Chapter 20 Carboxylic Acid Derivatives NAS Carboxylic acid
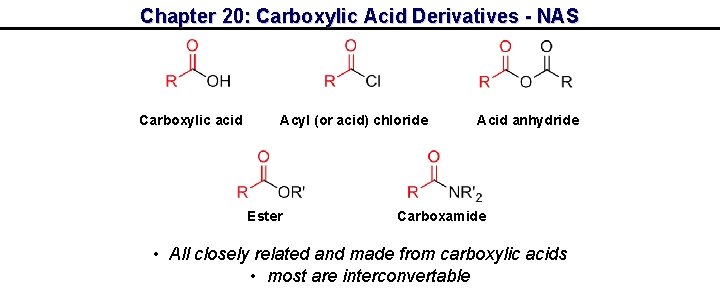
Chapter 20: Carboxylic Acid Derivatives - NAS Carboxylic acid Acyl (or acid) chloride Ester Acid anhydride Carboxamide • All closely related and made from carboxylic acids • most are interconvertable
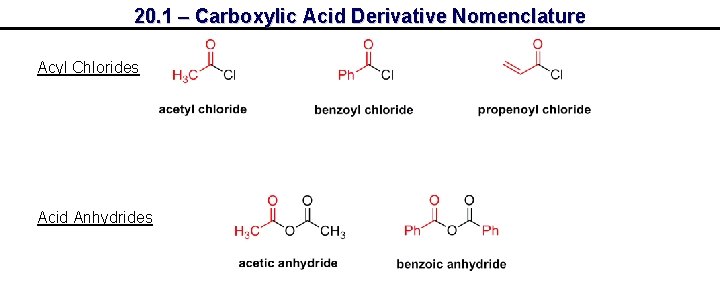
20. 1 – Carboxylic Acid Derivative Nomenclature Acyl Chlorides Acid Anhydrides
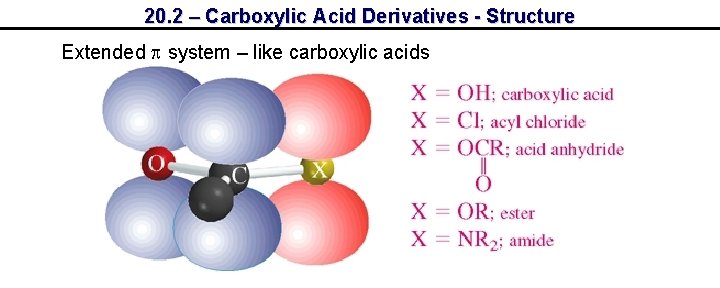
20. 2 – Carboxylic Acid Derivatives - Structure Extended system – like carboxylic acids
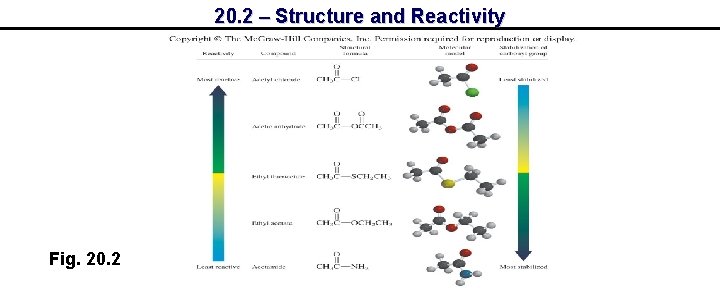
20. 2 – Structure and Reactivity Fig. 20. 2
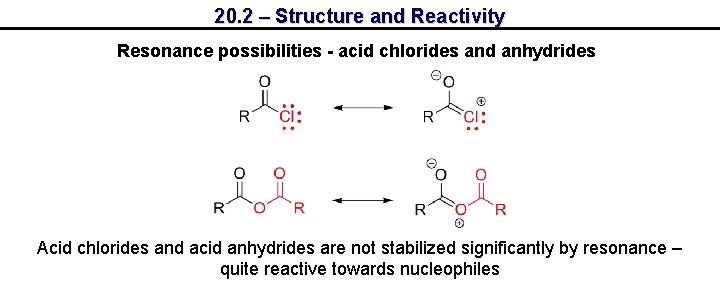
20. 2 – Structure and Reactivity Resonance possibilities - acid chlorides and anhydrides Acid chlorides and acid anhydrides are not stabilized significantly by resonance – quite reactive towards nucleophiles
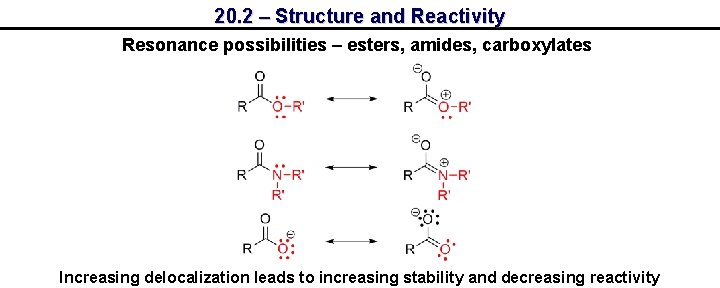
20. 2 – Structure and Reactivity Resonance possibilities – esters, amides, carboxylates Increasing delocalization leads to increasing stability and decreasing reactivity
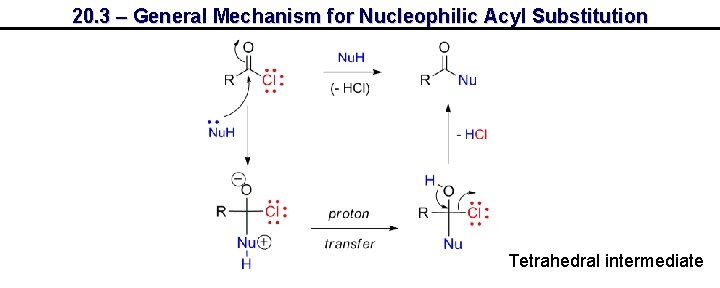
20. 3 – General Mechanism for Nucleophilic Acyl Substitution Tetrahedral intermediate
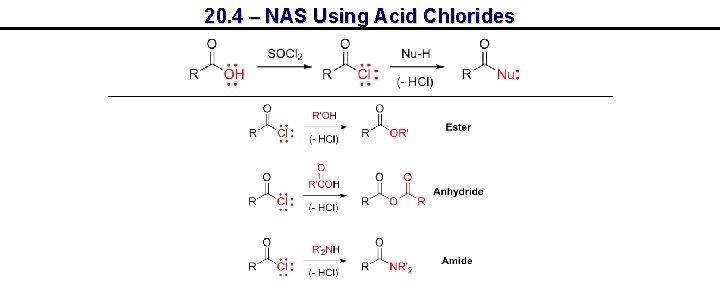
20. 4 – NAS Using Acid Chlorides
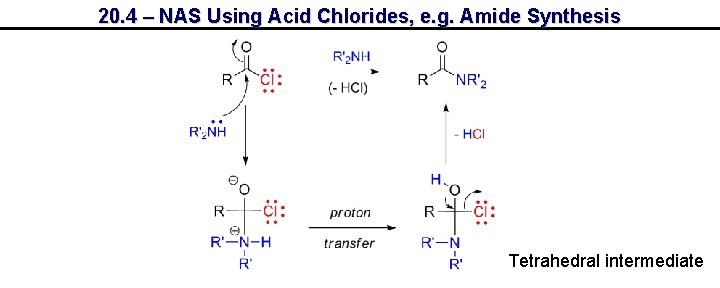
20. 4 – NAS Using Acid Chlorides, e. g. Amide Synthesis Tetrahedral intermediate
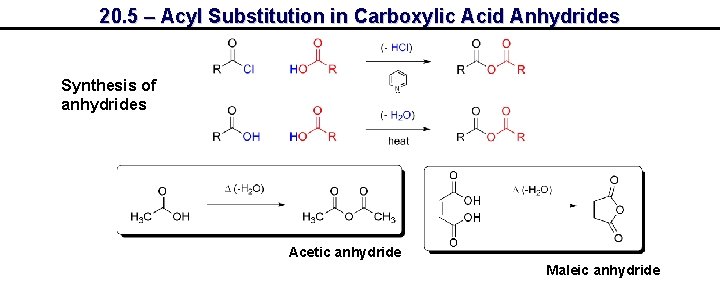
20. 5 – Acyl Substitution in Carboxylic Acid Anhydrides Synthesis of anhydrides Acetic anhydride Maleic anhydride
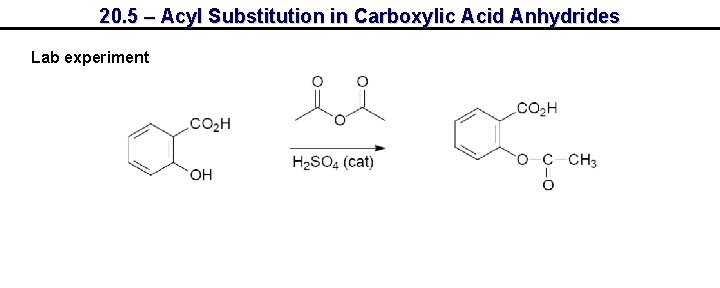
20. 5 – Acyl Substitution in Carboxylic Acid Anhydrides Lab experiment
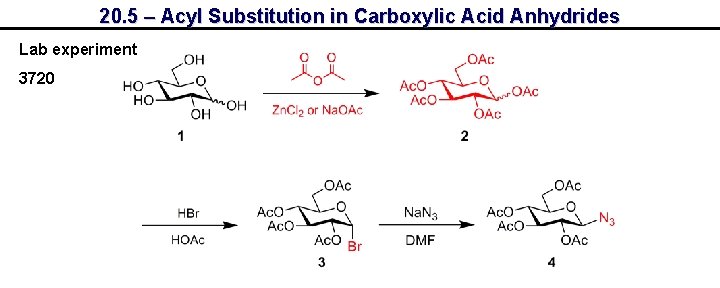
20. 5 – Acyl Substitution in Carboxylic Acid Anhydrides Lab experiment 3720
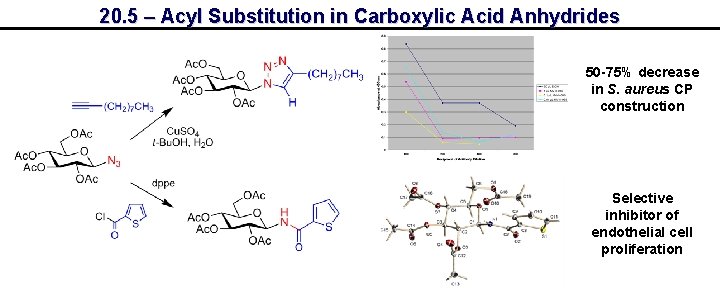
20. 5 – Acyl Substitution in Carboxylic Acid Anhydrides 50 -75% decrease in S. aureus CP construction Selective inhibitor of endothelial cell proliferation
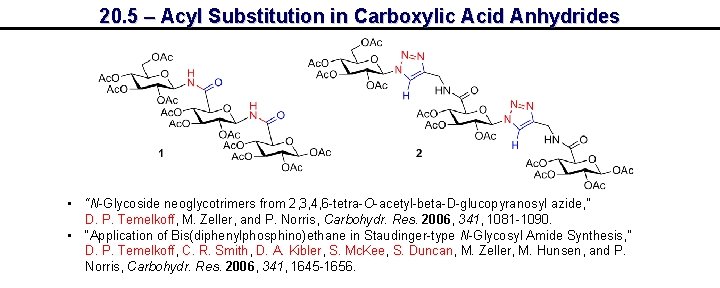
20. 5 – Acyl Substitution in Carboxylic Acid Anhydrides • “N-Glycoside neoglycotrimers from 2, 3, 4, 6 -tetra-O-acetyl-beta-D-glucopyranosyl azide, ” D. P. Temelkoff, M. Zeller, and P. Norris, Carbohydr. Res. 2006, 341, 1081 -1090. • “Application of Bis(diphenylphosphino)ethane in Staudinger-type N-Glycosyl Amide Synthesis, ” D. P. Temelkoff, C. R. Smith, D. A. Kibler, S. Mc. Kee, S. Duncan, M. Zeller, M. Hunsen, and P. Norris, Carbohydr. Res. 2006, 341, 1645 -1656.
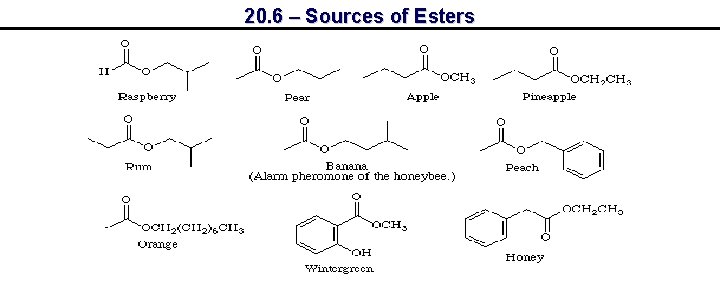
20. 6 – Sources of Esters
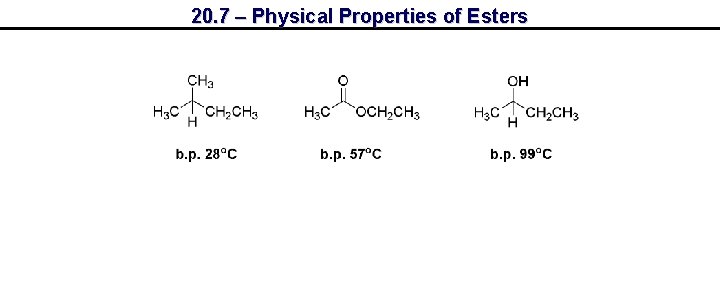
20. 7 – Physical Properties of Esters
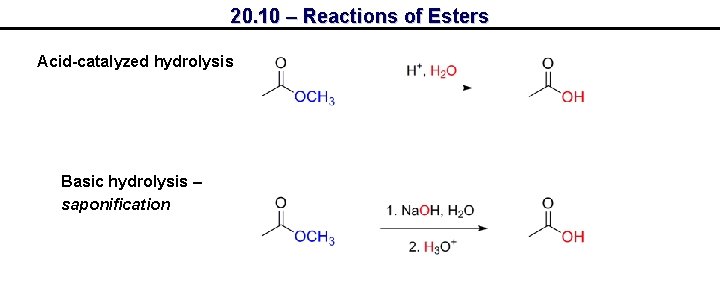
20. 10 – Reactions of Esters Acid-catalyzed hydrolysis Basic hydrolysis – saponification
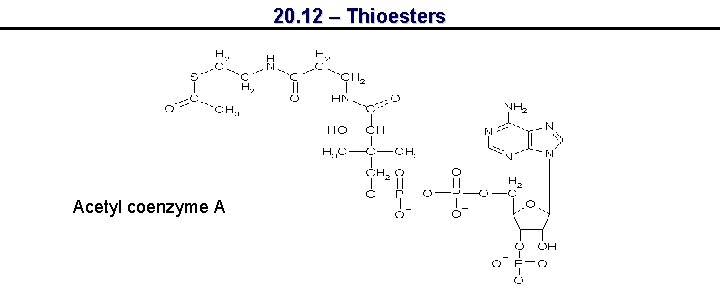
20. 12 – Thioesters Acetyl coenzyme A
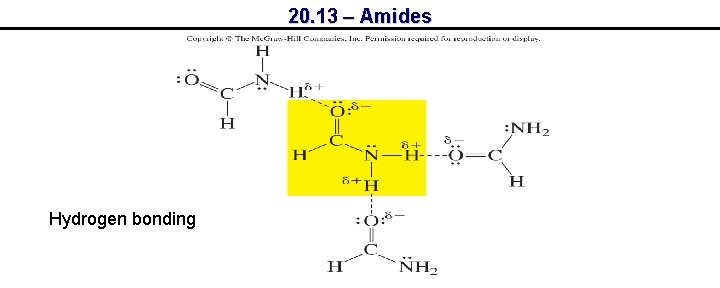
20. 13 – Amides Hydrogen bonding
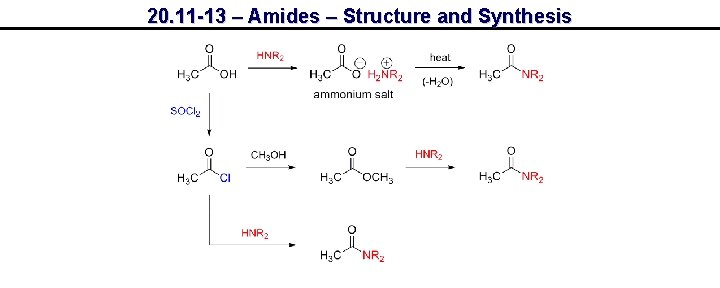
20. 11 -13 – Amides – Structure and Synthesis
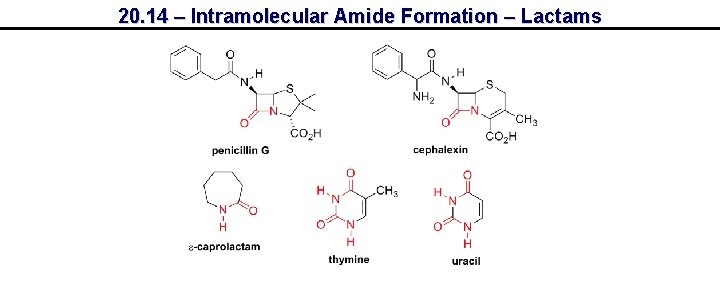
20. 14 – Intramolecular Amide Formation – Lactams

20. 15 – Hydrolysis of Amides – not covering
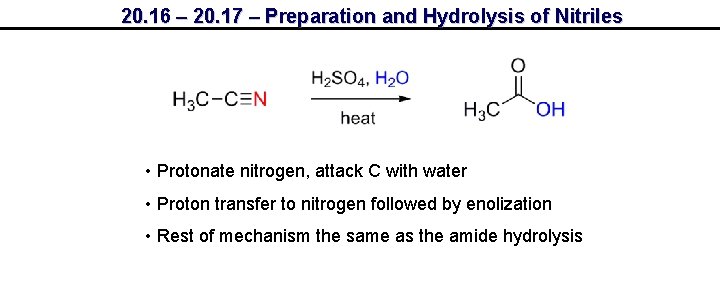
20. 16 – 20. 17 – Preparation and Hydrolysis of Nitriles • Protonate nitrogen, attack C with water • Proton transfer to nitrogen followed by enolization • Rest of mechanism the same as the amide hydrolysis

20. 18 – Addition of RMg. X to Nitriles – Not Covering
- Slides: 24