Chapter 22 Carbonates and other minerals with triangular

Chapter 22 Carbonates and other minerals with triangular anion groups. Sedimentary origins

Introduction Triangular anion group: (XO 3)n. Carbonates: CO 32 Simple salts of carbonic acid H 2 CO 3: calcite Ca. CO 3; dolomite Ca. Mg(CO 3)2 With additional anions: malachite Cu 2(CO 3)(OH)2 Mixed compounds: sulfate-carbonates; phosphate-carbonates, etc. Nitrates: NO 3 Borates: BO 33 - (some borates also tetrahedra BO 45 -) Study info for NB carbonates, nitrates and borates from Table 22. 1

Carbonates: Composition Forms by bonding of carbonic acid (H 2 CO 3) to: Na, Ca, Mg, Fe, Mn, Zn, Sr, Ba low ionization potential large cations For Na+, H 2 O is incorporated into structure Bi 2+, Cu 2+and rare earth elements (REEs) only when OH-, F- or O 2 - present to weaken the CO 32 - complex
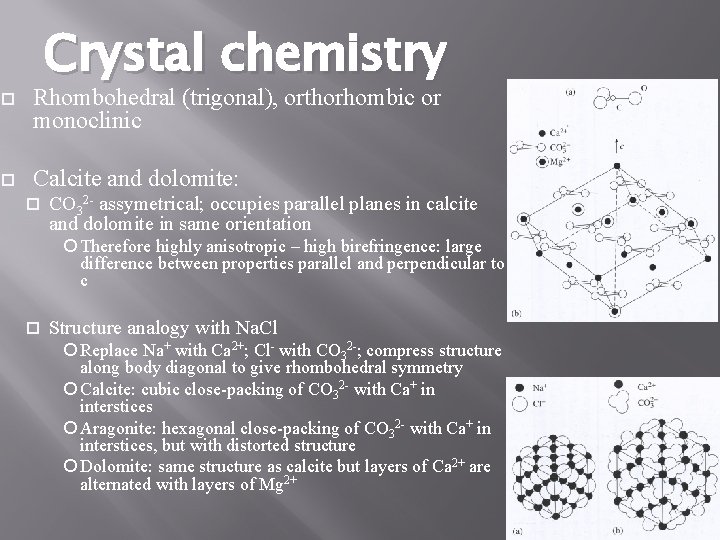
Crystal chemistry Rhombohedral (trigonal), orthorhombic or monoclinic Calcite and dolomite: CO 32 - assymetrical; occupies parallel planes in calcite and dolomite in same orientation Therefore highly anisotropic – high birefringence: large difference between properties parallel and perpendicular to c Structure analogy with Na. Cl Replace Na+ with Ca 2+; Cl- with CO 32 -; compress structure along body diagonal to give rhombohedral symmetry Calcite: cubic close-packing of CO 32 - with Ca+ in interstices Aragonite: hexagonal close-packing of CO 32 - with Ca+ in interstices, but with distorted structure Dolomite: same structure as calcite but layers of Ca 2+ are alternated with layers of Mg 2+
Crystal chemistry Carbonates display polymorphism and isomorphism Isomorphy: Small cations: isomorphy within calcite structure (calcite, magnesite, siderite, rhodochrosite, smithsonite) Large cations: isomorphy within aragonite structure (aragonite, witherite, strontianite, cerussite) Polymorphy of Ca. CO 3: High T; low P: calcite; Low T; high P: aragonite Sheet and chain like structures common in carbonates Polymerization of triangular groups common in borates: BO 33 - isolated Combined into B 2 O 54 - or B 2 O 42 - pairs, B 4 O 84 - rings or B 2 O 54 - chains
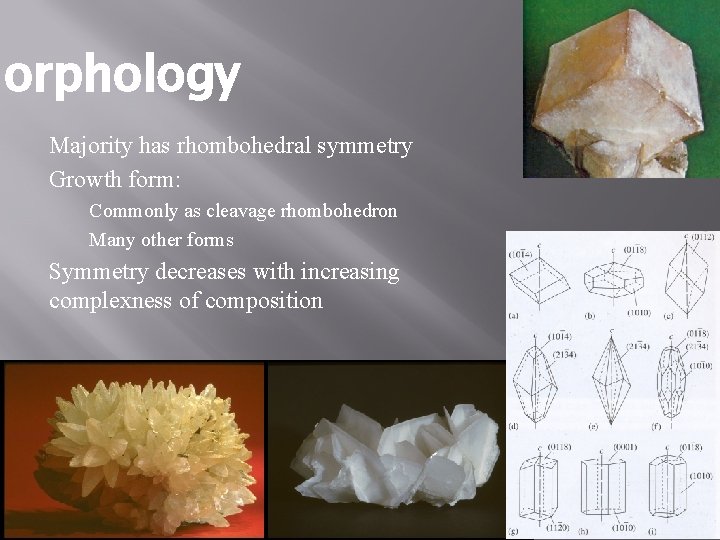
Morphology Majority has rhombohedral symmetry Growth form: Commonly as cleavage rhombohedron Many other forms Symmetry decreases with increasing complexness of composition

Physical and Optical Properties Colorless to white Mn: pink (rhodochrosite) Cu: green (malachite) or blue (azurite) Fe: yellow (ankerite, siderite) or brown Reaction with HCl 2 HCl + Ca(Pb, Zn, Mg…)CO 3 H 2 O + CO 2 + Ca(Pb, Zn, Mg…)Cl 2 Some only when concentrated acid or mineral heated High birefringence & high order interference colors

Classification Associations: Al-Mg-(Na) association Magnesite Group Hydrotalcite Group Na(K)-Ca-Ba association Calcite-Aragonite Group Dolomite-Barytocalcite Group Bastnaesite Group Zn-Cu-Pb(U) association Smithsonite Group Malachite Group Cerussite Group Structural type Calcite group Dolomite group Aragonite group Soda carbonate minerals Other carbonate minerals Nitrates Borates

Calcite Structural Type Calcite Ca. CO 3 Often with minor Mg, Fe, Mn Limited at room T Very diverse habit Limestone: chemical or biological origin Usually high in Mg when precipitate from seawater Forms marble when metamorphosed Skarns and hydrothermal deposits

Calcite Structural Type Magnesite Mg. CO 3 Endmember of magnesite-siderite isomorphic series Gray-white solid masses of granular texture Occur in: Dolostones affected by hydrothermal solutions – recrystallisation Veins formed during weathering of serpentinites

Calcite Structural Type Siderite Fe. CO 3 Brown crystals, highly vitreous luster Granular aggregates in hydrothermal veins Sedimentary rocks: hydrothermal alteration Chemical origin Fe ore Rusty color (white when nonoxidized); high density
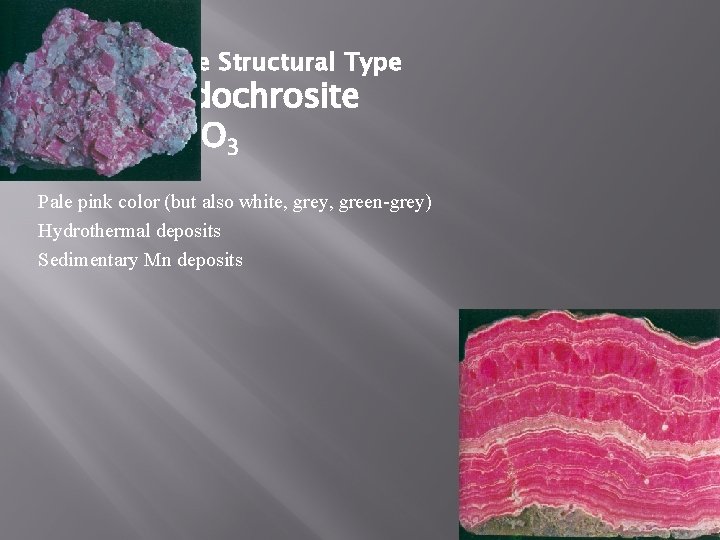
Calcite Structural Type Rhodochrosite Mn. CO 3 Pale pink color (but also white, grey, green-grey) Hydrothermal deposits Sedimentary Mn deposits

Calcite Structural Type Smithsonite Zn. CO 3 Limited solid solution, with Zn replaced by: Fe, Mn, Ca, Pb, Mg, Cd, Co Occurs as: Colloidal, botryoidal, earthy masses OR White, green, brown crystals Oxidation product of sphalerite (Zn. S) ores
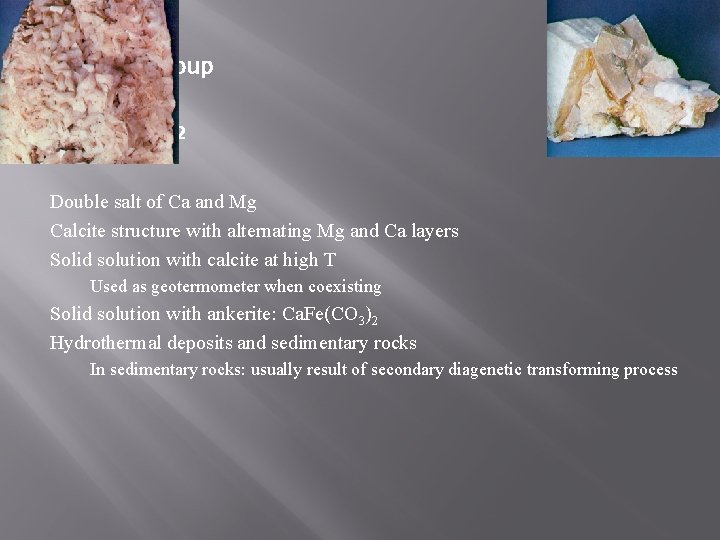
Dolomite Group Dolomite Ca. Mg(CO 3)2 Double salt of Ca and Mg Calcite structure with alternating Mg and Ca layers Solid solution with calcite at high T Used as geotermometer when coexisting Solid solution with ankerite: Ca. Fe(CO 3)2 Hydrothermal deposits and sedimentary rocks In sedimentary rocks: usually result of secondary diagenetic transforming process

Dolomite Group Ankerite Ca. Fe(CO 3)2 Double salt of Ca and Fe Calcite structure with alternating Fe and Ca layers – therefore very similar to dolomite – solid solution Also in this group: Kutnahorite - Ca. Mn(CO 3)2
- Slides: 15