Chapter 20 Overview OxidationReduction reactions Balancing Redox Reactions

Chapter 20

Overview • Oxidation-Reduction reactions » Balancing Redox Reactions • Half-Reaction method • Acidic Solution • Basic Solution • • Voltaic Cells Cell EMF--standard reduction potentials Oxidizing & Reducing reagents Spontaneity of Redox reactions

• Effect of Concentration » Nernst Equation » Equilibrium Constants • Commercial Voltaic Cells • Electrolysis » Quantitative Aspects » Electrical Work

Redox Reactions • Involve a transfer of electrons » Oxidation loss of one or more electron(s) • oxidation state will increase » Reduction gain of one or more electron(s) • oxidation state will decrease » Must occur simultaneously Zn(s) + Cu 2+(aq) Zn 2+(aq) + Cu(s) Zn 2+(aq) + 2 e. Cu 2+(aq) + 2 e- Cu(s) oxidation ½ rxn reduction ½ rxn

You must know oxidation states: (Review: Section 8. 10) • What are the oxidation states of each atom in the following: » H 2 » CO » Cl. O 2» HC 2 H 3 O 2 H 0 C +2, O -2 Cl +3, O -2 H +1, C 1 +3, C 2 -3, O

Balancing Redox Reactions • Mass balance must be observed • e--transfer must be balanced • Simple reactions: » Sn 2+ + Fe 3+ Sn 4+ + Fe 2+ • Sn 2+ Sn 4+ + 2 e • Fe 3+ + e- Fe 2+ x 2 2 Fe 3+ + 2 e- 2 Fe 2+ oxidation ½ rxn reduction ½ rxn Sn 2+ + 2 Fe 3+ Sn 4+ + 2 Fe 2+

• Reactions involving H & O in acid: » Mn. O 4 - + C 2 O 42 - Mn 2+ + CO 2 » write both ½ reactions • Mn. O 4 • C 2 O 42 - Mn 2+ CO 2 » mass balance (all except H & O) • Mn. O 4 • C 2 O 42 - Mn 2+ 2 CO 2 » add H 2 O & H+ to balance O & H • 8 H+ + Mn. O 4 • C 2 O 42 - 2 CO 2 Mn 2+ + 4 H 2 O

» balance charge by adding electrons • 5 e- + 8 H+ + Mn. O 4 • C 2 O 42 - 2 CO 2 + 2 e- Mn 2+ + 4 H 2 O » balance electrons transferred • 10 e- + 16 H+ + 2 Mn. O 4 • 5 C 2 O 42 - 10 CO 2 + 10 e- 2 Mn 2+ + 8 H 2 O » add half reactions • 16 H+ + 2 Mn. O 4 -+ 5 C 2 O 42 - 10 CO 2 + 2 Mn 2+ + 8 H 2 O » check the balance
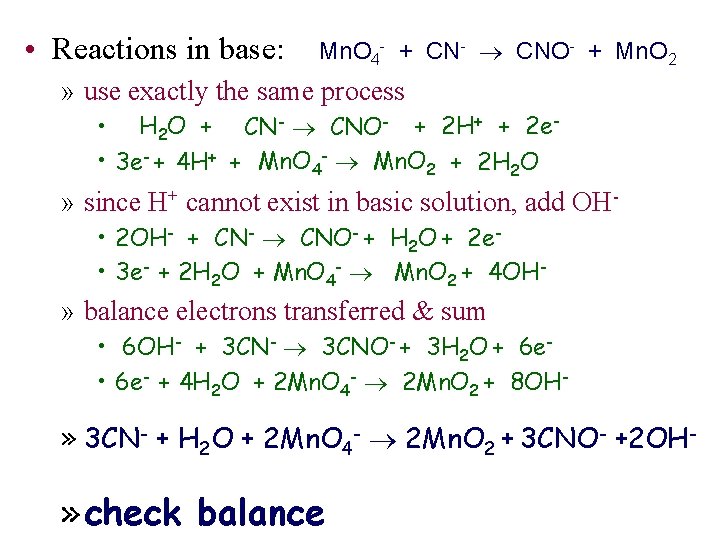
• Reactions in base: Mn. O 4 - + CN- CNO- + Mn. O 2 » use exactly the same process • H 2 O + CN- CNO- + 2 H+ + 2 e • 3 e- + 4 H+ + Mn. O 4 - Mn. O 2 + 2 H 2 O » since H+ cannot exist in basic solution, add OH • 2 OH- + CN- CNO- + H 2 O + 2 e • 3 e- + 2 H 2 O + Mn. O 4 - Mn. O 2 + 4 OH- » balance electrons transferred & sum • 6 OH- + 3 CN- 3 CNO- + 3 H 2 O + 6 e • 6 e- + 4 H 2 O + 2 Mn. O 4 - 2 Mn. O 2 + 8 OH- » 3 CN- + H 2 O + 2 Mn. O 4 - 2 Mn. O 2 + 3 CNO- +2 OH- » check balance

Voltaic Cells • A spontaneous redox reaction that does work • Anode » electrode at which oxidation occurs » loses mass » electrons released, sign is negative • Cathode » electrode at which reduction occurs » gains mass » electrons consumed, sign is positive
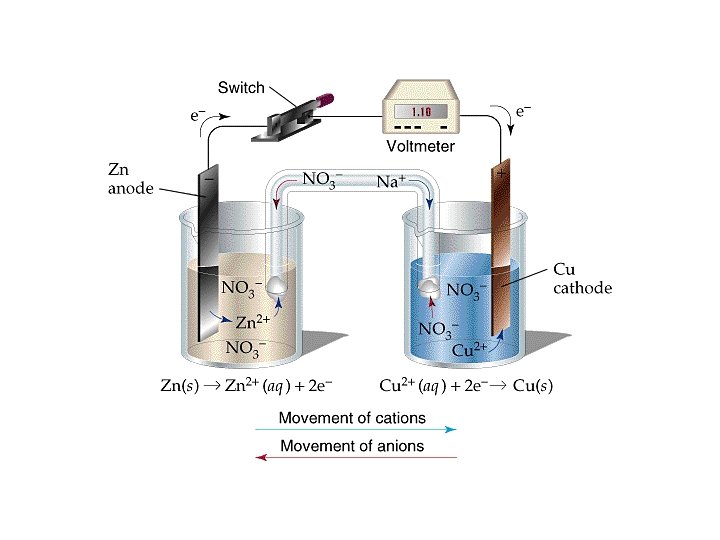

Cell EMF • Difference in potential energy of electrons at the anode and cathode » Diff. in potential energy per electrical charge measured in volts » 1 V = 1 J C • Potential difference = EMF, electromotive force • Ecell = cell potential = cell voltage » Eºcell = cell potential under std. conditions • 1 M, 1 atm, 25 ºC

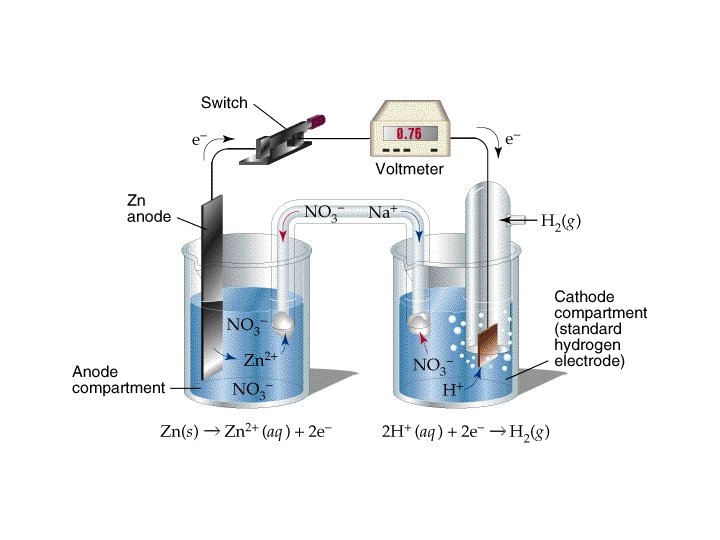
• Standard reduction potentials » E ºred in tables » E ºcell = E ºred (cathode) - E ºred (anode) • Based on “standard hydrogen electrode” » 2 H+(aq, 1 M) + 2 e- H 2(g, 1 atm) E ºred = 0 V » Zn(s) + 2 H+(aq) Zn 2+(aq) + H 2(g) E ºcell = 0. 76 V » 0. 76 V = 0 V - E ºred (anode) • Zn 2+(aq, 1 M) + 2 e- Zn(s) E ºred (anode) = -0. 76 V


Problem: • Calculate Eºcell for • 2 Al(s) + 3 I 2(s) 2 Al 3+(aq) + 6 I-(aq) » Anode: 2 Al 3+ + 6 e- » Cathode: 3 I 2 + 6 e- 6 I • Eºcell = E ºred (cathode) - E ºred (anode) • E ºcell = 0. 54 V - (-1. 66 V) • E ºcell = 2. 20 V

• Note: stoichiometric coefficient does not affect the value of the E ºred (it is an intensive property) • E ºox = - E ºred • 2 Al(s) + 3 I 2(s) 2 Al 3+(aq) + 6 I-(aq) • 2 Al 3+ + 6 e- E ºox = +1. 66 V • 3 I 2 + 6 e- 6 I- E ºred = +0. 54 V • E ºcell = E ºox + E ºred = 2. 20 V • The more positive the E ºcell the more driving force for the reaction

Oxidizing/Reducing Agents • Oxidizing agents cause oxidation » oxidizing agents are reduced » the more (+) the E ºred the better the ox. agent • Reducing agents cause reduction » reducing agents are oxidized » the more (-) the E ºred the better the red. agent
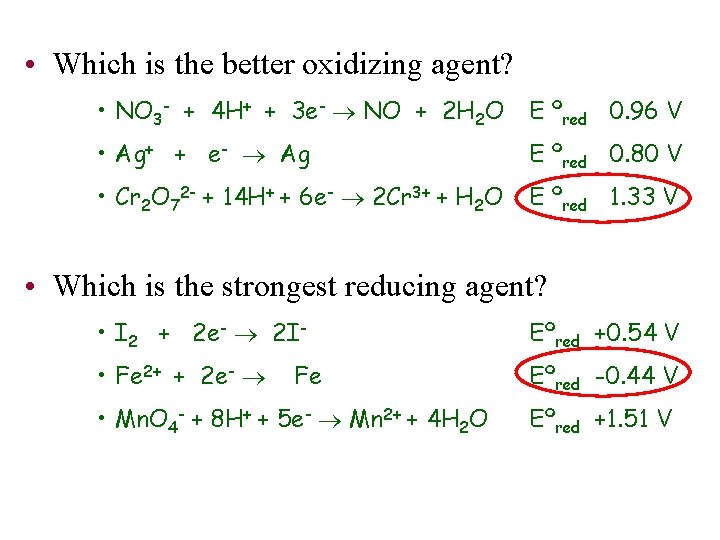
• Which is the better oxidizing agent? • NO 3 - + 4 H+ + 3 e- NO + 2 H 2 O E ºred 0. 96 V • Ag+ + e- Ag E ºred 0. 80 V • Cr 2 O 72 - + 14 H+ + 6 e- 2 Cr 3+ + H 2 O E ºred 1. 33 V • Which is the strongest reducing agent? • I 2 + 2 e- 2 I- Eºred +0. 54 V • Fe 2+ + 2 e- Eºred -0. 44 V Fe • Mn. O 4 - + 8 H+ + 5 e- Mn 2+ + 4 H 2 O Eºred +1. 51 V

Spontaneity of Redox Reactions • Spontaneous redox rxns have positive potentials • Non-spontaneous redox rxns have negative potentials • Is this rxn spont. or non-spont. ? » Mn. O 4 - + 8 H+ + 5 Fe 2+ 5 Fe 3+ + Mn 2+ + 4 H 2 O • Fe 2+ Fe 3+ + 1 e • Mn. O 4 - + 8 H+ + 5 e- Mn 2+ + 4 H 2 O • E ºox + E ºred = + 0. 74 v Yes Eºox = -0. 77 v E ºred = +1. 51 v

EMF & Free Energy • If both DG & E are a measure of spontaneity, they must be related » DG = - n. FE • F is Faraday’s constant 1 F = 96, 500 J/v mol e- • remember: 1 C = 1 J/v • n = mol e- transferred » In the standard state DGº = - n. FEº

• Calculate the standard free energy change for Hg + 2 Fe 3+ Hg 2+ + 2 Fe 2+ » n = 2 mol electrons transferred • Hg 2+ + 2 e- Eox = - 0. 854 v • 2 Fe 3+ +2 e- 2 Fe 2+ Ered= + 0. 771 v • Ecell = - 0. 083 v » DG = - (2 mol e-)(-0. 083 v)(96, 500 J/v mol e-) » = + 16 k. J

Concentration & Cell EMF • Nernst Equation » relationship between DG & concentrations • DG = DGº + RT ln Q Q = [prod]x/[react]y » substitute -n. FE for DG • E = Eº - (RT/n. F) ln Q or • E = Eº - (2. 303 RT/n. F) log Q • 2. 303 RT/F = 0. 0592 v-mol e- at std. temp. • E = Eº - (0. 0592/n) log Q
![• Calculate the emf that the following cell generates when [Mn 2+] = • Calculate the emf that the following cell generates when [Mn 2+] =](http://slidetodoc.com/presentation_image_h2/4aad7d64e1549b5190135dcaad03fe5c/image-24.jpg)
• Calculate the emf that the following cell generates when [Mn 2+] = 0. 10 M & [Al 3+] = 1. 5 M 2 Al + 3 Mn 2+ 2 Al 3+ + 3 Mn • Eº = + 0. 48 v • E = (+ 0. 48 v) - (0. 0592 v/ 6) log [(1. 5)2/(0. 10)3] • E = + 0. 45 v • when [Mn 2+] = 1. 5 M & [Al 3+] = 0. 10 M • E = (+ 0. 48 v) - (0. 0592 v/ 6) log [(0. 10)2/(1. 5)3] • E = + 0. 51 v

Equilibrium Constants • Remember DG = DGº + RT ln Q, if Q = K, then DG = 0, therefore -n. FE = 0 and • 0 = Eº - (RT/n. F) ln K or • 0 = Eº - (0. 0592/n) log K • K can be calculated from cell potentials • log K = n. E º/0. 0592

• Calculate the equilibrium constant, K, for 2 IO 3 - + 5 Cu + 12 H+ I 2 + 5 Cu 2+ + 6 H 2 O • Eº = + 0. 858 v • n = 10 mol e- transferred • log K = n. Eº/0. 0592 • log K = 145 • K = 1 x 10145

Voltaic Cells • Lead storage battery » Pb. O 2 + SO 4 -2 + 4 H+ + 2 e- Pb. SO 4 + H 2 O Pb + SO 42 - Pb. SO 4 + 2 e • Ecell = + 2. 041 v • Dry cell » NH 4+ + 2 Mn. O 2 + 2 e- Mn 2 O 3 + 2 NH 3 + H 2 O Zn 2+ + 2 e • In an alkaline cell the NH 4 Cl is replaced with KOH

• Ni-Cd » Ni. O 2 + 2 H 2 O + 2 e- Ni(OH)2 + 2 OHCd + 2 OH- Cd(OH)2 + 2 e- • Fuel cells » 4 e- + O 2 + 2 H 2 O 4 OH 2 H 2 + 4 OH- 4 H 2 O

Electrolytic Cells • Redox reactions that are not spontaneous • Must be driven by an outside source of electrical energy • Cathode » reduction occurs » by sign convention, is negative • Anode » oxidation occurs » by sign convention, is positive

Quantitative Aspects • Redox reactions occur in stoichiometric relationship to the transfer of electrons • Electrons put into a system through electrical energy, can be quantized » Coulomb = quantity of charge passing through electrical circuit in 1 s at 1 ampere (A) current • Coulomb = (amp) (seconds)

Problem: Calculate the mass of Mg formed upon passage of a current of 60. 0 A for a period of 4. 00 x 10 3 s. • Mg. Cl 2 Mg + Cl 2 » Mg 2+ + 2 e- Mg 2 Cl- Cl 2 + 2 e • we are concerned with the reduction • (60. 0 A)(4 x 103 s)(1 C/1 A-s) = 2. 4 x 105 C • (2. 4 x 105 C)(1 mol e-/ 96, 500 C) = 2. 49 mol e • (2. 49 mol e-)(1 mol Mg/2 mol e-) = 1. 24 mol Mg • (1. 24 mol Mg)(24. 3 g/mol) = 30. 1 Mg

Electrical Work • DG = wmax DG = - n. FE wmax = - n. FE • Max work proportional to potential • wmax • J = = - n F E (mol) (C/mol) (J/C) • Electrical work = (watt) (time) • 1 watt (W) = 1 J/s • 1 k. Wh = 3. 6 x 106 J or watt-s = J
- Slides: 32