Chapter 20 Chemical Bonds SECTION 3 WRITING FORMULAS

Chapter 20: Chemical Bonds SECTION 3: WRITING FORMULAS AND NAMING COMPOUNDS

BINARY IONIC COMPOUNDS � Binary compound- one that is composed of 2 elements � Example: � But KI, Na. Cl before you can write these we have to know a few pieces of information…

WHAT WILL I NEED TO KNOW TO WRITE AN IONIC FORMULA? � Are electrons gained or lost? � When you know this…you can figure out the oxidation number- tells you how many electrons an atom has gained, lost, or shared to become stable (IT’S THE CHARGE) � Example: Na has an oxidation # of +1 � When something looks like this: iron(III) oxide, the roman numeral is the oxidation number of iron � But…compounds � Example: are neutral Ca. F 2 and Al 2 O 3
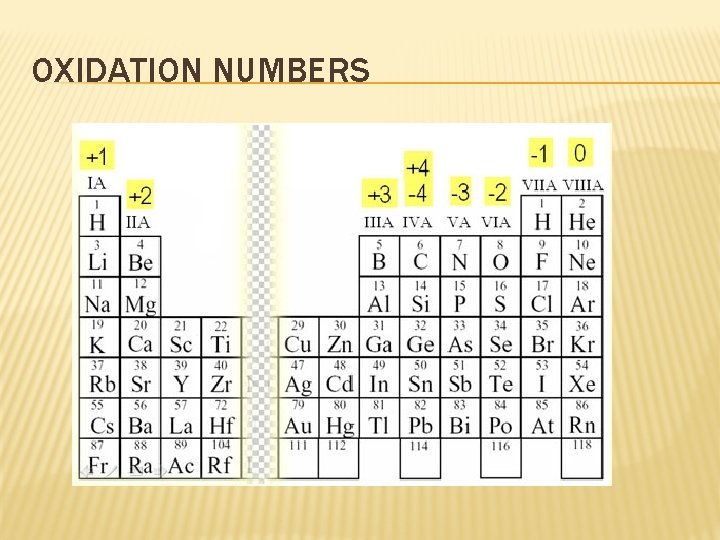
OXIDATION NUMBERS

WRITING IONIC FORMULAS � 1. Always write the positive ion first (metal) followed by the negative ion (nonmetal) � 2. (Hint) The charge of one ion (without the sign) becomes the subscript of the other ion � Example: You choose

WRITING NAMES OF IONIC COMPOUNDS 1. Write the name of the positive ion � 2. Use table 2 in your book to see if the positive ion is capable of forming more than one oxidation number. If so, determine the charge it is using by looking at the negative ion (only have 1 possible charge) � 3. Write the root name of the negative ion � � Example: Chlor- for chlorine and ox- for oxygen � 4. Add the ending –ide to the root � Example: Tell me the name of Ba. F 2

COMPOUNDS WITH COMPLEX IONS � Not all compounds are binary � Example: Na. HCO 3 � Contains a polyatomic ion- a positively or negatively charged, covalently bonded group of atoms � Table of these on p. 619
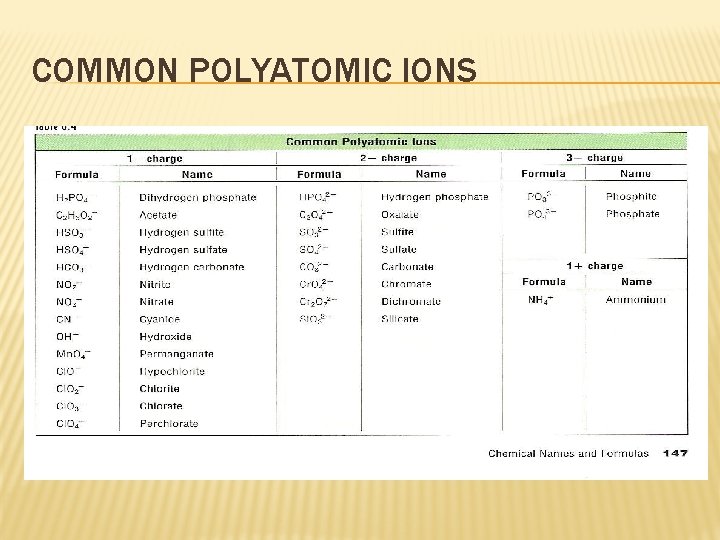
COMMON POLYATOMIC IONS

WRITING NAMES AND FORMULAS FOR COMPOUNDS WITH COMPLEX IONS � Writing Names � 1. Write the name of the positive ion � 2. Use Table 4 p. 619 to find the name of the polyatomic ion � 3. Write the name of the negative ion � Examples: � Writing K 2 SO 4 and Sr(OH)2 Formulas � Follow the rules for binary compounds, except when more than one polyatomic ion is needed, write parentheses around the polyatomic ion before adding the subscript � Example: Barium chlorate

COMPOUNDS WITH ADDED WATER � Some ionic compounds have water molecules as part of their structure � Called hydrates- a compound that has water chemically attached to its ions and written into its chemical formula � Naming � Example: Ca. SO 4 2 H 2 O

NAMING BINARY COVALENT COMPOUNDS � Remember, covalent compounds are formed between elements that are nonmetals � Using Prefixes � We use prefixes to indicate the number of atoms of each element are in the Compound � Example: N 2 O 5
- Slides: 11