Chapter 22 Carboxylic Acids and Their DerivativesNucleophilic Acyl

Chapter 22 Carboxylic Acids and Their Derivatives—Nucleophilic Acyl Substitution 1
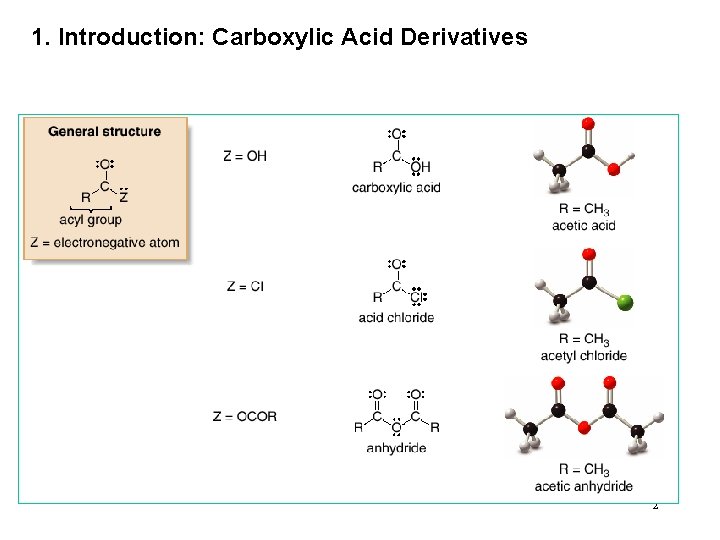
1. Introduction: Carboxylic Acid Derivatives 2
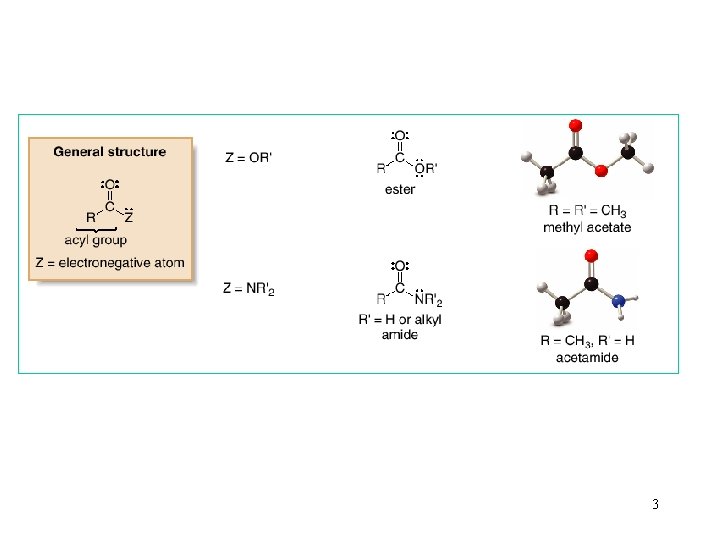
3
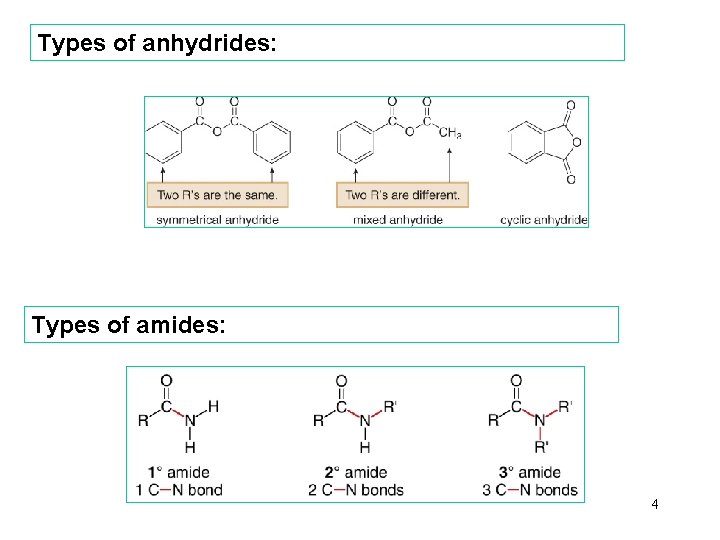
Types of anhydrides: Types of amides: 4
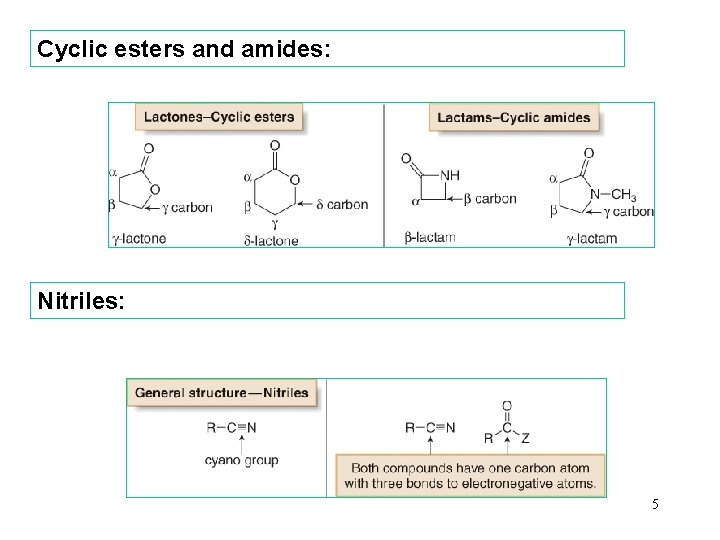
Cyclic esters and amides: Nitriles: 5
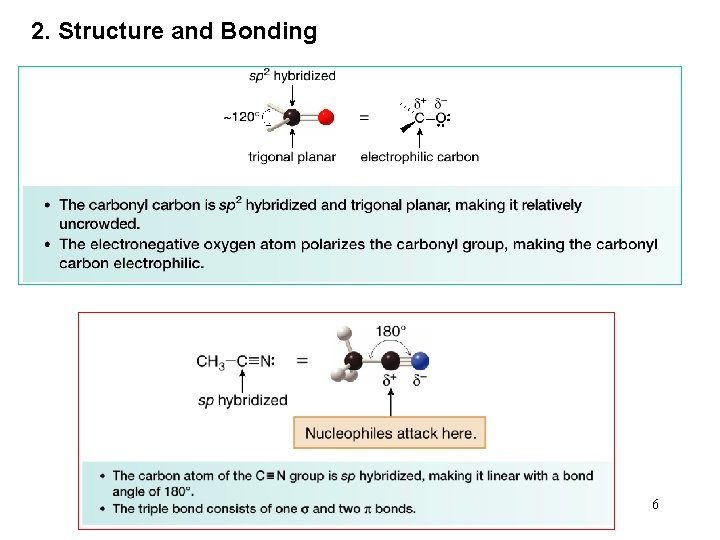
2. Structure and Bonding 6
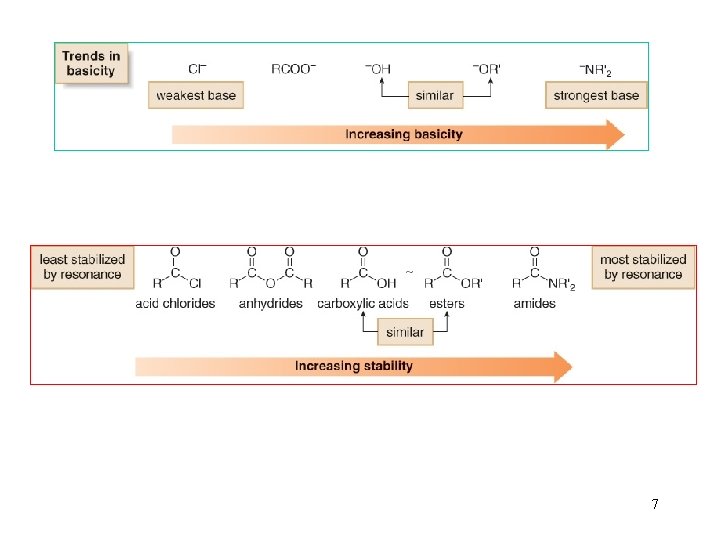
7
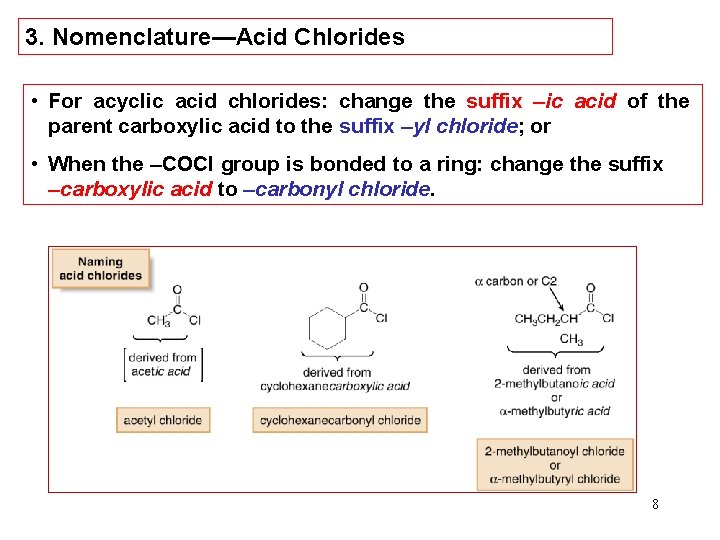
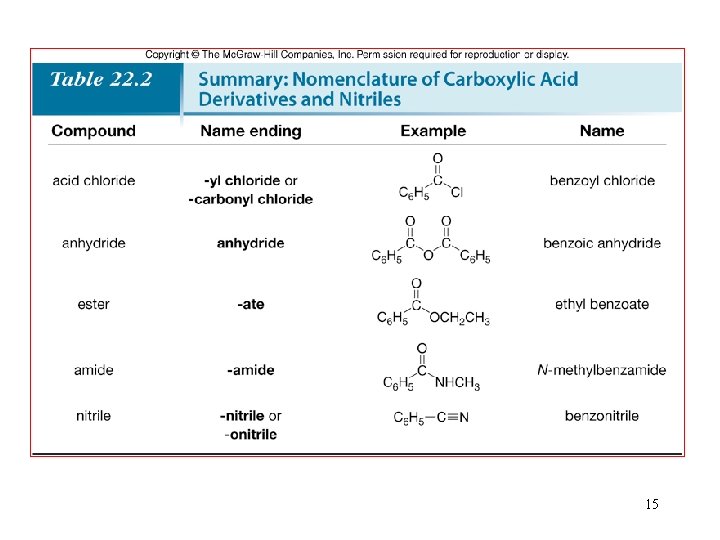
3. Nomenclature—Acid Chlorides • For acyclic acid chlorides: change the suffix –ic acid of the parent carboxylic acid to the suffix –yl chloride; or • When the –COCl group is bonded to a ring: change the suffix –carboxylic acid to –carbonyl chloride. 8
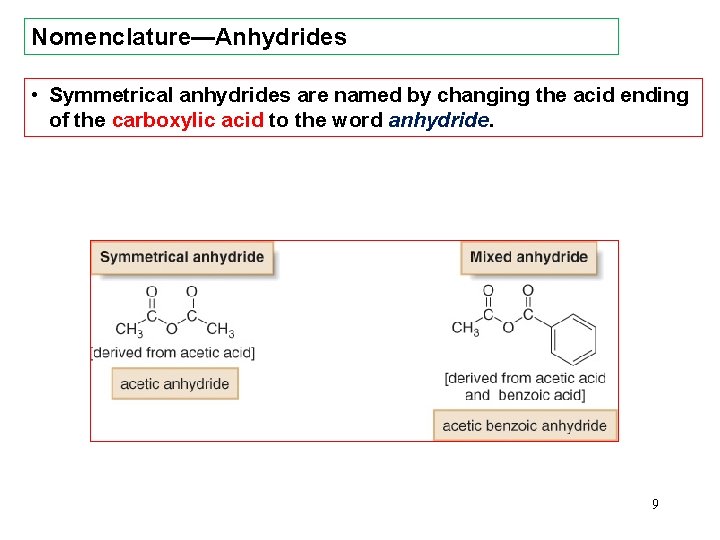
Nomenclature—Anhydrides • Symmetrical anhydrides are named by changing the acid ending of the carboxylic acid to the word anhydride. 9
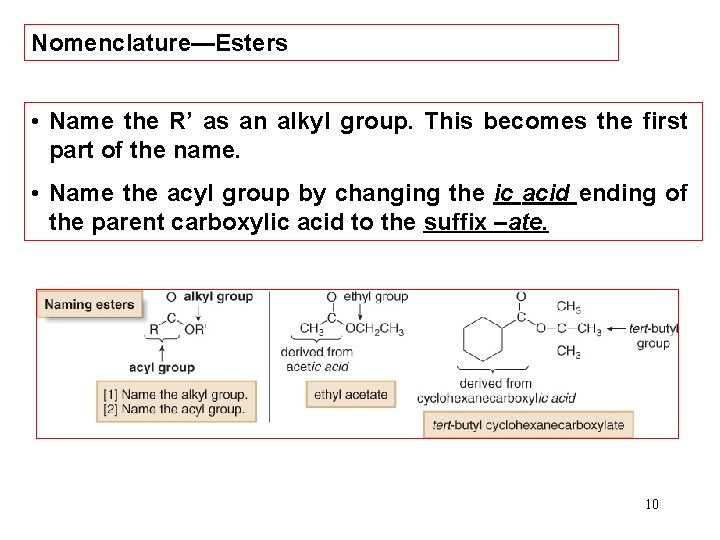
Nomenclature—Esters • Name the R’ as an alkyl group. This becomes the first part of the name. • Name the acyl group by changing the ic acid ending of the parent carboxylic acid to the suffix –ate. 10
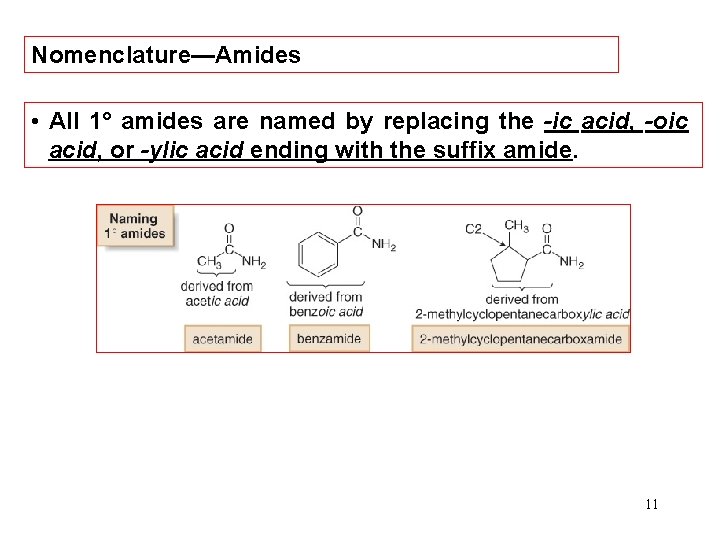
Nomenclature—Amides • All 1° amides are named by replacing the -ic acid, -oic acid, or -ylic acid ending with the suffix amide. 11
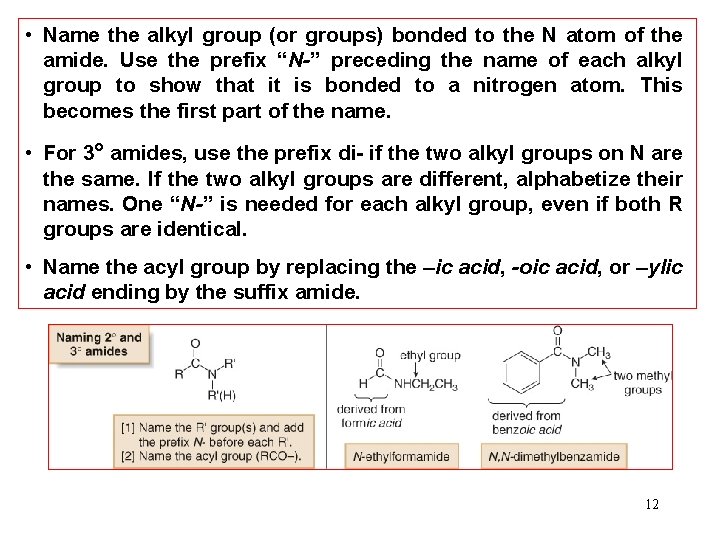
• Name the alkyl group (or groups) bonded to the N atom of the amide. Use the prefix “N-” preceding the name of each alkyl group to show that it is bonded to a nitrogen atom. This becomes the first part of the name. • For 3° amides, use the prefix di- if the two alkyl groups on N are the same. If the two alkyl groups are different, alphabetize their names. One “N-” is needed for each alkyl group, even if both R groups are identical. • Name the acyl group by replacing the –ic acid, -oic acid, or –ylic acid ending by the suffix amide. 12

Nomenclature—Nitriles • In contrast to the carboxylic acid derivatives, nitriles are named as alkane derivatives. • Find the longest chain that contains the CN and add the word nitrile to the name of parent alkane. Number the chain to put CN at C 1, but omit this number from the name. • Common names of nitriles are derived from the names of the carboxylic acid having the same number of carbon atoms by replacing the –ic acid ending of the carboxylic acid with the suffix –onitrile. • When the CN is named as a substituent it is called a 13 cyano group.
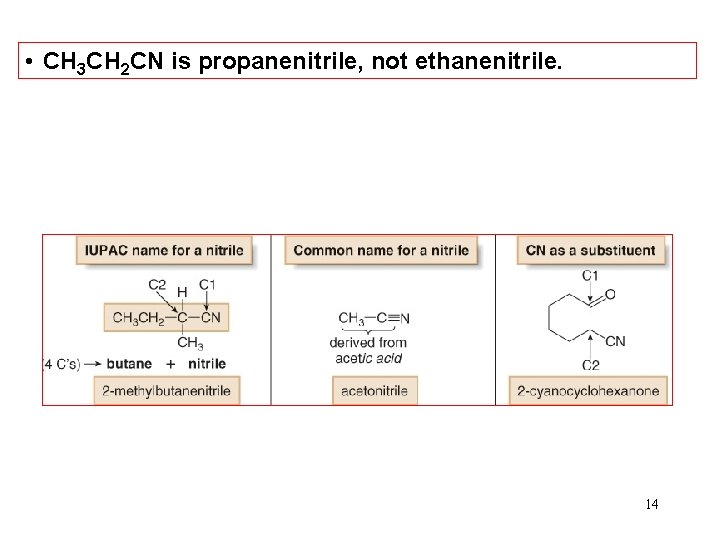
• CH 3 CH 2 CN is propanenitrile, not ethanenitrile. 14
15
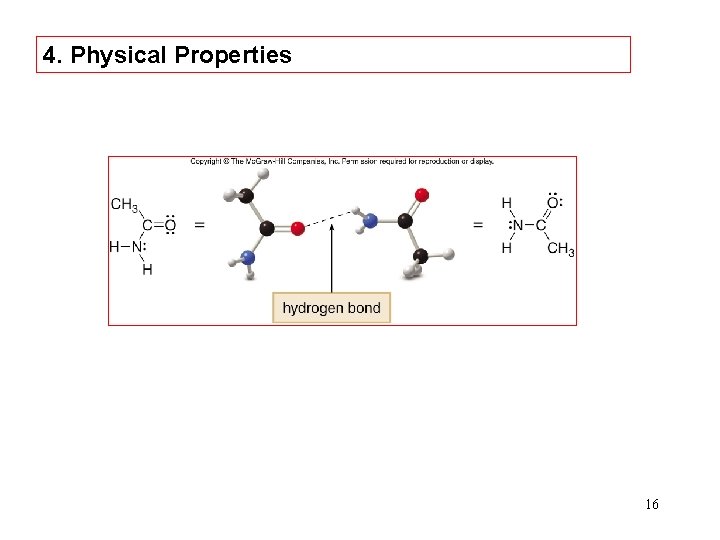
4. Physical Properties 16
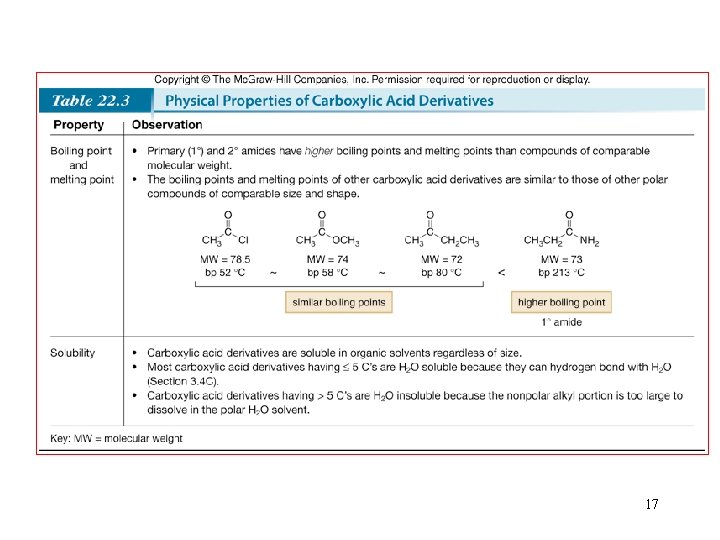
17
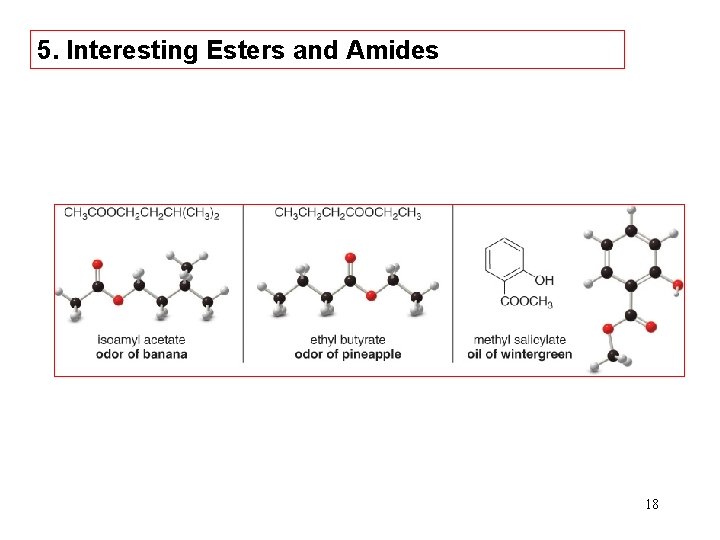
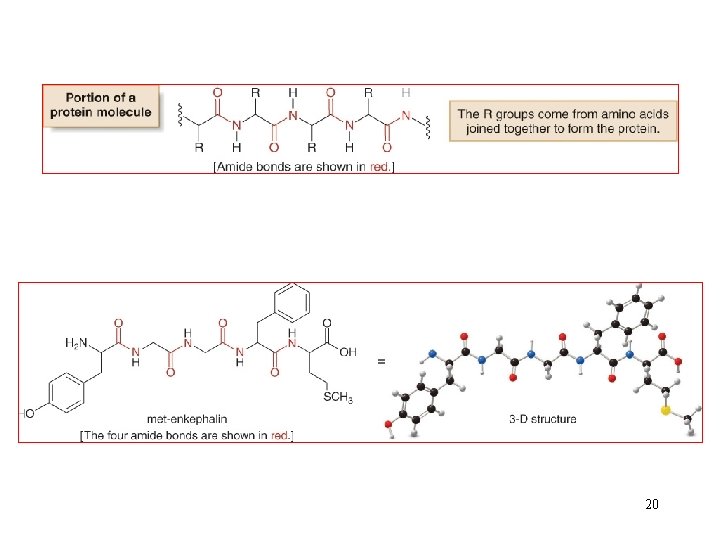
5. Interesting Esters and Amides 18
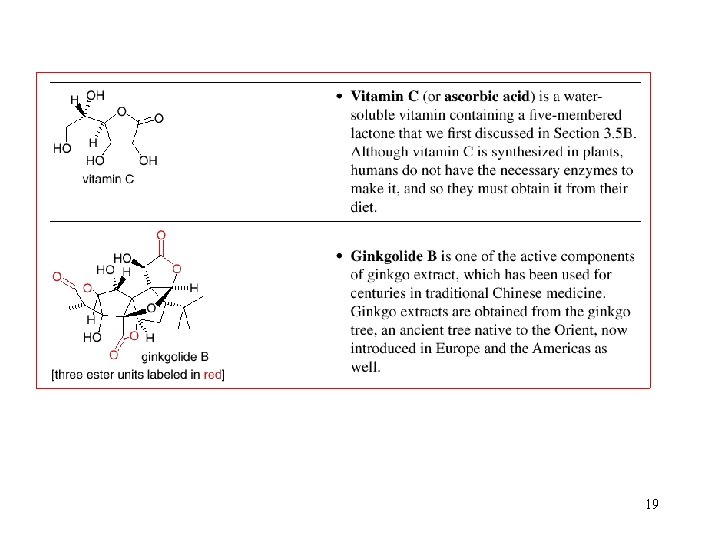
19
20
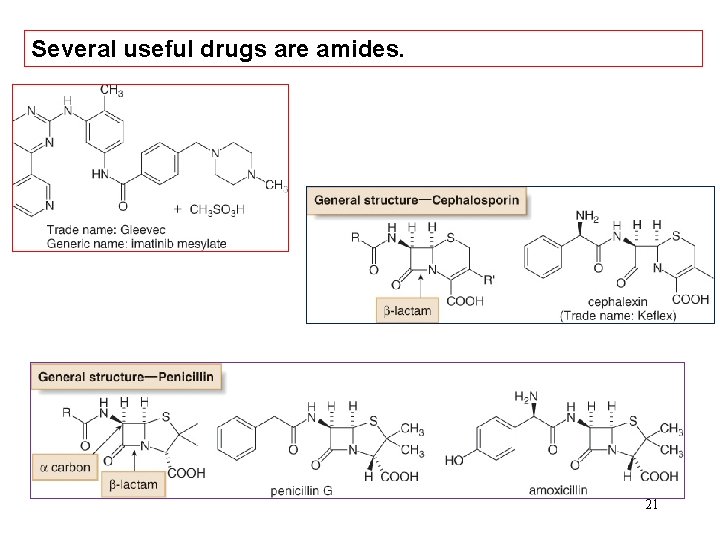
Several useful drugs are amides. 21
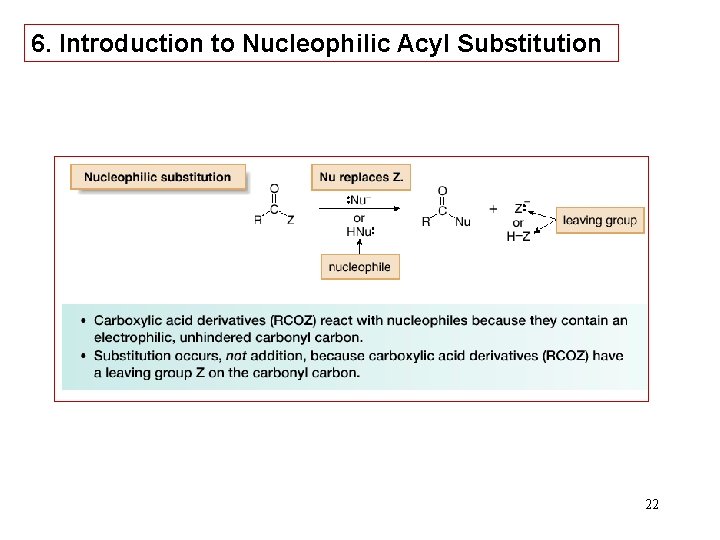
6. Introduction to Nucleophilic Acyl Substitution 22
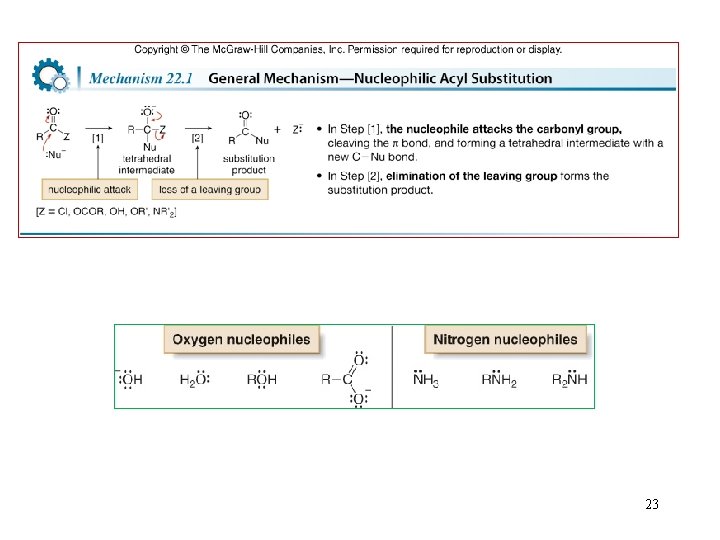
23
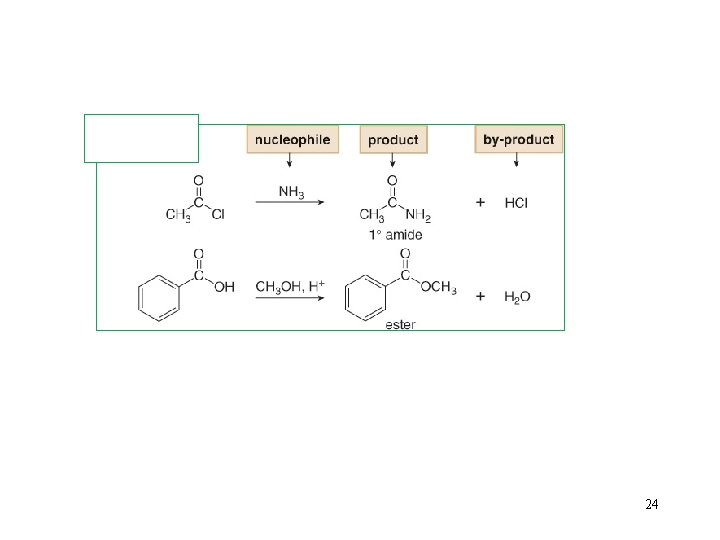
24
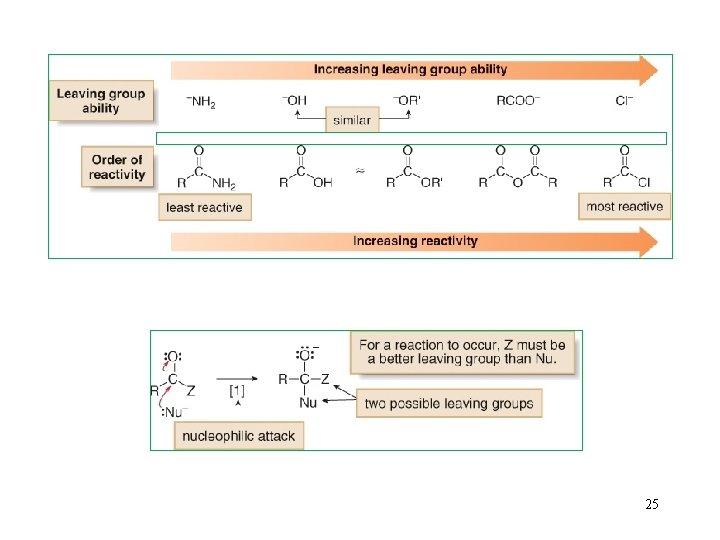
25
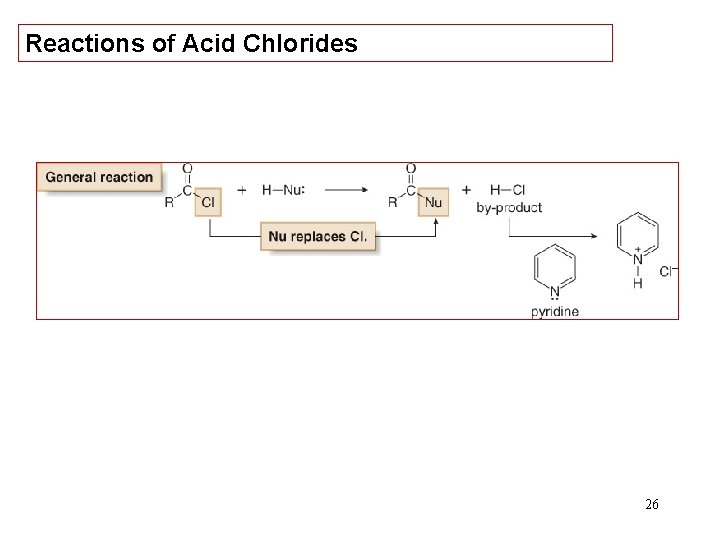
Reactions of Acid Chlorides 26
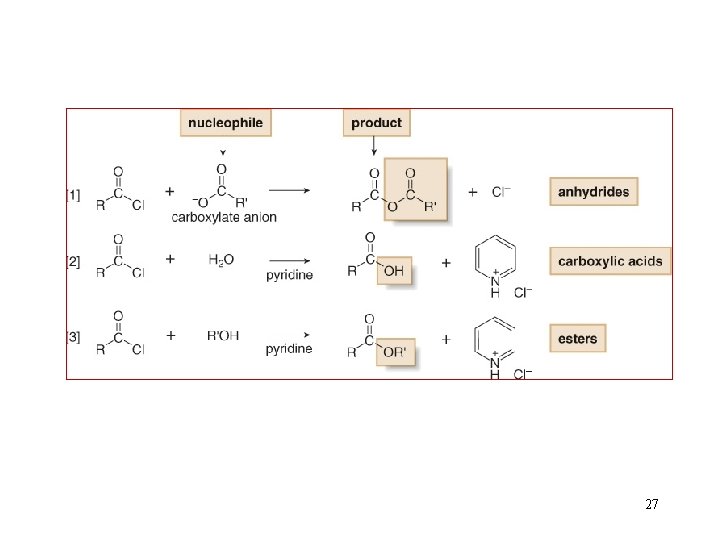
27
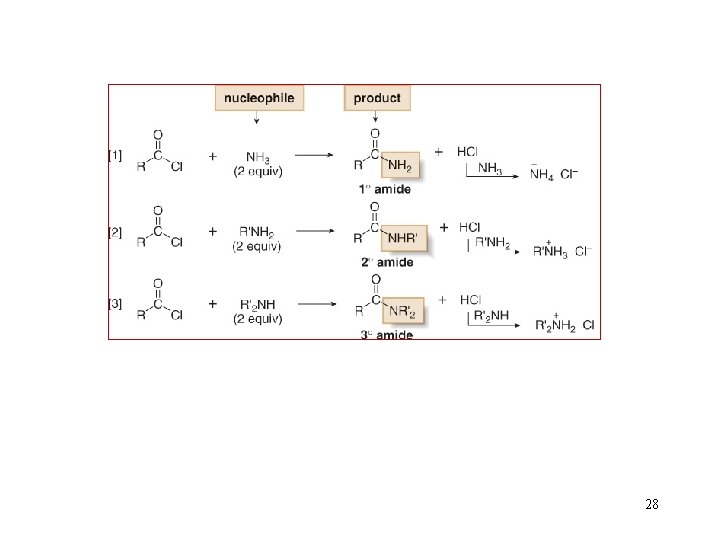
28
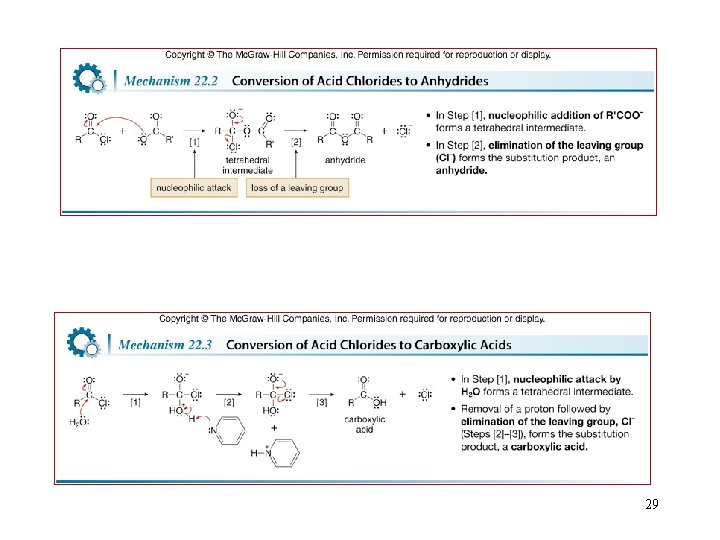
29

Reactions of Anhydrides 30
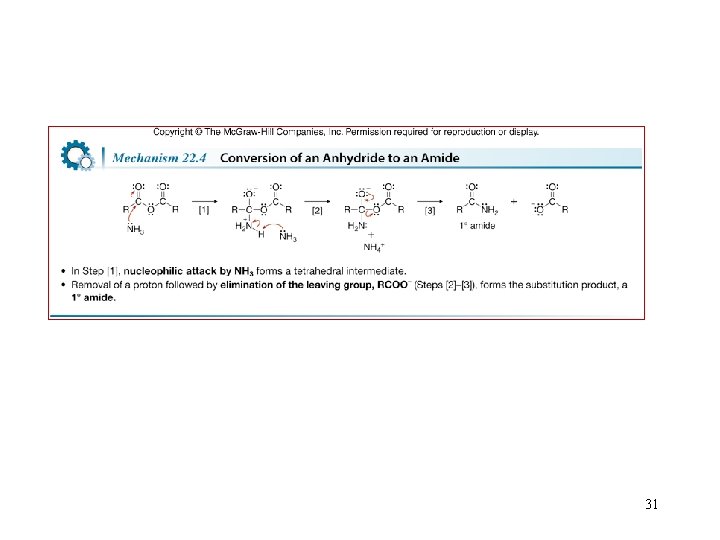
31
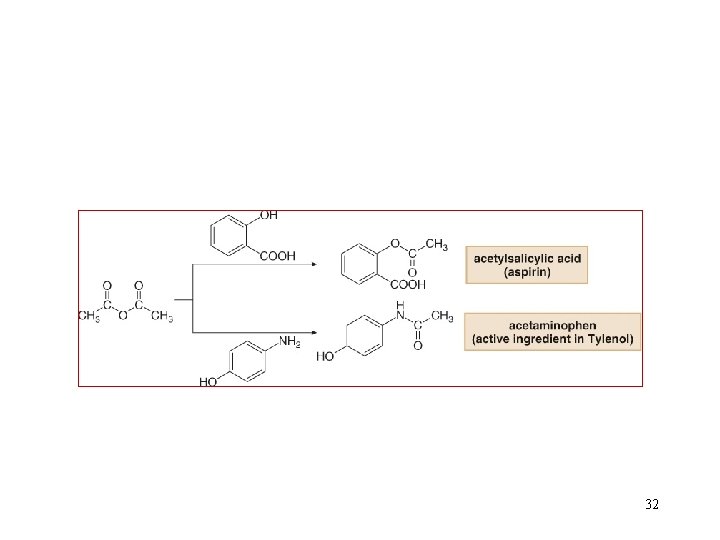
32
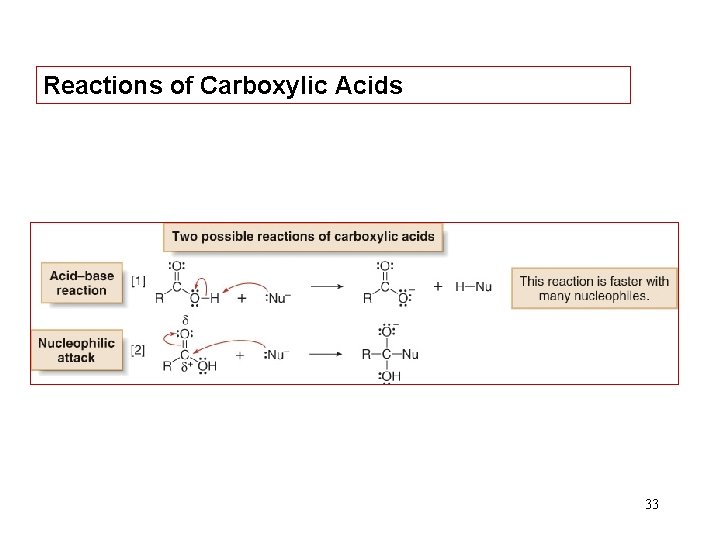
Reactions of Carboxylic Acids 33
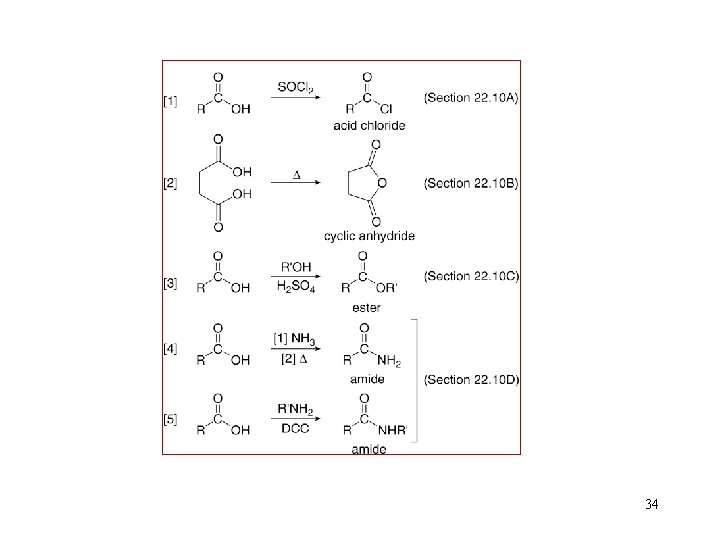
34
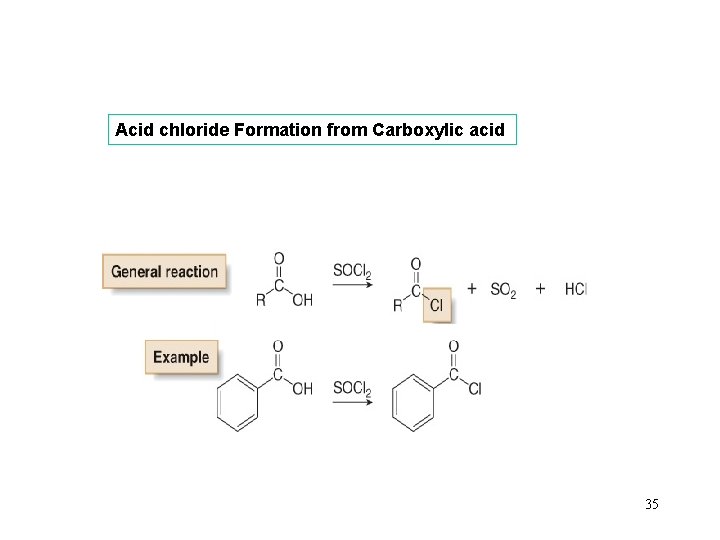
Acid chloride Formation from Carboxylic acid 35
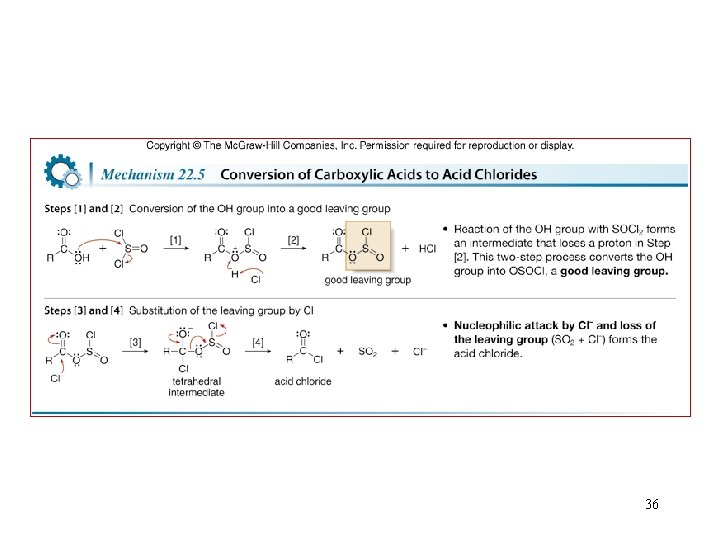
36
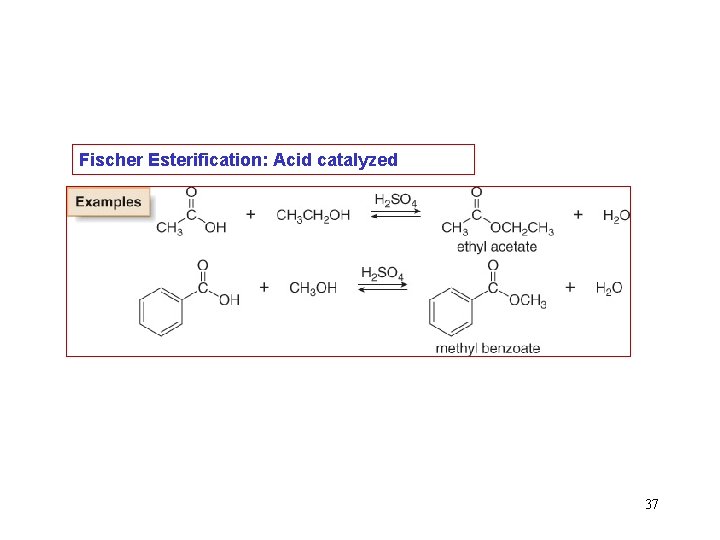
Fischer Esterification: Acid catalyzed 37
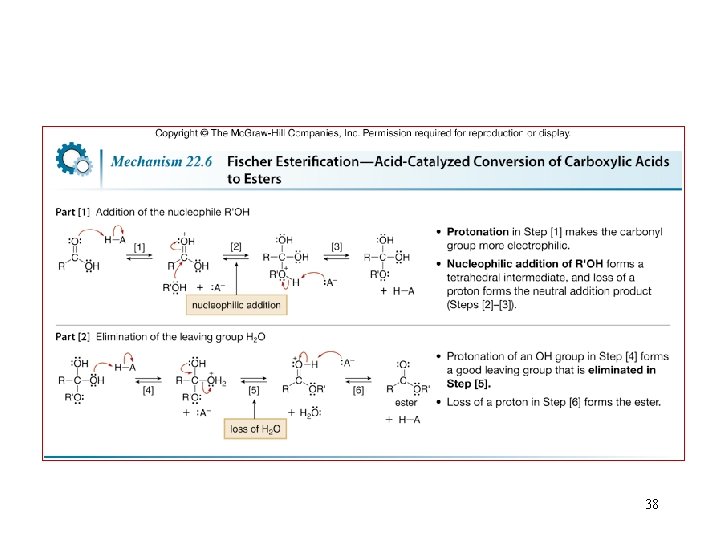
38
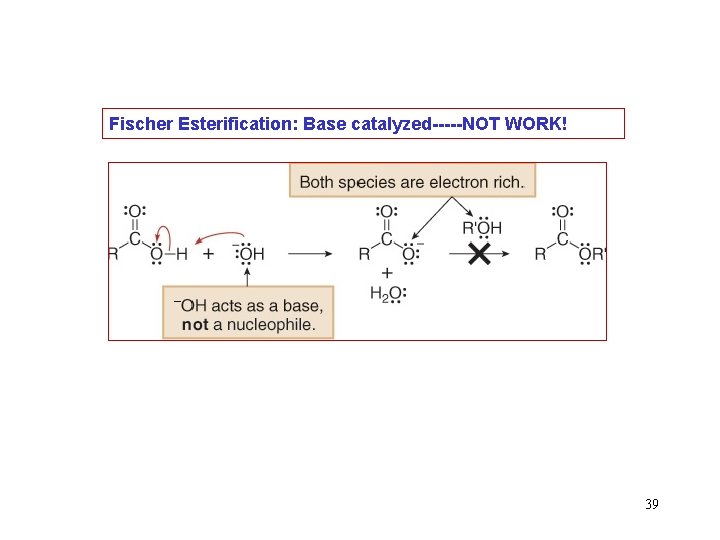
Fischer Esterification: Base catalyzed-----NOT WORK! 39
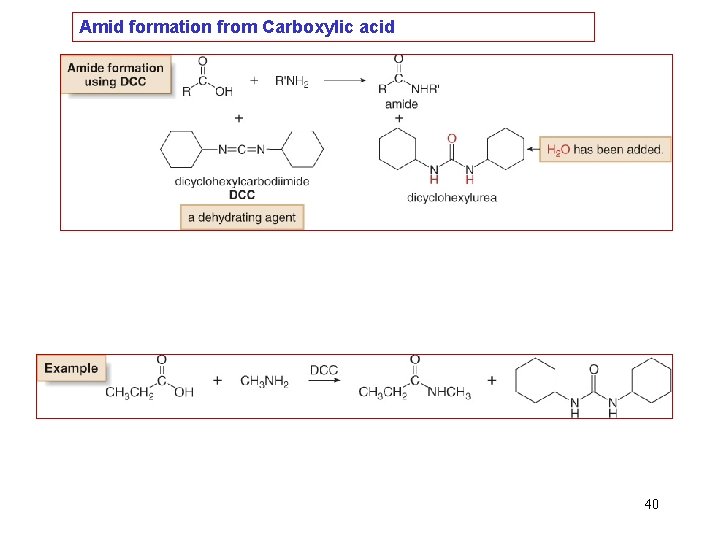
Amid formation from Carboxylic acid 40
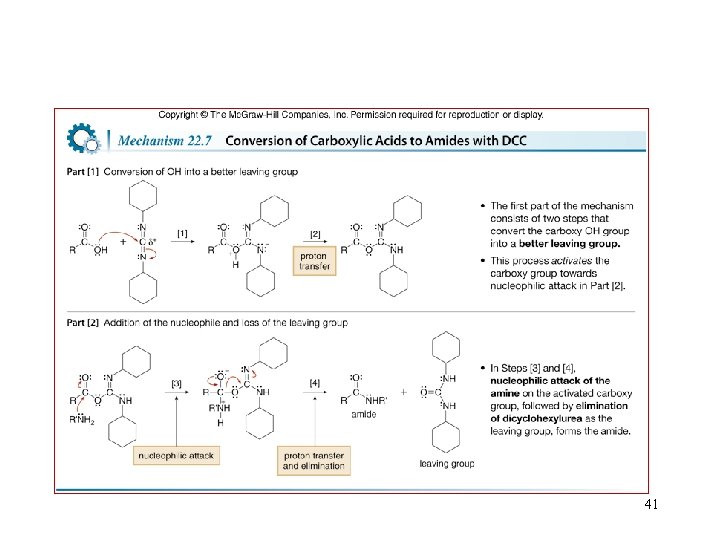
41
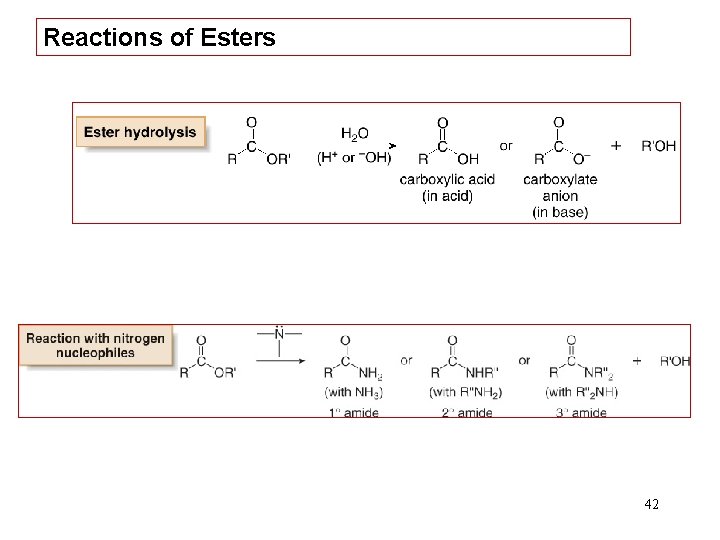
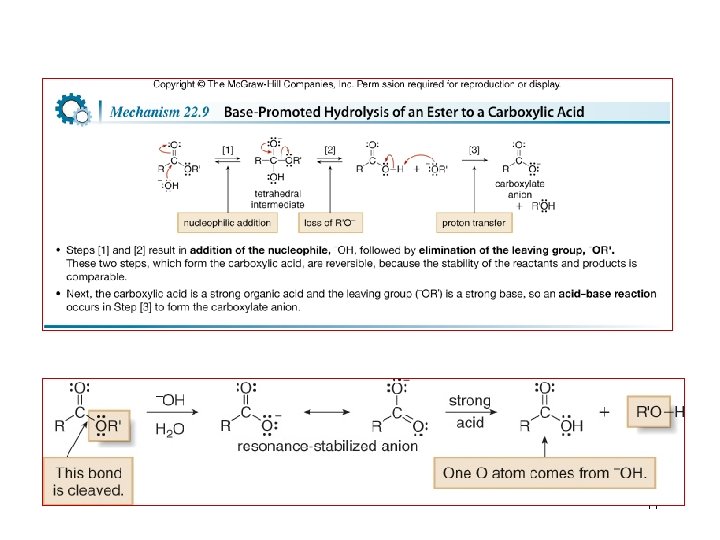
Reactions of Esters 42
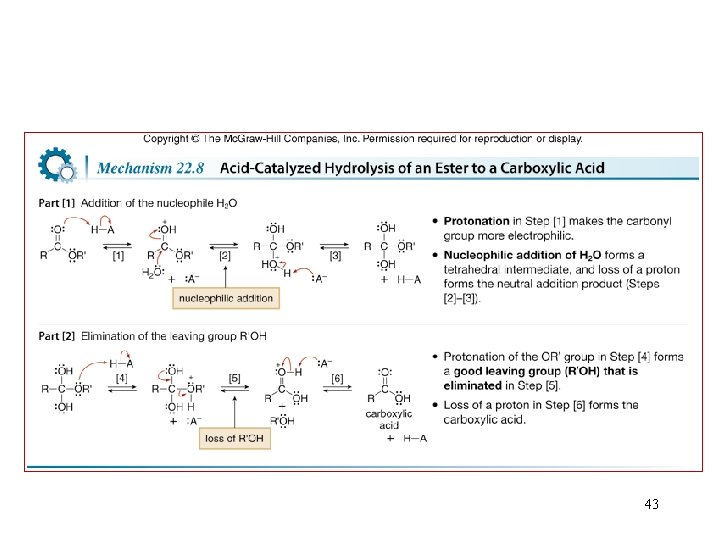
43
44
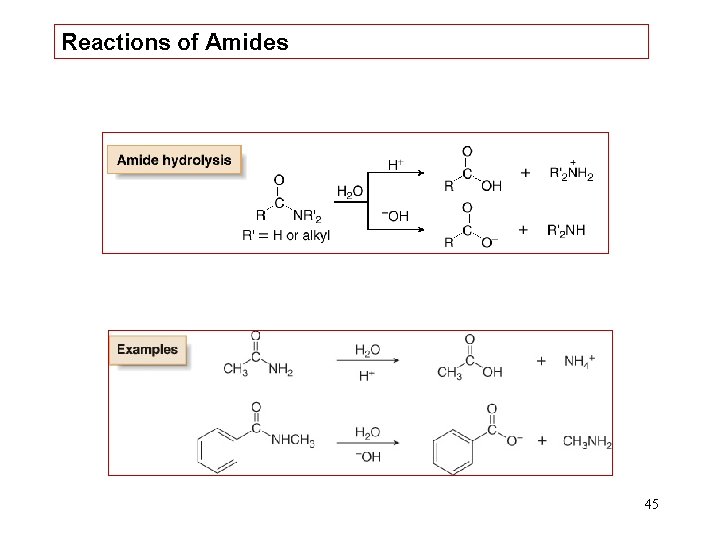
Reactions of Amides 45
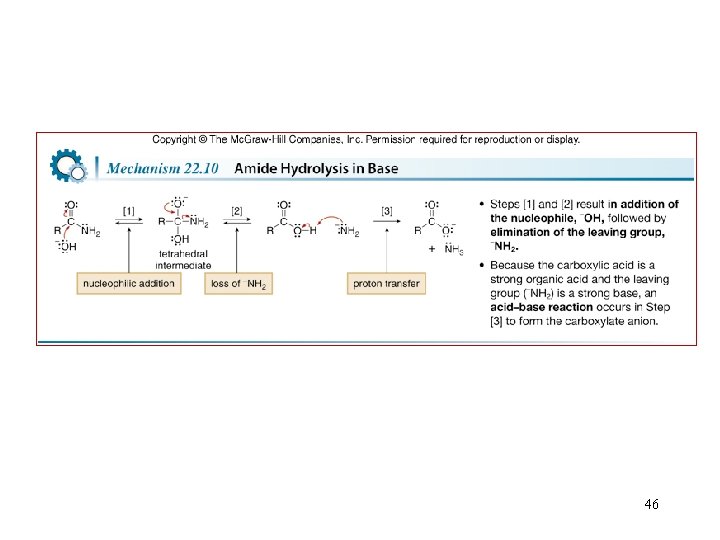
46
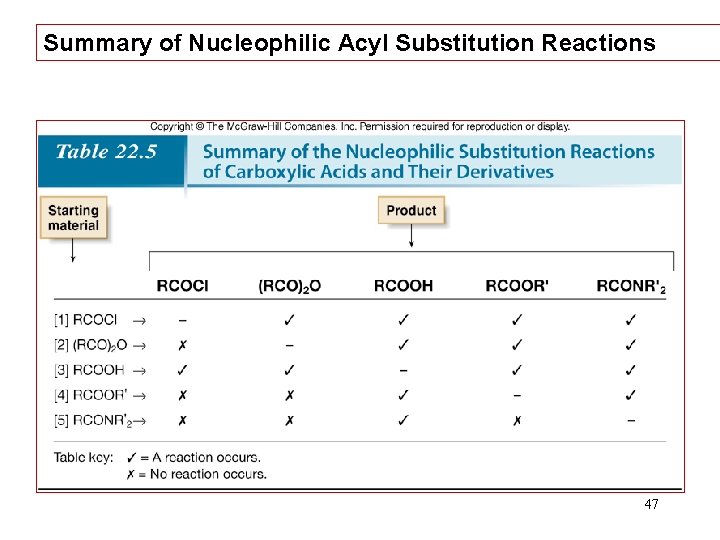
Summary of Nucleophilic Acyl Substitution Reactions 47
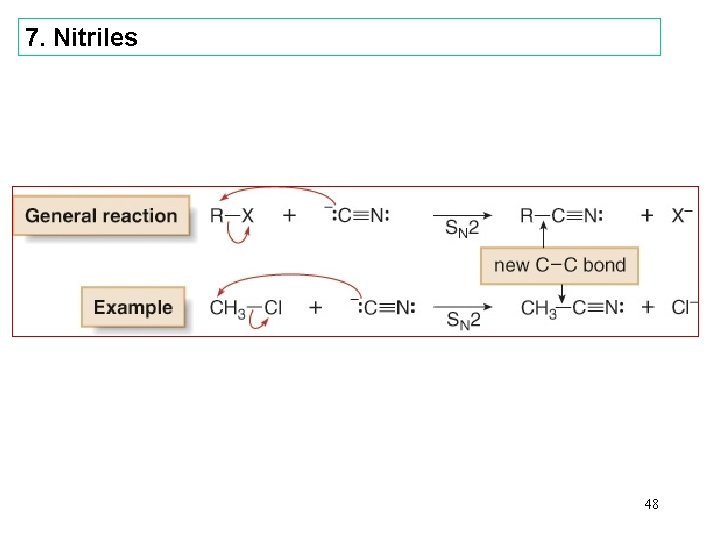
7. Nitriles 48
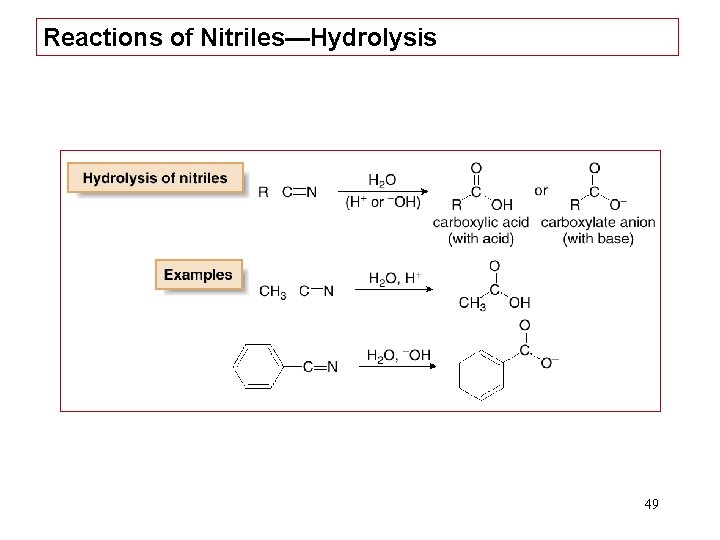
Reactions of Nitriles—Hydrolysis 49
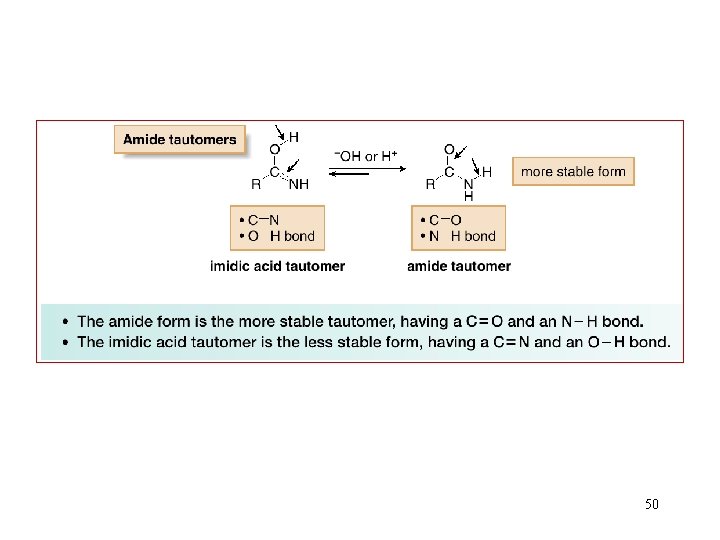
50
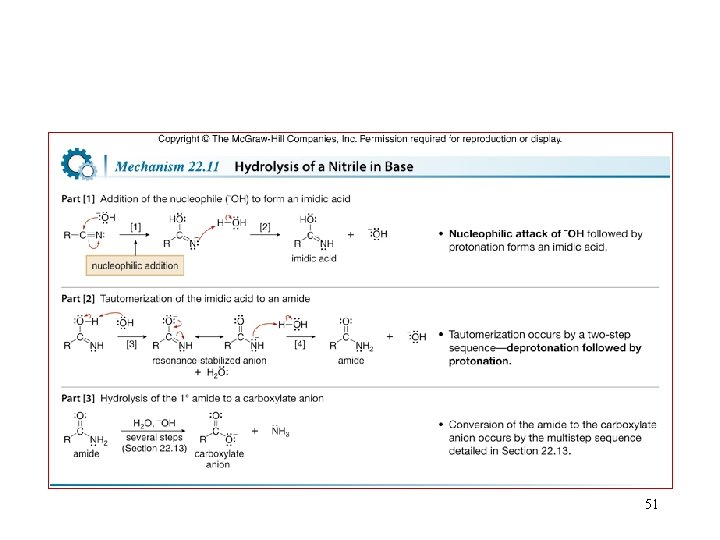
51
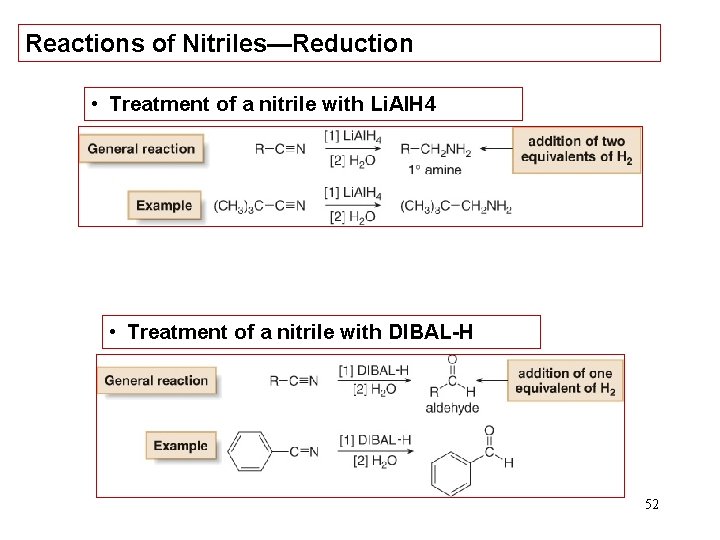
Reactions of Nitriles—Reduction • Treatment of a nitrile with Li. Al. H 4 • Treatment of a nitrile with DIBAL-H 52
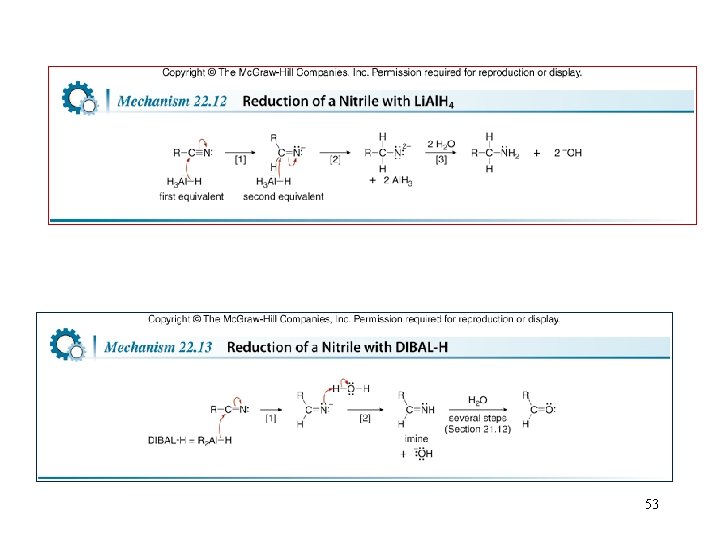
53
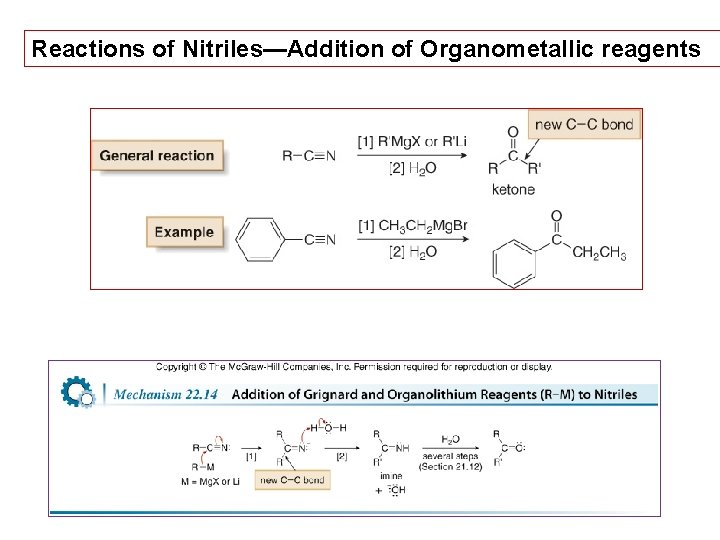
Reactions of Nitriles—Addition of Organometallic reagents 54
- Slides: 54