Chapter 20 Schaechters SBM 2044 Lecture 7 Tetanus

• Chapter 20 (Schaechter’s)

SBM 2044 Lecture 7 Tetanus and Botulinum Neurotoxins • Clostridium tetani Clostridium botulinum • Similar neurotoxins – different diseases Portrait by Charles Bell c. 1821

Clostridium botulinum Clostridium tetani • Gram-positive, spore forming, rods • Strict anaerobes • Normal habitat: soil

Diseases • • • Pseudomembranous colitis gangrene Cellulitis Tetanus Botulism Food poisoning

Pathogenesis Encounter, entry and multiplication: • C. botulinum spores found in soil or marine sediments, contaminated meats, vegetables and fish. • C. tetani in GIT of humans and in soil samples – most cases of tetanus are associated with a traumatic wound • C. perfringens in soil and the intestinal tract of many animals. Wound site has a low oxidation reduction potential, compromised blood supply, calcium ions and various peptides and amino acids that allows germination of spores. • Spores are heat resistant – survive food processing and canning – anaerobic spores germinate and release potent toxins – proteolytic enzymes cause spoilage of food

Damaging effects of Clostridium • Many clostridial diseases are serious and life threatening • All are caused by exotoxins secreted by the clostridia, often without colonization or invasion by the organism. • Heating canned foods insufficiently before sealing lids produces a nice anaerobic environment without killing spores – ideal conditions for germination and growth of C. botulinum. – e. g. boiling not sufficient, and many home pressure cookers also inadequate. • Even if toxin is present in food, boiling for 15 mins or so before eating is usually sufficient to inactivate toxin

Damage by C. botulinum • 1 microgram can kill a large family; 400 g could kill all the people on Earth • Botulinum toxin prevents the release of neurotransmitter Ach, muscle unable to contract • Flaccid paralysis within 12 -36 hrs after ingestion of toxin

Botulism • Most cases: Toxin ingested in food bloodstream (common source: home-canned foods) flaccid paralysis • Infant botulism: (Floppy baby syndrome) In absence of developed gut flora, C. botulinum can grow in colon (anaerobic environment) + produce toxin – less readily absorbed than ingested toxin, but can cause death. • Wound botulism: RARE – but can occur if deep wounds (anaerobic) very heavily contaminated (spores) with soil – e. g. war
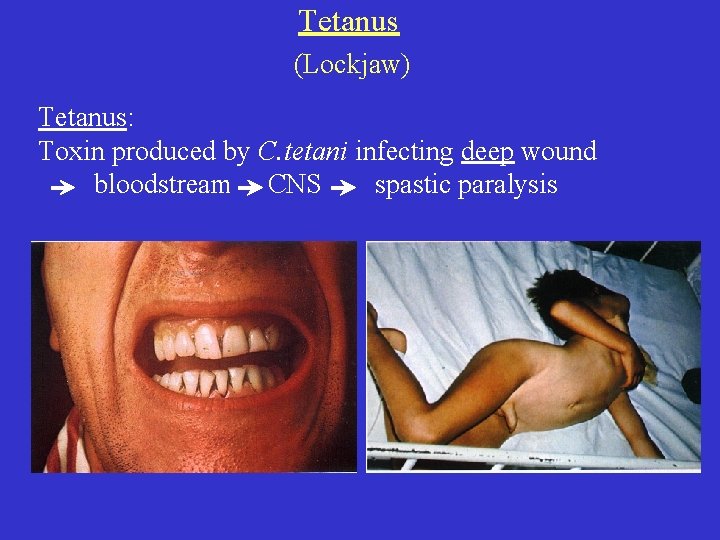
Tetanus (Lockjaw) Tetanus: Toxin produced by C. tetani infecting deep wound bloodstream CNS spastic paralysis

Clostridium neurotoxins Botulinum neurotoxin (Bo. NT): • produced by C. botulinum (C. barati, & C. butyricum) • 8 different variants (serotypes A to G) • toxin gene carried on phage or plasmid Tetanus neurotoxin (Te. NT): • produced by C. tetani • all strains produce same Tet. NT - no significant variants • gene located on a plasmid Can also produce other toxins – do not confuse the neurotoxins (e. g. C. botulinum C 2 & C 3 toxins; C. tetani tetanolysin)
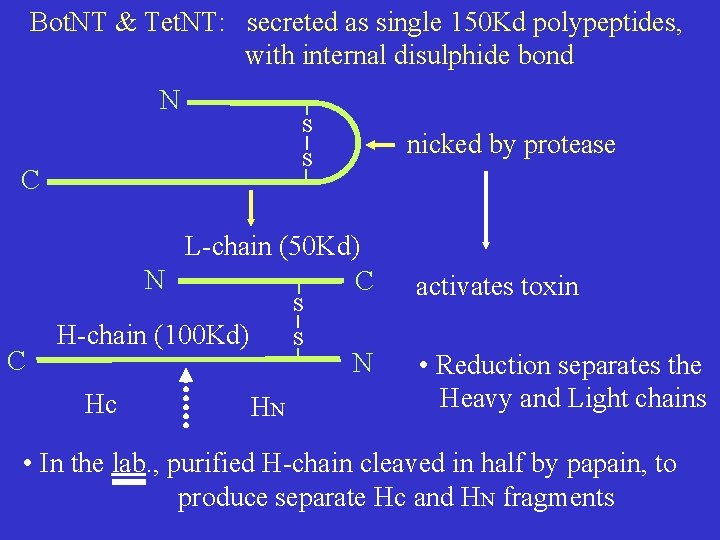
Bot. NT & Tet. NT: secreted as single 150 Kd polypeptides, with internal disulphide bond N S nicked by protease S C L-chain (50 Kd) N C S C H-chain (100 Kd) Hc S HN N activates toxin • Reduction separates the Heavy and Light chains • In the lab. , purified H-chain cleaved in half by papain, to produce separate Hc and HN fragments
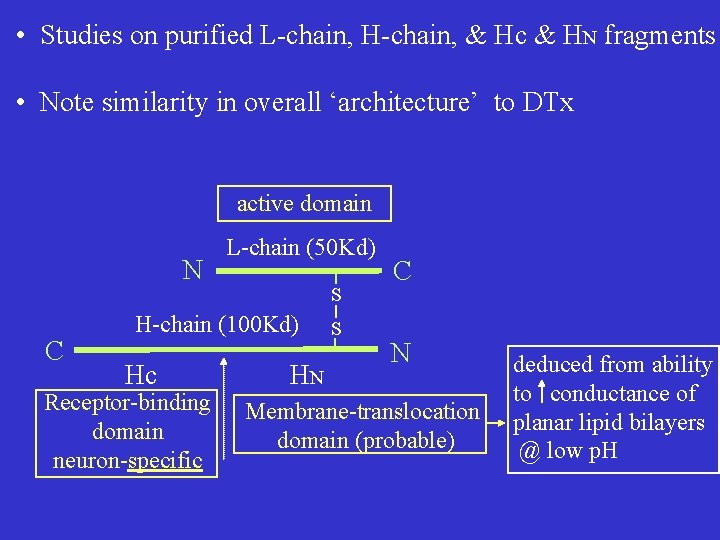
• Studies on purified L-chain, H-chain, & Hc & HN fragments • Note similarity in overall ‘architecture’ to DTx active domain N C L-chain (50 Kd) S H-chain (100 Kd) Hc Receptor-binding domain neuron-specific HN S C N Membrane-translocation domain (probable) deduced from ability to conductance of planar lipid bilayers @ low p. H

Mechanisms of action • By 1948: Bot. NT blocks neurotransmitter release at neuro-muscular junctions (NMJ) • In 1960 s: Tet. NT blocks neurotransmitter release by inhibitory interneurons in spinal cord. Both toxins very similar structures + very similar mechanisms BUT Different sites of action Very different diseases
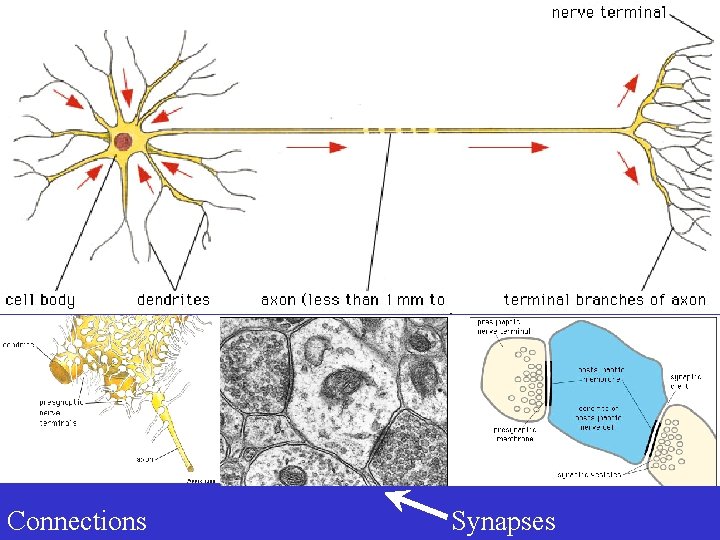
Neurons Connections Synapses
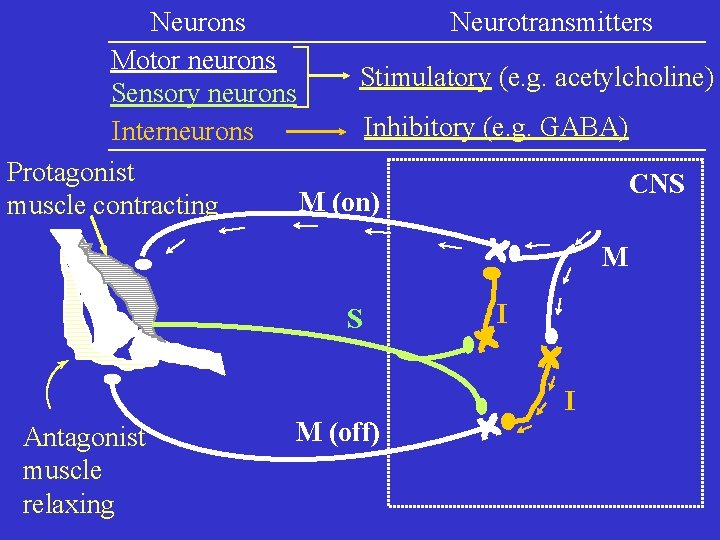
Neurons Neurotransmitters Motor neurons Stimulatory (e. g. acetylcholine) Sensory neurons Inhibitory (e. g. GABA) Interneurons Protagonist CNS M (on) muscle contracting M S I I Antagonist muscle relaxing M (off)
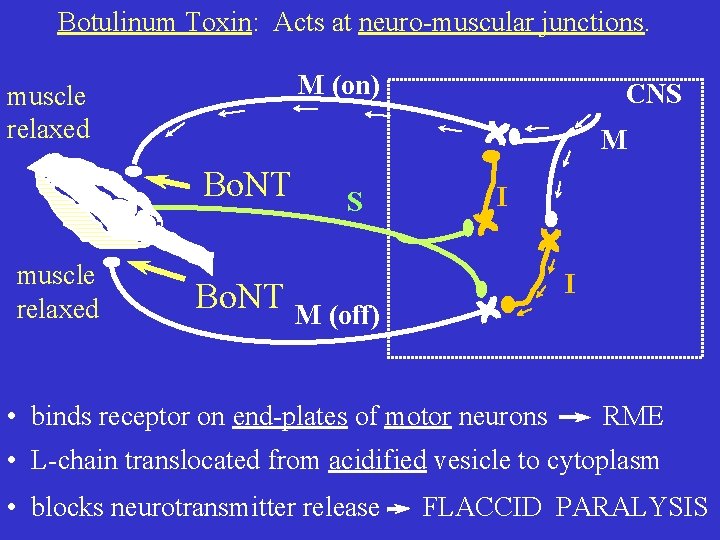
Botulinum Toxin: Acts at neuro-muscular junctions. M (on) muscle relaxed M Bo. NT muscle relaxed CNS S I I Bo. NT M (off) • binds receptor on end-plates of motor neurons RME • L-chain translocated from acidified vesicle to cytoplasm • blocks neurotransmitter release FLACCID PARALYSIS
JAMA 2001; 285; 1059 -1070

Mechanisms of action of Bot. TN

Tetanus Toxin: Acts in the CNS • binds receptor at terminals of peripheral nerves RME • Unlike Bot. NT, active domain NOT translocated at this site (reasons still unclear - distinct receptors/vesicles ? ? ) • Instead, retrograde axonal transport CNS vesicle inside axon CNS M M S I I M
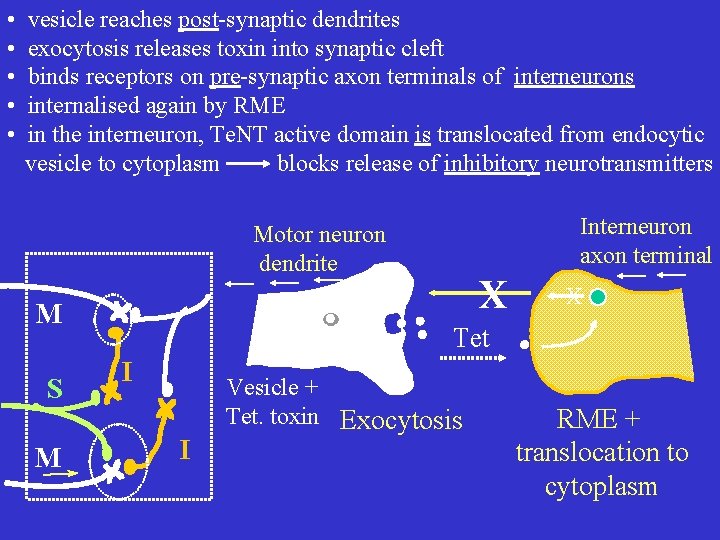
• • • vesicle reaches post-synaptic dendrites exocytosis releases toxin into synaptic cleft binds receptors on pre-synaptic axon terminals of interneurons internalised again by RME in the interneuron, Te. NT active domain is translocated from endocytic vesicle to cytoplasm blocks release of inhibitory neurotransmitters Interneuron axon terminal Motor neuron dendrite M S M X X Tet I I Vesicle + Tet. toxin Exocytosis RME + translocation to cytoplasm
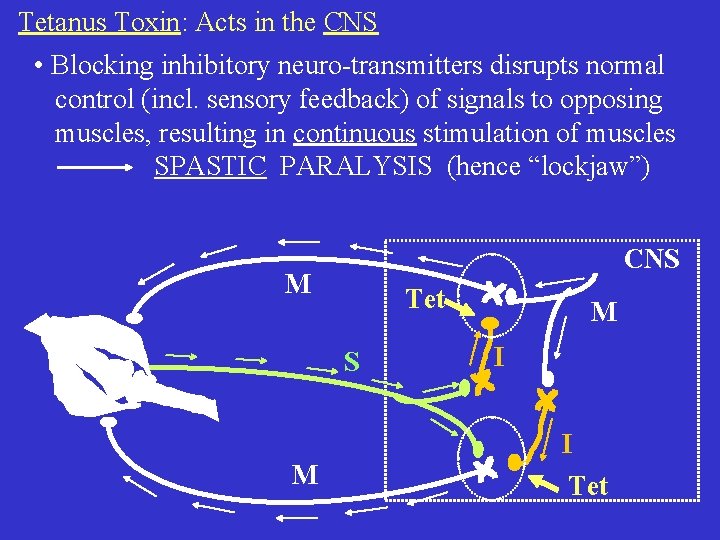
Tetanus Toxin: Acts in the CNS • Blocking inhibitory neuro-transmitters disrupts normal control (incl. sensory feedback) of signals to opposing muscles, resulting in continuous stimulation of muscles SPASTIC PARALYSIS (hence “lockjaw”) CNS M Tet S M I I M Tet

Mechanism of action of L-chain DNA sequencing provided key clue • Regions of strongest homology between amino acid sequences of the L-chains of Tet. NT & various Bot. NTs included a Zn-binding ‘motif’ similar to that in Zn-dependent proteases called Zn-endopeptidases Strongly suggested that L-chains act as proteases Must ‘target’ proteins involved in neurotransmitter release
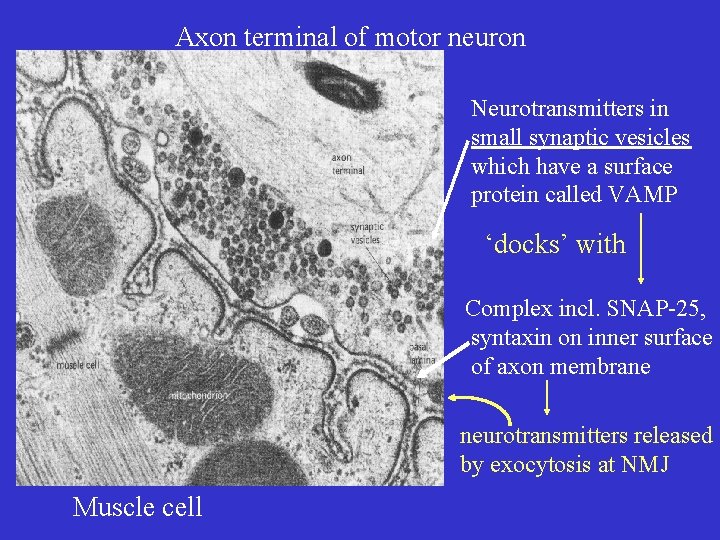
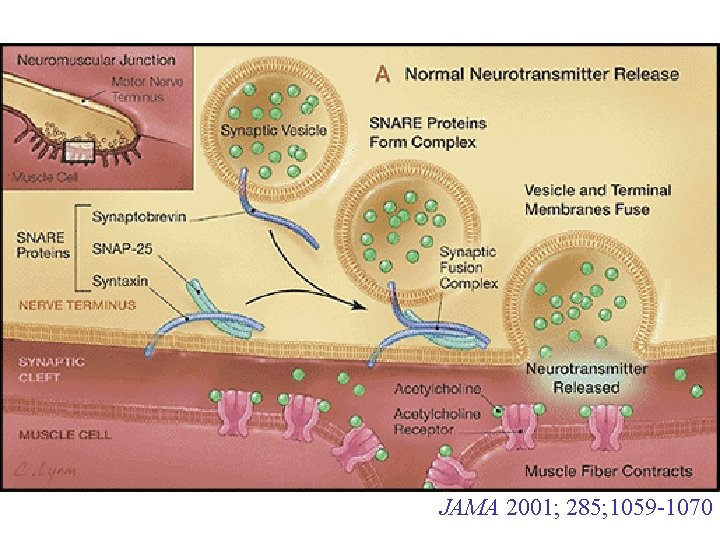
Axon terminal of motor neuron Neurotransmitters in small synaptic vesicles which have a surface protein called VAMP ‘docks’ with Complex incl. SNAP-25, syntaxin on inner surface of axon membrane neurotransmitters released by exocytosis at NMJ Muscle cell
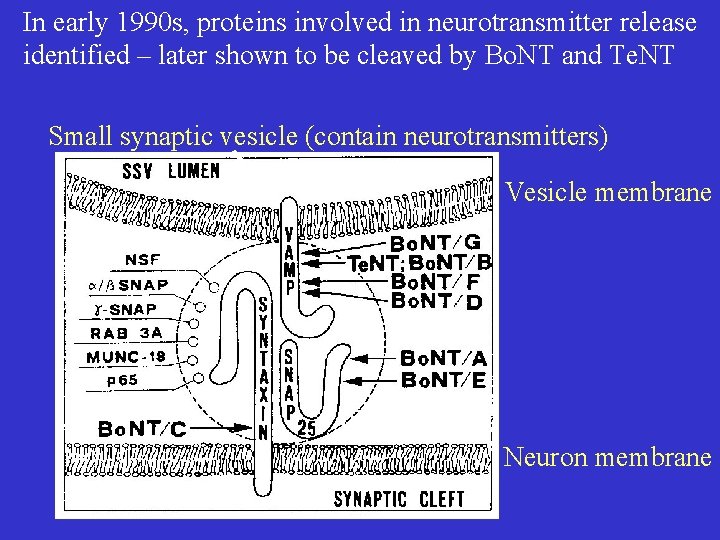
In early 1990 s, proteins involved in neurotransmitter release identified – later shown to be cleaved by Bo. NT and Te. NT Small synaptic vesicle (contain neurotransmitters) Vesicle membrane Neuron membrane
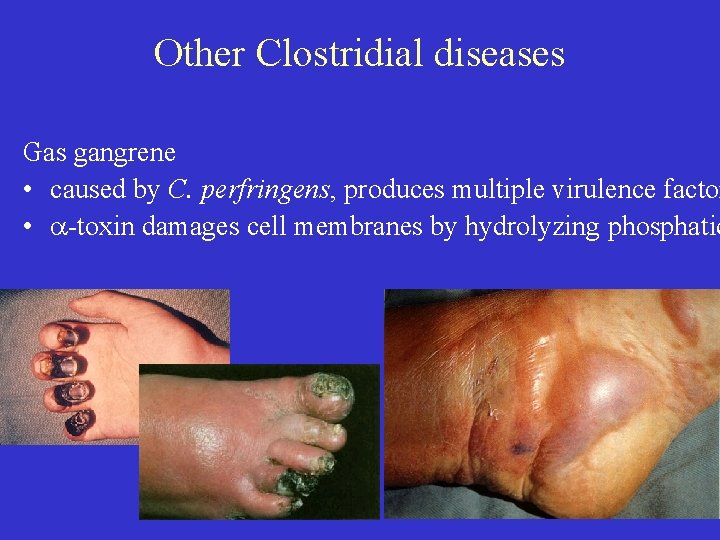
Other Clostridial diseases Gas gangrene • caused by C. perfringens, produces multiple virulence factor • -toxin damages cell membranes by hydrolyzing phosphatid

Other Clostridial diseases C. difficile • Cause severe ulcerating disease of the large bowel – pseudomembranous colitis (diarrhoeal disease) • Antibiotic-associated diarrhoea – milder and less severe than pseudomembranous colitis

Treatment and Prevention • Botulism – Trivalent antitoxin for types A, B and E. antitoxin from horses – Botulism can be prevented by proper canning methods • Tetanus – Tetanus toxoid vaccine – Antibiotic to kill the organism – Physiological exercise to limit complications • Gas gangrene – Prompt treatment of either surgical removal or penicillin administration to control wound infection • C. difficile diarrhoea – Antibiotics (cephalopsorins, ampicillin, clindamycin)

Term Paper 1. Discuss how sea cucumber protein could potentially help in infectious diseases. 2. How do animal slaughtering contribute to the spread of zoonosis diseases? 3. Vaccination can be unsafe and ineffective. Give your comments. 4. Tell us about ONE most interesting topic in the microbiology subject. Justify by giving reasons and examples to your chosen topic.

Term Paper • Choose your question number. (3 students per question) • Write in word-processed, fully formatted – PLUS legends for tables, figures, – and REFERENCES – 1. 5 or 2. 0 spacing • Write for ONLY 15 – 30 PAGES. • Remember to write not just a purely scientific paper, but a philosophical one! • Deadline: FRIDAY, 27 FEBRUARY 2009
- Slides: 29