CHAPTER 2 The Chemical Basis of Life Life

CHAPTER 2 The Chemical Basis of Life • Life depends on Chemistry • Organisms are composed of matter • Matter is made of elements • Elements are made of atoms Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

• Rattlebox moths release a chemical that spiders don’t like • This spider caught a rattlebox moth and then let it go Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
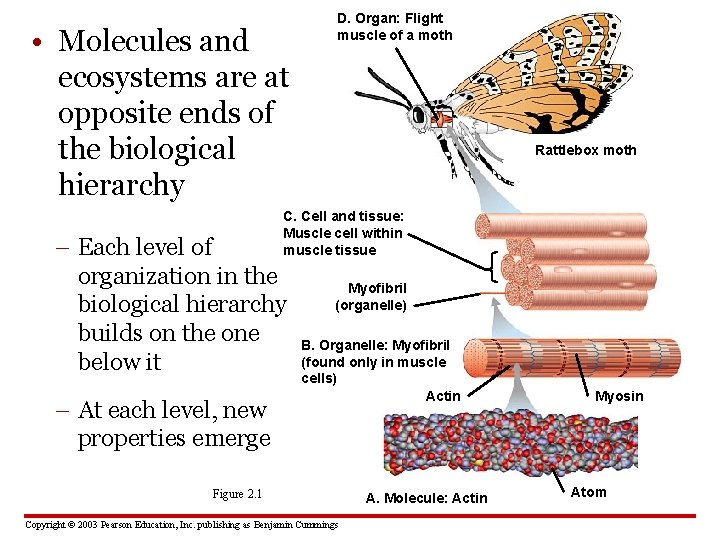
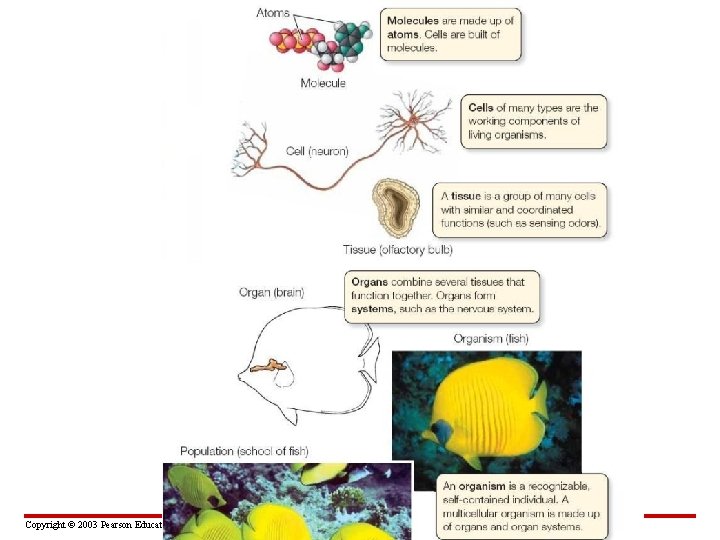
• Molecules and ecosystems are at opposite ends of the biological hierarchy D. Organ: Flight muscle of a moth Rattlebox moth C. Cell and tissue: Muscle cell within muscle tissue – Each level of organization in the biological hierarchy builds on the one below it – At each level, new properties emerge Myofibril (organelle) B. Organelle: Myofibril (found only in muscle cells) Actin Figure 2. 1 Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings A. Molecule: Actin Myosin Atom
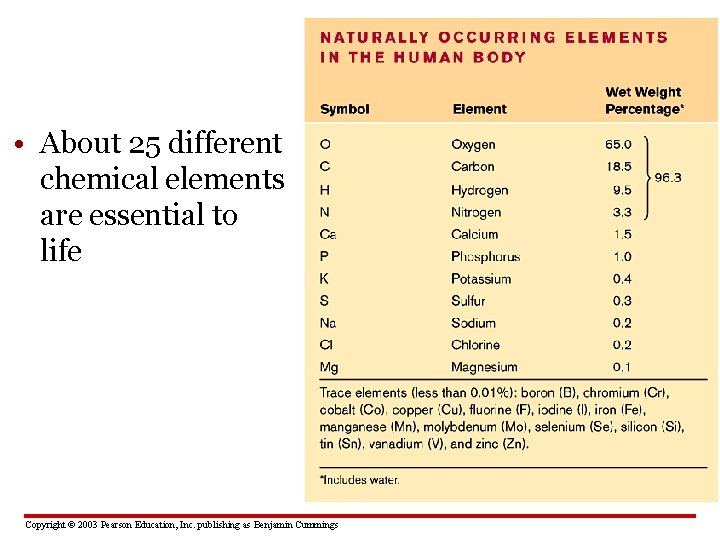
• About 25 different chemical elements are essential to life Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
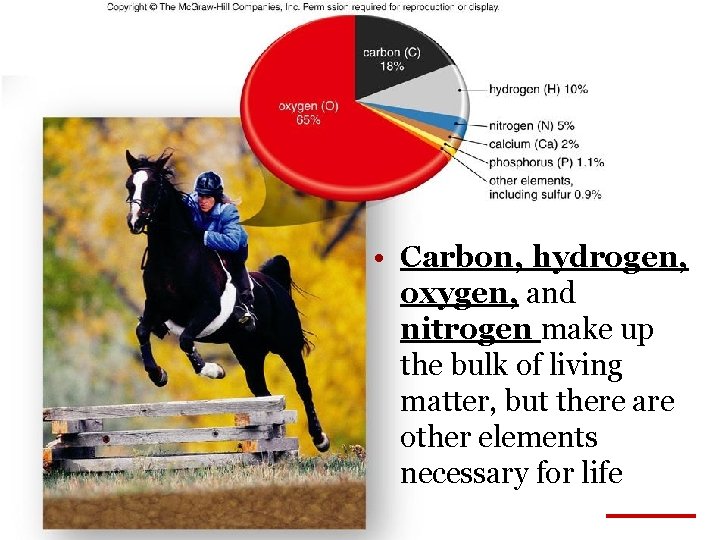
• Carbon, hydrogen, oxygen, and nitrogen make up the bulk of living matter, but there are other elements necessary for life Table 2. 2 Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

• Goiters are caused by iodine deficiency • Nitrogen deficiency in corn Figure 2. 2 Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

2. 3 Elements can combine to form compounds • Chemical elements combine in fixed ratios to form compounds • Example: sodium + chlorine sodium chloride Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
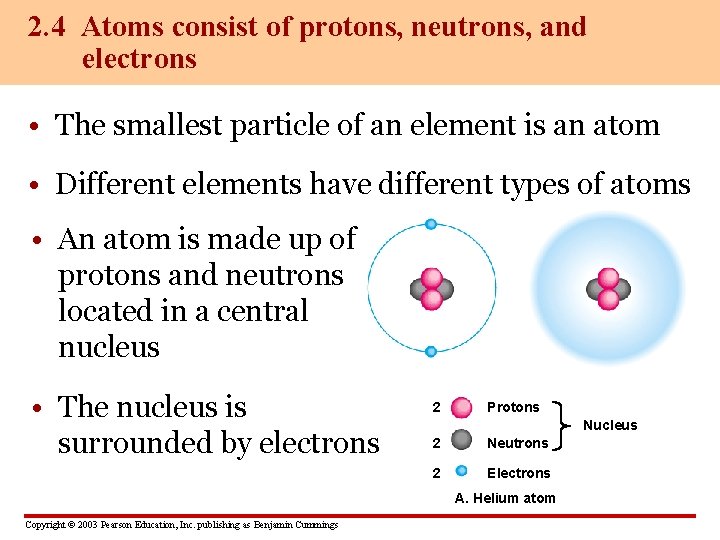
2. 4 Atoms consist of protons, neutrons, and electrons • The smallest particle of an element is an atom • Different elements have different types of atoms • An atom is made up of protons and neutrons located in a central nucleus • The nucleus is surrounded by electrons 2 Protons Nucleus 2 Neutrons 2 Electrons A. Helium atom Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
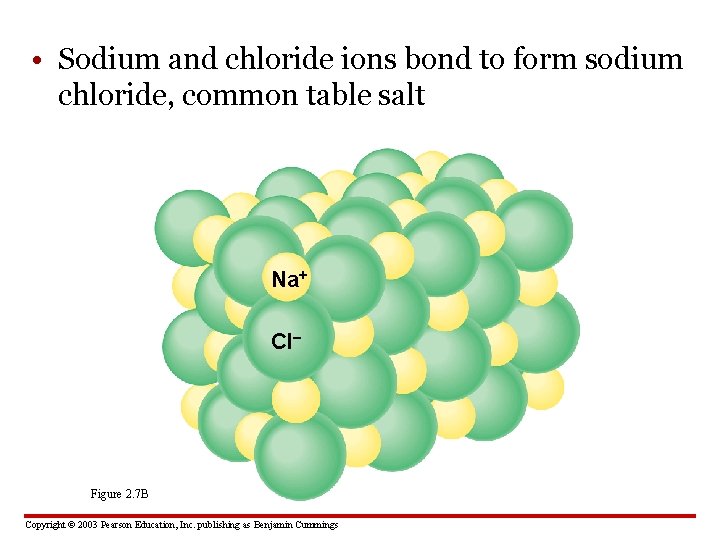
• Sodium and chloride ions bond to form sodium chloride, common table salt Na+ Cl– Figure 2. 7 B Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

THE PROPERTIES OF WATER 2. 9 Water is a polar molecule • Atoms in a covalently bonded molecule may share electrons equally, creating a nonpolar molecule • If electrons are shared unequally, a polar molecule is created Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
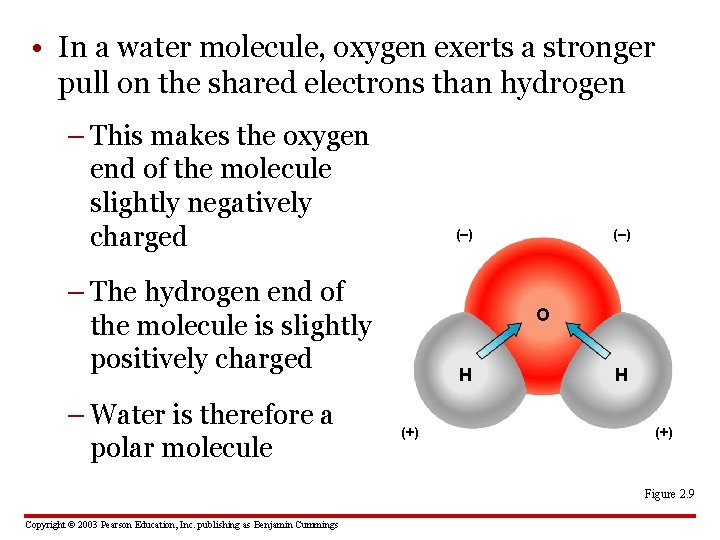
• In a water molecule, oxygen exerts a stronger pull on the shared electrons than hydrogen – This makes the oxygen end of the molecule slightly negatively charged (–) – The hydrogen end of the molecule is slightly positively charged – Water is therefore a polar molecule (–) O H (+) Figure 2. 9 Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
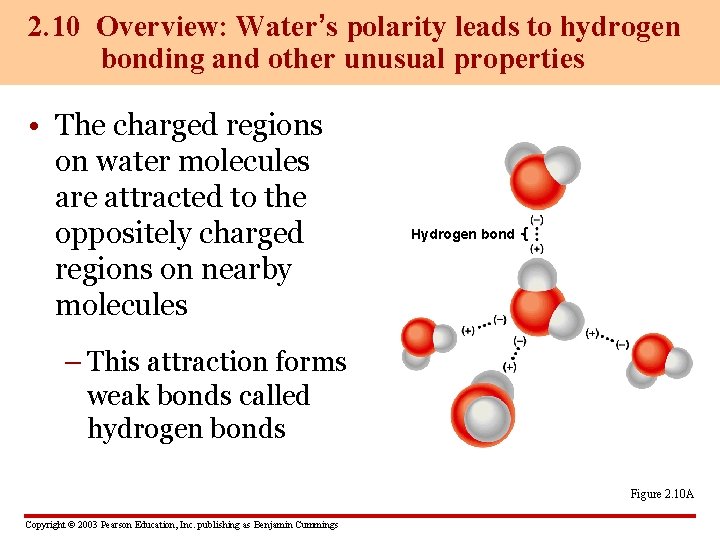
2. 10 Overview: Water’s polarity leads to hydrogen bonding and other unusual properties • The charged regions on water molecules are attracted to the oppositely charged regions on nearby molecules Hydrogen bond – This attraction forms weak bonds called hydrogen bonds Figure 2. 10 A Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

• Like no other common substance, water exists in nature in all three physical states: – as a solid – as a liquid – as a gas Figure 2. 10 B Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

2. 11 Hydrogen bonds make liquid water cohesive and adhesive Cohesion – water sticking to itself Adhesion – water sticking to other molecules • Cohesion among water molecules plays a key role in the transport of water against gravity in plants • Adhesion, clinging of one substance to another, contributes too, as water adheres to the wall of the vessels. Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

• Surface tension, a measure of the force necessary to stretch or break the surface of a liquid, is related to cohesion. – Water has a greater surface tension than most other liquids of cohesion. – Water behaves as if covered by an invisible film. – Some animals can stand, walk, or run on water without breaking the surface. Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Fig. 3. 3

2. 12 Water’s hydrogen bonds moderate temperature • It takes a lot of energy to disrupt hydrogen bonds Therefore water is able to absorb a great deal of heat energy without a large increase in temperature Three-fourths of the earth is covered by water. The water serves as a large heat sink responsible for: 1. Prevention of temperature fluctuations that are outside the range suitable for life. 2. Coastal areas having a mild climate 3. A stable marine environment Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

– A water molecule takes a large amount of energy with it when it evaporates – This leads to evaporative cooling Figure 2. 12 Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
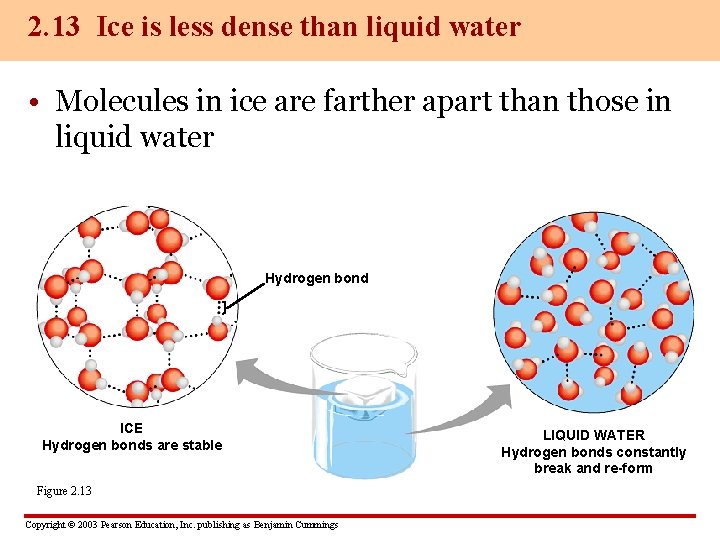
2. 13 Ice is less dense than liquid water • Molecules in ice are farther apart than those in liquid water Hydrogen bond ICE Hydrogen bonds are stable Figure 2. 13 Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings LIQUID WATER Hydrogen bonds constantly break and re-form

– Ice is therefore less dense than liquid water, which causes it to float – If ice sank, it would seldom have a chance to thaw – Ponds, lakes, and oceans would eventually freeze solid Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
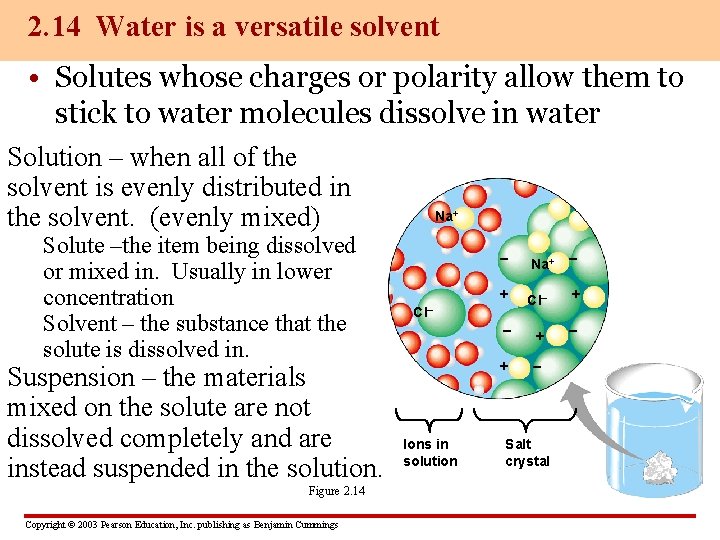
2. 14 Water is a versatile solvent • Solutes whose charges or polarity allow them to stick to water molecules dissolve in water Solution – when all of the solvent is evenly distributed in the solvent. (evenly mixed) Solute –the item being dissolved or mixed in. Usually in lower concentration Solvent – the substance that the solute is dissolved in. Suspension – the materials mixed on the solute are not dissolved completely and are instead suspended in the solution. Figure 2. 14 Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Na+ – + Cl– + – + – Cl– Ions in solution Salt crystal

2. 15 The chemistry of life is sensitive to acidic and basic conditions • A compound that releases H+ ions in solution is an acid, and one that accepts H+ ions in solution is a base • Acidity is measured on the p. H scale: – 0 -7 is acidic – 8 -14 is basic – Pure water and solutions that are neither basic nor acidic are neutral, with a p. H of 7 Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
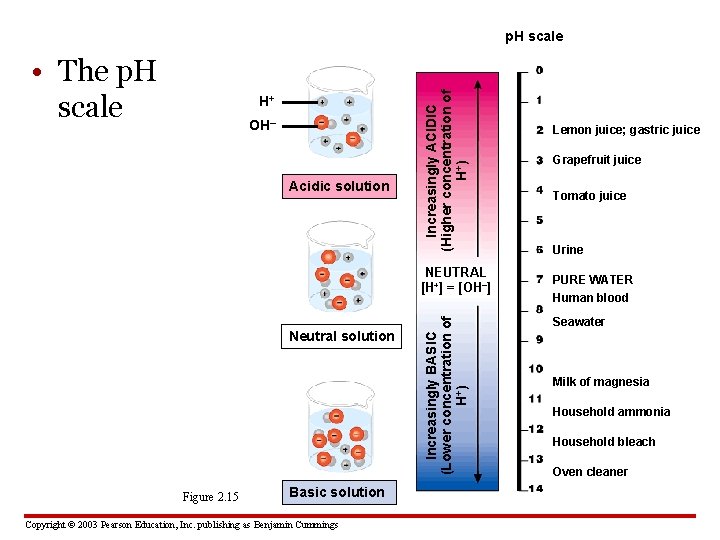
• The p. H scale H+ OH– Acidic solution Increasingly ACIDIC (Higher concentration of H+) p. H scale Neutral solution Figure 2. 15 Basic solution Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Increasingly BASIC (Lower concentration of H+) NEUTRAL [H+] = [OH–] Lemon juice; gastric juice Grapefruit juice Tomato juice Urine PURE WATER Human blood Seawater Milk of magnesia Household ammonia Household bleach Oven cleaner

• Cells are kept close to p. H 7 by buffers • Buffers are substances that resist p. H change – They accept H+ ions when they are in excess and donate H+ ions when they are depleted – Buffers are not foolproof Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

2. 16 Connection: Acid precipitation threatens the environment • Some ecosystems are threatened by acid precipitation • Acid precipitation is formed when air pollutants from burning fossil fuels combine with water vapor in the air to form sulfuric and nitric acids Figure 2. 16 A Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings
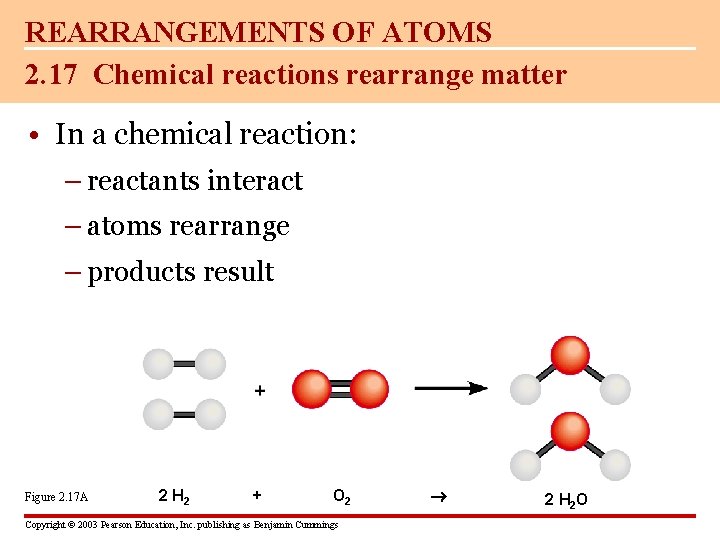
REARRANGEMENTS OF ATOMS 2. 17 Chemical reactions rearrange matter • In a chemical reaction: – reactants interact – atoms rearrange – products result Figure 2. 17 A 2 H 2 + O 2 Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings 2 H 2 O
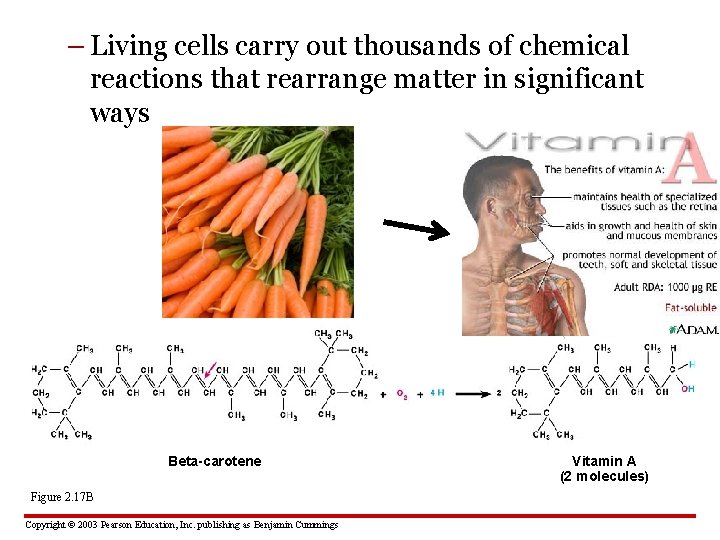
– Living cells carry out thousands of chemical reactions that rearrange matter in significant ways Beta-carotene Figure 2. 17 B Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Vitamin A (2 molecules)
- Slides: 28