CHAPTER 2 PROPERTIES OF MATTER Matter Pure Substances

CHAPTER 2 PROPERTIES OF MATTER
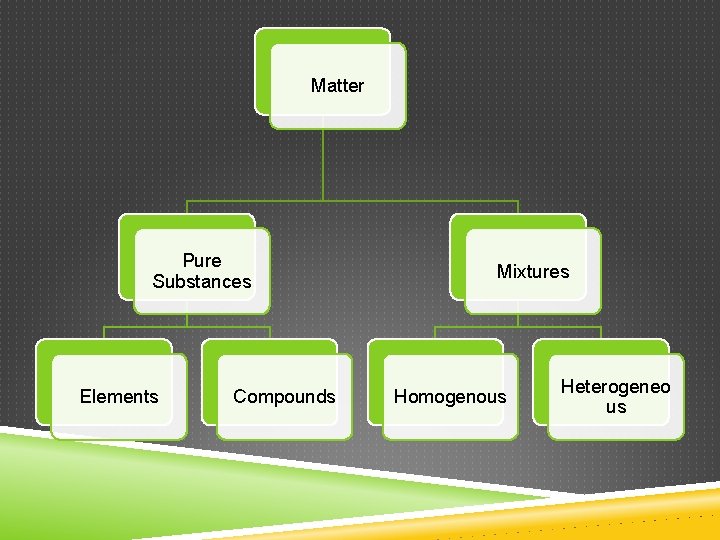
Matter Pure Substances Elements Compounds Mixtures Homogenous Heterogeneo us

PURE SUBSTANCES Matter that always has exactly the same composition is classified as a pure substance

TYPES OF PURE SUBSTANCES Element- simplest form of matter contains only one type of atom Compound- contains two or more elements chemically combined - can be broken down into two or more elements using chemical

ELEMENTS AND COMPOUNDS Elements are represented by symbols Ex: Na Compounds are represented by chemical formulas, groups of element symbols Ex: Na. Cl

CHEMICAL SYMBOLS 1 st letter always upper case Each element has its own 2 nd and 3 rd always lower case Represents one atom (or mole) of the element Co vs. CO Ni vs. NI

Chemical Formula – the “recipe” for a compound. • Tells what elements are in the compound. • Tells # of atoms of each element. – Ex. H 2 O = 2 atoms of hydrogen and 1 atom of oxygen – Ex. Na. Cl C 6 H 12 O 6 H 2 SO 4

COMPOUNDS Na. Cl C 6 H 12 O 6

MIXTURES Mixture-physical blend of two or more components 2 Types: Heterogeneous Homogenous (solution)

MIXTURES Heterogeneous – parts of the mixture are noticeably different form each other Homogeneous- parts of the mixture are evenly distributed

TYPES OF MIXTURES Homogeneous Heterogeneo us

SOLUTIONS, COLLOIDS, SUSPENSIONS Size of the particles in the mixture can have an effect on its properties Solution Suspension Colloid

SOLUTION, COLLOID, SUSPENSION Solution- when a substance dissolves in another substance and forms a homogeneous mixture Ex: sugar in coffee

SOLUTION, COLLOID, SUSPENSION Colloids have particles bigger than in a solution but smaller than in a suspension. Particles won’t settle out over time Examples: whipped cream, marshmallow, milk, mayo, fog

SOLUTION, COLLOID, SUSPENSION Suspension- heterogeneous mixture that separates into layers over time Can use a filter to separate particles Ex:

Solution Colloid . 1 -1 nm Not able to filter 1 – 1000 nm Not able to filter No Tyndall Effect Sometimes Tyndall effect Suspension < 1000 nm Will filter Sometimes Tyndall effect

Mixtures: Colloids, suspensions, solutions

PHYSICAL PROPERTIES Physical property- any characteristic of a material that can be observed or measured without changing the composition of the substances viscosity Conductivity Malleability Hardness Melting point Boiling point density

PHYSICAL PROPERTIES : VISCOSITY

PHYSICAL PROPERTIES: VISCOSITY Viscosity – resistance to flow “thickness” of a liquid The greater the viscosity, the slower the liquid moves

PHYSICAL PROPERTIES: CONDUCTIVITY Conductivity- a material’s ability to allow heat to flow Known as conductors Usually a good conductor of electricity

PHYSICAL PROPERTIES: MALLEABILITY malleability- ability of a solid to be hammered without shattering

PHYSICAL PROPERTIES: MELTING AND BOILING POINT Melting point – the temperature a substance turns from a solid to a liquid

PHYSICAL PROPERTIES: MELTING AND BOILING POINT Boiling point – the temperature a substance turns from a liquid to a gas

PHYSICAL PROPERTIES: MELTING AND BOILING POINT What is the melting point of water? What is the freezing point of water? What is the boiling point of water?

PHYSICAL PROPERTIES: DENSITY Density- the amount of matter in a given volume Density of copper 8. 96 g/cm 3 Density of zinc 7. 14 g/cm 3

USING PHYSICAL PROPERTIES Shoelaces

SEPARATING MIXTURES What are some ways that we can separate mixtures?

• • • Separating mixtures by physical means Filtering By hand Boiling- fractional distillation Freezing Melting Evaporating Dissolving Crystallizing Magnetism

FILTRATION AND DISTILLATION Filtration- Separates materials based on the size of the particles Distillation- separates materials based on their boiling points
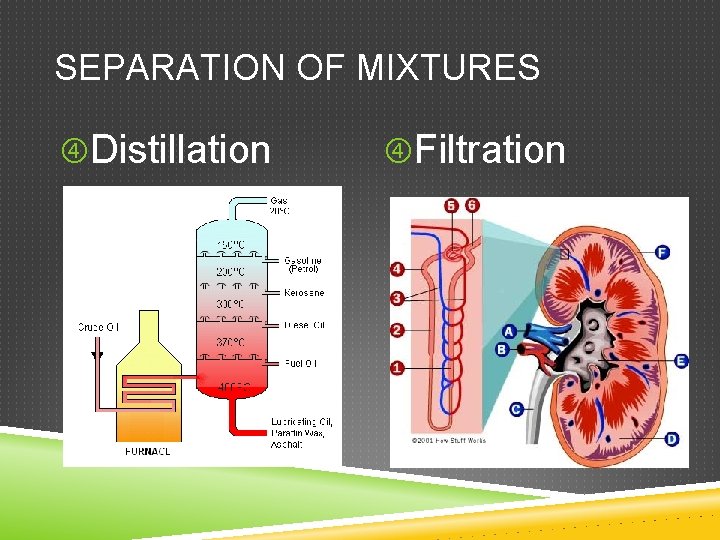
SEPARATION OF MIXTURES Distillation Filtration

PHYSICAL CHANGES Physical change – when some of the properties change but the substance in the material stay the same Ex: freezing water, shredding paper

FIRST FIVE QUESTIONS… List seven examples of physical properties 2. Name two ways we can separate mixtures. 3. When you describe a liquid as thick, are you saying that it has a high or low viscosity? 4. Explain why sharpening a pencil is an example of a physical change. 1.

CHEMICAL PROPERTIES Chemical property-any ability to produce a change in composition of matter Flammability Reactivity Toxicity Heat of combustion

CHEMICAL PROPERTIES : REACTIVITY Chemical reactivity- property that describes how easy it is for a substance to chemically combine with another substance Ex: oxygen – high reactivity nitrogen - low reactivity

CHEMICAL CHANGES (RXN) During a chemical change, the composition of matter always changes Reactant Product

INDICATORS OF CHEMICAL CHANGE Formation of precipitate A solid that forms and separates from a mixture Production of a gas Change in p. H Energy change Color change Light

CONSERVATION OF MASS - During a chemical reaction, the mass of the products is always equal to the mass of reactants - Law of Conservation of Mass
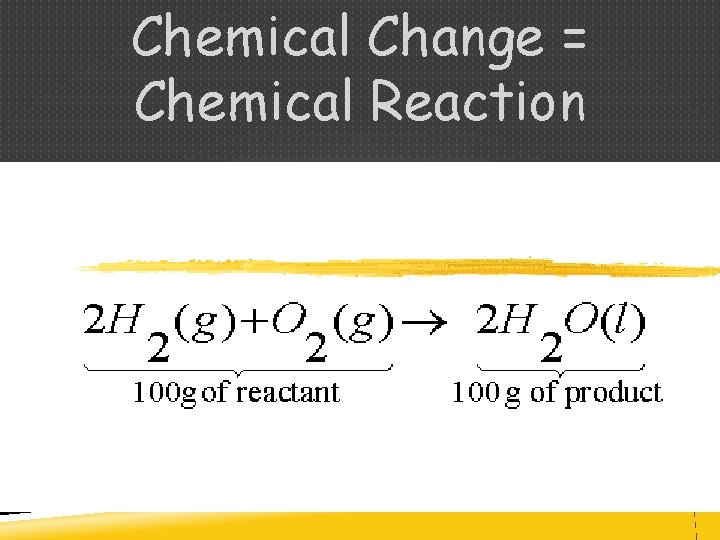
Chemical Change = Chemical Reaction

Which of the following is not a chemical change? A. Paper being shredded B. Steel rusting C. Charcoal burning D. Newspaper yellowing in the sun

Which of these properties could not be used to distinguish between table salt and sugar A. Boiling point B. Melting point C. Density D. Color
- Slides: 41