CHAPTER 2 Review Essential Chemistry for Biology Power

CHAPTER 2 Review Essential Chemistry for Biology Power. Point® Lecture Slides for Essential Biology, Second Edition & Essential Biology with Physiology Neil Campbell, Jane Reece, and Eric Simon Presentation prepared by Chris C. Romero Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings

Matter: Elements and Compounds u. Atoms are the basic units of matter u. An Element is a substance of only one type of atom Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings
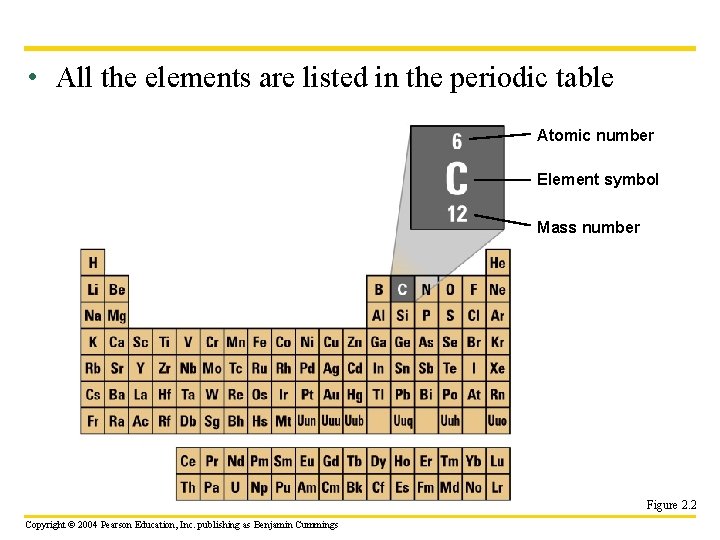
• All the elements are listed in the periodic table Atomic number Element symbol Mass number Figure 2. 2 Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings
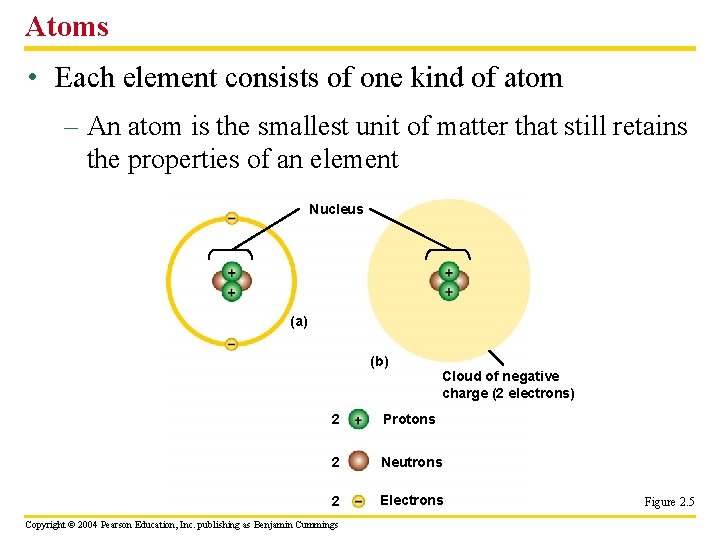
Atoms • Each element consists of one kind of atom – An atom is the smallest unit of matter that still retains the properties of an element Nucleus (a) (b) Cloud of negative charge (2 electrons) 2 Protons 2 Neutrons 2 Electrons Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings Figure 2. 5

The Structure of Atoms • Three particles that make up an atom – A proton is positively charged – found in atomic nucleus – An electron is negatively charged – found in space outside atomic nucleus called shells – A neutron is electrically neutral - found in atomic nucleus Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings

• Most atoms have protons and neutrons packed tightly into the nucleus – The nucleus is the atom’s central core (protons and neutrons) – The electrons orbit the nucleus in “shells” (electrons) Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings

• Elements differ in the number of subatomic particles in their atoms – The number of protons = the atomic number and determines which element it is – protons + neutrons = An atom’s mass number (atomic mass) – Mass is a measure of the amount of matter in an object • Example: Carbon has 6 protons = atomic # and 6 neutrons, Carbons atomic mass is 12 Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings

Question • If and atom contains 15 protons and 12 neutrons, its Atomic Number is? 15 • What is its Mass Number or Atomic Mass? 27 Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings
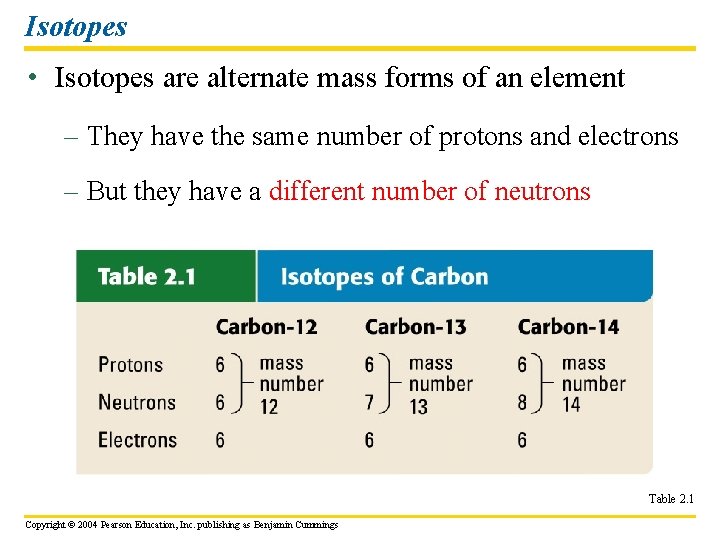
Isotopes • Isotopes are alternate mass forms of an element – They have the same number of protons and electrons – But they have a different number of neutrons Table 2. 1 Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings

Electron Arrangement and the Chemical Properties of Atoms • Electrons determine how an atom behaves when it encounters other atoms • Electrons orbit the nucleus of an atom in specific electron shells – The number of electrons in the outermost shell determines the chemical properties of an atom Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings

Chemical Bonding and Molecules • Chemical reactions enable atoms to give up or acquire electrons in order to complete their outer shells Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings

Ionic Bonds • When an atom loses or gains electrons, it becomes electrically charged – Charged atoms are called IONS – Ionic bonds are formed between oppositely charged ions Sodium atom (Na) Chlorine atom (Cl) Complete outer shells Sodium ion (Na ) Chloride ion (Cl ) Sodium chloride (Na. Cl) Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings Figure 2. 8

Covalent Bonds • A covalent bond forms when two atoms share one or more pairs of outer-shell electrons • SHARING 2 ELECTRONS IS A SINGLE BOND • SHARING 4 ELECTRONS IS A DOUBLE BOND Figure 2. 9 Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings
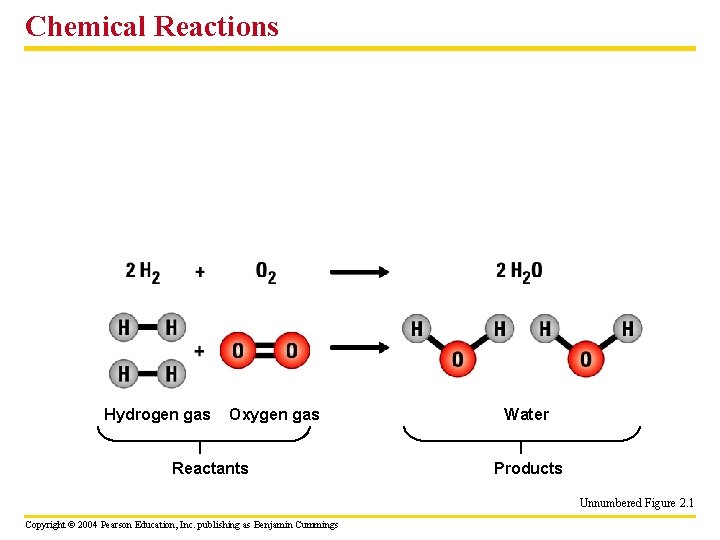
Chemical Reactions Hydrogen gas Oxygen gas Reactants Water Products Unnumbered Figure 2. 1 Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings

WATER AND LIFE • The most abundant compound in most living things is WATER Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings

The Structure of Water • Water is Polar – it has charges – Its two hydrogen atoms are joined to one oxygen atom by single covalent bonds H H O Unnumbered Figure 2. 2 Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings
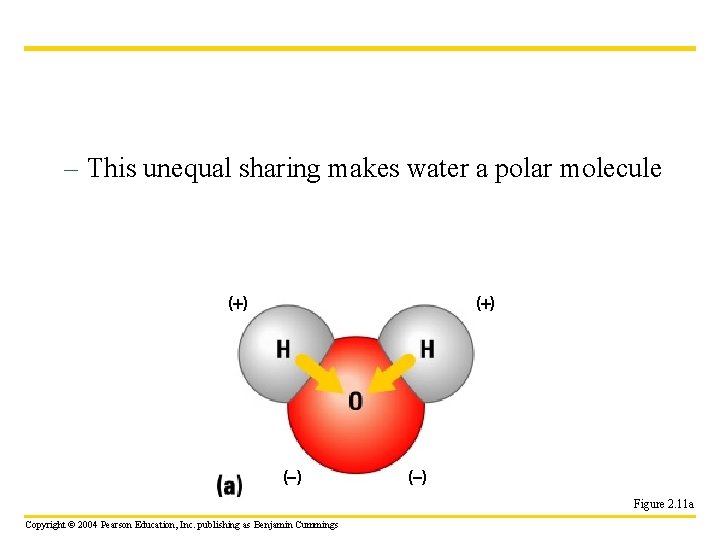
– This unequal sharing makes water a polar molecule ( ) ( ) Figure 2. 11 a Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings

• The polarity of water gives rise to waters special properties of ( ) • Adhesion • Cohesion ( ) ( ) • Surface tension Hydrogen bond ( ) ( ) – These interactions are called hydrogen bonds Figure 2. 11 b Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings

The Cohesion of Water • Water molecules stick together as a result of hydrogen bonding Microscopic tubes Figure 2. 12 Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings

• Surface tension is the measure of how difficult it is to stretch or break the surface of a liquid – Hydrogen bonds give water an unusually high surface tension Figure 2. 13 Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings

Water as the Solvent of Life • Water is the solvent • Salt is the solute • Suspensions are mixtures of water and an undissolved material – Oil and water Figure 2. 16 Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings

Acids, Bases, and p. H • Acid H+ > OH- p. H below 7 • Base H+ < OH- p. H above 7 Water is neutral – p. H of 7 H+ = OH- Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings

p. H Scale Examples of Acids and Basis Acid – Stomach Acid Base – Oven Cleaner Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings

• Buffers are substances that resist p. H change – They accept H+ ions when they are in excess – They donate H+ ions when they are depleted – Buffers are weak acids or bases that react with strong acids and basis to prevent sudden p. H changes. Figure 2. 18 Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings

The End Power. Point® Lecture Slides for Essential Biology, Second Edition & Essential Biology with Physiology Neil Campbell, Jane Reece, and Eric Simon Presentation prepared by Chris C. Romero Copyright © 2004 Pearson Education, Inc. publishing as Benjamin Cummings
- Slides: 25