Chapter 21 Proteins 41509 Spider Silk a fibrous

Chapter 21 Proteins 4/15/09 Spider Silk, a fibrous protein. . . tough on bugs Not on you. . . (soft and smooth)

Proteins • Proteins many have functions: • 1. Structure: collagen and keratin are the chief constituents of skin, bone, hair, and nails. • 2. Catalysts: virtually all reactions in living systems are catalyzed by • proteins called enzymes. • 3. Movement: muscles are made up of proteins called myosin and actin. • 4. Transport: Transport hemoglobin transports oxygen from the lungs to cells; other proteins transport molecules across cell membranes. • 5. Hormones: many hormones are proteins, among them insulin, • oxytocin, and human growth hormone.

Proteins 6. Protection: blood clotting involves the protein fibrinogen; the body used proteins called antibodies to fight disease. 7. Storage: casein in milk and ovalbumin in eggs store nutrients for newborn infants and birds; ferritin, a protein in the liver, stores iron. 8. Regulation: certain proteins not only control the expression of genes, but also control when gene expression takes place. Proteins are divided into two types: 1. fibrous proteins (e. g. collagen, keratin) 2. globular proteins (e. g. Hemoglobin)
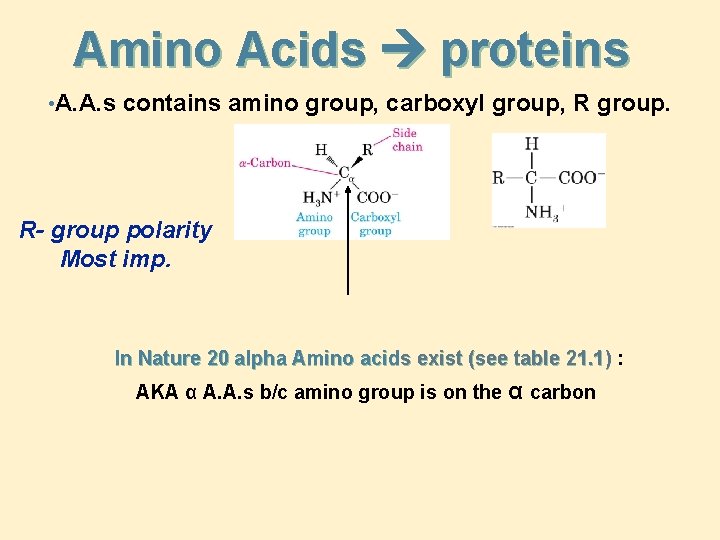
Amino Acids proteins • A. A. s contains amino group, carboxyl group, R group. R- group polarity Most imp. In Nature 20 alpha Amino acids exist (see table 21. 1) : AKA α A. A. s b/c amino group is on the α carbon
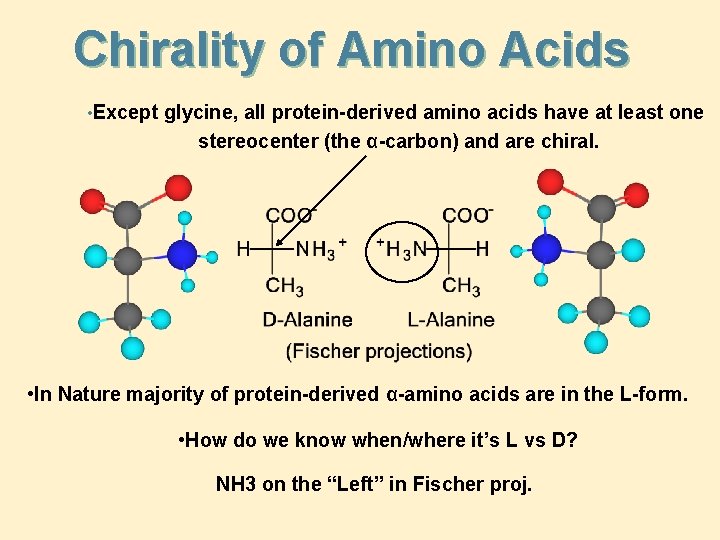
Chirality of Amino Acids • Except glycine, all protein-derived amino acids have at least one stereocenter (the α-carbon) and are chiral. • In Nature majority of protein-derived α-amino acids are in the L-form. • How do we know when/where it’s L vs D? NH 3 on the “Left” in Fischer proj.

Chirality of Amino Acids • A comparison of the stereochemistry of L-alanine and D-glyceraldehyde (as Fischer projections):

See table 21. 1 R-group polarity classification: 1. Nonpolar (hydrophobic-repel H 2 O 2. Polar (hydrophillic) 3. Basic (hydrophillic) 4. Acidic (hydrophillic) Determines structure + function of protein

20 Protein-Derived AA Red = R Groups, AKA Side Chains Nonpolar side chains (at p. H 7. 0)

20 Protein-Derived AA • Polar side chains (at p. H 7. 0) Red = R Groups, AKA Side Chains

20 Protein-Derived AA • Acidic and basic side chains (at p. H 7. 0) Red = R Groups, AKA Side Chains
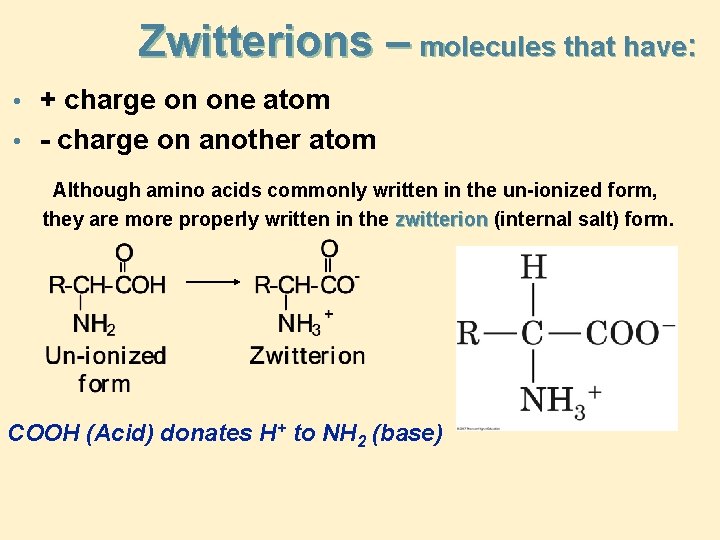
Zwitterions – molecules that have: • + charge on one atom • - charge on another atom Although amino acids commonly written in the un-ionized form, they are more properly written in the zwitterion (internal salt) form. COOH (Acid) donates H+ to NH 2 (base)
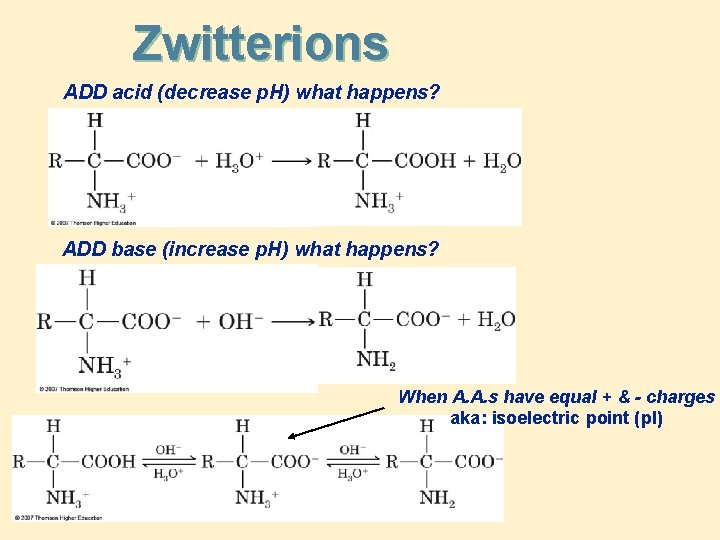
Zwitterions ADD acid (decrease p. H) what happens? ADD base (increase p. H) what happens? When A. A. s have equal + & - charges aka: isoelectric point (p. I)
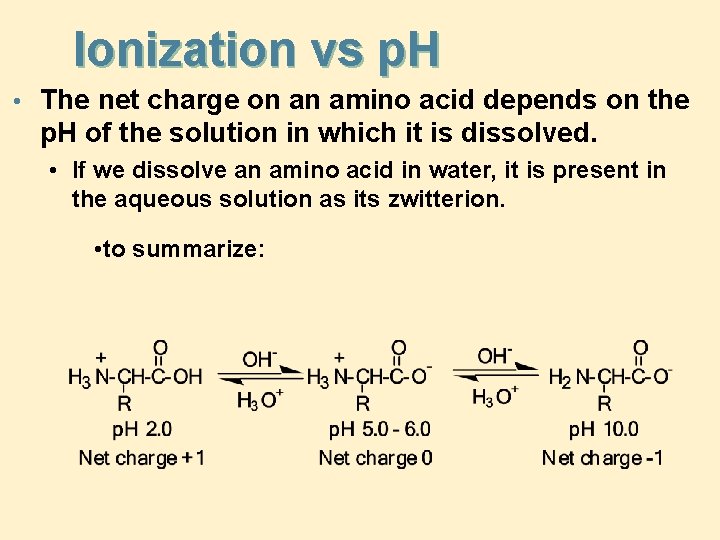
Ionization vs p. H • The net charge on an amino acid depends on the p. H of the solution in which it is dissolved. • If we dissolve an amino acid in water, it is present in the aqueous solution as its zwitterion. • to summarize:

Isoelectric Point • Isoelectric point, p. I: The p. H at which the majority of molecules of a compound in solution have no net charge.

Cysteine • The -SH (sulfhydryl) group of cysteine is easily oxidized to an -S-S- (disulfide).
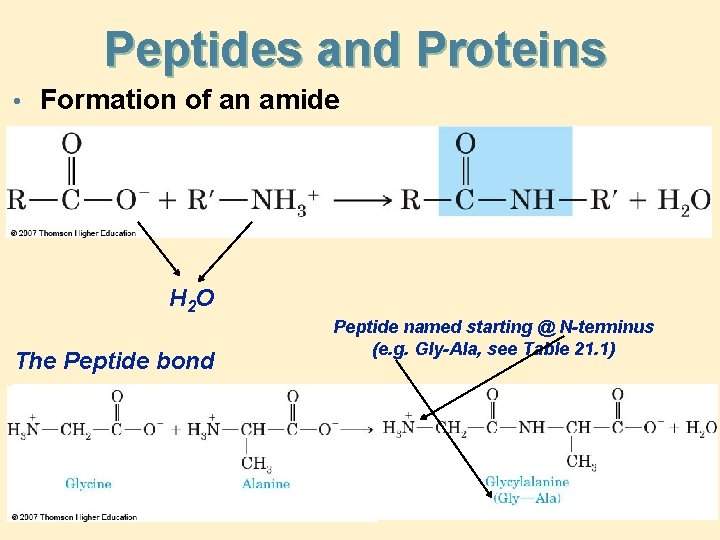
Peptides and Proteins • Formation of an amide H 2 O The Peptide bond Peptide named starting @ N-terminus (e. g. Gly-Ala, see Table 21. 1)
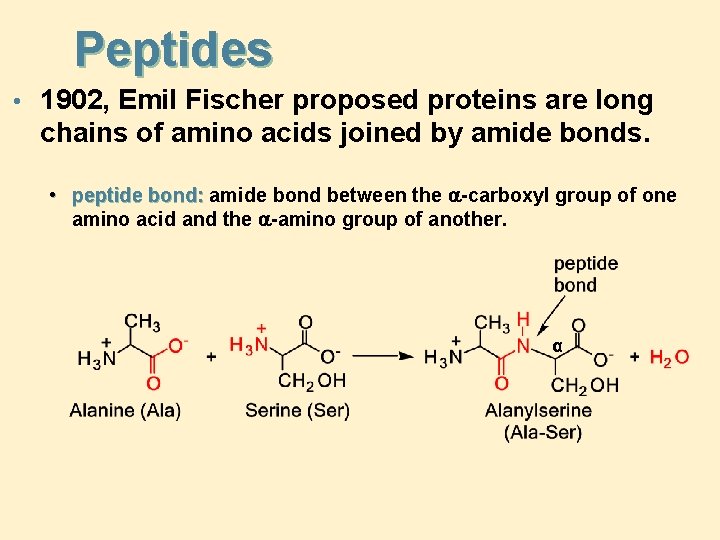
Peptides • 1902, Emil Fischer proposed proteins are long chains of amino acids joined by amide bonds. • peptide bond: amide bond between the -carboxyl group of one amino acid and the -amino group of another. α
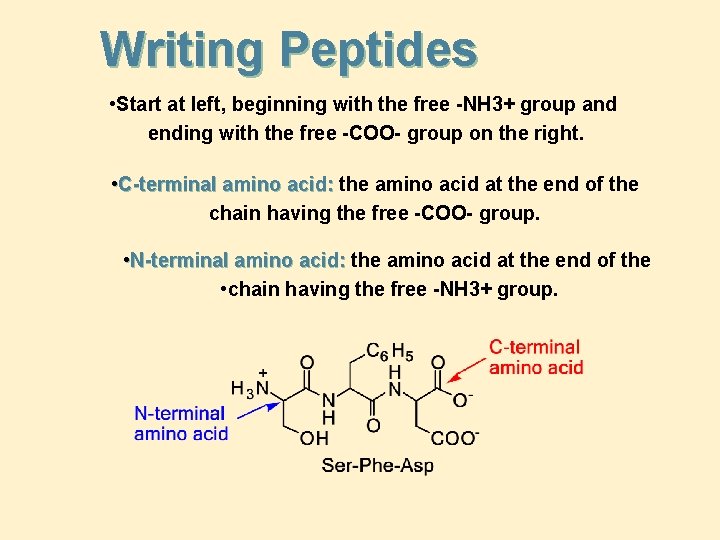
Writing Peptides • Start at left, beginning with the free -NH 3+ group and ending with the free -COO- group on the right. • C-terminal amino acid: the amino acid at the end of the chain having the free -COO- group. • N-terminal amino acid: the amino acid at the end of the • chain having the free -NH 3+ group.

Peptides and Proteins • What would this tri-peptide be called? Note: short chain A. A. s = peptides Longer chained 10 -20 A. A. s = polypeptides > 30 A. A. chains = proteins (residues)
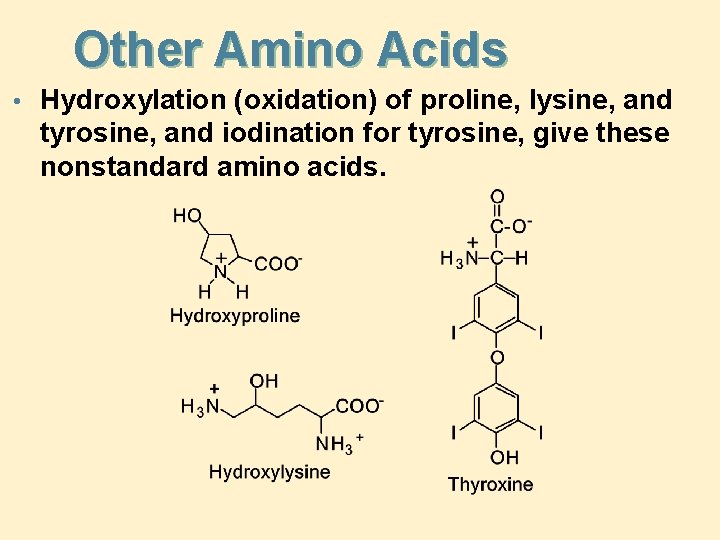
Other Amino Acids • Hydroxylation (oxidation) of proline, lysine, and tyrosine, and iodination for tyrosine, give these nonstandard amino acids.
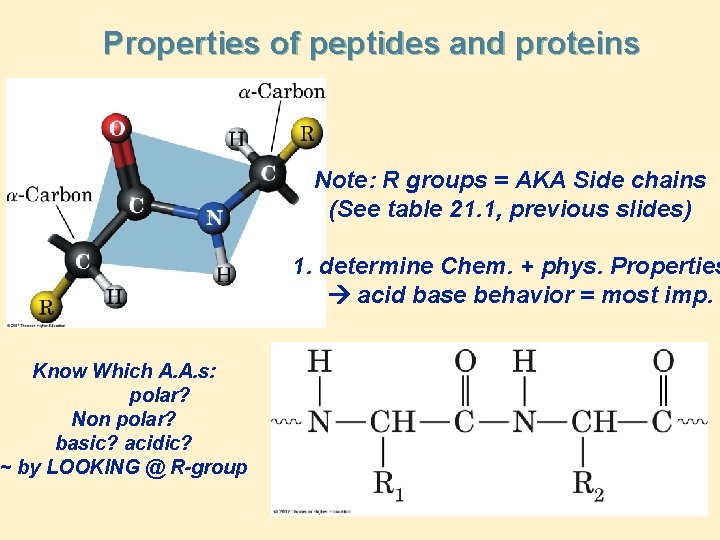
Properties of peptides and proteins Note: R groups = AKA Side chains (See table 21. 1, previous slides) 1. determine Chem. + phys. Properties acid base behavior = most imp. Know Which A. A. s: polar? Non polar? basic? acidic? ~ by LOOKING @ R-group
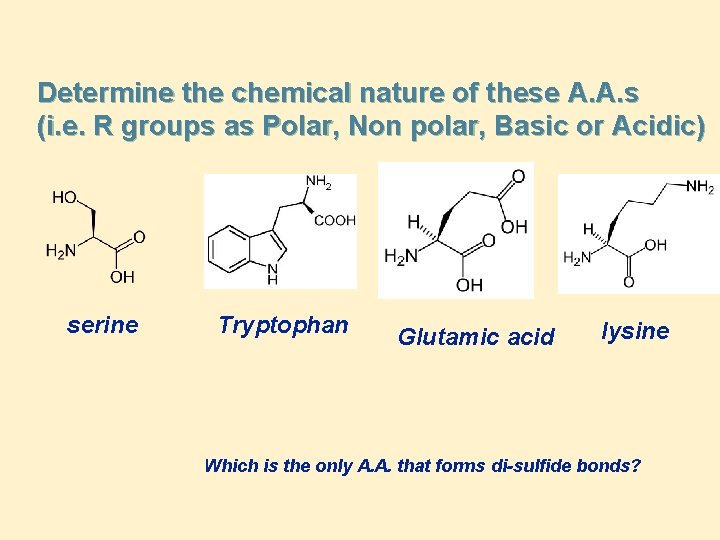
Determine the chemical nature of these A. A. s (i. e. R groups as Polar, Non polar, Basic or Acidic) serine Tryptophan Glutamic acid lysine Which is the only A. A. that forms di-sulfide bonds?

Peptides and Proteins • Proteins behave as zwitterions. • Proteins isoelectric point, p. I • At its p. I, the protein has no net charge. • At higher p. H (more basic) than its p. I, has net (-)charge. • At lower p. H (more acidic) than its p. I, has net (+)charge. • Hemoglobin, almost equal number of acidic and basic side chains; its p. I is 6. 8. ~ 7 • Serum albumin has acidic side chains; its p. I is 4. 9. • Proteins are least soluble in water at their isoelectric points and can be precipitated from solution at this p. H. Changing the p. H changes “the Nature” of Protein, )
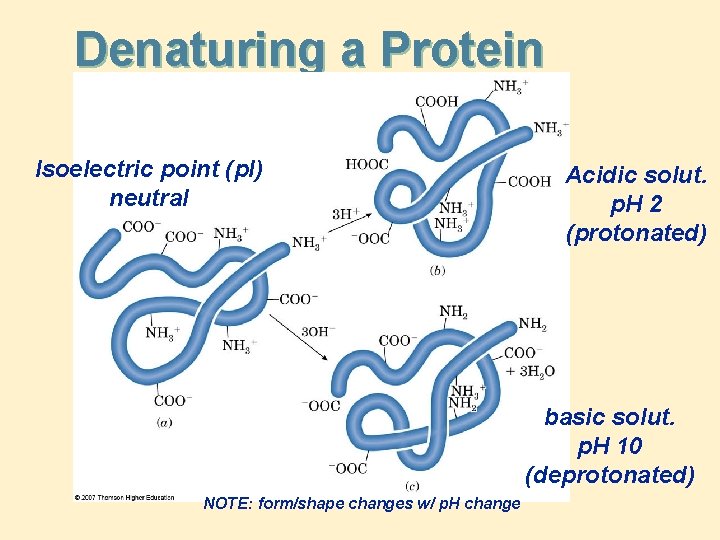
Denaturing a Protein Isoelectric point (p. I) neutral Acidic solut. p. H 2 (protonated) basic solut. p. H 10 (deprotonated) NOTE: form/shape changes w/ p. H change
Levels of Structure • Primary structure: the sequence of amino acids in a polypeptide chain; read from the N-terminal amino acid to the C-terminal amino acid. • Secondary structure: conformations of amino acids in localized regions of a polypeptide chain; examples are -helix, b-pleated sheet, and random coil. • Tertiary structure: the complete three-dimensional arrangement of atoms of a polypeptide chain. • Quaternary structure: the spatial relationship and interactions between subunits in a protein that has more than one polypeptide chain. » See Hemoglobin next slide. . .
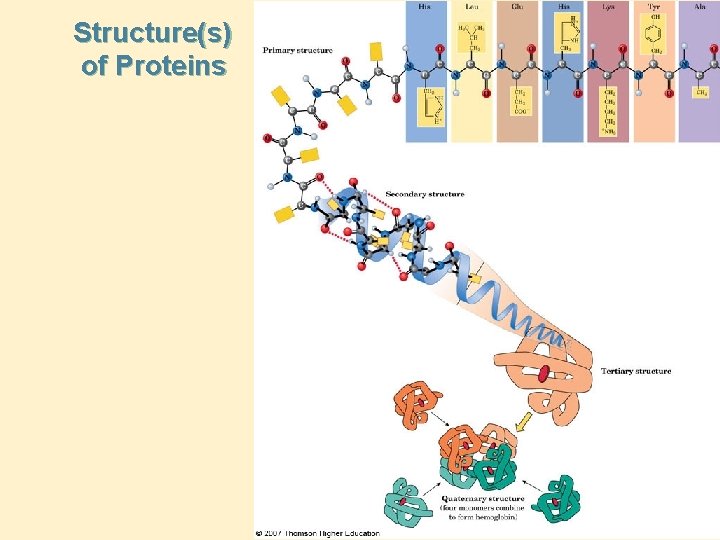
Structure(s) of Proteins
- Slides: 26