Chapter 23 The first law of thermodynamics 23




































- Slides: 43

Chapter 23 The first law of thermodynamics

23 -1 Heat: energy in transit(传输) 1. “Heat is energy that flows between a system and its environment because of a temperature difference between them” We choose our sign convention so that Q is positive in the case that the internal energy of the system tends to be increased. Like other forms of energy, heat can be expressed in the SI unit of Joules (J).

2. Misconceptions(误解) about heat Neither heat nor work is an intrinsic property of a system. We cannot say that a system “contains” a certain amount of heat or work. They are not state functions. Both heat and work associated with a “thermodynamic process” 3. The understanding of heat in history See动画库力学夹5 -03作功与传热

23 -2 The transfer of heat 1. Thermal conduction Consider a thin slab of a homogeneous material of thickness and area A (Fig 23 -2). One face is held at T and the other at a somewhat higher constant temperature. Fig 23 -2 A T Q

Experiment shows that is (23 -1) H is the “rate of heat transfer” k is the “thermal conductivity” of the material A is the area of the slab is the thickness of the slab Considering the direction of H and infinitesimal thickness of the slab, we have: (23 -4)

Sample Problem 23 -2 A thin, cylindrical metal pipe is carrying steam at a temperature of Ts=1000 c. The pipe has a diameter of 5. 4 cm and is wrapped with a thickness of 5. 2 cm of fiberglass(玻璃丝) insulation. A length D=6. 2 m of the pipe passes through a room in which the temperature is TR=110 c. At what rate does heat energy pass through the insulation?

2. Convection(对流) 3. Radiation

Problem Two identical rectangular rods of metal are welded end to end as shown in Fig. a), and 10 J of heat flows through the rods in 2. 0 min. How long would it take for 30 J to flow throught the rods if they are welded as shown in Fig. b? a) b) 00 C 1000 C

23 -3 The first law of thermodynamics 1. For a thermodynamic system, internal energy is the only type of energy the system may have. The law of conservation of energy of the system can be expressed as (First law of thermodynamics) (23 -6) (i) Q is the energy transferred (as heat) between the system and its environment because of a temperature different.

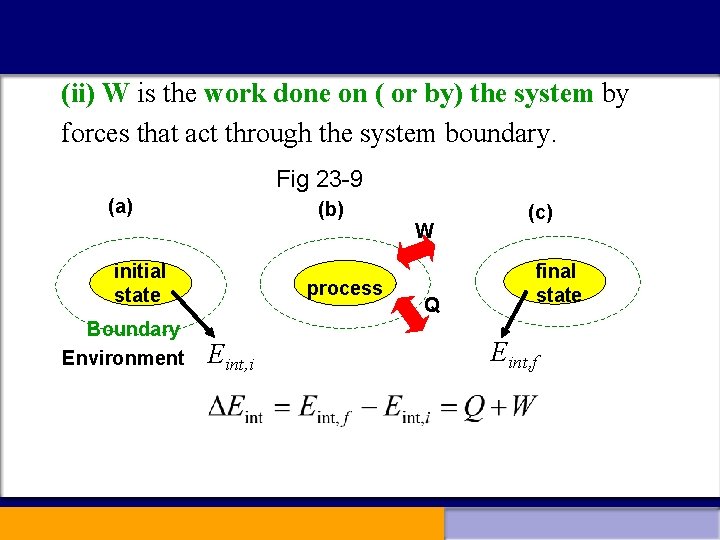
(ii) W is the work done on ( or by) the system by forces that act through the system boundary. Fig 23 -9 (a) (b) W initial state Boundary Environment process Eint, i Q (c) final state Eint, f

(iii) In any thermodynamic process between equilibrium state i and f, the quantity Q+W has the same value. This quantity is equal to the change in the internal energy. (iv) The first law of thermodynamics is a general result that is thought to apply to every process in nature that proceeds between equilibrium states.

23 -4 Heat capacity (热容)and specific heat(比热) 1. Heat capacity C: (23 -7) 2. Specific heat: The heat capacity per unit mass of a body (23 -8) The heat capacity (C) is characteristic of a particular object, but the specific heat (c) characterizes a kind of substance.

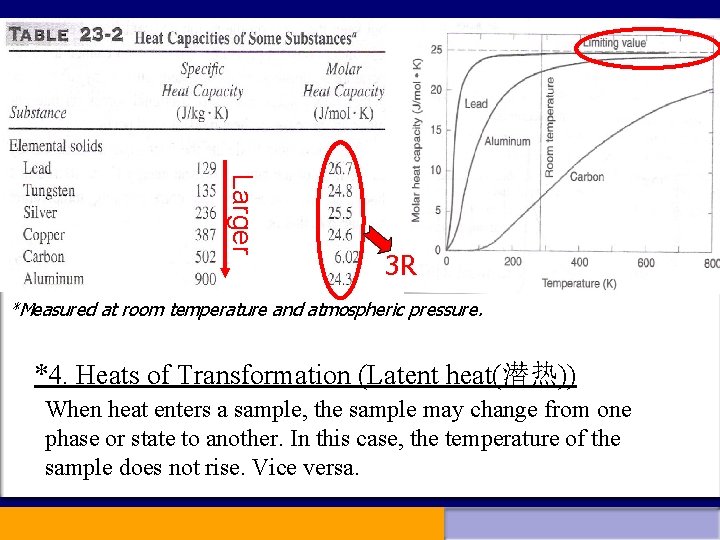
Usually both C and c depend on the temperature and condition under which the heat Q is added to the material. (23 -10) 3. Molar heat capacity If we multiply the specific heat by the molar mass M, we obtain the “molar heat capacity”. n--- the molar number

Larger 3 R *Measured at room temperature and atmospheric pressure. *4. Heats of Transformation (Latent heat(潜热)) When heat enters a sample, the sample may change from one phase or state to another. In this case, the temperature of the sample does not rise. Vice versa.

Sample Problem 23 -3 A cube of copper of mass mc=75 g is placed in an oven at a temperature of T 0=3120 C until it comes to thermal equilibrium. The cube is then dropped quickly into an insulated beaker(烧杯) containing a quantity of water of mass mw=220 g. The heat capacity of the beaker alone is Cb=190 J/K. Initially the water and the beaker are at a temperature of Ti=12. 00 c. What is the final equilibrium temperature Tf of the system consisting of the copper +water+beaker?

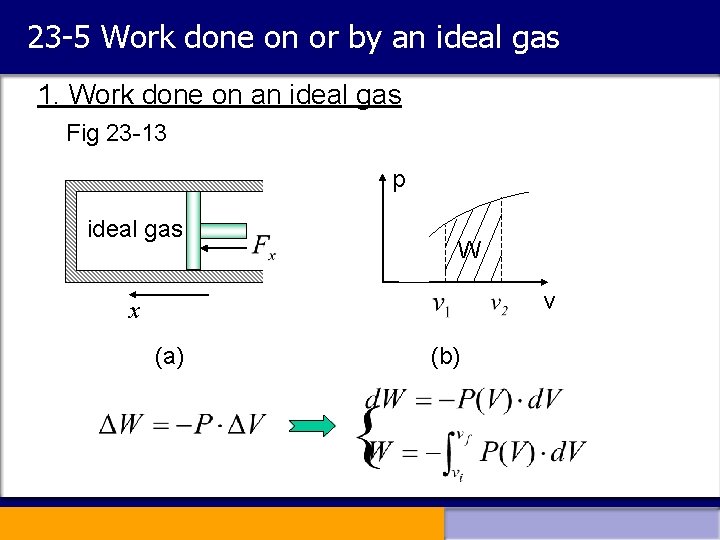
23 -5 Work done on or by an ideal gas 1. Work done on an ideal gas Fig 23 -13 p ideal gas W v x (a) (b)

Work done on gas in a more general form: (23 -15) (a) If PV relationship is known, the work done on the gas is equal to the area under the curve representing the process. (b) The pressure force is not a conservative force. P Fig 23 -14 B D A C V Two paths: A B D A C D

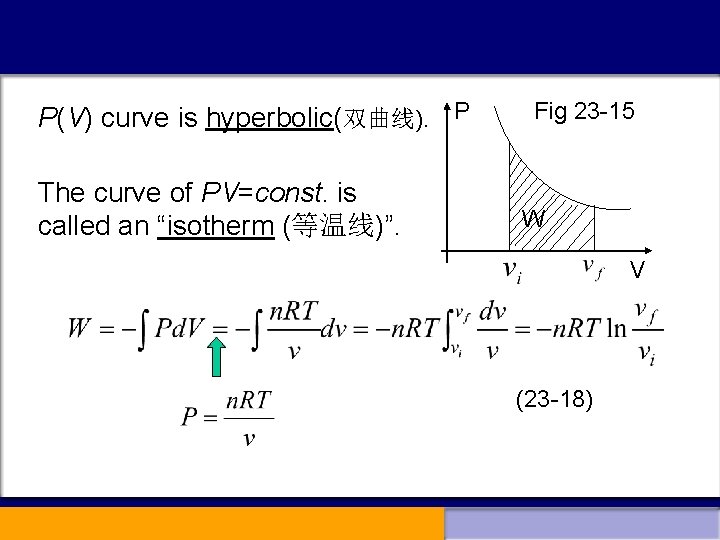
2. Several typical thermal processes (a). Work done at constant volume (V is const. ) p p W 0 (23 -16) W=0 v v (b). Work done at constant pressure (P is const. ) p (23 -17) v (c). Work done at constant temperature (T is const. ) namely “isothermal (等温) process”,

P(V) curve is hyperbolic(双曲线). P The curve of PV=const. is called an “isotherm (等温线)”. Fig 23 -15 W V (23 -18)

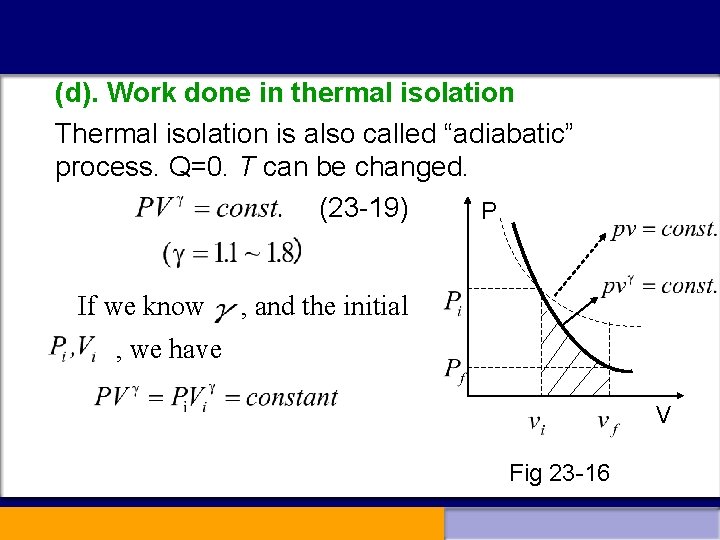
(d). Work done in thermal isolation Thermal isolation is also called “adiabatic” process. Q=0. T can be changed. (23 -19) P If we know , and the initial , we have V Fig 23 -16

(23 -20) We can now find the adiabatic work: (23 -21) By further using , Eq(23 -21) becomes (23 -22)

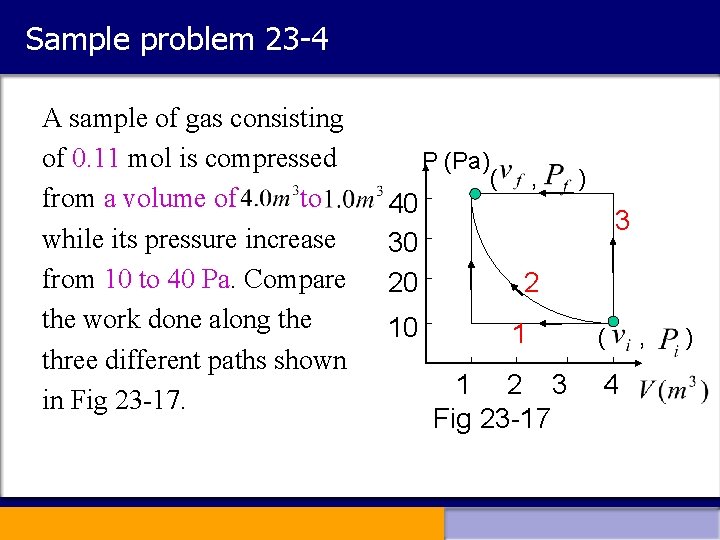
Sample problem 23 -4 A sample of gas consisting of 0. 11 mol is compressed from a volume of to while its pressure increase from 10 to 40 Pa. Compare the work done along the three different paths shown in Fig 23 -17. P (Pa) 40 30 20 10 ( , ) 3 2 1 1 2 3 Fig 23 -17 ( 4 , )

Solution: Path 1 Path 2 represents an isothermal process Path 3

23 -6 The internal energy of an ideal gas 1. Internal energy of ideal monatomic gas Translational kinetic energy is (23 -23) No potential energy. No rotational kinetic energy. So is the entire store of internal energy. The total internal energy of n moles of an ideal monatomic gas is (23 -24)

y 2. Internal energy of molecule consisting of two particles such as Fig 23 -18 d z X The total kinetic energy of a diatomic molecule is (23 -26) U=0 Etot=K

3. Degrees of freedom The five terms in Eq 23 -26 represent independent ways in which a molecule can absorb energy and are called ‘degree of freedom”. 4. Equipartition of energy theorem (能量均分定理) Maxwell derived a theorem: “When the number of molecule is large, the average energy per molecule is for each independent degree of freedom” See动画库力学夹4 -13能量均分定理

5. The internal energy for different kinds of ideal gases: (a) Monatomic ideal gas (23 -27) (b) Diatomic gas (23 -28) (c) A polyatomic gas generally has six degrees of freedom, (23 -29) (d) If the molecule is not a rigid one, there also oscillating degrees of freedom. Notes: 1* here dependents only on T and degree of freedom. 2* Equipartition of energy theorem only apply in classical physics.

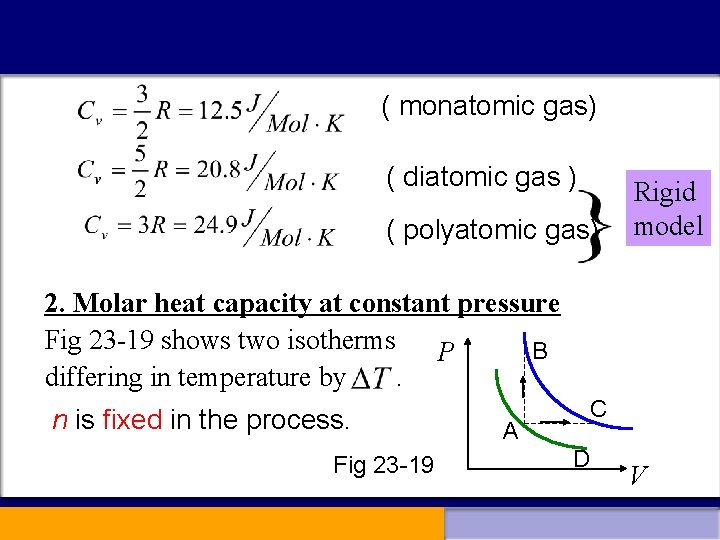
23 -7 Heat capacities of an ideal gas 1. Molar heat capacity at constant volume Constant volume (23 -30) If represents the molar heat capacity at constant volume, (23 -31) Using Eqs(23 -27), (23 -28), (23 -29), we have

( monatomic gas) ( diatomic gas ) ( polyatomic gas) Rigid model 2. Molar heat capacity at constant pressure Fig 23 -19 shows two isotherms P B differing in temperature by. n is fixed in the process. Fig 23 -19 C A D V

Path AB is the constant-volume process; P Path AC is a constant pressure process. B A C The change in internal energy is the same for path AB V and AC. Along AB: Along AC: (23 -35) (23 -36) (23 -27)

Using the ideal gas law (23 -38) or (23 -39) ( monatomic gas) (23 -40) ( diatomic gas ) (23 -41) ( polyatomic gas) (23 -42)

Sample problem 23 -7 An ideal gas with 0. 11 mole begins at the initial point with volume and pressure. Let us compress the gas adiabatically until volume is. Find the change in internal energy of the gas, assuming it to be helium ( a monatomic gas with ).

Solution: Using Eq(23 -19) The initial and final temperature are The change in internal energy is

23 -8 Applications of the first law of thermodynamics (for ideal gases) 1. Adiabatic process ( ) Let’s derive: (23 -19) (23 -47) can always be expressed as: (23 -48) (23 -49)

Take the ratio between Eqs(23 -49) and (23 -48) (23 -51)

Since i and f are arbitrary points, then we obtain (23 -52) We can rewrite these results in terms of temperature, using the ideal gas equation of state (PV=n. RT) (23 -53) or (23 -54) 2. Isothermal process or (23 -55)

3. Constant-volume processes (23 -56) In this case all the heat that enters the gas (Q>0) is stored as internal energy. A P 4. Cyclical processes In a cyclical process, the system 1 restores to its initial state (A-B-C-A). B or (23 -57) 3 W>0 2 Fig 23 -21 C

5. Free expansion Stopcock ( The gas in Fig(23 -22) is initially insulating 隔离阀) in one side of the container, and when the stopcock is opened, the gas vacu. expands into the previously evacuated half. In this process, no work is done. The container is insulated, so the process is adiabatic. Fig 23 -22

Hence (23 -58) The initial state ( all gas on one side ) is an equilibrium state, as is the final state. But the intermediate process during intial and final state is nonequilibrium. T and P do not have unique value, and we can not plot this process on a PV diagram.

Summary of typical thermal processes:

Sample problem 23 -9 The cycle shown in Fig 23 -21 consists of three processes, starting at point A: A->B: constant-volume; P A B->C: constant-pressure; C->A: isothermal compression. 1 3 n=0. 75 mol diatomic gas with B 2 , , . Find Q, W and. Fig 23 -21 C V

Solution: at point C: process 1 (A->B)

process 2 (B->C) For the cycle, we have: process 3 C->A