Chapter 22 Enzymes Cyclooxygenase Aspirin Inhibits Cycloxygenase Work
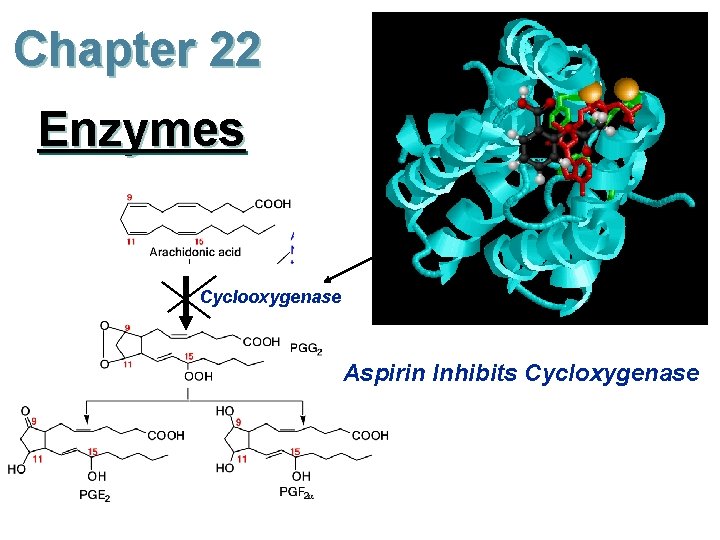
Chapter 22 Enzymes Cyclooxygenase Aspirin Inhibits Cycloxygenase
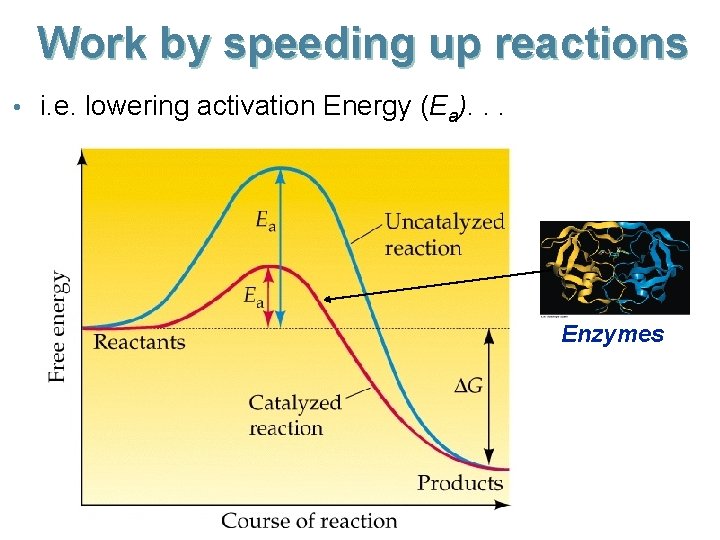
Work by speeding up reactions • i. e. lowering activation Energy (Ea). . . Enzymes
Enzyme Catalysis • Enzyme: a biological catalyst. • ~all enzymes are proteins. • increase rate of a reaction by 109 - 1020 over vs uncatalyzed reaction. • Some catalyze the reaction of only one compound. • Some are stereospecific; for example, enzymes that catalyze the reactions of only L-amino acids (natural). • Some catalyze reactions of specific types of compounds or bonds; e. g. trypsin catalyzes hydrolysis of peptide bonds of Lys and Arg.

Classification of Enzymes • Enzymes are commonly named after the reaction or reactions they catalyze. • example: lactate dehydrogenase, alchohol dehydrogenase. • Enzymes are classified into six major groups. • Oxidoreductases: oxidation-reduction reactions. • Transferases: group transfer reactions. • Hydrolases: hydrolysis reactions. • Lyases: addition of groups to a double bond, or removal of groups to create a double bond. • Isomerases: isomerization reactions. • Ligases: the joining to two molecules.
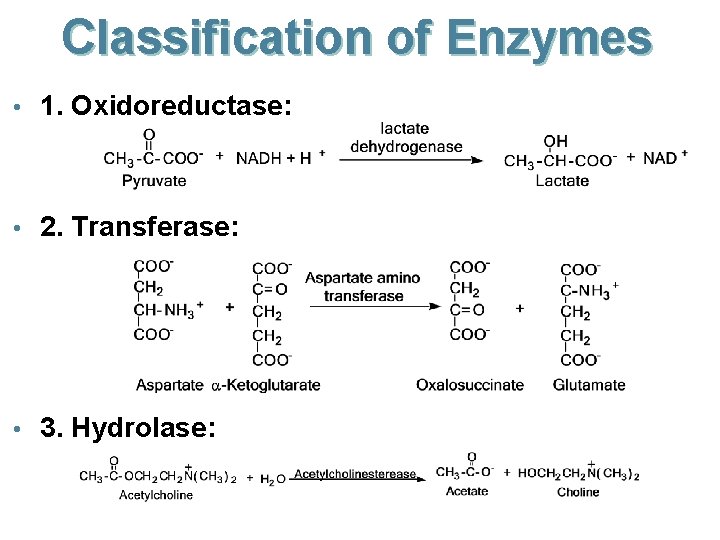
Classification of Enzymes • 1. Oxidoreductase: • 2. Transferase: • 3. Hydrolase:
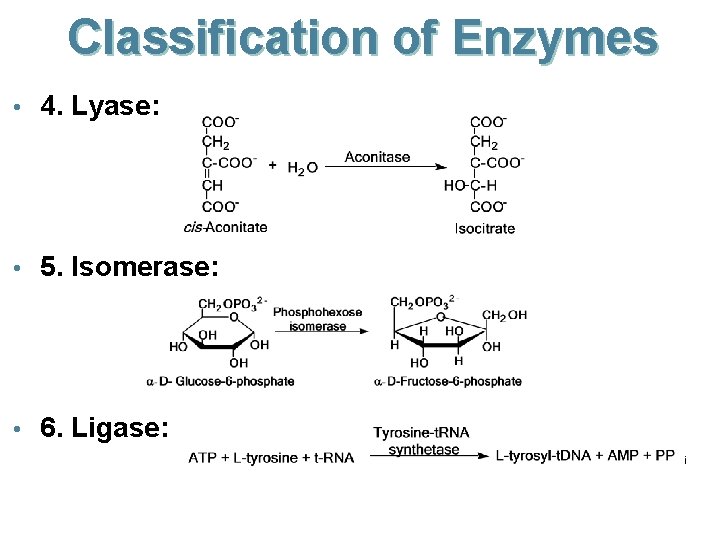
Classification of Enzymes • 4. Lyase: • 5. Isomerase: • 6. Ligase:
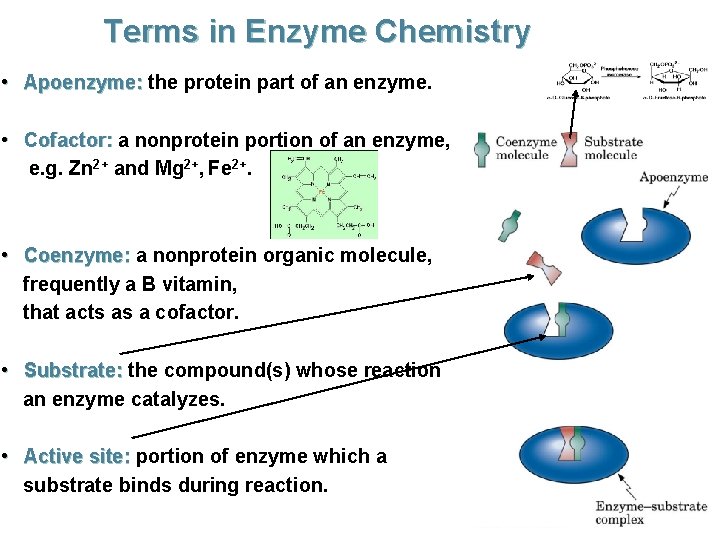
Terms in Enzyme Chemistry • Apoenzyme: the protein part of an enzyme. • Cofactor: a nonprotein portion of an enzyme, e. g. Zn 2+ and Mg 2+, Fe 2+. • Coenzyme: a nonprotein organic molecule, frequently a B vitamin, that acts as a cofactor. • Substrate: the compound(s) whose reaction an enzyme catalyzes. • Active site: portion of enzyme which a substrate binds during reaction.
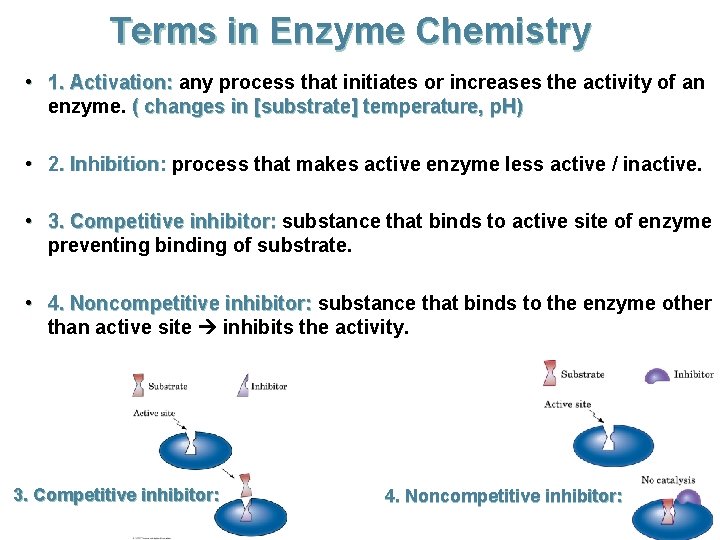
Terms in Enzyme Chemistry • 1. Activation: any process that initiates or increases the activity of an enzyme. ( changes in [substrate] temperature, p. H) • 2. Inhibition: process that makes active enzyme less active / inactive. • 3. Competitive inhibitor: substance that binds to active site of enzyme preventing binding of substrate. • 4. Noncompetitive inhibitor: substance that binds to the enzyme other than active site inhibits the activity. 3. Competitive inhibitor: 4. Noncompetitive inhibitor:

Challenge Question • What is the difference between reversible and Irreversible non competitive inhibition? 4. Noncompetitive inhibitor:

Enzyme Activity • a measure of how much a reaction rate is increased. • We examine how the rate of an enzyme-catalyzed reaction is effected by: • enzyme concentration • substrate concentration • temperature • p. H
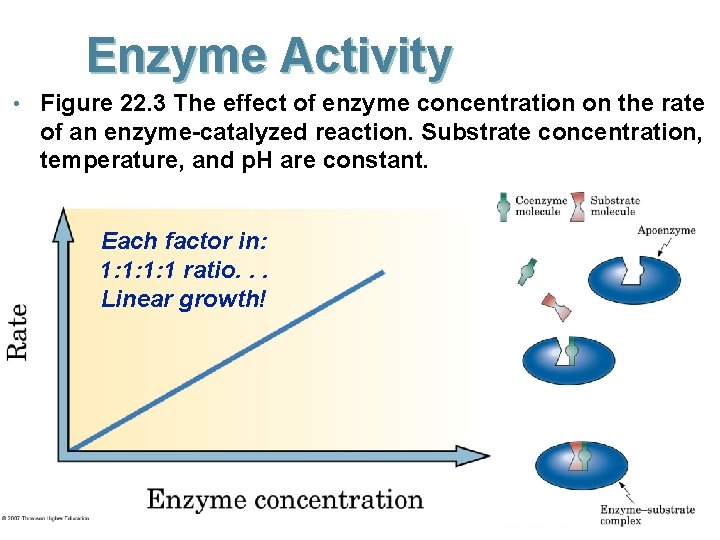
Enzyme Activity • Figure 22. 3 The effect of enzyme concentration on the rate of an enzyme-catalyzed reaction. Substrate concentration, temperature, and p. H are constant. Each factor in: 1: 1 ratio. . . Linear growth!
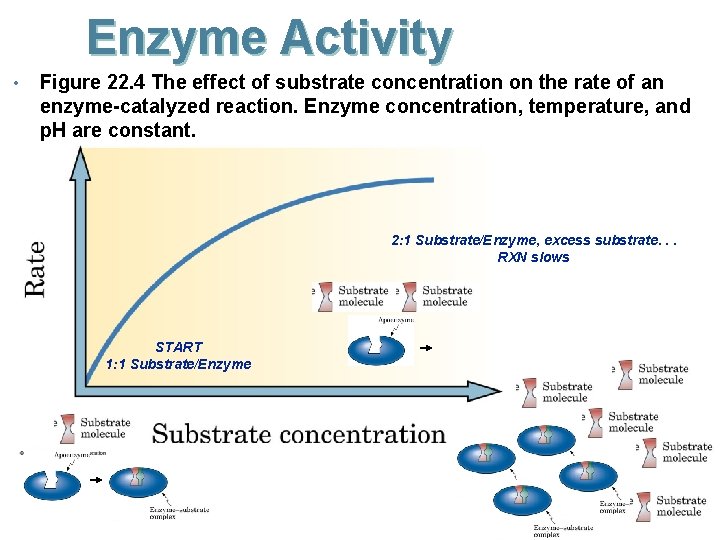
Enzyme Activity • Figure 22. 4 The effect of substrate concentration on the rate of an enzyme-catalyzed reaction. Enzyme concentration, temperature, and p. H are constant. 2: 1 Substrate/Enzyme, excess substrate. . . RXN slows START 1: 1 Substrate/Enzyme
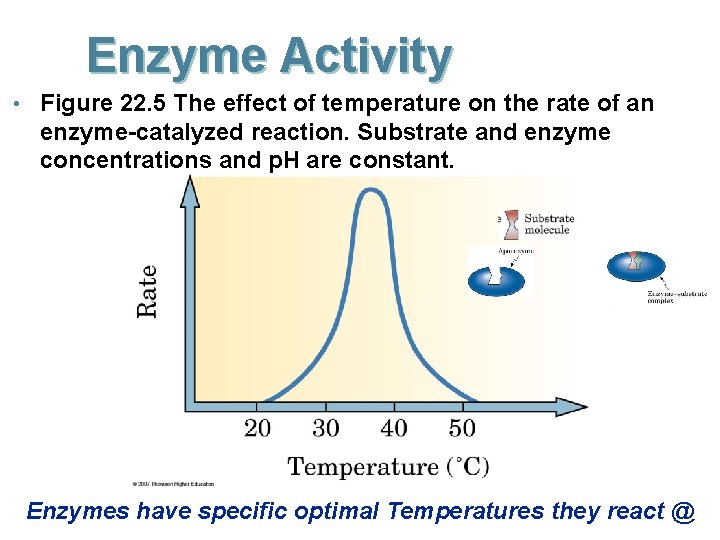
Enzyme Activity • Figure 22. 5 The effect of temperature on the rate of an enzyme-catalyzed reaction. Substrate and enzyme concentrations and p. H are constant. Enzymes have specific optimal Temperatures they react @
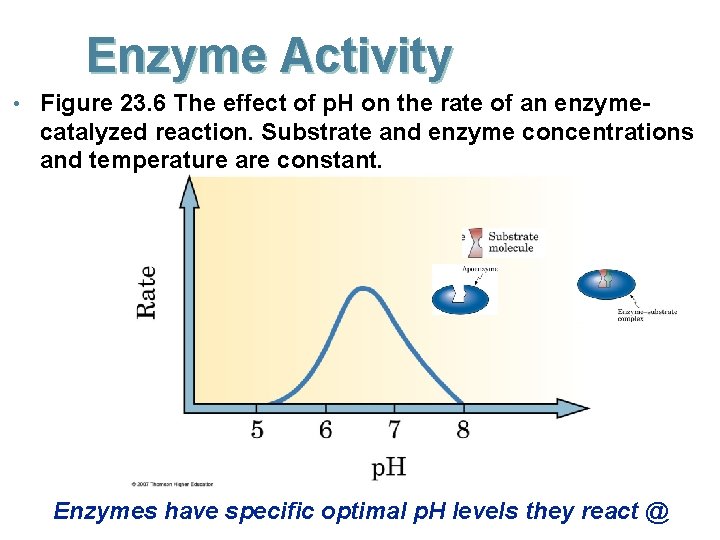
Enzyme Activity • Figure 23. 6 The effect of p. H on the rate of an enzyme- catalyzed reaction. Substrate and enzyme concentrations and temperature are constant. Enzymes have specific optimal p. H levels they react @

Confirming your knowledge The activity of pepsin was measured at various p. H values. When the temp, concentration of pepsin and substrate were held constant the following data was obtained: p. H Enzyme reaction Activity 1. 0 0. 5 1. 5 2. 6 2. 0 4. 8 3. 0 2. 0 4. 0 0. 4 5. 0 0. 0 What is the optimal p. H? Predict the activity of pepsin in the blood (physiological p. H ~7. 4)

Mechanism of Action • Lock-and-key model of enzyme mechanism. • The enzyme is a rigid three-dimensional body. • The enzyme surface contains the active site.
- Slides: 16