Chapter 2 The Chemistry of Life I Properties





































- Slides: 49

Chapter 2 The Chemistry of Life

I. Properties: 2. 2 Properties of Water A. Polarity: uneven distribution of electrons between O and H atoms B. Hydrogen Bonds: weaker than ionic and covalent bonds, gives water many of its properties C. Cohesion: attraction of molecules of same substances (ex. water and water) D. Adhesion: attraction of molecules of different substances (ex. water and glass) 1. capillary action: tendency of water to rise in a thin tube due to adhesion E. All life processes need water to take place F. Helps transport materials (ex. blood)

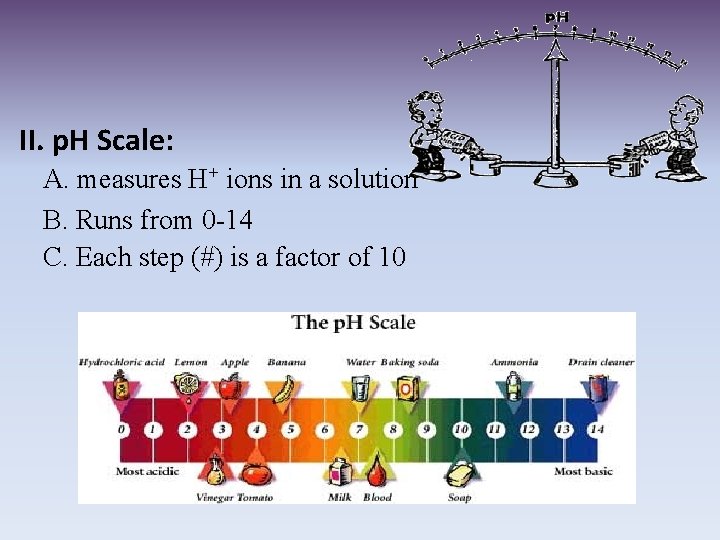
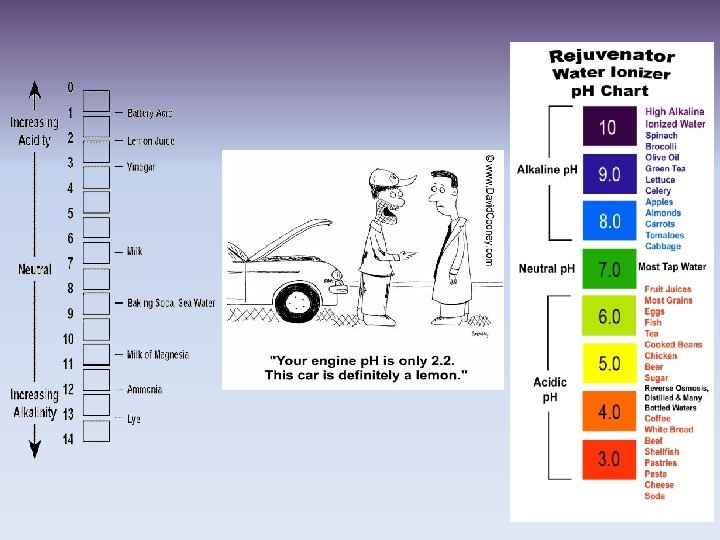
II. p. H Scale: A. measures H+ ions in a solution B. Runs from 0 -14 C. Each step (#) is a factor of 10


D. Acid: 1. Found on the ph scale from 0 -6. 9 2. Increase in strength as you get closer to zero 3. High concentration of H+ (hydrogen ions) 4. Taste sour, can burn you 5. Examples: lemons, tomatoes, stomach acid, battery acid

E. Base: 1. Found on the p. H scale from 7. 1 -14 2. Increase in strength as you get closer to 14 3. Low concentration of H+ ions, high concentration of OHions (hydroxide ions) 4. Taste bitter, can burn you 5. Examples: antacid tablets, drano, soap, bleach F. Neutral 1. Found at 7 on the p. H scale 2. Example: pure water

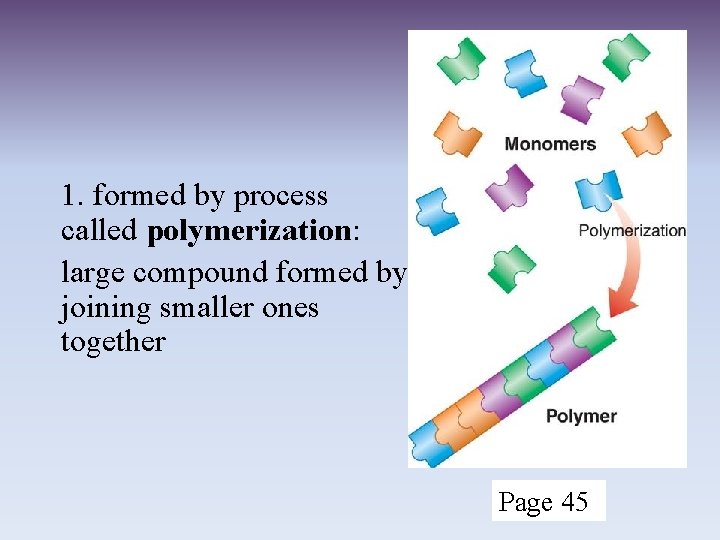
2. 3 Carbon Compounds III. Carbon: A. Has four valence electrons allowing carbon to form MANY different bonds (single, double, or triple bonds) B. making MANY different compounds C. Compounds can be in a line, ring, or branched (page 44) IV. Macromolecules (page 45) A. monomers polymers 0 00000000 macromolecules

1. formed by process called polymerization: large compound formed by joining smaller ones together Page 45

V. Organic Compounds A. All contain Carbon B. Four groups in living things 1. Carbohydrates 2. Lipids 3. Nucleic acids 4. Proteins C. May be referred to as biomolecules

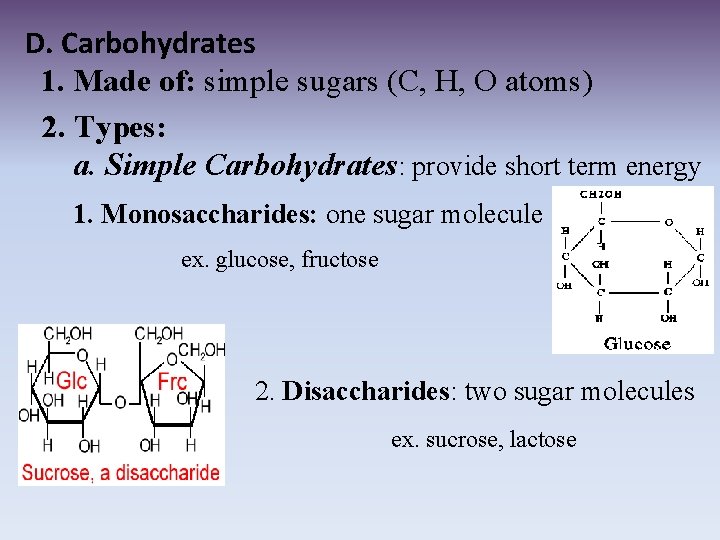
D. Carbohydrates 1. Made of: simple sugars (C, H, O atoms) 2. Types: a. Simple Carbohydrates: provide short term energy 1. Monosaccharides: one sugar molecule ex. glucose, fructose 2. Disaccharides: two sugar molecules ex. sucrose, lactose

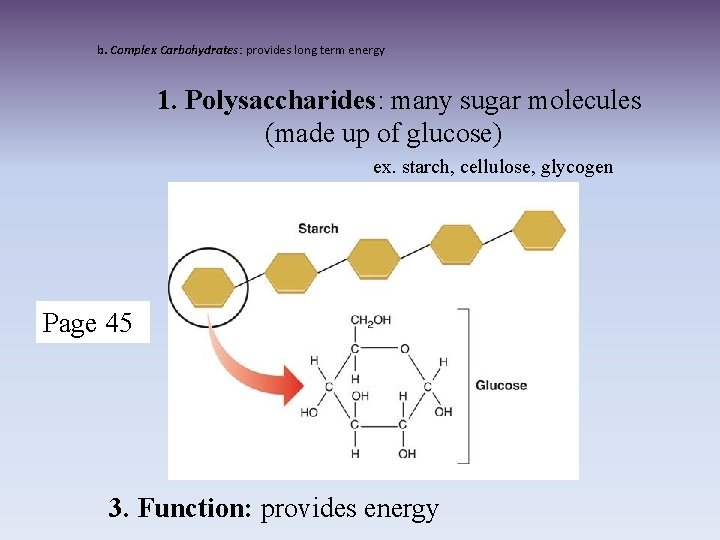
b. Complex Carbohydrates: provides long term energy 1. Polysaccharides: many sugar molecules (made up of glucose) ex. starch, cellulose, glycogen Page 45 3. Function: provides energy

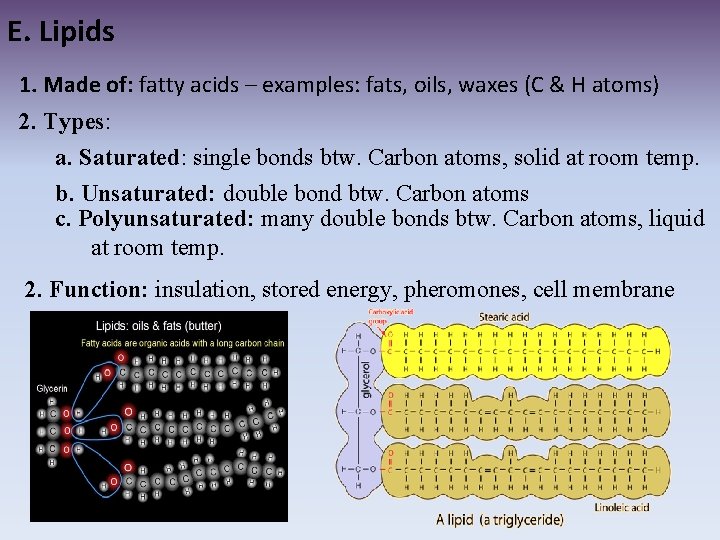
E. Lipids 1. Made of: fatty acids – examples: fats, oils, waxes (C & H atoms) 2. Types: a. Saturated: single bonds btw. Carbon atoms, solid at room temp. b. Unsaturated: double bond btw. Carbon atoms c. Polyunsaturated: many double bonds btw. Carbon atoms, liquid at room temp. 2. Function: insulation, stored energy, pheromones, cell membrane

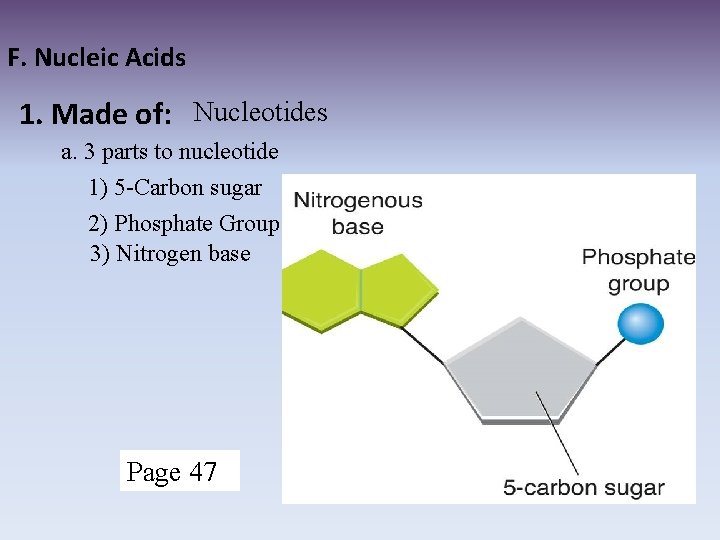
F. Nucleic Acids 1. Made of: Nucleotides a. 3 parts to nucleotide 1) 5 -Carbon sugar 2) Phosphate Group 3) Nitrogen base Page 47

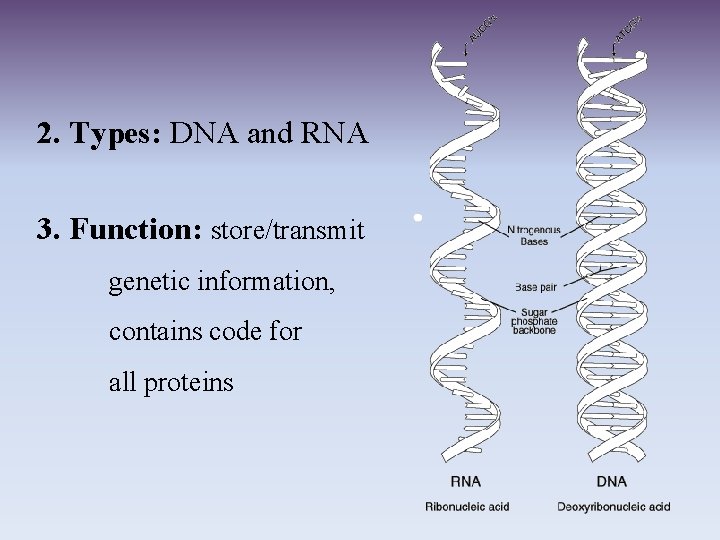
2. Types: DNA and RNA 3. Function: store/transmit genetic information, contains code for all proteins

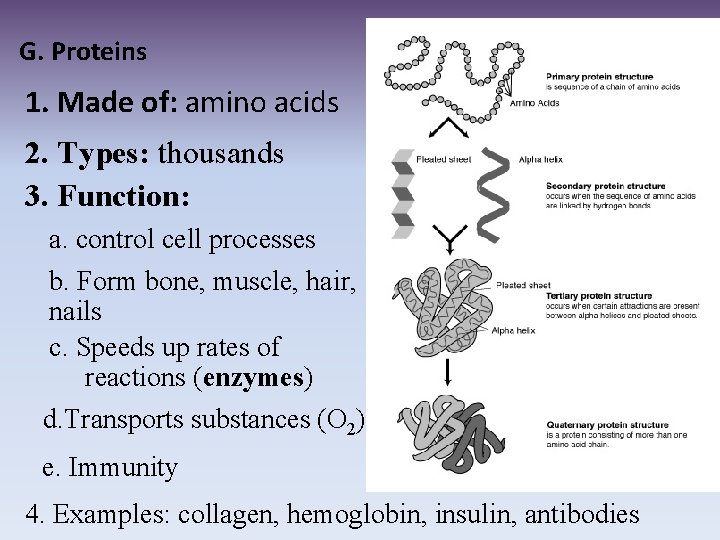
G. Proteins 1. Made of: amino acids 2. Types: thousands 3. Function: a. control cell processes b. Form bone, muscle, hair, nails c. Speeds up rates of reactions (enzymes) d. Transports substances (O 2) e. Immunity 4. Examples: collagen, hemoglobin, insulin, antibodies

2. 4 Chemical Reactions and Enzymes VI. Chemical Reactions: A. changes in chemical bonds that join atoms and compounds B. Parts of chemical equation: 1. Reactants: • Enter reaction (ex: substrate) 2. Products: • Produced in reaction 3. Coefficient: • # in front of substance used to balance equation b/c matter cannot be created or destroyed

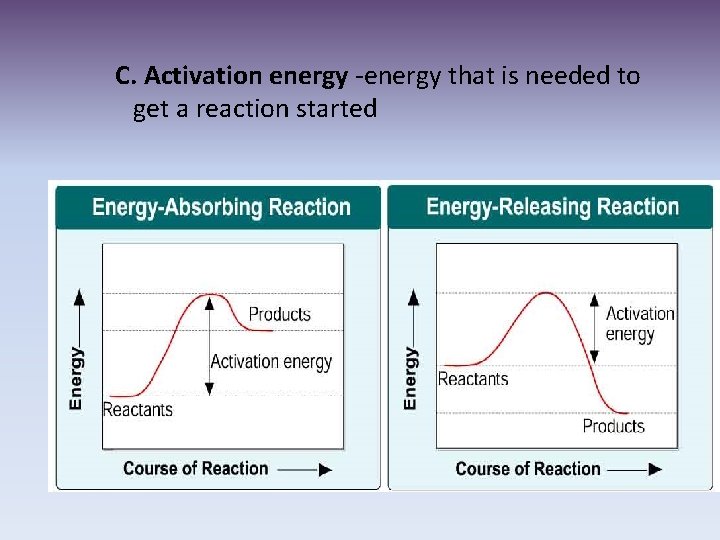
C. Activation energy -energy that is needed to get a reaction started

VII. Enzymes A. Biological catalyst (that speeds up chemical reactions by lowering activation energy) B. Made up of protein C. Function: Speeds up chemical reactions that would take place on their own (ex: breathe out CO 2 but would take 6 hrs. without the enzyme carbonic anhydrase) D. Affected by: 1. Temperature – if too hot or too cold, the protein will not work 2. p. H – if too high or too low, the protein will not work 3. Concentration – small amount = slower reaction rate

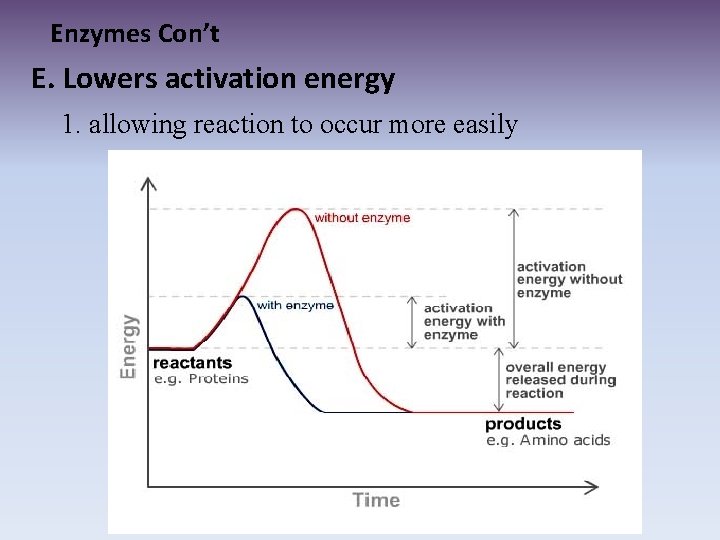
Enzymes Con’t E. Lowers activation energy 1. allowing reaction to occur more easily

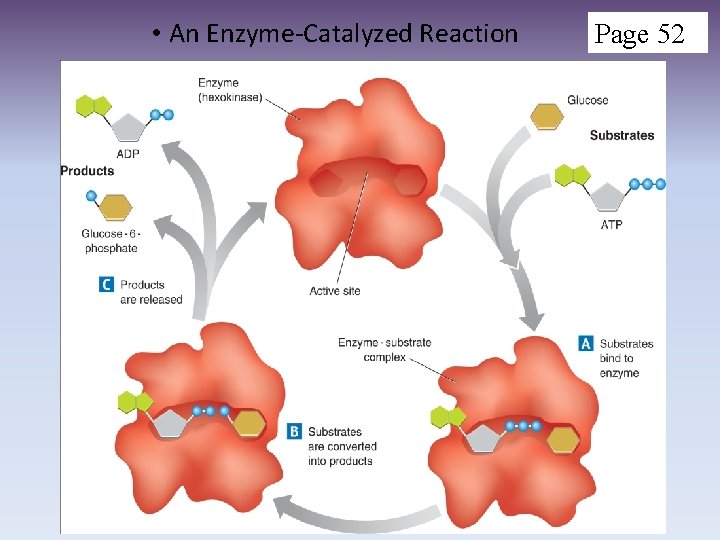
Enzymes Con’t 2. HOW? a. Provides active site where reactants (aka. Substrate) can join together called enzyme-substrate complex (lock and key mechanism) b. E-S Complex remains together until the reaction is done (products are released) c. The enzyme is not permanently changed or used up and can be used again for the same reaction (very specific) Active site Enzyme substrate

• An Enzyme-Catalyzed Reaction Page 52

2 -1 1. The particles that move around the nucleus of an atom are called A. neutrons. B. protons. C. electrons. D. isotopes.

2 -2 2. A compound that produces hydroxide ions in solution is called a(an) A. base. B. buffer. C. acid. D. salt.

2 -2 3. Hydrogen bonds between water molecules result from A. adhesion between water molecules. B. magnetic attractions between water molecules. C. uneven electron distribution in each water molecule. D. ionic bonds in the water molecule.

2 -2 4. On a p. H scale, a value of 2 means that the solution has A. equal concentrations of H+ and OH- ions. B. the same concentration of H+ ions as pure water. C. higher concentration of H+ than in pure water. D. lower concentration of H+ than in pure water.

2– 3 5. Large carbohydrate molecules such as starch are known as A. lipids. B. monosaccharides. C. proteins. D. polysaccharides.

2– 3 6. Many lipids are formed from glycerol and A. fatty acids. B. monosaccharides. C. amino acids. D. nucleic acids.

2– 3 7. Proteins are among the most diverse macromolecules because A. they contain both amino groups and carboxyl groups. B. they can twist and fold into many different and complex structures. C. they contain nitrogen as well as carbon, hydrogen, and oxygen. D. their R groups can be either acidic or basic.

2– 3 8. Which of the following statements about cellulose is true? A. Animals make it and use it to store energy. B. Plants make it and use it to store energy. C. Animals make it and use it as part of the skeleton. D. Plants make it and use it to give structural support to cells.

2– 3 9. A major difference between polysaccharides and proteins is that A. plants make polysaccharides, while animals make proteins. B. proteins are made of monomers, while polysaccharides are not. C. polysaccharides are made of monosaccharides, while proteins are made of amino acids. D. proteins carry genetic information, while polysaccharides do not.

2– 4 10. The elements or compounds produced by a chemical reaction are known as A. reactants. B. enzymes. C. products. D. waste.

2– 4 11. Chemical reactions always involve A. rearrangement of bonds between atoms. B. enzymes. C. catalysts. D. changes in the atomic number of the reactants.

2– 4 12. The factor that prevents many energyreleasing reactions from occurring at relatively low temperatures is called A. catalytic energy. B. chemical bond energy. C. enzyme energy. D. activation energy.

2– 4 13. Which of the following statements is true? A. All proteins are enzymes. B. All catalysts are enzymes. C. All enzymes are catalysts. D. All catalysts are proteins.

2– 4 14. What happens to an enzyme after the reaction it catalyzes has taken place? A. The enzyme is destroyed, and the cell must make another. B. The enzyme holds on to the product until another enzyme removes it. C. The enzyme is unchanged and ready to accept substrate molecules. D. The enzyme changes shape so it can accept a different kind of substrate.

2 -1 1. The particles that move around the nucleus of an atom are called A. neutrons. B. protons. C. electrons. D. isotopes.

2 -2 2. A compound that produces hydroxide ions in solution is called a(an) A. base. B. buffer. C. acid. D. salt.

2 -2 3. Hydrogen bonds between water molecules result from A. adhesion between water molecules. B. magnetic attractions between water molecules. C. uneven electron distribution in each water molecule. D. ionic bonds in the water molecule.

2 -2 4. On a p. H scale, a value of 2 means that the solution has A. equal concentrations of H+ and OH- ions. B. the same concentration of H+ ions as pure water. C. higher concentration of H+ than in pure water. D. lower concentration of H+ than in pure water.

2– 3 5. Large carbohydrate molecules such as starch are known as A. lipids. B. monosaccharides. C. proteins. D. polysaccharides.

2– 3 6. Many lipids are formed from glycerol and A. fatty acids. B. monosaccharides. C. amino acids. D. nucleic acids.

2– 3 7. Proteins are among the most diverse macromolecules because A. they contain both amino groups and carboxyl groups. B. they can twist and fold into many different and complex structures. C. they contain nitrogen as well as carbon, hydrogen, and oxygen. D. their R groups can be either acidic or basic.

2– 3 8. Which of the following statements about cellulose is true? A. Animals make it and use it to store energy. B. Plants make it and use it to store energy. C. Animals make it and use it as part of the skeleton. D. Plants make it and use it to give structural support to cells.

2– 3 9. A major difference between polysaccharides and proteins is that A. plants make polysaccharides, while animals make proteins. B. proteins are made of monomers, while polysaccharides are not. C. polysaccharides are made of monosaccharides, while proteins are made of amino acids. D. proteins carry genetic information, while polysaccharides do not.

2– 4 10. The elements or compounds produced by a chemical reaction are known as A. reactants. B. enzymes. C. products. D. waste.

2– 4 11. Chemical reactions always involve A. rearrangement of bonds between atoms. B. enzymes. C. catalysts. D. changes in the atomic number of the reactants.

2– 4 12. The factor that prevents many energyreleasing reactions from occurring at relatively low temperatures is called A. catalytic energy. B. chemical bond energy. C. enzyme energy. D. activation energy.

2– 4 13. Which of the following statements is true? A. All proteins are enzymes. B. All catalysts are enzymes. C. All enzymes are catalysts. D. All catalysts are proteins.

2– 4 14. What happens to an enzyme after the reaction it catalyzes has taken place? A. The enzyme is destroyed, and the cell must make another. B. The enzyme holds on to the product until another enzyme removes it. C. The enzyme is unchanged and ready to accept substrate molecules. D. The enzyme changes shape so it can accept a different kind of substrate.