Chapter 21 The Organic Chemistry of Carbohydrates Paula

Chapter 21 The Organic Chemistry of Carbohydrates Paula Yurkanis Bruice University of California, Santa Barbara © 2014 Pearson Education, Inc.
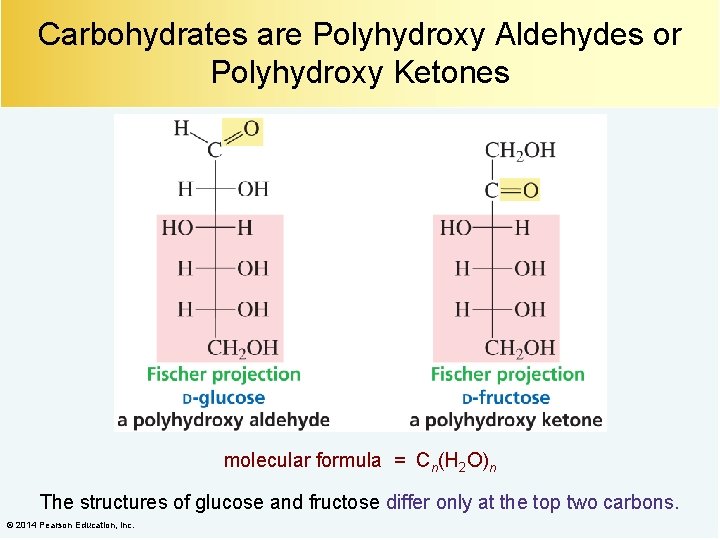
Carbohydrates are Polyhydroxy Aldehydes or Polyhydroxy Ketones molecular formula = Cn(H 2 O)n The structures of glucose and fructose differ only at the top two carbons. © 2014 Pearson Education, Inc.
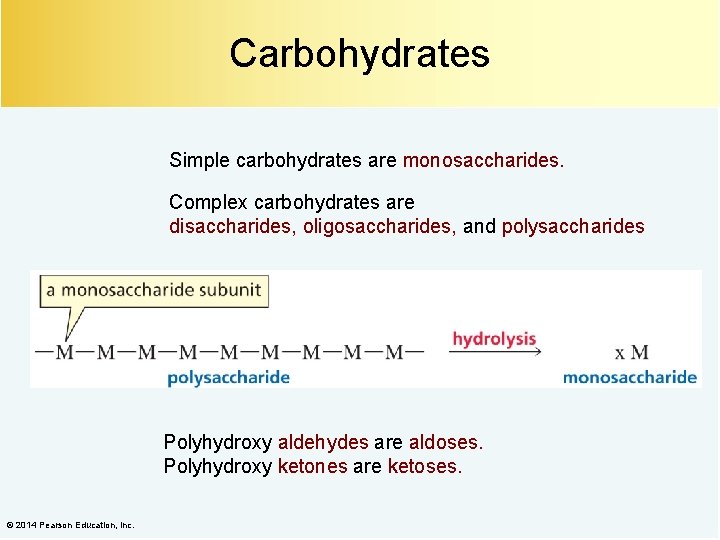
Carbohydrates Simple carbohydrates are monosaccharides. Complex carbohydrates are disaccharides, oligosaccharides, and polysaccharides Polyhydroxy aldehydes are aldoses. Polyhydroxy ketones are ketoses. © 2014 Pearson Education, Inc.
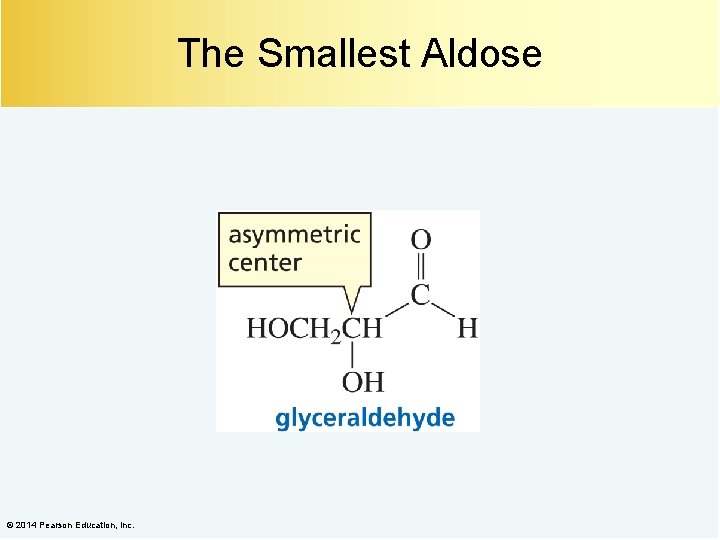
The Smallest Aldose © 2014 Pearson Education, Inc.
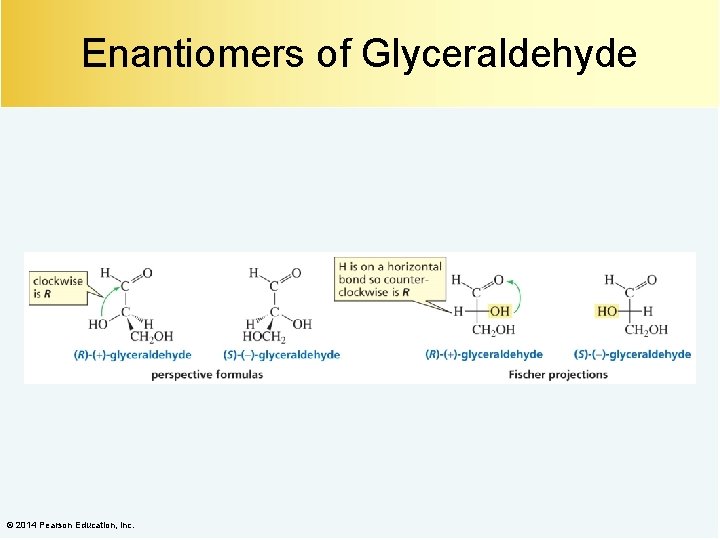
Enantiomers of Glyceraldehyde © 2014 Pearson Education, Inc.
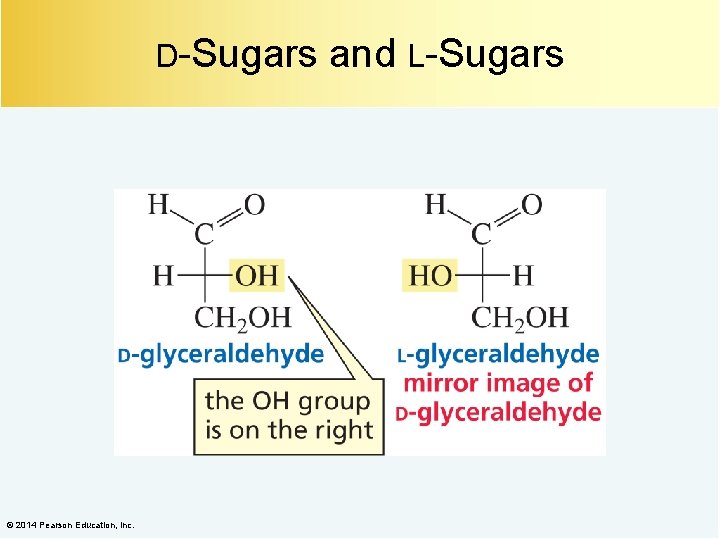
D-Sugars © 2014 Pearson Education, Inc. and L-Sugars
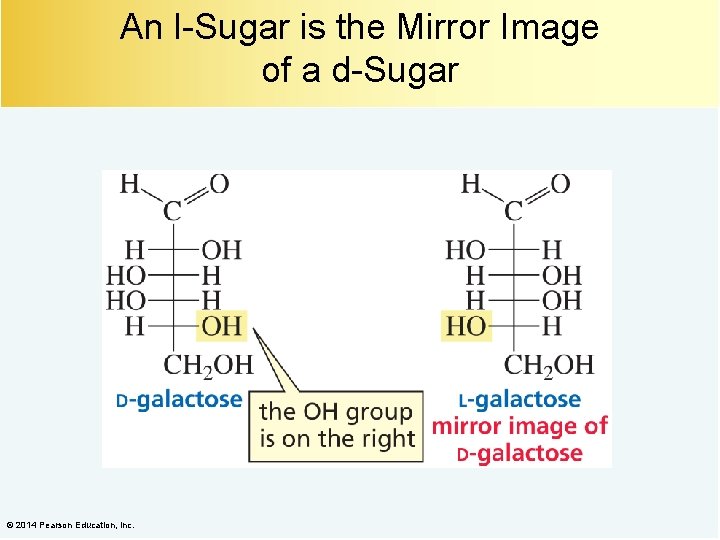
An l-Sugar is the Mirror Image of a d-Sugar © 2014 Pearson Education, Inc.
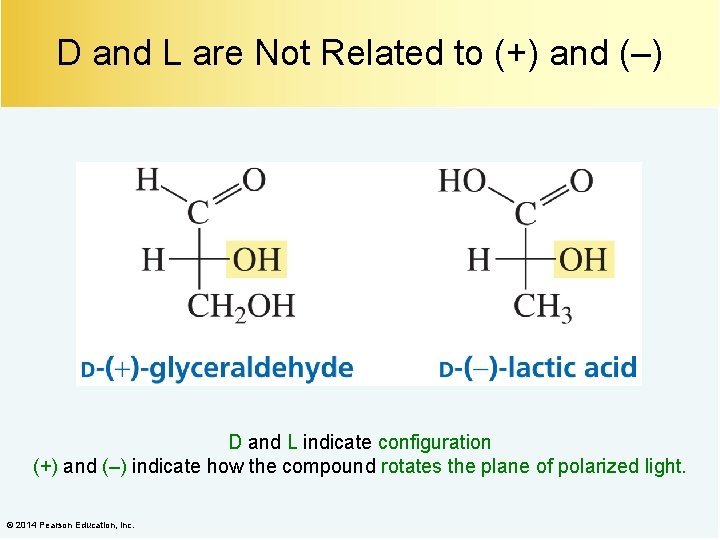
D and L are Not Related to (+) and (–) D and L indicate configuration (+) and (–) indicate how the compound rotates the plane of polarized light. © 2014 Pearson Education, Inc.
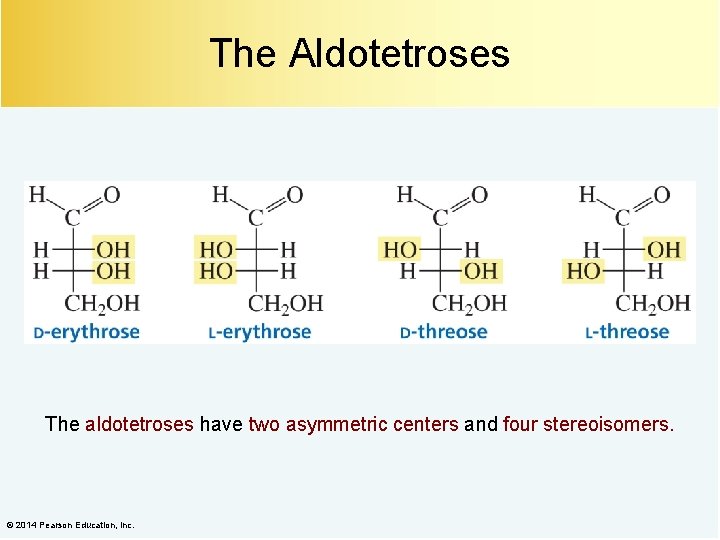
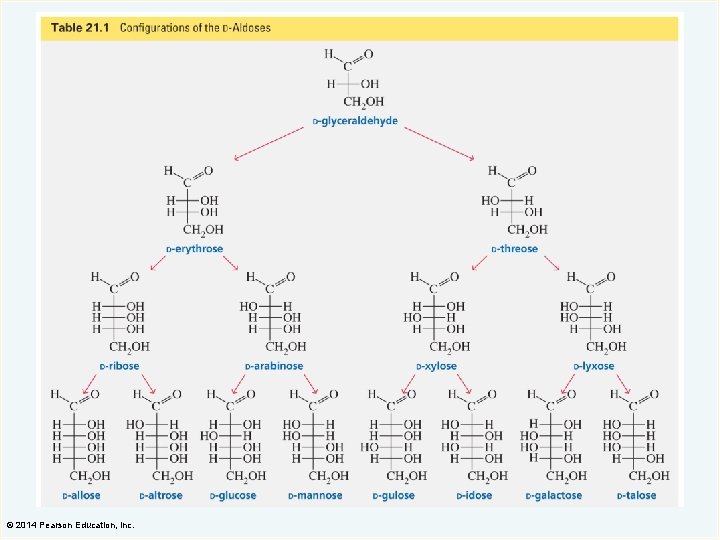
The Aldotetroses The aldotetroses have two asymmetric centers and four stereoisomers. © 2014 Pearson Education, Inc.
© 2014 Pearson Education, Inc.
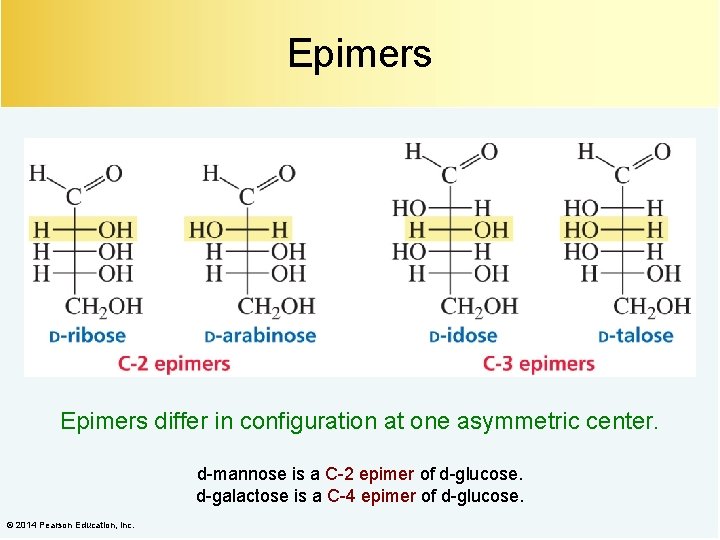
Epimers differ in configuration at one asymmetric center. d-mannose is a C-2 epimer of d-glucose. d-galactose is a C-4 epimer of d-glucose. © 2014 Pearson Education, Inc.
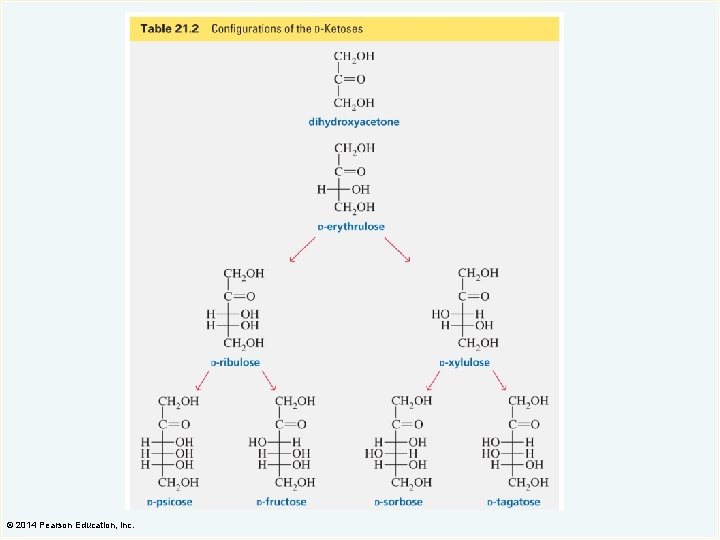
© 2014 Pearson Education, Inc.
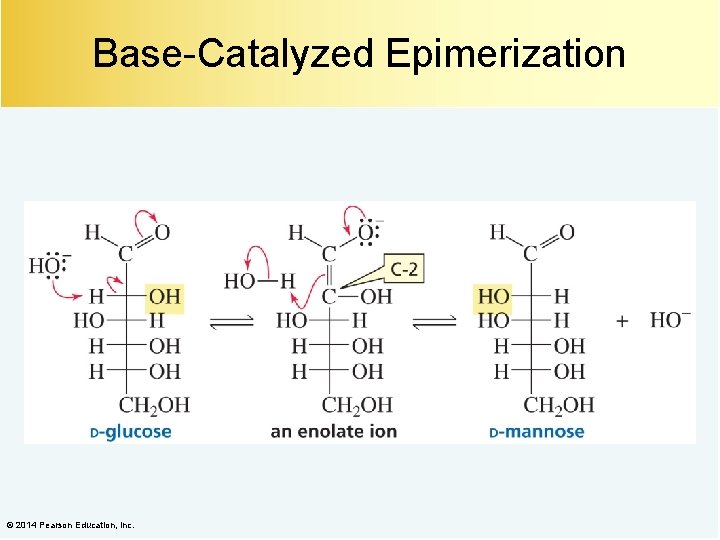
Base-Catalyzed Epimerization © 2014 Pearson Education, Inc.
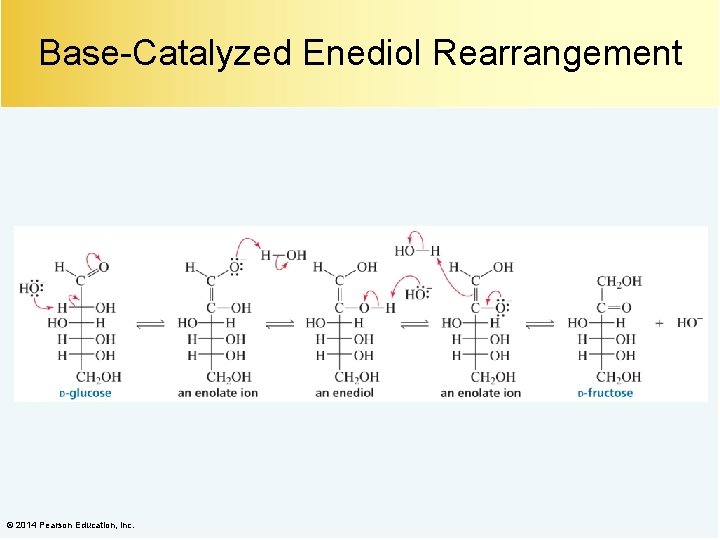
Base-Catalyzed Enediol Rearrangement © 2014 Pearson Education, Inc.
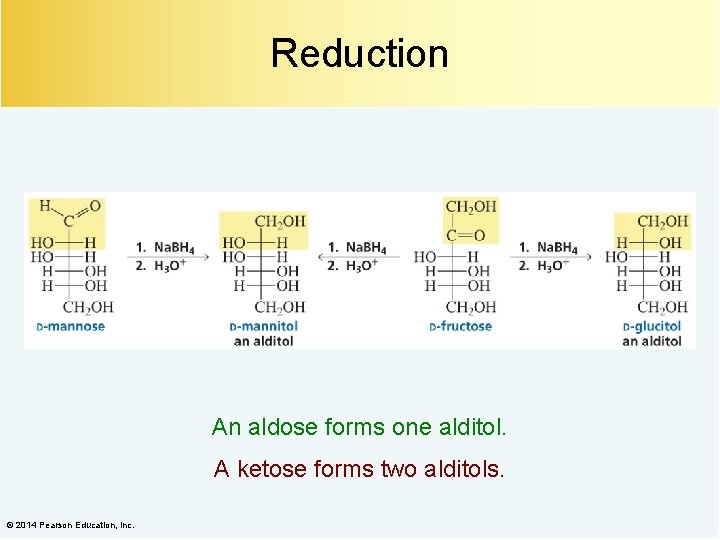
Reduction An aldose forms one alditol. A ketose forms two alditols. © 2014 Pearson Education, Inc.
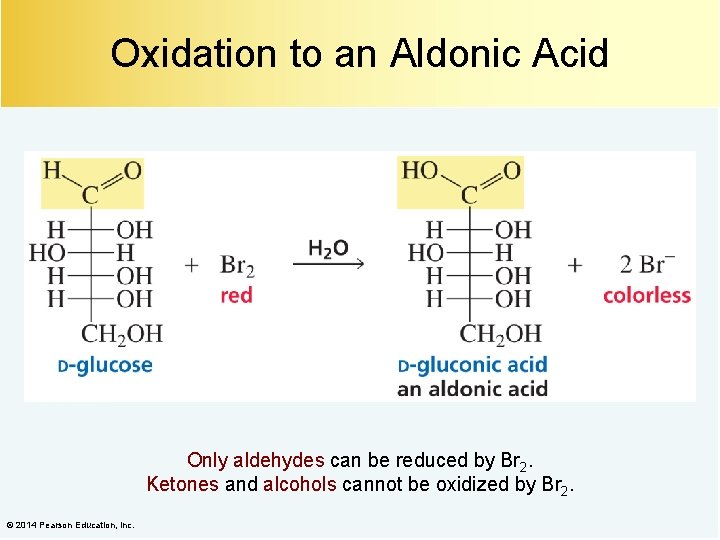
Oxidation to an Aldonic Acid Only aldehydes can be reduced by Br 2. Ketones and alcohols cannot be oxidized by Br 2. © 2014 Pearson Education, Inc.
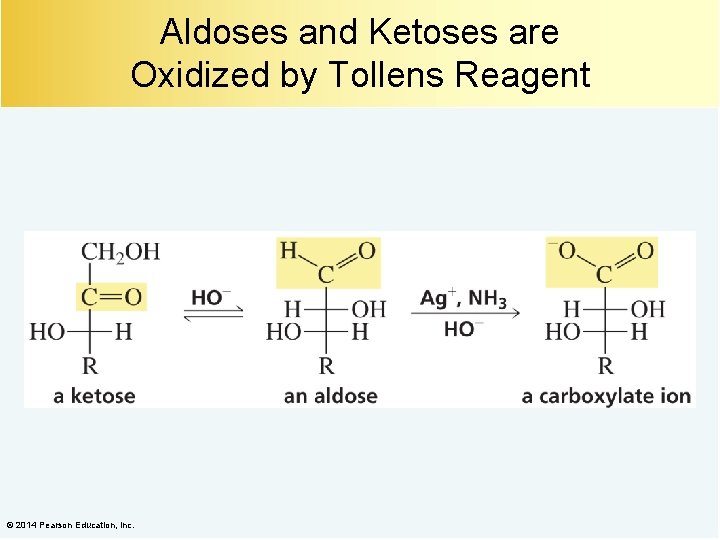
Aldoses and Ketoses are Oxidized by Tollens Reagent © 2014 Pearson Education, Inc.
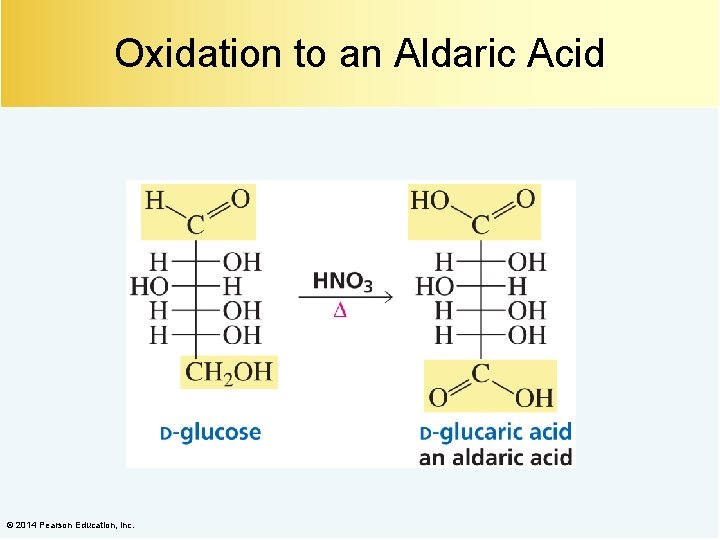
Oxidation to an Aldaric Acid © 2014 Pearson Education, Inc.
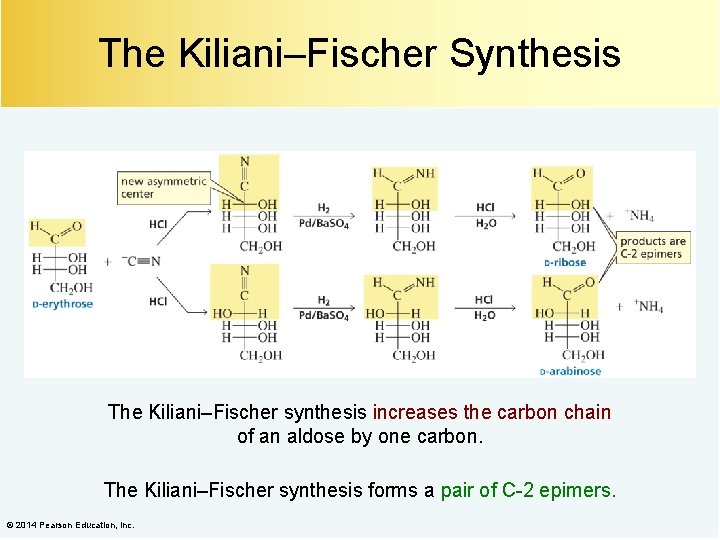
The Kiliani–Fischer Synthesis The Kiliani–Fischer synthesis increases the carbon chain of an aldose by one carbon. The Kiliani–Fischer synthesis forms a pair of C-2 epimers. © 2014 Pearson Education, Inc.

The Wohl Degradation The Wohl degradation decreases the carbon chain of an aldose by one carbon. © 2014 Pearson Education, Inc.
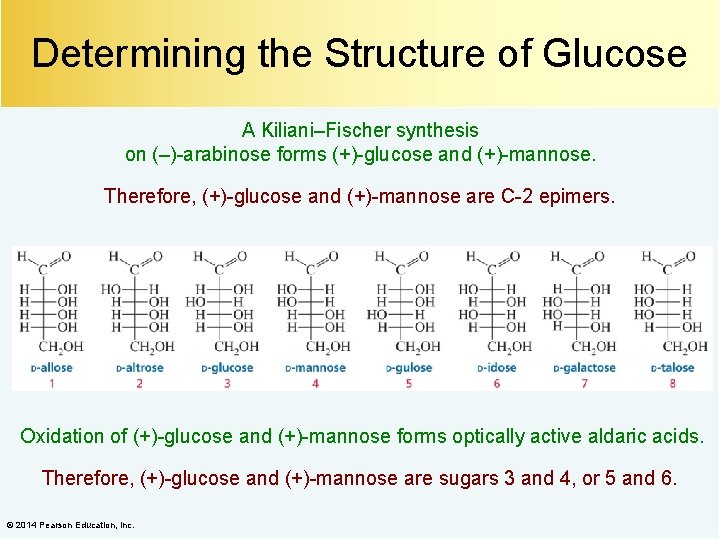
Determining the Structure of Glucose A Kiliani–Fischer synthesis on (–)-arabinose forms (+)-glucose and (+)-mannose. Therefore, (+)-glucose and (+)-mannose are C-2 epimers. Oxidation of (+)-glucose and (+)-mannose forms optically active aldaric acids. Therefore, (+)-glucose and (+)-mannose are sugars 3 and 4, or 5 and 6. © 2014 Pearson Education, Inc.
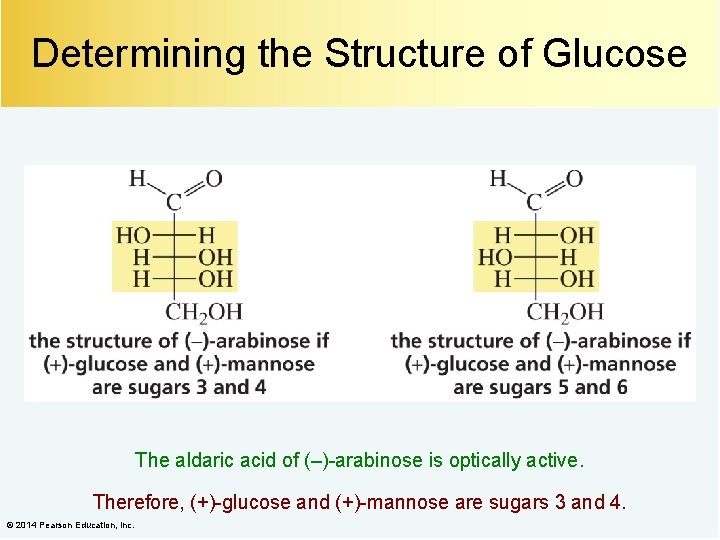
Determining the Structure of Glucose The aldaric acid of (–)-arabinose is optically active. Therefore, (+)-glucose and (+)-mannose are sugars 3 and 4. © 2014 Pearson Education, Inc.
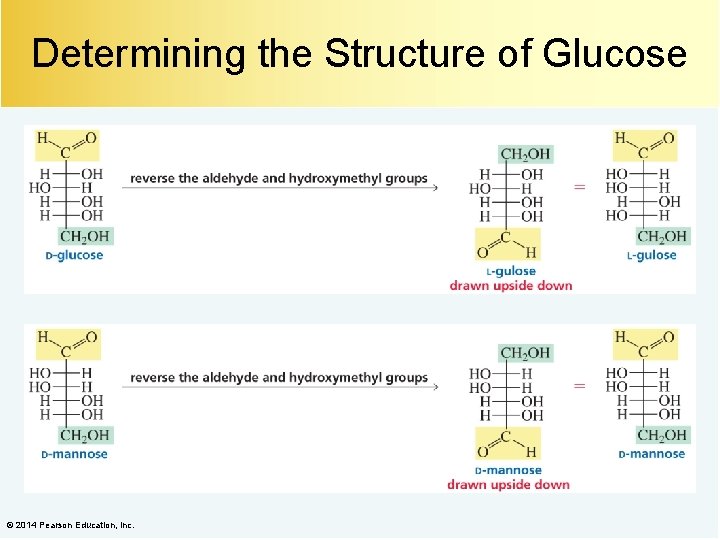
Determining the Structure of Glucose © 2014 Pearson Education, Inc.
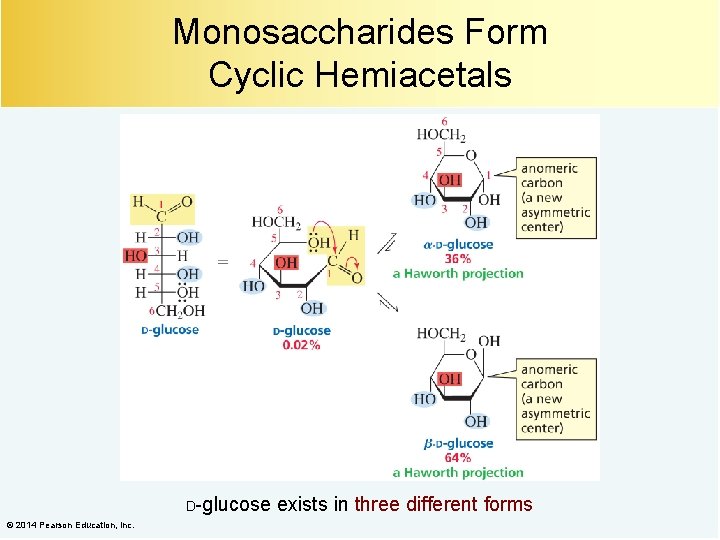
Monosaccharides Form Cyclic Hemiacetals D-glucose © 2014 Pearson Education, Inc. exists in three different forms
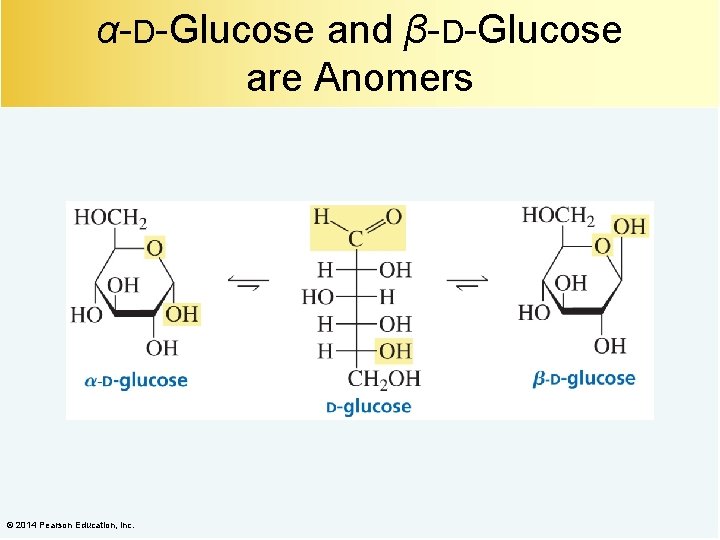
α-D-Glucose and β-D-Glucose are Anomers © 2014 Pearson Education, Inc.
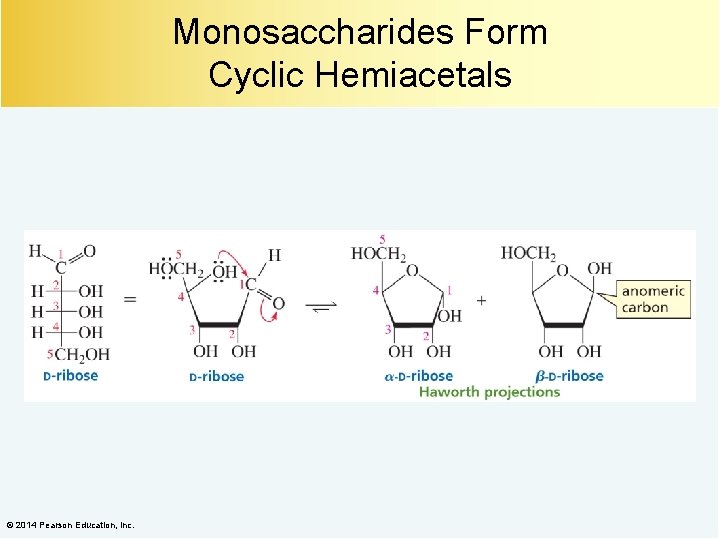
Monosaccharides Form Cyclic Hemiacetals © 2014 Pearson Education, Inc.
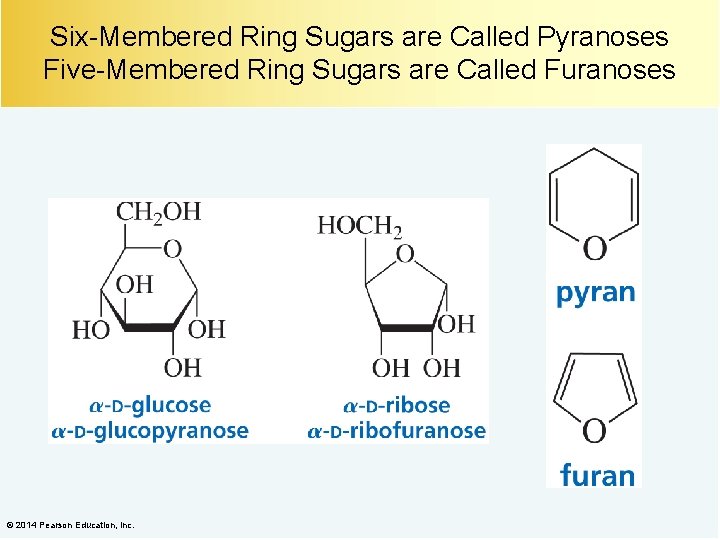
Six-Membered Ring Sugars are Called Pyranoses Five-Membered Ring Sugars are Called Furanoses © 2014 Pearson Education, Inc.
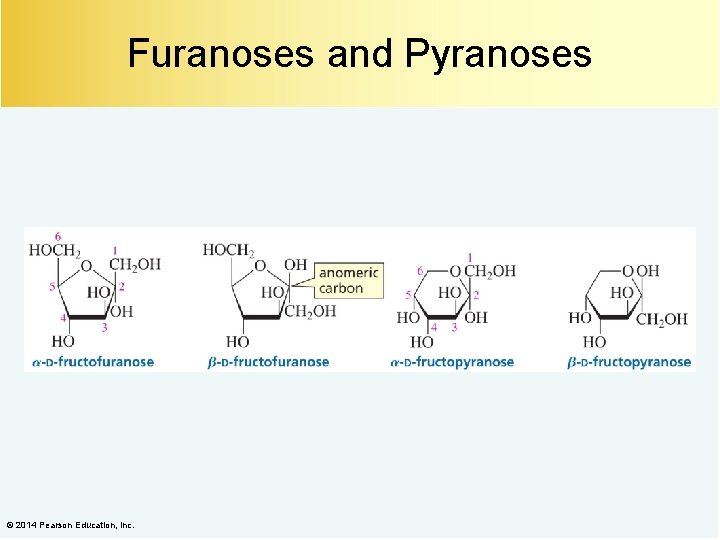
Furanoses and Pyranoses © 2014 Pearson Education, Inc.
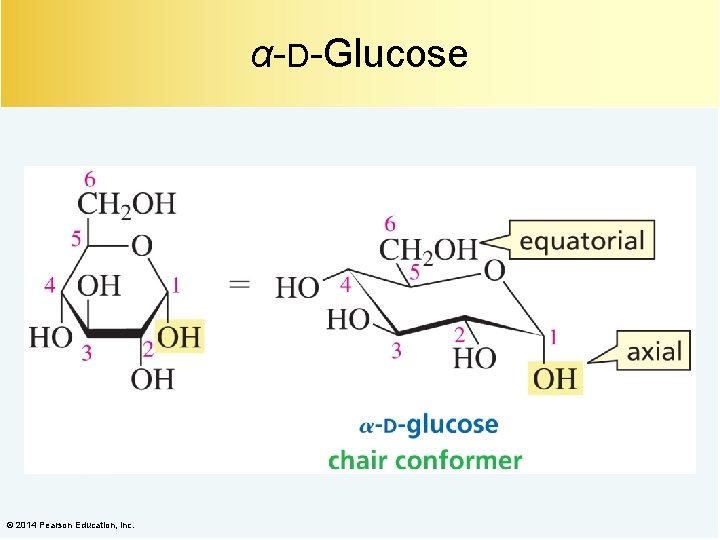
α-D-Glucose © 2014 Pearson Education, Inc.
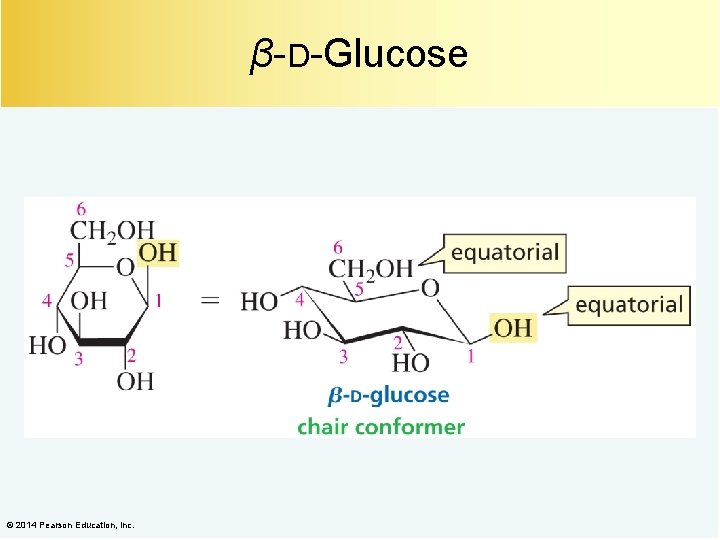
β-D-Glucose © 2014 Pearson Education, Inc.
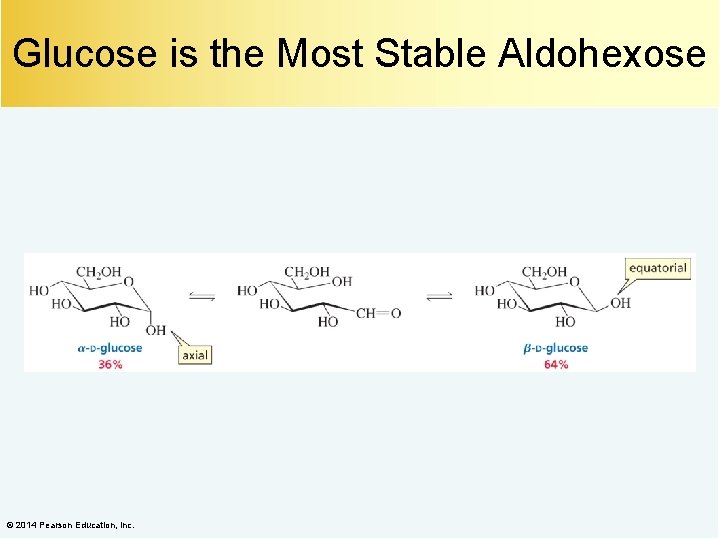
Glucose is the Most Stable Aldohexose © 2014 Pearson Education, Inc.
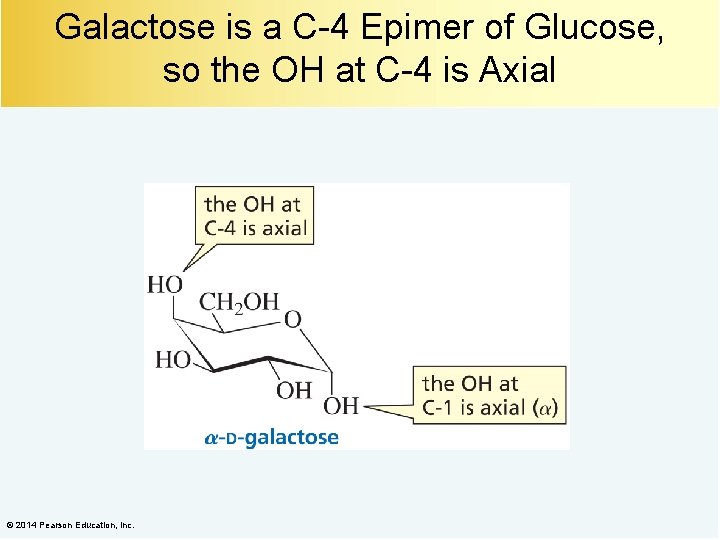
Galactose is a C-4 Epimer of Glucose, so the OH at C-4 is Axial © 2014 Pearson Education, Inc.
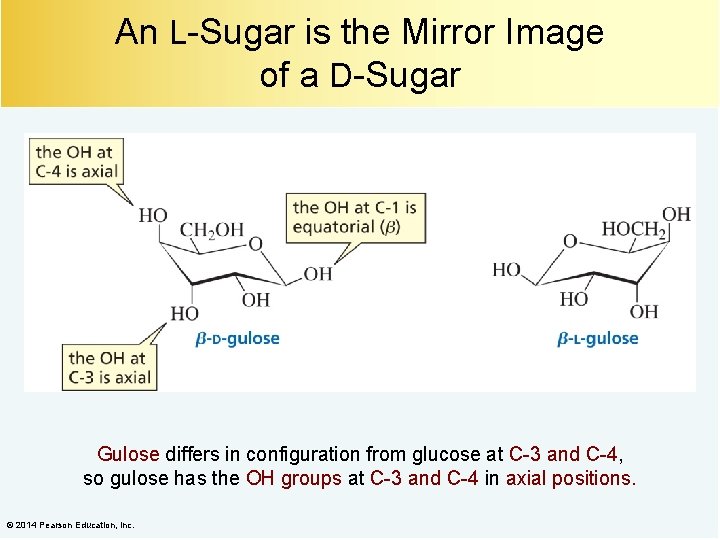
An L-Sugar is the Mirror Image of a D-Sugar Gulose differs in configuration from glucose at C-3 and C-4, so gulose has the OH groups at C-3 and C-4 in axial positions. © 2014 Pearson Education, Inc.
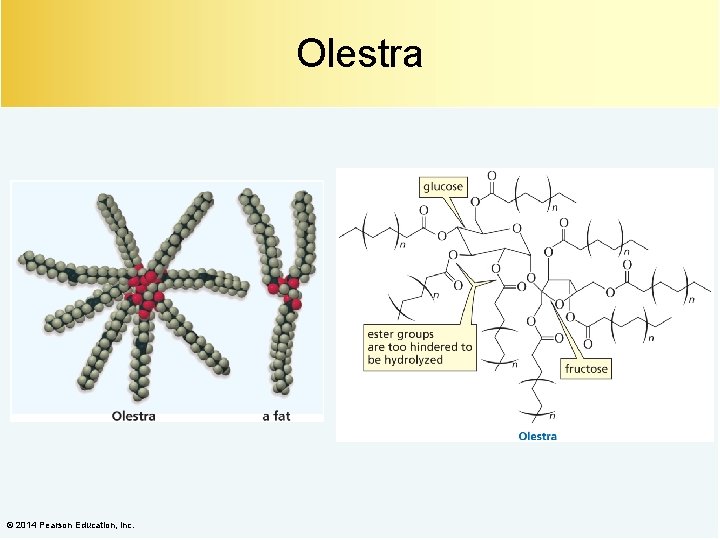
Olestra © 2014 Pearson Education, Inc.
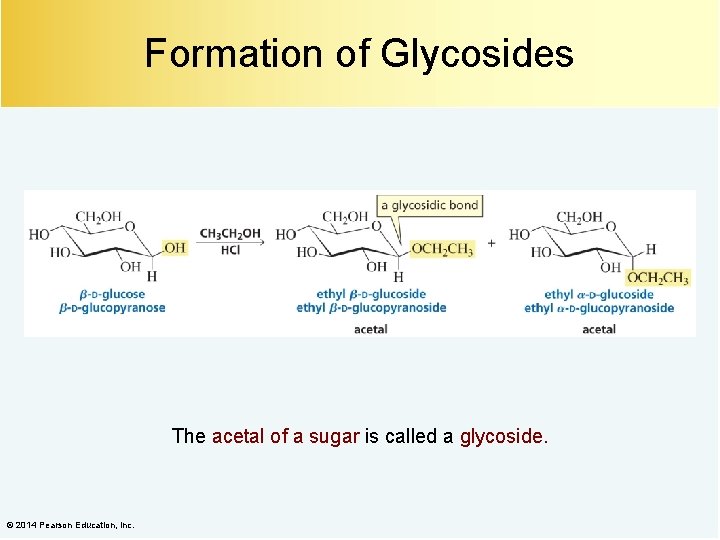
Formation of Glycosides The acetal of a sugar is called a glycoside. © 2014 Pearson Education, Inc.
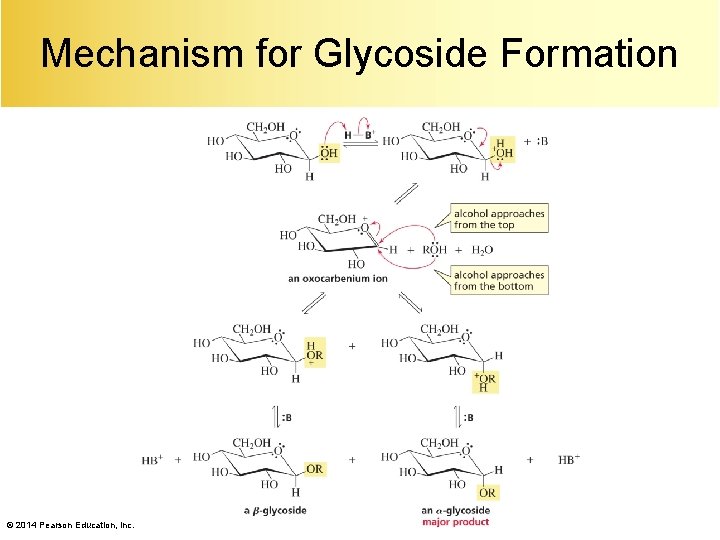
Mechanism for Glycoside Formation © 2014 Pearson Education, Inc.
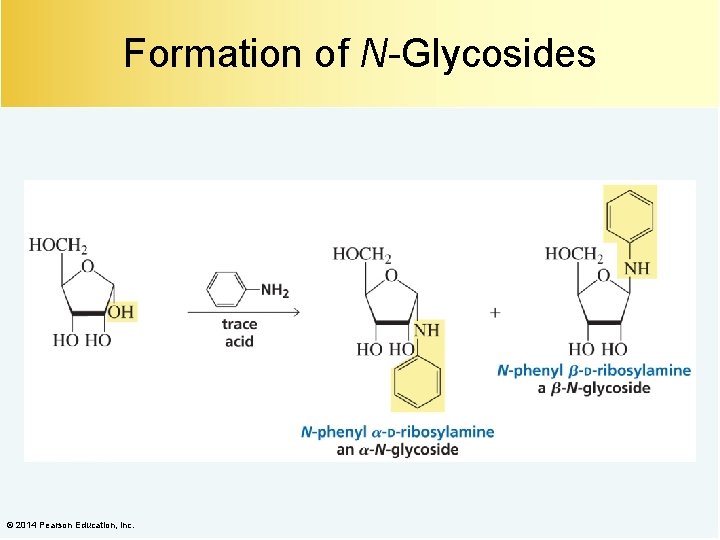
Formation of N-Glycosides © 2014 Pearson Education, Inc.
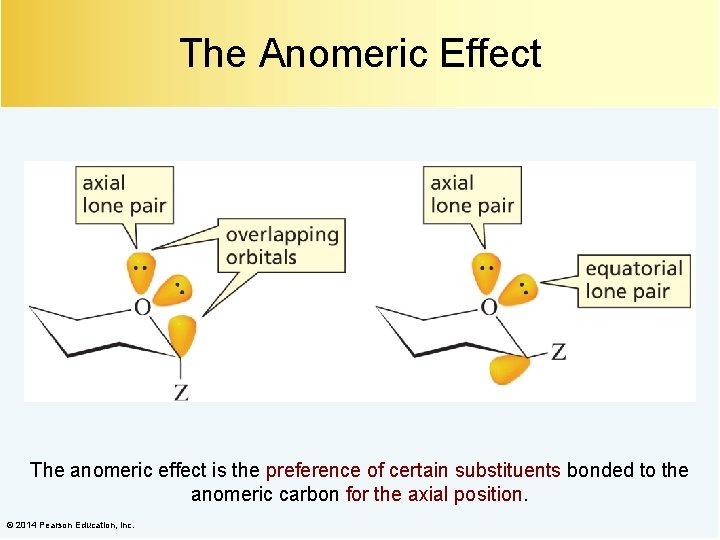
The Anomeric Effect The anomeric effect is the preference of certain substituents bonded to the anomeric carbon for the axial position. © 2014 Pearson Education, Inc.

Reducing and Nonreducing Sugars A reducing sugar can reduce Ag+ or Br 2. A nonreducing sugar cannot reduce Ag+ or Br 2. A sugar with an aldehyde, a ketone, or a hemiacetal group is a reducing sugar. A sugar without one of these groups is a nonreducing sugar. © 2014 Pearson Education, Inc.
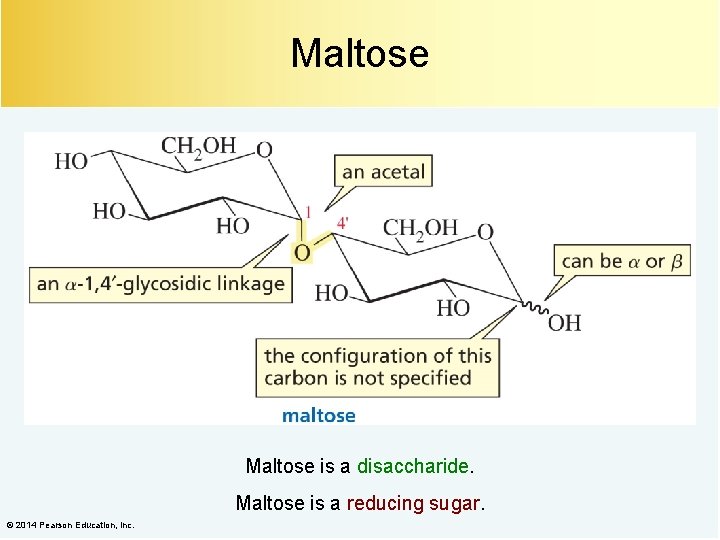
Maltose is a disaccharide. Maltose is a reducing sugar. © 2014 Pearson Education, Inc.
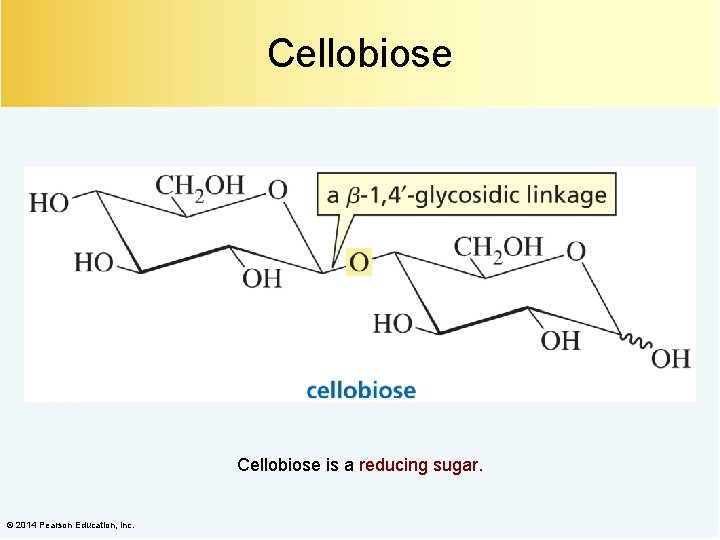
Cellobiose is a reducing sugar. © 2014 Pearson Education, Inc.
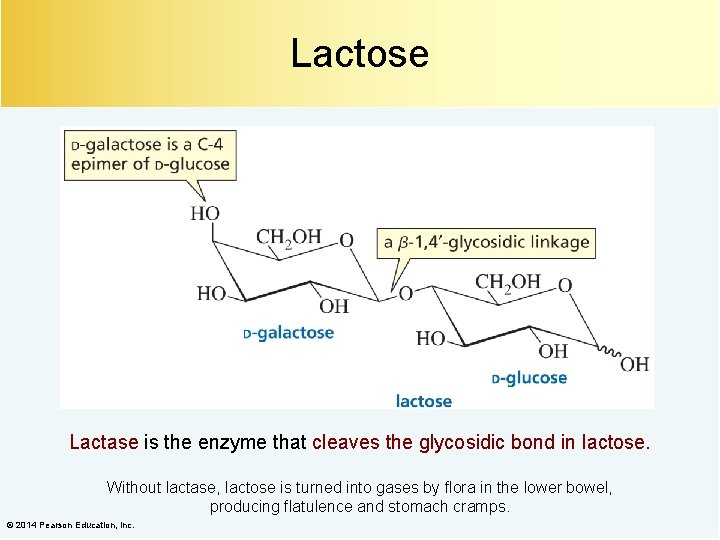
Lactose Lactase is the enzyme that cleaves the glycosidic bond in lactose. Without lactase, lactose is turned into gases by flora in the lower bowel, producing flatulence and stomach cramps. © 2014 Pearson Education, Inc.
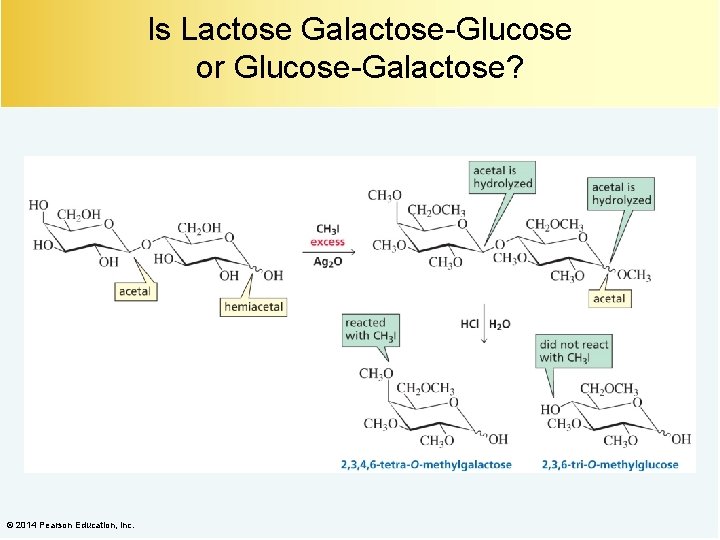
Is Lactose Galactose-Glucose or Glucose-Galactose? © 2014 Pearson Education, Inc.
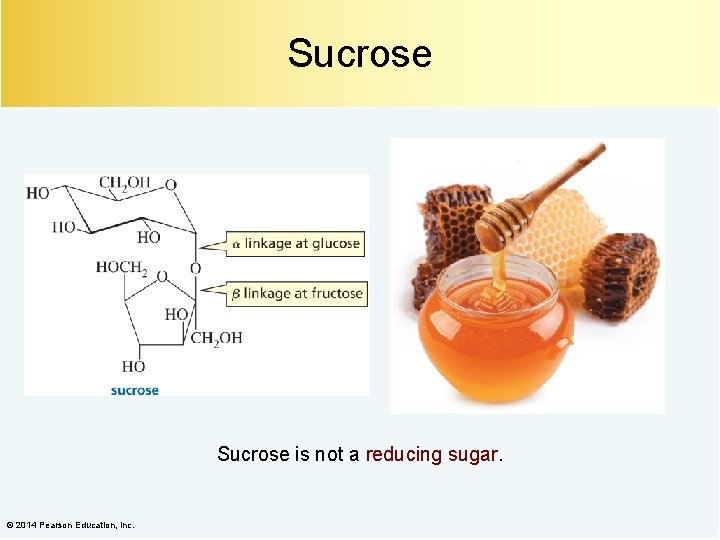
Sucrose is not a reducing sugar. © 2014 Pearson Education, Inc.
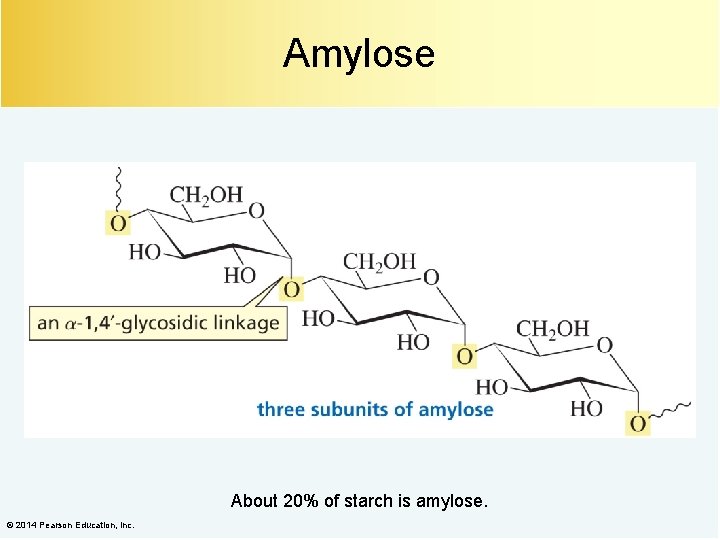
Amylose About 20% of starch is amylose. © 2014 Pearson Education, Inc.
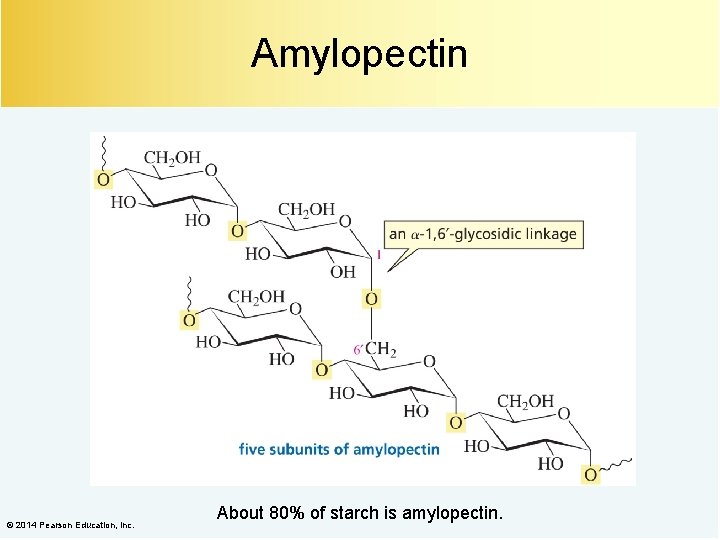
Amylopectin © 2014 Pearson Education, Inc. About 80% of starch is amylopectin.
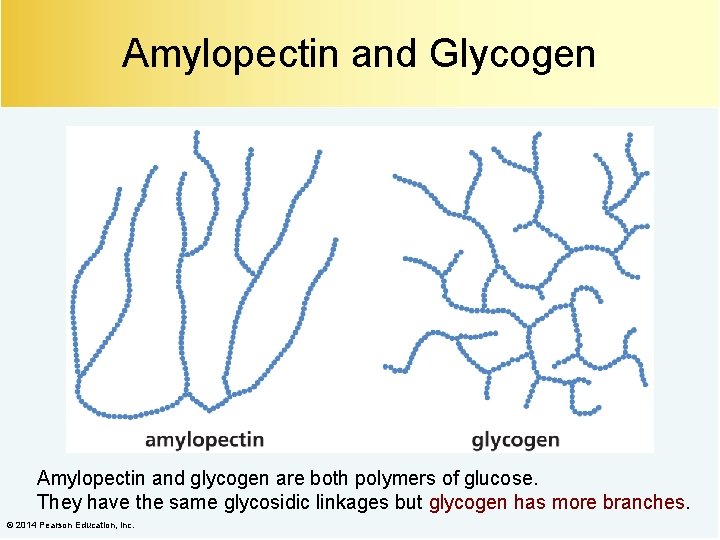
Amylopectin and Glycogen Amylopectin and glycogen are both polymers of glucose. They have the same glycosidic linkages but glycogen has more branches. © 2014 Pearson Education, Inc.
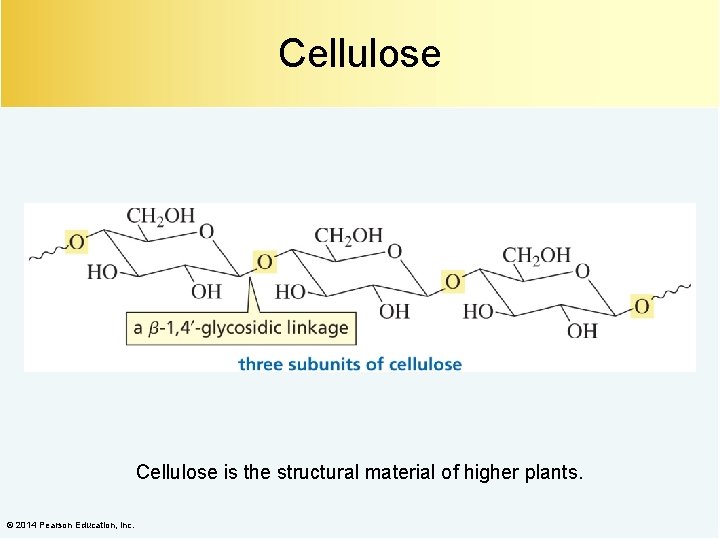
Cellulose is the structural material of higher plants. © 2014 Pearson Education, Inc.
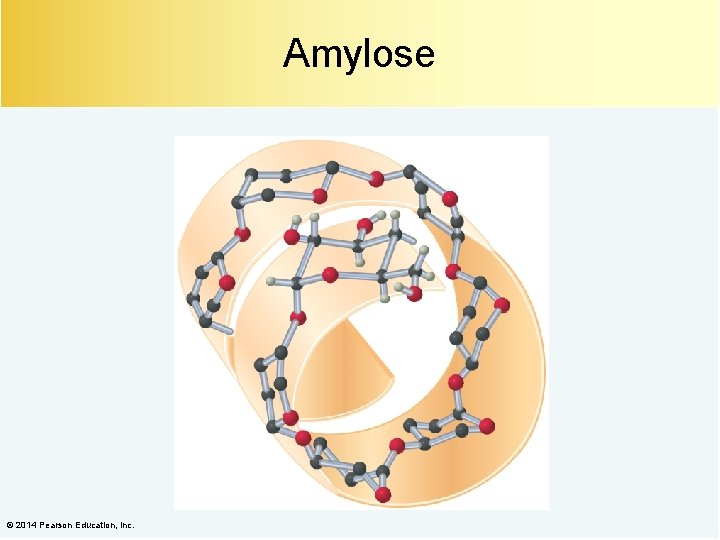
Amylose © 2014 Pearson Education, Inc.
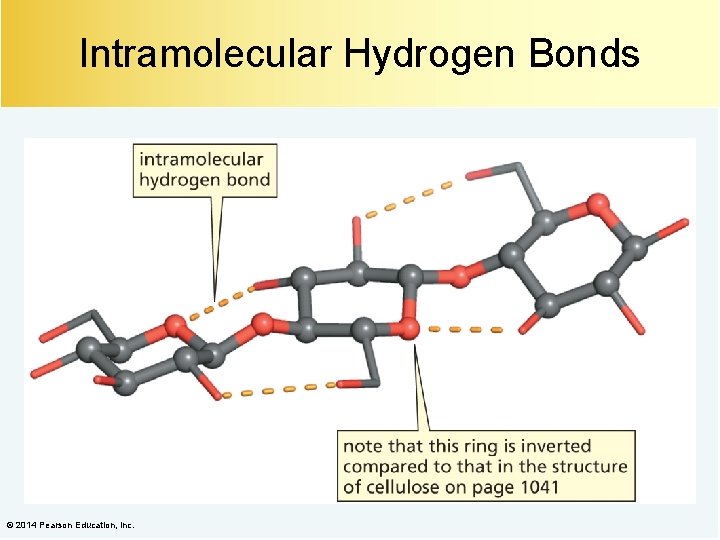
Intramolecular Hydrogen Bonds © 2014 Pearson Education, Inc.
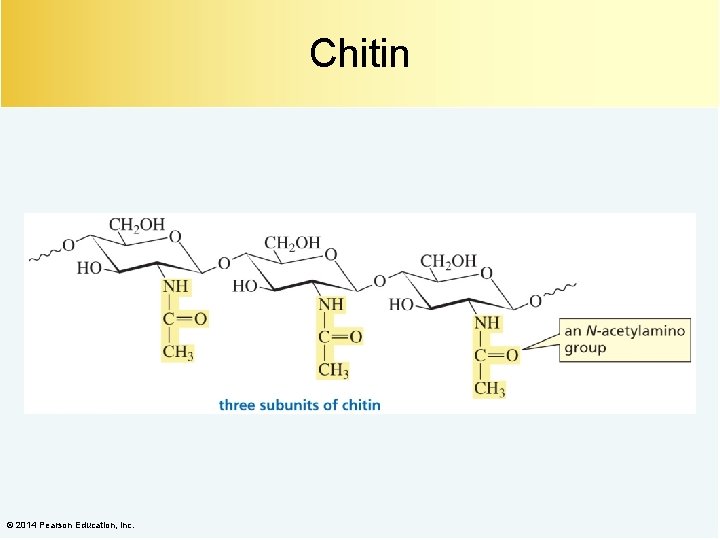
Chitin © 2014 Pearson Education, Inc.
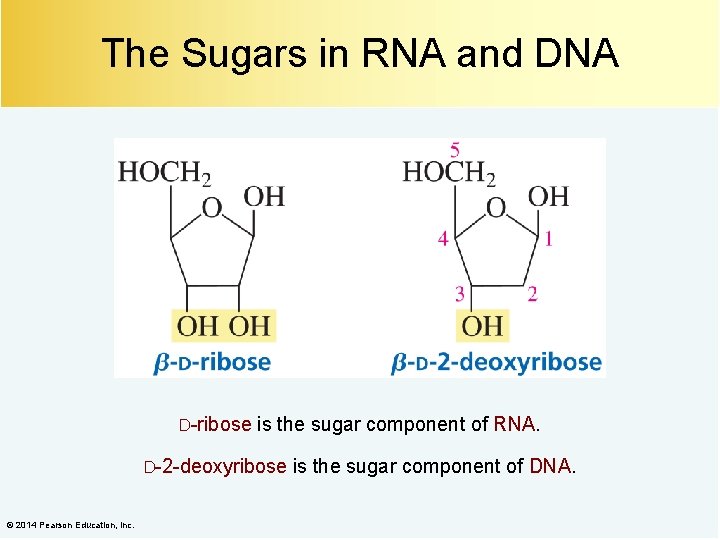
The Sugars in RNA and DNA D-ribose is the sugar component of RNA. D-2 -deoxyribose © 2014 Pearson Education, Inc. is the sugar component of DNA.
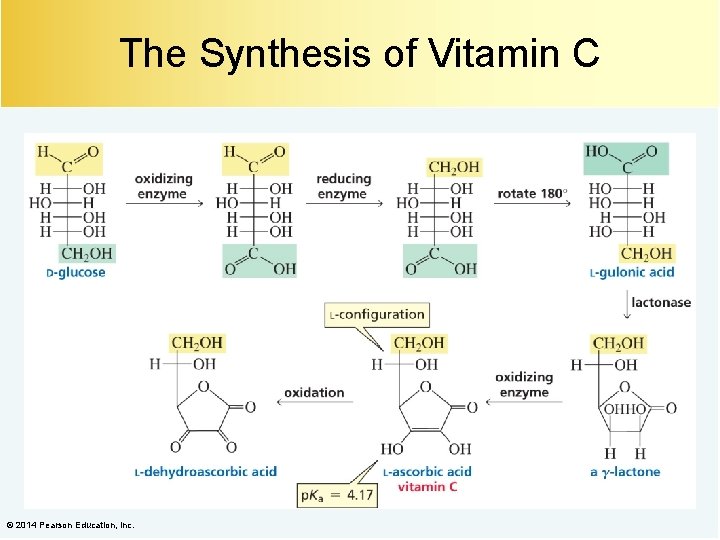
The Synthesis of Vitamin C © 2014 Pearson Education, Inc.
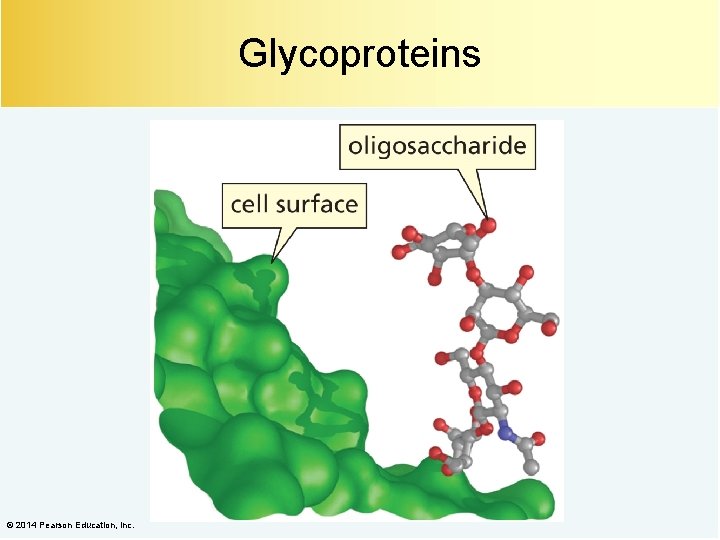
Glycoproteins © 2014 Pearson Education, Inc.
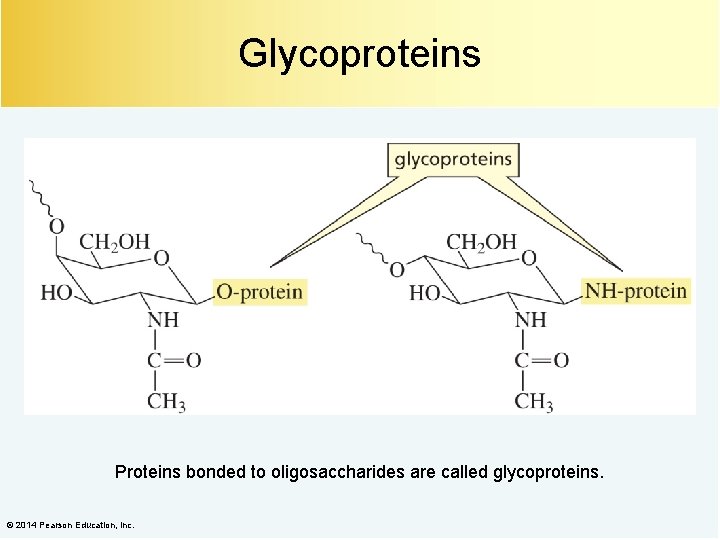
Glycoproteins Proteins bonded to oligosaccharides are called glycoproteins. © 2014 Pearson Education, Inc.
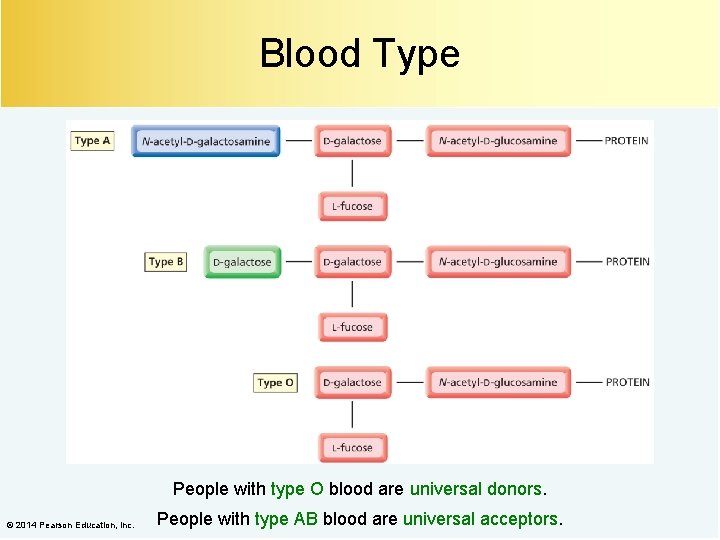
Blood Type People with type O blood are universal donors. © 2014 Pearson Education, Inc. People with type AB blood are universal acceptors.
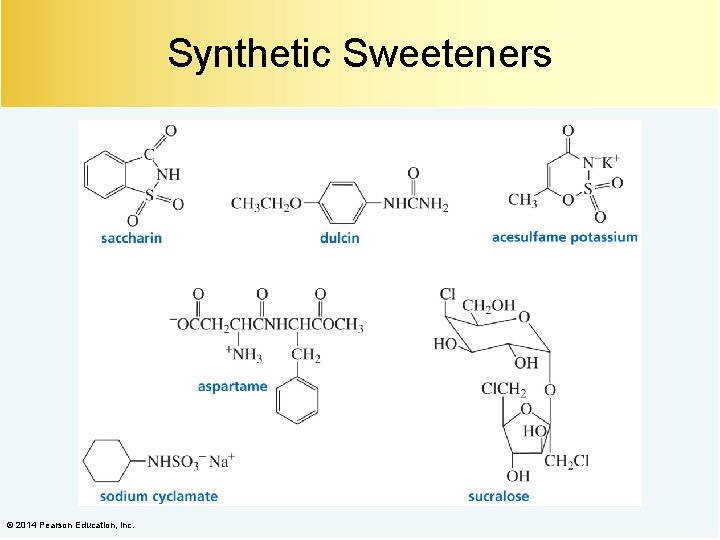
Synthetic Sweeteners © 2014 Pearson Education, Inc.
- Slides: 57