Chapter 2 Section 1 Composition of Matter Objectives

Chapter 2 Section 1 Composition of Matter Objectives • Define the term matter. • Explain the relationship between elements and atoms. • Draw and label a model of the structure of an atom. • Explain how compounds affect an atom’s stability. • Contrast covalent and ionic bonds.
Chapter 2 Section 1 Composition of Matter • Matter is anything that occupies space and has mass. • Mass is the quantity of matter an object has.

Chapter 2 Section 1 Composition of Matter Elements and Atoms • Elements are made of a single kind of atom and cannot be broken down by chemical means into simpler substances. • Atoms are composed of protons, neutrons, and electrons.

Chapter 2 Section 1 Composition of Matter Elements and Atoms, continued • The Nucleus – Protons and neutrons make up the nucleus of the atom.

Chapter 2 Section 1 Composition of Matter Elements and Atoms, continued • Electrons – Electrons move about the nucleus in orbitals. – An orbital is a threedimensional region around a nucleus that indicates the probable location of an electron.

Chapter 2 Section 1 Composition of Matter Elements and Atoms, continued • Isotopes – Atoms of the same element that have a different number of neutrons are called isotopes.

Chapter 2 Section 1 Composition of Matter Compounds • Compounds consist of atoms of two or more elements that are joined by chemical bonds in a fixed proportion.

Chapter 2 Section 1 Composition of Matter Compounds, continued • Covalent Bonds – A covalent bond is formed when two atoms share electrons.

Chapter 2 Section 1 Composition of Matter Compounds, continued • Ionic Bonds – An ionic bond is formed when one atom gives up an electron to another. The positive ion is then attracted to a negative ion to form the ionic bond.

Chapter 2 Ionic Bonding Section 1 Composition of Matter

Chapter 2 Section 2 Energy Objectives • Describe the physical properties of each state of matter. • Describe the role of reactants and products in chemical reactions. • Explain the relationship between enzymes and activation energy. • Explain how oxidation and reduction reactions are linked.

Chapter 2 Section 2 Energy and Matter • States of Matter – Addition of energy to a substance can cause its state to change from a solid to a liquid and from a liquid to a gas.

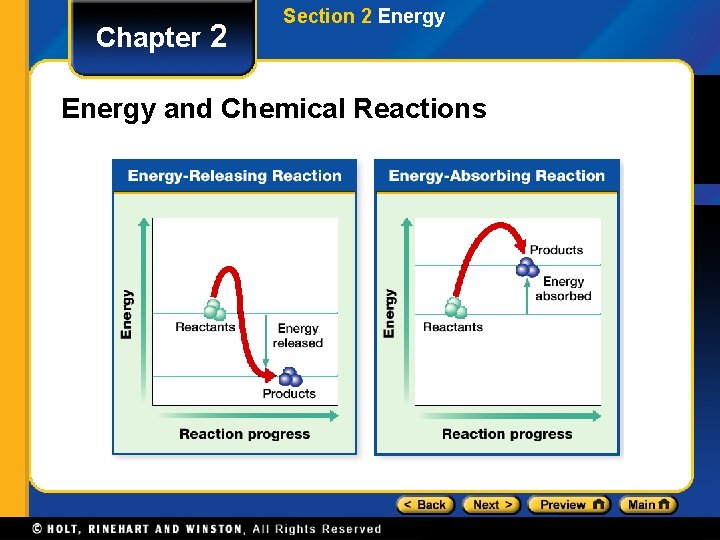
Chapter 2 Section 2 Energy and Chemical Reactions • Reactants are substances that enter chemical reactions. • Products are substances produced by chemical reactions.
Chapter 2 Section 2 Energy and Chemical Reactions

Chapter 2 Section 2 Energy and Chemical Reactions, continued • Activation Energy – Enzymes lower the amount of activation energy necessary for a reaction to begin in living systems.

Chapter 2 Section 2 Energy and Chemical Reactions, continued • Oxidation Reduction Reactions – A chemical reaction in which electrons are exchanged between atoms is called an oxidationreduction reaction.

Chapter 2 Section 3 Water and Solutions Objectives • Describe the structure of a water molecule. • Explain how water’s polar nature affects its ability to dissolve substances. • Outline the relationship between hydrogen bonding and the different properties of water. • Identify the roles of solutes and solvents in solutions. • Differentiate between acids and bases.

Chapter 2 Section 3 Water and Solutions Polarity • Water is considered to be a polar molecule due to an uneven distribution of charge. • The electrons in a water molecule are shared unevenly between hydrogen and oxygen.

Chapter 2 Section 3 Water and Solutions Polarity, continued • Solubility of Water – The polarity of water makes it effective at dissolving other polar substances such as sugars, ionic compounds, and some proteins.
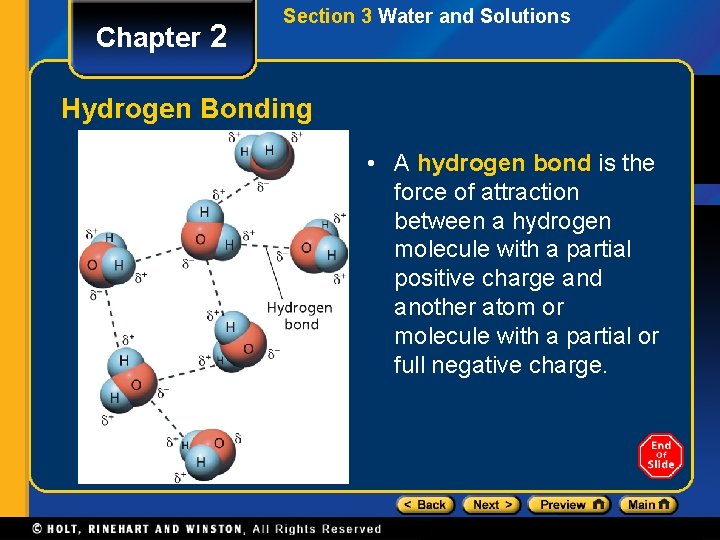
Chapter 2 Section 3 Water and Solutions Hydrogen Bonding • A hydrogen bond is the force of attraction between a hydrogen molecule with a partial positive charge and another atom or molecule with a partial or full negative charge.

Chapter 2 Section 3 Water and Solutions Hydrogen Bonding, continued • Cohesion and Adhesion – Cohesion is an attractive force that holds molecules of a single substance together, such as water molecules. – Adhesion is the attractive force between two particles of different substances, such as water molecules and glass molecules.

Chapter 2 Section 3 Water and Solutions Hydrogen Bonding, continued • Temperature Moderation – Water has the ability to absorb a relatively large amount of energy as heat and the ability to cool surfaces through evaporation.

Chapter 2 Section 3 Water and Solutions Hydrogen Bonding, continued • Density of Ice – Solid water is less dense than liquid water due to the shape of the water molecule and hydrogen bonding.

Chapter 2 Section 3 Water and Solutions • A solution consists of a solute dissolved in a solvent.

Chapter 2 Section 3 Water and Solutions Acids and Bases • Ionization of Water – Water ionizes into hydronium ions (H 3 O+) and hydroxide ions (OH–).

Chapter 2 Section 3 Water and Solutions Acids and Bases, continued • Acids – Acidic solutions contain more hydronium ions than hydroxide ions.

Chapter 2 Section 3 Water and Solutions Acids and Bases, continued • Bases – Basic solutions contain more hydroxide ions than hydronium ions.

Chapter 2 Section 3 Water and Solutions Acids and Bases, continued • p. H – Scientists have developed a scale for comparing the relative concentrations of hydronium ions and hydroxide ions in a solution. This scale is called the p. H scale, and it ranges from 0 to 14.

Chapter 2 The p. H Scale Section 3 Water and Solutions

Chapter 2 Section 3 Water and Solutions Acids and Bases, continued • Buffers – Buffers are chemicals that neutralize the effects of adding small amounts of either an acid or a base to a solution.
- Slides: 30