Chapter 2 Section 2 UNITS OF MEASUREMENT Objectives

Chapter 2 : Section 2 UNITS OF MEASUREMENT

Objectives Distinguish between a quantity, a unit, and a measurement standard Name SI units for length, mass, time, volume, and density Distinguish between mass and weight Perform density calculations Transform a statement of equality to a conversion factor

Units of measurement Quantity – magnitude, size, or amount 5 2, 000 0. 030 o Units – compare what is measured with defined size m. L g feet Quantity has no meaning without the unit 5 m. L 100 g 2, 000 feet
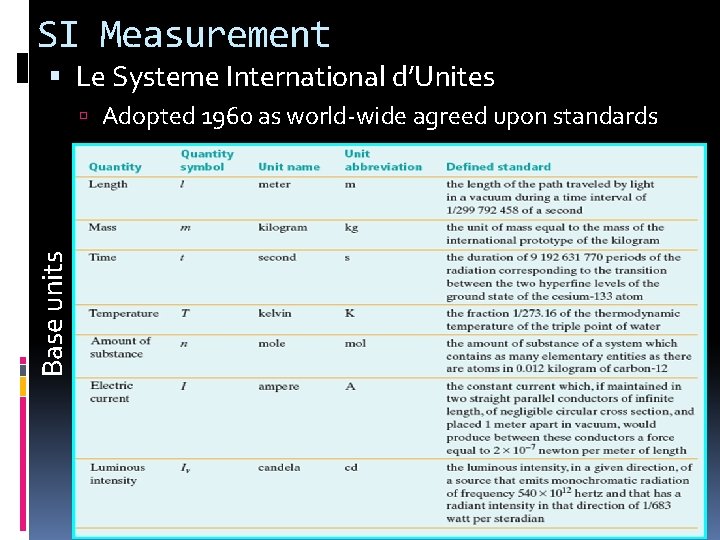
SI Measurement Le Systeme International d’Unites Base units Adopted 1960 as world-wide agreed upon standards

Some SI Base Units Mass – measure of the amount of matter in an object Grams (g) Electronic scale or triple beam balance Volume – amount of space an object takes up Liters L V = L X W X H (cm 3) Graduated cylinder Water displacement Visual Concept 1 – Click Here Visual Concept 2 – Click Here Weight – measure of the gravitational pull on matter Example : Moon has 1/6 weight
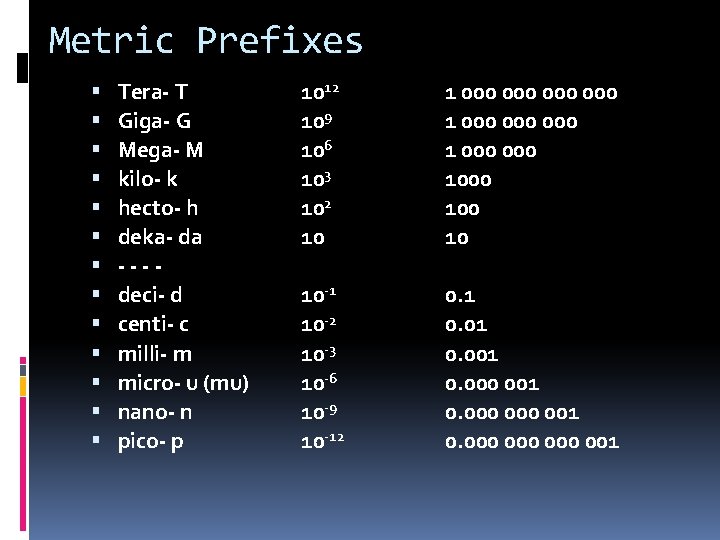
Metric Prefixes Tera- T Giga- G Mega- M kilo- k hecto- h deka- da ---deci- d centi- c milli- m micro- u (mu) nano- n pico- p 1012 109 106 103 102 10 1 000 000 1 000 1000 10 10 -1 10 -2 10 -3 10 -6 10 -9 10 -12 0. 1 0. 001 0. 000 000 001

Derived SI units Combination of SI base units Examples Area Volume Density Molar mass m 2 m 3 Kg/m 2 Kg/mol L x W x H mass/volume mass/mole Speed : 2 measurements distance divided by time

Volume and Density Volume = Length x Width x Height 1 cm x 1 cm = 1 cm 3 Density = mass / Volume Sample Problem 2 -1 A sample of aluminum metal has a mass of 8. 4 g. The volume of the sample is 3. 1 cm 3. Calculate the density of aluminum. Step 1: Write density equation Step 2: List your variables Step 3: Rearrange the equation D = ? m = 8. 4 g V = 3. 1 cm 3 m V = 8. 4 g 3. 1 cm 3 = 2. 7 g/cm 3 Step 4: Replace variables in the equation Step 5: Solve

Rearranging Equations D = m V Solve for m: m V x D = Step 1: V x V Multiply both sides by V Step 3: Step 2: V’s will cancel out leaving m by itself and on top Solve for V: Step 1: Multiply both sides by V Step 2: Divide both side by D m = Dx V so: D = Rewrite the equation m V x D = V V x D = D so: Step 3: Rewrite the equation x V m D V = m D

Practice pg. 40 #1 -3 1. What is the density of a block of marble that occupies 310. cm 3 and has a mass of 853 g? 2. 75 g/cm 3 2. Diamond has a density of 3. 26 g/cm 3. What is the mass of a diamond that has a volume of 0. 351 cm 3? 1. 14 g 3. What is the volume of a sample of liquid mercury that has a mass of 76. 2 g, given that the density of mercury is 13. 6 g/m. L? 5. 60 m. L

Density of Some Common Substances Substance Density (g/cm 3) Air 0. 0013 Feathers 0. 0025 Wood(Oak) 0. 6 - 0. 9 Ice 0. 92 Water 1. 00 Bricks 1. 84 Aluminum 2. 70 Steel 7. 80 Silver 10. 50 Gold 19. 30

Conversion Factors Ratio derived from the equality between two different units that can be used to convert from one unit to the other Visual Concept – Click Here Equalities 1 dollar = 4 quarters 1 kg = 1000 g 1 Km = 0. 6213 miles 1 dollar 4 quarter or 4 quarters 1 dollar So: How many quarter are there in $55 ? = 55 dollars x 4 quarters 1 dollars = 220 quarters

Making conversions Video Concept – Click Here How many miles are in 15 Km? 1. Write given value: 2. Choose conversion factor 3. Multiply top and divide bottom 11. 0 miles x 1 km = 17. 7 km 0. 6213 miles

Sample Problem 2. 2 Express a mass of 5. 712 grams in milligrams and in kilograms. 5. 172 g x x 1000 mg 1 kg 1000 g = 5172 mg = 0. 005172 kg

Practice 1 & 2 pg. 42 1. Express a length of 16. 45 m in centimeters and in kilometers. 2. Express a mass of 0. 014 mg in grams. Answers: 1. 1645 cm, 0. 01645 km 2. 0. 000 014 g
- Slides: 15