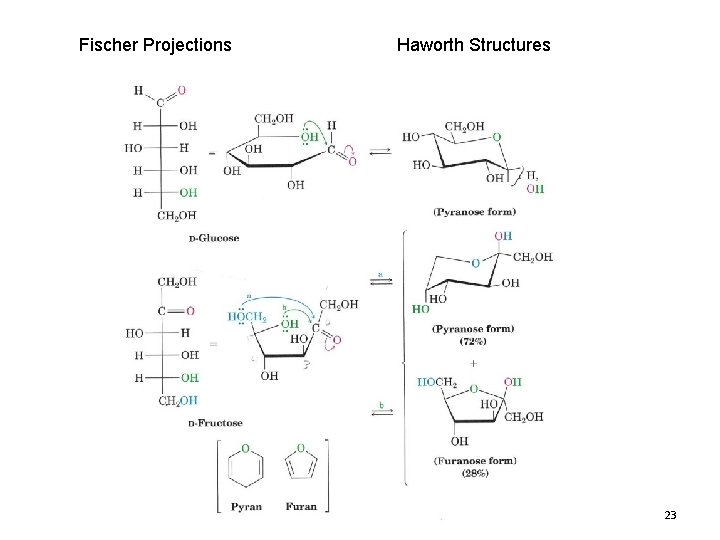
Fischer Projections Haworth Structures 23
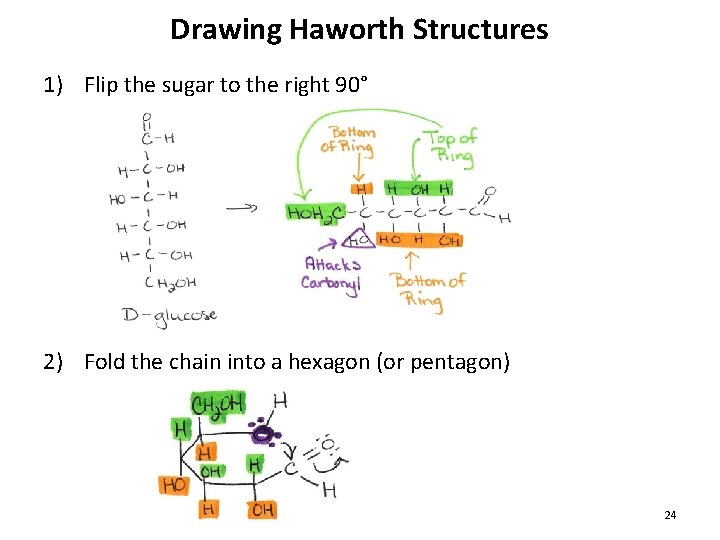
Drawing Haworth Structures 1) Flip the sugar to the right 90° 2) Fold the chain into a hexagon (or pentagon) 24
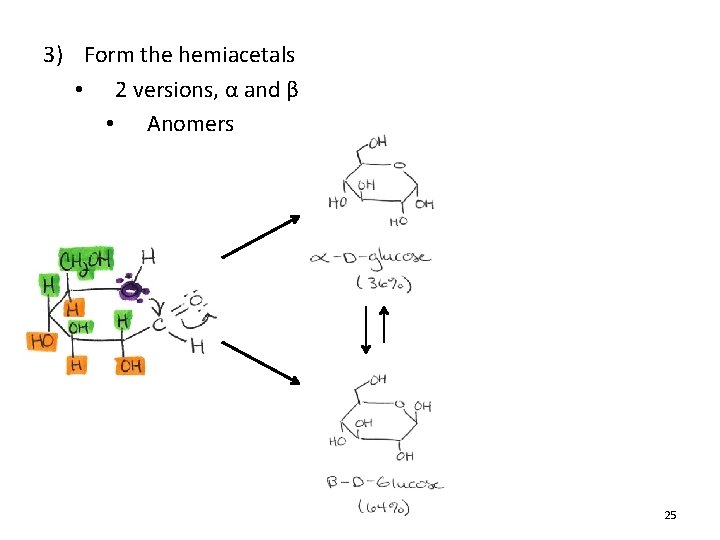
3) Form the hemiacetals • 2 versions, α and β • Anomers 25

Problems • Draw the cyclic structures for the following sugars 26
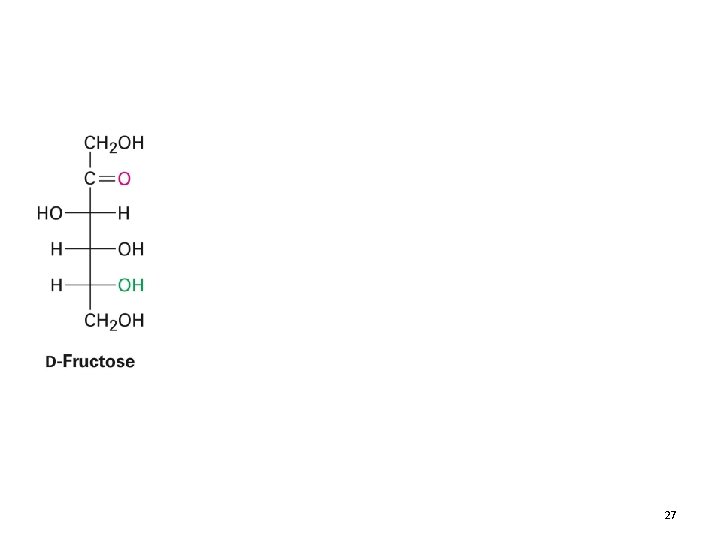
27

Monosaccharide Anomers: Mutarotation • The two anomers of D-glucopyranose can be crystallized and purified • -D-glucopyranose melts at 146° and its specific rotation, [ ]D = +112. 2° • b-D-glucopyranose melts at 148– 155°C with a specific rotation of [ ]D = +18. 7° • Rotation of solutions of either pure anomer slowly changes due to slow conversion of the pure anomers into a 37: 63 equilibrium mixture of : b with a [ ]D = +52. 6° • called mutarotation 28
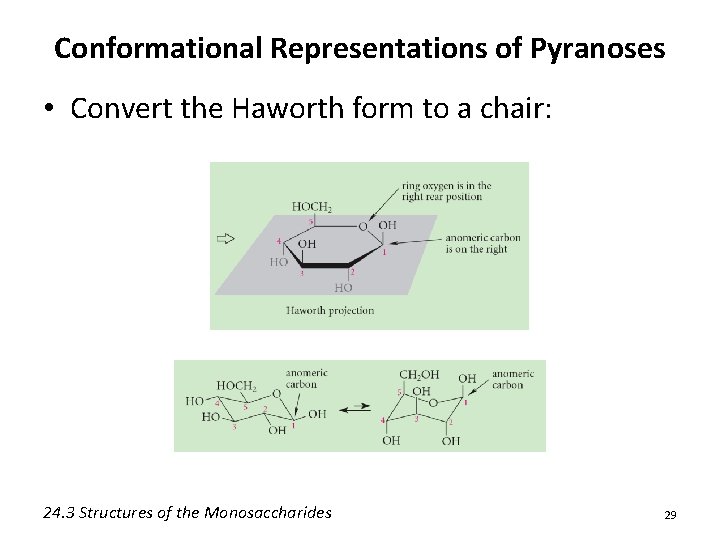
Conformational Representations of Pyranoses • Convert the Haworth form to a chair: 24. 3 Structures of the Monosaccharides 29

Oxidation and Reduction of Carbohydrates • The aldehydes of aldoses may be reduced or selectively oxidized without impacting the other alcohols • Selective oxidation of the primary alcohol group may also be realized 24. 8 Oxidation and Reduction Reactions of Carbohydrates 30
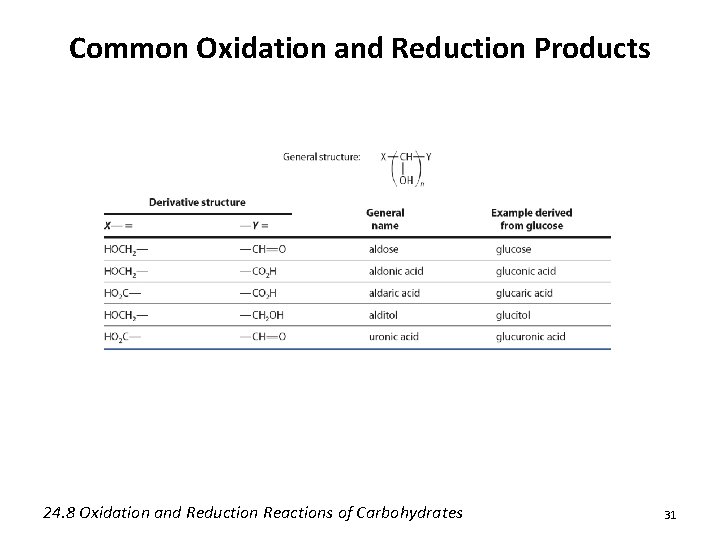
Common Oxidation and Reduction Products 24. 8 Oxidation and Reduction Reactions of Carbohydrates 31
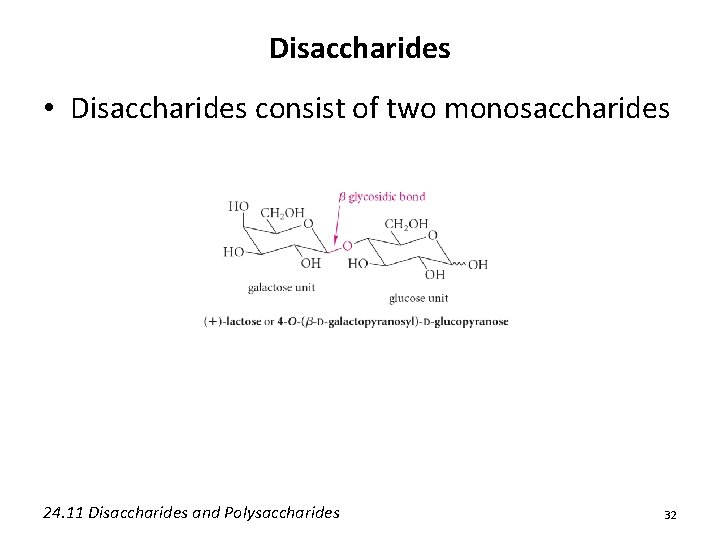
Disaccharides • Disaccharides consist of two monosaccharides 24. 11 Disaccharides and Polysaccharides 32
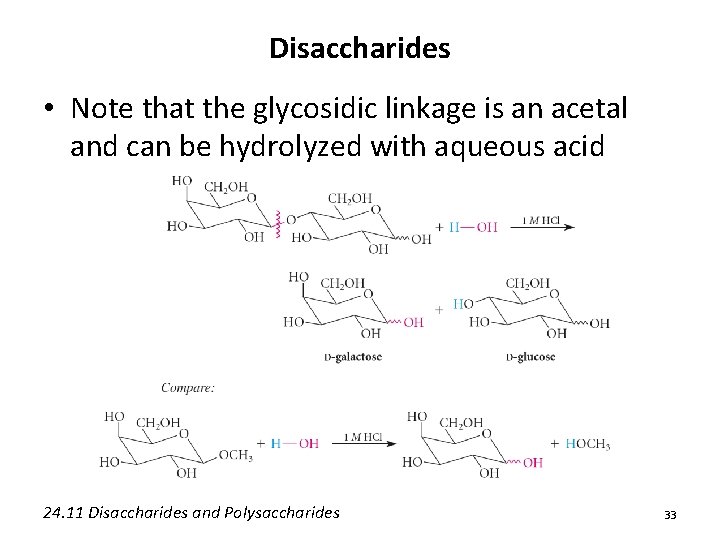
Disaccharides • Note that the glycosidic linkage is an acetal and can be hydrolyzed with aqueous acid 24. 11 Disaccharides and Polysaccharides 33
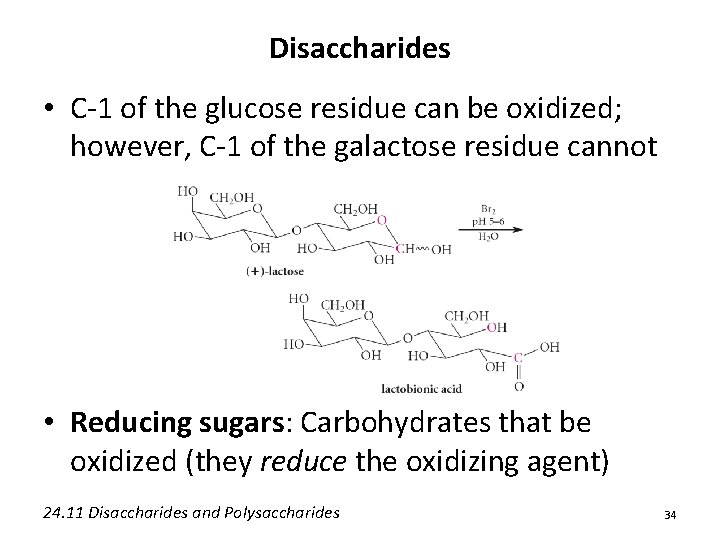
Disaccharides • C-1 of the glucose residue can be oxidized; however, C-1 of the galactose residue cannot • Reducing sugars: Carbohydrates that be oxidized (they reduce the oxidizing agent) 24. 11 Disaccharides and Polysaccharides 34
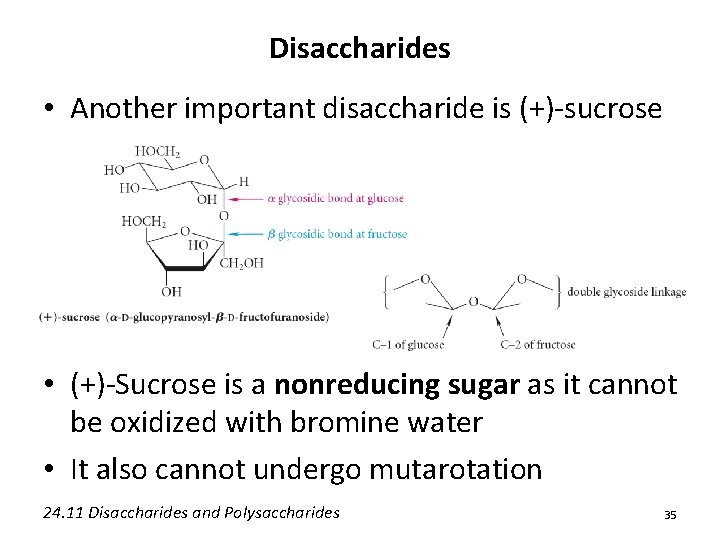
Disaccharides • Another important disaccharide is (+)-sucrose • (+)-Sucrose is a nonreducing sugar as it cannot be oxidized with bromine water • It also cannot undergo mutarotation 24. 11 Disaccharides and Polysaccharides 35

Polysaccharides • Sugars with many monosaccharide residues connected by glycosidic linkages are called polysaccharides • Cellulose is polymer of glucose 24. 11 Disaccharides and Polysaccharides 36
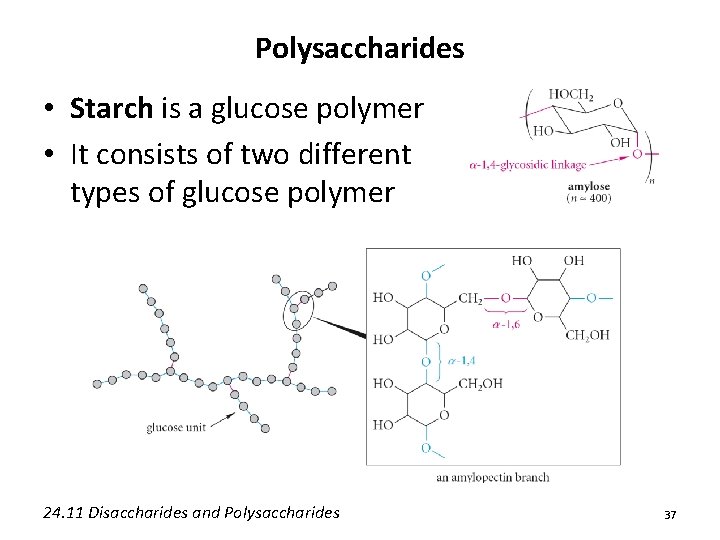
Polysaccharides • Starch is a glucose polymer • It consists of two different types of glucose polymer 24. 11 Disaccharides and Polysaccharides 37

Polysaccharides • Chitin is a polysaccharide that occurs widely in nature (e. g. , shells of lobsters and crabs) 24. 11 Disaccharides and Polysaccharides 38
- Slides: 38